Abstract
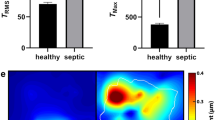
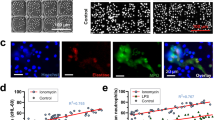
Neutrophil swarms protect healthy tissues by sealing off sites of infection. In the absence of swarming, microbial invasion of surrounding tissues can result in severe infections. Recent observations in animal models have shown that swarming requires rapid neutrophil responses and well-choreographed neutrophil migration patterns. However, in animal models, physical access to the molecular signals coordinating neutrophil activities during swarming is limited. Here, we report the development and validation of large microscale arrays of zymosan particle clusters for the study of human neutrophils during swarming ex vivo. We characterized the synchronized swarming of human neutrophils under the guidance of neutrophil-released chemokines, and measured the mediators released at different phases of human-neutrophil swarming against targets simulating infections. We found that the network of mediators coordinating human-neutrophil swarming includes start and stop signals, proteolytic enzymes and enzyme inhibitors, as well as modulators of activation of other immune and non-immune cells. We also show that the swarming behaviour of neutrophils from patients following major trauma is deficient and gives rise to smaller swarms than those of neutrophils from healthy individuals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ng, L. G. et al. Visualizing the neutrophil response to sterile tissue injury in mouse dermis reveals a three-phase cascade of events. J. Invest. Dermatol. 131, 2058–2068 (2011).
Lammermann, T. et al. Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature 498, 371–375 (2013).
Afonso, P. V. et al. LTB4 is a signal-relay molecule during neutrophil chemotaxis. Dev. Cell 22, 1079–1091 (2012).
Kienle, K. & Lammermann, T. Neutrophil swarming: an essential process of the neutrophil tissue response. Immunol. Rev. 273, 76–93 (2016).
Jones, C. N. et al. Microfluidic assay for precise measurements of mouse, rat, and human neutrophil chemotaxis in whole-blood droplets. J. Leukoc. Biol. 100, 241–247 (2016).
Malawista, S. E., de Boisfleury Chevance, A., van Damme, J. & Serhan, C. N. Tonic inhibition of chemotaxis in human plasma. Proc. Natl Acad. Sci. USA 105, 17949–17954 (2008).
Lammermann, T. In the eye of the neutrophil swarm-navigation signals that bring neutrophils together in inflamed and infected tissues. J. Leukoc. Biol. 100, 55–63 (2016).
Liese, J., Rooijakkers, S. H., van Strijp, J. A., Novick, R. P. & Dustin, M. L. Intravital two-photon microscopy of host-pathogen interactions in a mouse model of Staphylococcus aureus skin abscess formation. Cell Microbiol. 15, 891–909 (2013).
Kamenyeva, O. et al. Neutrophil recruitment to lymph nodes limits local humoral response to Staphylococcus aureus. PLoS Pathog. 11, e1004827 (2015).
Kreisel, D. et al. In vivo two-photon imaging reveals monocyte-dependent neutrophil extravasation during pulmonary inflammation. Proc. Natl Acad. Sci. USA 107, 18073–18078 (2010).
Chtanova, T. et al. Dynamics of neutrophil migration in lymph nodes during infection. Immunity 29, 487–496 (2008).
Bruns, S. et al. Production of extracellular traps against Aspergillus fumigatus in vitro and in infected lung tissue is dependent on invading neutrophils and influenced by hydrophobin RodA. PLoS Pathog. 6, e1000873 (2010).
Jones, C. N. et al. Human neutrophils are primed by chemoattractant gradients for blocking the growth of Aspergillus fumigatus. J. Infect. Dis. 213, 465–475 (2016).
Leliefeld, P. H., Wessels, C. M., Leenen, L. P., Koenderman, L. & Pillay, J. The role of neutrophils in immune dysfunction during severe inflammation. Crit. Care 20, 73 (2016).
Malawista, S. E., de Boisfleury, A. C. & Naccache, P. H. Inflammatory gout: observations over a half-century. FASEB J. 25, 4073–4078 (2011).
Boneschansker, L., Yan, J., Wong, E., Briscoe, D. M. & Irimia, D. Microfluidic platform for the quantitative analysis of leukocyte migration signatures. Nat. Commun. 5, 4787 (2014).
