Abstract
Technologies that allow for removal and subsequent recovery and reuse of phosphorus from polluted streams are imperative. One such technology is hybrid ion exchange nanotechnology (HIX-Nano), which may allow to produce a valuable nutrient solution following phosphorus desorption of the saturated media. This study evaluated the potential of four regeneration chemistries to desorb phosphorus from a commercially available HIX-Nano resin hybridized with iron oxide nanoparticles using a design of experiments (DoE) approach. More sustainable and less harmful regeneration solutions using a KOH/K2SO4 blend or a recovered NH4OH alkaline solution, along with tap water instead of synthetic acid, were compared to a control solution of KOH and H2SO4. Among the four regeneration methods studied, using the combination of recovered NH4OH and tap water shows the highest phosphorus recovery potential because: (i) it involves low cost and sustainable products, (ii) it showed a relatively high recovery efficiency (75 ± 15% as compared to the control at 89 ± 13%), and (iii) it did not demonstrate any significant dampening of the resin longevity after five adsorption and desorption cycles. Based on the DoE data, a series of regression models was developed to generate understanding of the effect of important operational parameters (volume of the regenerant solution, rinse speed, strength of the alkaline solution) on the phosphorus concentration in the recovered nutrient solution. Overall, this study indicates that HIX-Nano may contribute to providing a cost-effective and sustainable technological solution to tackle the phosphorus problem in wastewater treatment applications across the globe.
Similar content being viewed by others
Introduction
With the rapid increase in industrialization and human population since the turn of the industrial revolution, civilization finds itself in increasingly dire need of the Earth’s resources to sustain its way of life in terms of production and consumption. Since 1960, the use of synthetic phosphorus (P) fertilizers has tripled, damaging continuously further the natural phosphorus cycle1. The pathways of phosphorus pollution are numerous, including wastewater treatment plant discharge (municipal and industrial); farm fertilizer losses due to erosion, runoff, drainage; or leaching; urban runoff (stormwater pollution); and phosphate rock fertilizer production.
The soluble reactive phosphorus component of dissolved phosphorus is a known contributor to eutrophication and the proliferation of cyanobacteria when this nutrient pollutant overloads fresh waterways2,3,4. Legislative action to combat nutrient pollution has been enacted across North America in the form of tightening total maximum discharge limits (TMDLs) and maximum allowable phosphorus limits in waterbodies across the continent. In the United States, these TMDLs span from the deep ocean waters off the coast of Hawaii to the Great Lakes and have a minimum target of 0.5 µg PO4-P/L5. The Everglades target as low as 0.019 mg/L in the effluent in order to reduce eutrophication6. In Canada, the new Pan-Canadian effluent limits go down to 0.1 mg PO4-P/L7. Similar regulations are being implemented elsewhere in the world.
Different methods by which to reduce the phosphorus introduced into water systems exist, but common technologies struggle to achieve so low a concentration as is required by legislation8. Moreover, when a chemical treatment approach is attempted for very low discharge standards (<0.5 mg/L total P), the stoichiometric requirement of metal salt (e.g., iron or aluminum salts) to phosphorus is costly. Large wastewater treatment plants with high phosphorus loads typically opt for an enhanced biological phosphorus removal process and anaerobic digestion system8. Whether chemical or biological, the above current practices focus primarily on phosphorus removal and not recovery.
Struvite precipitation has seen a growing interest over the past decade for the recovery of phosphorus from wastewater as a valuable fertilizer end-product9. However, the technology is typically only economically feasible at large wastewater treatment plants with high dissolved phosphorus loads, where it can reduce maintenance costs associated with nuisance struvite scaling10,11. The reduction cost per year taking into account a 3.56 × 107 L/day flow with a P concentration of 0.63 mg/L is about 87,600 USD if the target P concentration is 0.5 mg/L and 438,730 USD if this target concentration is 0.1 mg/L. These numbers assume that the recovered phosphorus can be sold12. Moreover, struvite precipitation typically only removes around 90% of the soluble phosphorus13, which is often not sufficient to meet stringent discharge criteria.
