Abstract
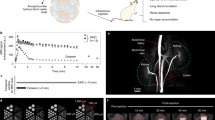
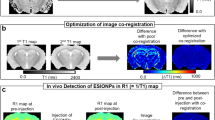
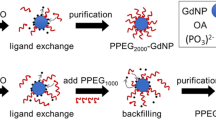
Iron-oxide-based contrast agents for magnetic resonance imaging (MRI) had been clinically approved in the United States and Europe, yet most of these nanoparticle products were discontinued owing to failures to meet rigorous clinical requirements. Significant advances have been made in the synthesis of magnetic nanoparticles and their biomedical applications, but several major challenges remain for their clinical translation, in particular large-scale and reproducible synthesis, systematic toxicity assessment, and their preclinical evaluation in MRI of large animals. Here, we report the results of a toxicity study of iron oxide nanoclusters of uniform size in large animal models, including beagle dogs and the more clinically relevant macaques. We also show that iron oxide nanoclusters can be used as T 1 MRI contrast agents for high-resolution magnetic resonance angiography in beagle dogs and macaques, and that dynamic MRI enables the detection of cerebral ischaemia in these large animals. Iron oxide nanoclusters show clinical potential as next-generation MRI contrast agents.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Michalet, X. et al. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 307, 538–544 (2005).
Nam, J. M., Thaxton, C. S. & Mirkin, C. A. Nanoparticle-based bio-bar codes for the ultrasensitive detection of proteins. Science 301, 1884–1886 (2003).
Chou, L. Y. T., Zagorovsky, K. & Chan, W. C. W. DNA assembly of nanoparticle superstructures for controlled biological delivery and elimination. Nat. Nanotech. 9, 148–155 (2014).
Howes, P. D., Chandrawati, R. & Stevens, M. M. Colloidal nanoparticles as advanced biological sensors. Science 346, 1247390 (2014).
Maggiorella, L. et al. Nanoscale radiotherapy with hafnium oxide nanoparticles. Future Oncol. 8, 1167–1181 (2012).
Libutti, S. K. et al. Phase I and pharmacokinetic studies of CYT-6091, a novel PEGylated colloidal gold-rhTNF nanomedicine. Clin. Cancer Res. 16, 6139–6149 (2010).
Zhao, Y. Z. et al. Bioengineered magnetoferritin nanoprobes for single-dose nuclear-magnetic resonance tumor imaging. ACS Nano 10, 4184–4191 (2016).
Lee, H., Sun, E., Ham, D. & Weissleder, R. Chip-NMR biosensor for detection and molecular analysis of cells. Nat. Med. 14, 869–874 (2008).
Zhang, C. et al. Magnesium silicide nanoparticles as a deoxygenation agent for cancer starvation therapy. Nat. Nanotech. 12, 378–386 (2017).
Fan, K. L. et al. Magnetoferritin nanoparticles for targeting and visualizing tumour tissues. Nat. Nanotech 7, 459–464 (2012).
Ghosh, D. et al. M13-templated magnetic nanoparticles for targeted in vivo imaging of prostate cancer. Nat. Nanotech. 7, 677–682 (2012).
Etoc, F. et al. Subcellular control Rac-GTPase signalling by magnetogenetic manipulation inside living cells. Nat. Nanotech. 8, 193–198 (2013).
Chan, K. W. Y. et al. MRI-detectable pH nanosensors incorporated into hydrogels for in vivo sensing of transplanted-cell viability. Nat. Mater. 12, 268–275 (2013).
Kircher, M. F. et al. A brain tumor molecular imaging strategy using a new triple-modality MRI-photoacoustic-Raman nanoparticle. Nat. Med. 18, 829–834 (2012).
Chapman, S. et al. Nanoparticles for cancer imaging: the good, the bad, and the promise. Nano Today 8, 454–460 (2013).
Lewin, M. et al. Tat peptide-derivatized magnetic nanoparticles allow in vivo tracking and recovery of progenitor cells. Nat. Biotechnol. 18, 410–414 (2000).