Serhan, C. N. Novel lipid mediators and resolution mechanisms in acute inflammation: to resolve or not? Am. J. Pathol. 177, 1576–1591 (2010).
Levy, B. D., Clish, C. B., Schmidt, B., Gronert, K. & Serhan, C. N. Lipid mediator class switching during acute inflammation: signals in resolution. Nat. Immunol. 2, 612–619 (2001).
Serhan, C. N. et al. Anti-inflammatory actions of neuroprotectin D1/protectin D1 and its natural stereoisomers: assignments of dihydroxy-containing docosatrienes. J. Immunol. 176, 1848–1859 (2006).
Hammerschmidt, D. E., Bowers, T. K., Lammi-Keefe, C. J., Jacob, H. S. & Craddock, P. R. Granulocyte aggregometry: a sensitive technique for the detection of C5a and complement activation. Blood 55, 898–902 (1980).
Wagner, J. L. & Hugli, T. E. Radioimmunoassay for anaphylatoxins: a sensitive method for determining complement activation products in biological fluids. Anal. Biochem. 136, 75–88 (1984).
Harvie, E. A., Green, J. M., Neely, M. N. & Huttenlocher, A. Innate immune response to Streptococcus iniae infection in zebrafish larvae. Infect. Immun. 81, 110–121 (2013).
Bazzoni, F. et al. Phagocytosing neutrophils produce and release high amounts of the neutrophil-activating peptide 1/interleukin 8. J. Exp. Med. 173, 771–774 (1991).
Guerrero, A. T. et al. Involvement of LTB4 in zymosan-induced joint nociception in mice: participation of neutrophils and PGE2 . J. Leukoc. Biol. 83, 122–130 (2008).
Bhaumik, P., St-Pierre, G., Milot, V., St-Pierre, C. & Sato, S. Galectin-3 facilitates neutrophil recruitment as an innate immune response to a parasitic protozoa cutaneous infection. J. Immunol. 190, 630–640 (2013).
Lopez-Dee, Z., Pidcock, K. & Gutierrez, L. S. Thrombospondin-1: multiple paths to inflammation. Mediators Inflamm. 2011, 296069 (2011).
Schenk, B. I., Petersen, F., Flad, H. D. & Brandt, E. Platelet-derived chemokines CXC chemokine ligand (CXCL)7, connective tissue-activating peptide III, and CXCL4 differentially affect and cross-regulate neutrophil adhesion and transendothelial migration. J. Immunol. 169, 2602–2610 (2002).
Senior, R. M., Gresham, H. D., Griffin, G. L., Brown, E. J. & Chung, A. E. Entactin stimulates neutrophil adhesion and chemotaxis through interactions between its Arg-Gly-Asp (RGD) domain and the leukocyte response integrin. J. Clin. Invest. 90, 2251–2257 (1992).
Baltus, T. et al. Differential and additive effects of platelet-derived chemokines on monocyte arrest on inflamed endothelium under flow conditions. J. Leukoc. Biol. 78, 435–441 (2005).
Dziarski, R., Platt, K. A., Gelius, E., Steiner, H. & Gupta, D. Defect in neutrophil killing and increased susceptibility to infection with nonpathogenic gram-positive bacteria in peptidoglycan recognition protein-S (PGRP-S)-deficient mice. Blood 102, 689–697 (2003).
Bornfeldt, K. E. et al. Platelet-derived growth factor. Distinct signal transduction pathways associated with migration versus proliferation. Ann. NY Acad. Sci. 766, 416–430 (1995).
Markowska, A. I., Liu, F. T. & Panjwani, N. Galectin-3 is an important mediator of VEGF- and bFGF-mediated angiogenic response. J. Exp. Med. 207, 1981–1993 (2010).
Yang, J. et al. An iron delivery pathway mediated by a lipocalin. Mol. Cell 10, 1045–1056 (2002).
Garlanda, C. et al. Non-redundant role of the long pentraxin PTX3 in anti-fungal innate immune response. Nature 420, 182–186 (2002).
Iyer, R. P., Patterson, N. L., Fields, G. B. & Lindsey, M. L. The history of matrix metalloproteinases: milestones, myths, and misperceptions. Am. J. Physiol. Heart Circ. Physiol. 303, H919– H930 (2012).