Alternative emerging methods to remove phosphorus down to very low concentrations and potentially recover it involve adsorption14,15 and ion exchange16,17. However, one major bottleneck of both technologies is their limited selectivity toward phosphorus due to the presence of competing anions, mainly sulfates, in wastewaters. The use of hybrid ion exchange nanotechnology (HIX-Nano), using nano-enhanced (adsorptive) ion exchange materials, may be a way to overcome this problem. Indeed, HIX-Nano uses metallic nanoparticles to greatly enhance the selectivity and adsorptive capacity of the ion exchange media, thereby providing significant benefits over traditional media18. Examples are the FerrIX A33e resin by Purolite and LayneRT by Layne, i.e., both strong anionic exchange resins (polystyrene cross-linked with divinylbenzene) impregnated with iron oxide nanoparticles. The active adsorption agents for the removal of phosphorus are the nano iron oxides, but without a host media, i.e., the ion exchange resin, these nanoparticles would wash away and potentially toxify the environment18.
The limited available literature on HIX-Nano provides evidence of the capability of ferric oxides impregnated on an ion exchange resin to adsorb and desorb phosphorus19,20. In the available studies, regeneration of the resin required an alkaline wash (typically NaOH) to remove the phosphorus and then an acid rinse (typically H2SO4) to condition the resin for a new adsorption cycle. The protonation and deprotonation of the metal oxides, in this case ferric oxide, occurs due to shifts in the solution pH around the point of zero charge (PZC)18,21. For hydrous ferric oxide, the PZC was determined experimentally to be around 7.922. A major limitation for the above studies to become economically viable at a larger scale is the high amount of chemicals needed for regeneration, along with the lack of valorization potential of the used regenerant solutions. Indeed, the current available literature targets the removal of phosphorus rather than its recovery as a formulated product. Moreover, the effect and interactions of important operational parameters such as regenerant solution volume, concentration, and rinsing speed on the regeneration efficiency have not yet been studied throughout literature, thereby limiting the potential for process optimization and hence reduction of operational costs.
To address these gaps in the understanding of phosphorus recovery via nano-enhanced absorptive media, this research aimed at designing and performing experiments using a HIX-Nano microcolumn to study the effect of different innovative and sustainable regenerant solutions on the efficiency of nutrient removal and recovery. To this end, a commercially available nano-enhanced adsorptive anion exchange resin was used. The studied regenerant solutions involve chemicals chosen to yield an N-P-K (nitrogen–phosphorus–potassium) hydroponic fertilizer post-desorption, as well as low-cost by-products obtained from other resource recovery processes implemented at wastewater treatment plants, along with tap water instead of synthetic acid. A design of experiments approach was used in order to study the effect of important operational parameters on the P concentration in the regeneration effluent solution, to evaluate its valorization potential, and to provide recommendations for optimization, all with the ultimate goal of fostering technology scale-up and implementation in industry. These operational parameters include the empty bed contact time (EBCT), the bed volume, the type of regenerant, and the regenerant concentration. Finally, a preliminary economic assessment was performed to obtain insights in the potential feasibility of the technology by means of a case study.
Results
Resin characterization
The static beaker tests yielded a maximum adsorption capacity of 21.92 ± 4.13 mg PO4-P (n = 3) adsorbed per gram of wet resin. This value was calculated after 168 h of adsorption. Figure 1 shows the breakthrough curve based on the results of the continuous adsorption experiment as concentration of PO4-P in the synthetic wastewater effluent with respect to the number of bed volumes of wastewater treated. At 500 bed volumes, the column began to experience saturation, and by 1300 bed volumes, the column was effectively saturated. Based on testing in these conditions, the resin demonstrated a maximum adsorption capacity of 3 mg PO4-P per gram of wet resin. The effluent concentration was below the detection limit (<0.001 mg PO4-P/L) until about 500 bed volumes of synthetic wastewater were treated.
Regeneration experiments
Supplementary Table 1 provides a summary of the results (concentrations of P in the recovered effluent and P recovery efficiency) of the desorption experiments conducted for all of the experimental sets, and Fig. 2 provides a graphical representation of the effluent P concentrations and recovery efficiencies with respect to the experimental set and conditions for each experiment as described in Tables 1–3.