Bulte, J. W. M. et al. Magnetodendrimers allow endosomal magnetic labeling and in vivo tracking of stem cells. Nat. Biotechnol. 19, 1141–1147 (2001).
Cho, M. H. et al. A magnetic switch for the control of cell death signalling in in vitro and in vivo systems. Nat. Mater. 11, 1038–1043 (2012).
Lee, J. H. et al. Exchange-coupled magnetic nanoparticles for efficient heat induction. Nat. Nanotech. 6, 418–422 (2011).
Gao, J. H., Gu, H. W. & Xu, B. Multifunctional magnetic nanoparticles: design, synthesis, and biomedical applications. Acc. Chem. Res. 42, 1097–1107 (2009).
Della Rocca, J., Liu, D. M. & Lin, W. B. Nanoscale metal-organic frameworks for biomedical imaging and drug delivery. Acc. Chem. Res. 44, 957–968 (2011).
Lee, N. et al. Iron oxide based nanoparticles for multimodal imaging and magnetoresponsive therapy. Chem. Rev. 115, 10637–10689 (2015).
Long, M. J. C., Pan, Y., Lin, H. C., Hedstrom, L. & Xu, B. Cell compatible trimethoprim-decorated iron oxide nanoparticles bind dihydrofolate reductase for magnetically modulating focal adhesion of mammalian cells. J. Am. Chem. Soc. 133, 10006–10009 (2011).
Pan, Y., Du, X. W., Zhao, F. & Xu, B. Magnetic nanoparticles for the manipulation of proteins and cells. Chem. Soc. Rev. 41, 2912–2942 (2012).
Issadore, D. et al. Self-assembled magnetic filter for highly efficient immunomagnetic separation. Lab Chip 11, 147–151 (2011).
Min, Y. Z., Caster, J. M., Eblan, M. J. & Wang, A. Z. Clinical translation of nanomedicine. Chem. Rev. 115, 11147–11190 (2015).
Lee, N. & Hyeon, T. Designed synthesis of uniformly sized iron oxide nanoparticles for efficient magnetic resonance imaging contrast agents. Chem. Soc. Rev. 41, 2575–2589 (2012).
Arami, H., Khandhar, A., Liggitt, D. & Krishnan, K. M. In vivo delivery, pharmacokinetics, biodistribution and toxicity of iron oxide nanoparticles. Chem. Soc. Rev. 44, 8576–8607 (2015).
Balakrishnan, V. S. et al. Physicochemical properties of ferumoxytol, a new intravenous iron preparation. Eur. J. Clin. Invest. 39, 489–496 (2009).
Yilmaz, A., Rosch, S., Yildiz, H., Klumpp, S. & Sechtem, U. First multiparametric cardiovascular magnetic resonance study using ultrasmall superparamagnetic iron oxide nanoparticles in a patient with acute myocardial infarction: new vistas for the clinical application of ultrasmall superparamagnetic iron oxide. Circulation 126, 1932–1934 (2012).
Penfield, J. G. & Reilly, R. F. What nephrologists need to know about gadolinium. Nat. Clin. Pract. Nephrol. 3, 654–668 (2007).
White, G. W., Gibby, W. A. & Tweedle, M. F. Comparison of Gd(DTPA-BMA) (Omniscan) versus Gd(HP-DO3A) (ProHance) relative-to gadolinium retention in human bone tissue by inductively coupled plasma mass spectroscopy. Invest. Radiol. 41, 272–278 (2006).
Kanda, T. et al. Gadolinium-based contrast agent accumulates in the brain even in subjects without severe renal dysfunction: evaluation of autopsy brain specimens with inductively coupled plasma mass spectroscopy. Radiology 276, 228–232 (2015).
Ghazani, A. A. et al. Molecular characterization of scant lung tumor cells using iron-oxide nanoparticles and micro-nuclear magnetic resonance. Nanomedicine 10, 661–668 (2014).
Shao, H. L. et al. Magnetic nanoparticles and microNMR for diagnostic applications. Theranostics 2, 55–65 (2012).
Shao, H. L., Yoon, T. J., Liong, M., Weissleder, R. & Lee, H. Magnetic nanoparticles for biomedical NMR-based diagnostics. Beilstein J. Nanotechnol. 1, 142–154 (2010).
