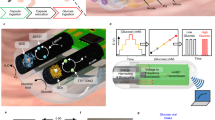
Abstract
Ingestible electronics have revolutionized the standard of care for a variety of health conditions. Extending the capacity and safety of these devices, and reducing the costs of powering them, could enable broad deployment of prolonged-monitoring systems for patients. Although previous biocompatible power-harvesting systems for in vivo use have demonstrated short (minute-long) bursts of power from the stomach, little is known about the potential for powering electronics in the longer term and throughout the gastrointestinal tract. Here, we report the design and operation of an energy-harvesting galvanic cell for continuous in vivo temperature sensing and wireless communication. The device delivered an average power of 0.23 μW mm−2 of electrode area for an average of 6.1 days of temperature measurements in the gastrointestinal tract of pigs. This power-harvesting cell could provide power to the next generation of ingestible electronic devices for prolonged periods of time inside the gastrointestinal tract.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Iddan, G., Meron, G., Glukhovsky, A. & Swain, P. Wireless capsule endoscopy. Nature 405, 417 (2000).
van der Schaar, P. J. et al. A novel ingestible electronic drug delivery and monitoring device. Gastrointest. Endosc. 78, 520–528 (2013).
Maqbool, S., Parkman, H. P. & Friedenberg, F. K. Wireless capsule motility: comparison of the SmartPill(R) GI monitoring system with scintigraphy for measuring whole gut transit. Dig. Dis. Sci. 54, 2167–2174 (2009).
Traverso, G. et al. Physiologic status monitoring via the gastrointestinal tract. PLoS ONE 10, e0141666 (2015).
Ramadass, Y. K. & Chandrakasan, A. P. A battery-less thermoelectric energy harvesting interface circuit with 35 mV startup voltage. IEEE J. Solid-State Circuits 46, 333–341 (2011).
Dagdeviren, C. et al. Conformal piezoelectric energy harvesting and storage from motions of the heart, lung, and diaphragm. Proc. Natl Acad. Sci. USA 111, 1927–1932 (2014).
Waters, B. H., Sample, A. P., Bonde, P. & Smith, J. R. Powering a ventricular assist device (VAD) with the free-range resonant electrical energy delivery (FREE-D) system. Proc. IEEE 100, 138–149 (2012).
Ho, J. S. et al. Wireless power transfer to deep-tissue microimplants. Proc. Natl Acad. Sci. USA 111, 7974–7979 (2014).
Laulicht, B., Traverso, G., Deshpande, V., Langer, R. & Karp, J. M. Simple battery armor to protect against gastrointestinal injury from accidental ingestion. Proc. Natl Acad. Sci. USA 111, 16490–16495 (2014).
Mercier, P. P., Lysaght, A. C., Bandyopadhyay, S., Chandrakasan, A. P. & Stankovic, K. M. Energy extraction from the biologic battery in the inner ear. Nat. Biotechnol. 30, 1240–1243 (2012).
Jung, W. et al. An ultra-low power fully integrated energy harvester based on self-oscillating switched-capacitor voltage doubler. IEEE J. Solid-State Circuits 49, 2800–2811 (2014).
El-Damak, D. & Chandrakasan, A. P. A 10 nW–1 μW power management IC with integrated battery management and self-startup for energy harvesting applications. IEEE J. Solid-State Circuits 51, 943–954 (2016).
Zhang, S. et al. A pH-responsive supramolecular polymer gel as an enteric elastomer for use in gastric devices. Nat. Mater. 14, 1065–1071 (2015).
Yin, L. et al. Materials, designs, and operational characteristics for fully biodegradable primary batteries. Adv. Mater. 26, 3879–3884 (2014).
Lee, K. B. & Lin, L. Electrolyte-based on-demand and disposable microbattery. IEEE J. Microelectromechanical Syst. 12, 840–847 (2003).
Garay, E. F. & Bashirullah, R. Biofluid activated microbattery for disposable microsystems. IEEE J. Microelectromechanical Syst. 24, 70–79 (2015).
Kim, Y. J., Chun, S.-E., Whitacre, J. & Bettinger, C. J. Self-deployable current sources fabricated from edible materials. J. Mater. Chem. B 1, 3781–3788 (2013).
Hafezi, H. et al. An ingestible sensor for measuring medication adherence. IEEE Trans. Biomed. Eng. 62, 99–109 (2015).
Jimbo, H. & Miki, N. Gastric-fluid-utilizing micro battery for micro medical devices. Sens. Actuat. B. 134, 219–224 (2008).
Mostafalu, P. & Sonkusale, S. Flexible and transparent gastric battery: energy harvesting from gastric acid for endoscopy application. Biosens. Bioelectron. 54, 292–296 (2014).
Di Maio, S. & Carrier, R. L. Gastrointestinal contents in fasted state and post-lipid ingestion: in vivo measurements and in vitro models for studying oral drug delivery. J. Control. Release 151, 110–122 (2011).
Roy, O. Z. & Wehnert, R. W. Improvements in biogalvanic energy sources. Med. Biol. Eng. 12, 50–56 (1974).
She, D., Tsang, M., Kim, J. K. & Allen, M. G. Immobilized electrolyte biodegradable batteries for implantable MEMS. In 18th Int. Conf. Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS) 494–497 (IEEE, 2015).
Dietary Reference Intakes for Vitamin A, K, Aresenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc (Food and Nutrition Board, Institute of Medicine, USA, 2001).
Haynes, W. M. CRC Handbook of Chemistry and Physics (CRC, 2015).
Kear, G., Barker, B. D. & Walsh, F. C. Electrochemical corrosion of unalloyed copper in chloride media—a critical review. Corros. Sci. 46, 109–135 (2004).