Arita, M. et al. Metabolic inactivation of resolvin E1 and stabilization of its anti-inflammatory actions. J. Biol. Chem. 281, 22847–22854 (2006).
Serhan, C. N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 510, 92–101 (2014).
Murata, T. et al. Anti-inflammatory role of PGD2 in acute lung inflammation and therapeutic application of its signal enhancement. Proc. Natl Acad. Sci. USA 110, 5205–5210 (2013).
Crea, A. E., Nakhosteen, J. A. & Lee, T. H. Mediator concentrations in bronchoalveolar lavage fluid of patients with mild asymptomatic bronchial asthma. Eur. Respir. J. 5, 190–195 (1992).
Montuschi, P. & Barnes, P. J. Exhaled leukotrienes and prostaglandins in asthma. J. Allergy Clin. Immunol. 109, 615–620 (2002).
Dalli, J. et al. Human sepsis eicosanoid and proresolving lipid mediator temporal profiles: correlations with survival and clinical outcomes. Crit. Care Med. 45, 58–68 (2017).
Jones, C. N. et al. Spontaneous neutrophil migration patterns during sepsis after major burns. PLoS ONE 9, e114509 (2014).
Vincent, J. L. Nosocomial infections in adult intensive-care units. Lancet 361, 2068–2077 (2003).
Colas, R. A., Shinohara, M., Dalli, J., Chiang, N. & Serhan, C. N. Identification and signature profiles for pro-resolving and inflammatory lipid mediators in human tissue. Am. J. Physiol. Cell Physiol. 307, C39–C54 (2014).
Reátegui, E. et al. Dataset for ‘Microscale arrays for the profiling of start and stop signals coordinating human-neutrophil swarming’. figsharehttp://dx.doi.org/10.6084/m9.figshare.5024567 (2017).
Acknowledgements
We thank B. Hamza and J. M. Martel of the BioMEMS Resource Center for help with microfabrication and stimulating discussions. This work was supported by a grant from the National Institute of General Medical Sciences (GM092804) and funding from the Harvard Medical School Tools and Technology Fund. Microfabrication was conducted at the BioMEMS Resource Center at Massachusetts General Hospital, supported by a grant from the National Institute of Biomedical Imaging and Bioengineering (EB002503). Work in the CNS laboratories was supported by the National Institutes of Health (GM095467 and GM38765).
Author information
Authors and Affiliations
Contributions
E.R. and D.I. designed the research; E.R., A.H.K., F.J., H.C. and E.W. designed and prepared the microfluidic devices; E.R. conducted the experiments; E.R., F.J., H.E., J.L. and D.I. analysed the results; C.N.S. and J.D. performed the metabololipidomic analysis; H.E. developed the biophysical model for cell swarming; and E.R. and D.I. prepared the manuscript, with significant contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures, tables and video captions. (PDF 2255 kb)
Supplementary Video 1
Human-neutrophil swarming on arrays of clusters of zymosan particles. (MOV 1384 kb)
Supplementary Video 2
Human-neutrophil swarming on a single cluster of zymosan particles. (MOV 2031 kb)
Supplementary Video 3
Human neutrophils do not swarm on solitary zymosan particles. (MOV 1491 kb)
Supplementary Video 4
Human-neutrophil tracking during swarming. (MOV 2644 kb)
Supplementary Video 5
Human-neutrophil tracking during swarming in the presence of LTB4 receptor. (MOV 5597 kb)
Rights and permissions
About this article
Cite this article
Reátegui, E., Jalali, F., Khankhel, A. et al. Microscale arrays for the profiling of start and stop signals coordinating human-neutrophil swarming. Nat Biomed Eng 1, 0094 (2017). https://doi.org/10.1038/s41551-017-0094
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-017-0094
This article is cited by
-
A genetically encoded sensor for visualizing leukotriene B4 gradients in vivo
Nature Communications (2023)
-
ComplexEye: a multi-lens array microscope for high-throughput embedded immune cell migration analysis
Nature Communications (2023)
-
Chemotaxis and swarming in differentiated HL-60 neutrophil-like cells
Scientific Reports (2021)
-
TICAM2-related pathway mediates neutrophil exhaustion
Scientific Reports (2020)
-
Neutrophil swarming delays the growth of clusters of pathogenic fungi
Nature Communications (2020)