Issadore, D. et al. Miniature magnetic resonance system for point-of-care diagnostics. Lab Chip 11, 2282–2287 (2011).
Ghazani, A. A., Castro, C. M., Gorbatov, R., Lee, H. & Weissleder, R. Sensitive and direct detection of circulating tumor cells by multimarker µ-nuclear magnetic resonance. Neoplasia 14, 388–395 (2012).
De Vries, I. J. M. et al. Magnetic resonance tracking of dendritic cells in melanoma patients for monitoring of cellular therapy. Nat. Biotechnol. 23, 1407–1413 (2005).
Valencia, P. M., Farokhzad, O. C., Karnik, R. & Langer, R. Microfluidic technologies for accelerating the clinical translation of nanoparticles. Nat. Nanotech. 7, 623–629 (2012).
Cheng, Z. L., Al Zaki, A., Hui, J. Z., Muzykantov, V. R. & Tsourkas, A. Multifunctional nanoparticles: cost versus benefit of adding targeting and imaging capabilities. Science 338, 903–910 (2012).
Yoon, T. J., Lee, H., Shao, H. L., Hilderbrand, S. A. & Weissleder, R. Multicore assemblies potentiate magnetic properties of biomagnetic nanoparticles. Adv. Mater. 23, 4793–4797 (2011).
Yoon, T. J., Lee, H., Shao, H. L. & Weissleder, R. Highly magnetic core-shell nanoparticles with a unique magnetization mechanism. Angew. Chem. Int. Ed. 50, 4663–4666 (2011).
Park, J. et al. Ultra-large-scale syntheses of monodisperse nanocrystals. Nat. Mater. 3, 891–895 (2004).
Laurent, S. et al. Magnetic iron oxide nanoparticles: synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 108, 2064–2110 (2008).
Choi, H. S. et al. Renal clearance of quantum dots. Nat. Biotechnol. 25, 1165–1170 (2007).
Jiang, W., Kim, B. Y. S., Rutka, J. T. & Chan, W. C. W. Nanoparticle-mediated cellular response is size-dependent. Nat. Nanotech. 3, 145–150 (2008).
Choi, H. S. et al. Design considerations for tumour-targeted nanoparticles. Nat. Nanotech. 5, 42–47 (2010).
Ye, L. et al. A pilot study in non-human primates shows no adverse response to intravenous injection of quantum dots. Nat. Nanotech. 7, 453–458 (2012).
Yan, G. M. et al. Genome sequencing and comparison of two nonhuman primate animal models, the cynomolgus and Chinese rhesus macaques. Nat. Biotechnol. 29, 1019–1023 (2011).
Kim, B. H. et al. Large-scale synthesis of uniform and extremely small-sized iron oxide nanoparticles for high-resolution T 1 magnetic resonance imaging contrast agents. J. Am. Chem. Soc. 133, 12624–12631 (2011).
Boles, M. A., Ling, D., Hyeon, T. & Talapin, D. V. The surface science of nanocrystals. Nat. Mater. 15, 141–153 (2016).
Na, H. B. et al. Versatile PEG-derivatized phosphine oxide ligands for water-dispersible metal oxide nanocrystals. Chem. Commun. 5167–5169 (2007).
Rowe, R. C., Sheskey, P. J. & Quinn, M. E. Handbook of Pharmaceutical Excipients 6th edn (Pharmaceutical Press, London, UK, and American Pharmacists Association, Washington, USA, 2009).
Van der Meel, R., Vehmeijer, L. J. C., Kok, R. J., Storm, G. & van Gaal, E. V. B. Ligand-targeted particulate nanomedicines undergoing clinical evaluation: current status. Adv. Drug Deliv. Rev. 65, 1284–1298 (2013).
Stirland, D. L., Nichols, J. W., Miura, S. & Bae, Y. H. Mind the gap: a survey of how cancer drug carriers are susceptible to the gap between research and practice. J. Control. Release 172, 1045–1064 (2013).