Low Voltage Digitially Controlled Potentiometer, ISL23315 (Intersil, 2015).
8-Bit Flash Microcontroller with XLP Technology, PIC12LF1840T39A (Microchip, 2014).
Snoeck, V. et al. Gastrointestinal transit time of nondisintegrating radio-opaque pellets in suckling and recently weaned piglets. J. Control. Release 94, 143–153 (2004).
Hossain, M., Abramowitz, W., Watrous, B. J., Szpunar, G. J. & Ayres, J. W. Gastrointestinal transit of nondisintegrating, nonerodible oral dosage forms in pigs. Pharm. Res. 7, 1163–1166 (1990).
Traverso, G. et al. Microneedles for drug delivery via the gastrointestinal tract. J. Pharm. Sci. 104, 362–367 (2015).
Ultra Low Power Boost Converter with Battery Management for Energy Harverster Applications, BQ25504 (Texas Instruments, 2015).
Santini, J. T. Jr, Cima, M. J. & Langer, R. A controlled-release microchip. Nature 397, 335–338 (1999).
Santini J. T. Jr, Richards, A. C., Scheidt, R., Cima, M. J. & Langer, R. Microchips as controlled drug-delivery devices. Angew. Chem. Int. Ed. 39, 2396–2407 (2000).
Singeap, A.-M. Capsule endoscopy: the road ahead. World J. Gastroenterol. 22, 369 (2016).
Reardon, S. Electroceuticals spark interest. Nature 511, 18 (2014).
Traverso, G. & Langer, R. Perspective: special delivery for the gut. Nature 519, S19 (2015).
Niven, D. J. et al. Accuracy of peripheral thermometers for estimating temperature: a systematic review and meta-analysis. Ann. Intern. Med. 163, 768–777 (2015).
Harpe, P., Gao, H., Dommele, R. V., Cantatore, E. & van Roermund, A. H. M . A 0.20 mm2 3 nW signal acquisition IC for miniature sensor nodes in 65 nm CMOS. IEEE J. Solid-State Circuits 51, 240–248 (2016).
Yaul, F. M. & Chandrakasan, A. P. A 10 bit SAR ADC with data-dependent energy reduction using LSB-first successive approximation. IEEE J. Solid-State Circuits 49, 2825–2834 (2014).
Paidimarri, A., Ickes, N. & Chandrakasan, A. P. A +10dBm 2.4GHz transmitter with sub-400pW leakage and 43.7% system efficiency. In 2015 IEEE Int. Solid-State Circuits Conf. (ISSCC) Digest of Technical Papers 1–3 (IEEE, 2015).
Fojtik, M. et al. A millimeter-scale energy-autonomous sensor system with stacked battery and solar cells. IEEE. J. Solid-State Circuits 48, 801–813 (2013).
Kethu, S. R. et al. Endoluminal bariatric techniques. Gastrointest. Endosc. 76, 1–7 (2012).
Rapoport, B. I., Kedzierski, J. T. & Sarpeshkar, R. A glucose fuel cell for implantable brain-machine interfaces. PLoS ONE 7, e38436 (2012).
Schoellhammer, C. M. et al. Ultrasound-mediated gastrointestinal drug delivery. Sci. Transl. Med. 7, 310ra168 (2015).
Nadeau, P. et al. Data for Prolonged energy harvesting for ingestible devices. figsharehttp://dx.doi.org/10.6084/m9.figshare.4451420 (2017).
Acknowledgements
We thank J. Haupt, M. Jamiel and A. Hayward for help with the in vivo porcine work. We also thank A. Paidimarri for helpful discussions. A.P.C. was funded by Texas Instruments, the Semiconductor Research Corporation’s Center of Excellence for Energy Efficient Electronics, and the Hong Kong Innovation and Technology Commission. R.L. was funded by a National Institutes of Health grant, EB-000244; a Max Planck Research Award, Ltr Dtd. 2/11/08; and the Alexander von Humboldt-Stiftung Foundation. G.T. was funded in part by the Division of Gastroenterology, Brigham and Women’s Hospital.
Author information
Authors and Affiliations
Contributions
P.N., D.E-D., D.G., Y.L.K., N.R., R.L., A.P.C. and G.T. conceived and designed the research. P.N., D.E-D., S.M., Y.L.K. and N.R. constructed the prototypes for testing. P.N., D.E-D., D.G. and Y.L.K. conducted the in vitro characterization. P.N. wrote the software for the capsules and offline processing of the packets. P.N., D.E-D., D.G., Y.L.K., C.C., L.B. and G.T. performed the in vivo pig experiments. P.N., D.E-D., D.G., Y.L.K., N.R., R.L., A.P.C. and G.T. analysed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that provisional patent application no. 62/328,084, covering a portion of this work, was filed with the United States Patent and Trademark Office on 27 April 2016.
Supplementary information
Supplementary Information
Supplementary Figures (PDF 3420 kb)
Rights and permissions
About this article
Cite this article
Nadeau, P., El-Damak, D., Glettig, D. et al. Prolonged energy harvesting for ingestible devices. Nat Biomed Eng 1, 0022 (2017). https://doi.org/10.1038/s41551-016-0022
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-016-0022
This article is cited by
-
Accelerated intestinal wound healing via dual electrostimulation from a soft and biodegradable electronic bandage
Nature Electronics (2024)
-
Digital automation of transdermal drug delivery with high spatiotemporal resolution
Nature Communications (2024)
-
Effect of bias potential and dimension on electrochemical migration of capacitors for implantable devices
npj Materials Degradation (2024)
-
A soil-inspired dynamically responsive chemical system for microbial modulation
Nature Chemistry (2023)
-
A swallowable X-ray dosimeter for the real-time monitoring of radiotherapy
Nature Biomedical Engineering (2023)