Ling, D., Lee, N. & Hyeon, T. Chemical synthesis and assembly of uniformly sized iron oxide nanoparticles for medical applications. Acc. Chem. Res. 48, 1276–1285 (2015).
Wang, Y.-X. J. Superparamagnetic iron oxide based MRI contrast agents: current status of clinical application. Quant. Imaging Med. Surg 1, 35–40 (2011).
Chen, R. et al. Parallel comparative studies on mouse toxicity of oxide nanoparticle- and gadolinium-based T 1 MRI contrast agents. ACS Nano 9, 12425–12435 (2015).
Saleem, K. S. & Logothetis, N. K. A Combined MRI and Histology Atlas of the Rhesus Monkey Brain in Stereotaxic Coordinates 1st edn (Academic Press, London, UK, 2007).
Varallyay, C. G. et al. Dynamic MRI using iron oxide nanoparticles to assess early vascular effects of antiangiogenic versus corticosteroid treatment in a glioma model. J. Cereb. Blood Flow Metab. 29, 853–860 (2009).
Kim, S. G. et al. Cerebral blood volume MRI with intravascular superparamagnetic iron oxide nanoparticles. NMR Biomed. 26, 949–962 (2013).
Coelho, O. R., Rickers, C., Kwong, R. Y. & Jerosch-Herold, M. MR myocardial perfusion imaging. Radiology 266, 701–715 (2013).
Acknowledgements
T.H. and S.H.C. acknowledge financial support from the Research Center Program of the Institute for Basic Science (IBS-R006-D1) in Korea. Y.L., D.-H.Z., D.L. and L.Z. acknowledge financial support from the National Key Research and Development Program of China (2016YFA0203600), National Natural Science Foundation of China (51572067, 21501039, 51503180, 31370983, 81401518, 31430028 and 5161101036), Fundamental Research Funds for the Central Universities (2015HGCH0009), Anhui Province Funds for Distinguished Young Scientists (1508085J08), Natural Science Foundation of Anhui Province (1708085ME114) and Young Top-Notch Talent Support Scheme at Anhui Medical University.
Author information
Authors and Affiliations
Contributions
T.H., S.H.C., D.-H.Z., Y.L., D.L., Y.-J.X. and G.-b.Z. conceived the idea and designed the experiments. T.H., S.H.C. and D.-H.Z. supervised the research. Y.L., D.L., B.H.K., H.C., J.K. and N.L. processed the samples. D.-H.Z., T.W., Y.Z., J.-C.H. and L.Z. worked on the macaque breeding. G.-b.Z. and M.-q.W. worked on the cerebral ischaemia model. Y.-J.X., Y.L., D.L., N.L. and S.H.C. investigated the MR performance. T.W., Y.Z., L.Z. and H.-Q.W. processed the biocompatibility evaluation. Y.-D.W., L.D., F.L. and B.Y. performed the biodistribution and pharmacokinetic study in vivo. X.-T.H. performed the pathological analysis. Y.L., D.L., M.J.H., Y.-J.X., G.-b.Z., N.L., T.H., S.H.C. and D.-H.Z. wrote the paper. All authors analysed and discussed the results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lu, Y., Xu, YJ., Zhang, Gb. et al. Iron oxide nanoclusters for T 1 magnetic resonance imaging of non-human primates. Nat Biomed Eng 1, 637–643 (2017). https://doi.org/10.1038/s41551-017-0116-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-017-0116-7
This article is cited by
-
Hypersensitive MR angiography based on interlocking stratagem for diagnosis of cardiac-cerebral vascular diseases
Nature Communications (2023)
-
Advances in magnetic nanoparticle-based magnetic resonance imaging contrast agents
Nano Research (2023)
-
Biodegradable and biocompatible exceedingly small magnetic iron oxide nanoparticles for T1-weighted magnetic resonance imaging of tumors
Journal of Nanobiotechnology (2022)
-
Effects of iron oxide nanoparticles as T2-MRI contrast agents on reproductive system in male mice
Journal of Nanobiotechnology (2022)
-
A hepatocyte-targeting nanoparticle for enhanced hepatobiliary magnetic resonance imaging
Nature Biomedical Engineering (2022)