For experimental set A, the average phosphorus concentration in the alkaline rinse effluent was 67.92 mg PO4-P/L, with a minimum phosphorus concentration of 21.70 mg PO4-P/L [Experiment A7 in Table 2] and a maximum concentration of 128.00 mg PO4-P/L [A2]. In the acid rinse, the average phosphorus concentration was 15.49 mg PO4-P/L, with a minimum of 0.00 mg PO4-P/L [A3] and a maximum of 44.00 mg PO4-P/L [A6]. As can be observed in Fig. 2, the high-volume regeneration experiments [A5–A8] showed an average recovery efficiency of 89 ± 13% compared to the low-volume average recovery efficiency of 46 ± 10% [A1–A4]. The highest recovery efficiency was obtained for experiment A6.
For experimental set B, the average concentration of phosphate in the effluent solution from the alkaline rinse was 86.79 mg PO4-P/L, with a minimum of 21.47 mg PO4-P/L [B7] and a maximum of 193.33 mg PO4-P/L [B1]. The average recovery concentration in the acid rinse step was 20.98 mg PO4-P/L, with a minimum of 0.00 mg PO4-P/L [B5] and a maximum of 125.33 mg PO4-P/L [B3]. As can be observed in Fig. 2, the high-volume experiments [B5–B8] showed an average recovery efficiency of 43 ± 13%, while the low-volume experiments [B1–B4] showed an average recovery efficiency of 24 ± 6.7%. The highest recovery efficiency was obtained for experiment B5 (75.11%).
For experimental set C, the average concentration of phosphate in the alkaline wash was 91.62 mg PO4-P/L, with a minimum value of 30.50 mg PO4-P/L [C8] and a maximum value of 168.33 mg PO4-P/L [C2]. In the acid rinse, the average phosphorus concentration recovered was 14.50 mg PO4-P/L, with a minimum of 0.00 mg PO4-P/L [C5 and C6] and a maximum of 57.33 mg PO4-P/L [C4]. As can be observed in Fig. 2, the overall average recovery efficiency of the high-volume experiments [C5–C8] was 79 ± 68% and the average recovery efficiency of the low-volume experiments [C1–C4] was 38 ± 27%. The highest recovery efficiency was obtained for experiment C6.
In the alkaline wash for experiment D, the average phosphorus concentration was 73.25 mg PO4-P/L, with a minimum of 18.83 mg PO4-P/L [D8] and a maximum of 154.00 mg PO4-P/L [D2]. In the acid rinse phase, the average phosphorus concentration was 9.29 mg PO4-P/L, with a minimum of 2.31 mg PO4-P/L [D6] and a maximum of 26.33 mg PO4-P/L [D2]. As can be observed in Fig. 2, the overall average recovery efficiency for the high-volume experiments [D4, D8] was 75 ± 54%, while the low-volume experiments [D2, D6] demonstrated an efficiency of 50 ± 32%. The highest recovery efficiency was obtained for experiment D6.
Resin longevity
Figure 3 shows the amount of phosphorus adsorbed onto the resin for each of the five repeated cycles for experimental sets C and D.
The plot shown in Fig. 3a, depicting the adsorptions for experimental set C, shows a general downward trend in efficiency across the five adsorption cycles for experimental conditions 2, 4, 5, and 7, although no clear general pattern can be observed when comparing the 8 different treatment conditions. It must be remarked that only four adsorption cycles were executed for condition 3 of experimental set C. The experimental sets and conditions are explained in Tables 1–3. The fifth measurement could not be executed due to the presence of degraded resin in the liquid sample. The discoloration caused by the leaching of iron particles from the resin matrix as well as the degradation of the resin itself made the colorimetric phosphorus measurement impossible for this sample. Resin degradation could be caused by resin swelling and shrinking. This phenomenon occurs when the resins absorb water due to a low concentration environment and desorb the water in a high concentration environment (cfr. osmosis)23. The resin might have soaked during a couple of days between experiments, and when a concentrated stream flowed through the resin bed, iron oxide followed the water through the pores23.
Concerning experimental set D, again, no significant general pattern can be observed in the resin longevity over the five adsorption–desorption cycles for the various experimental conditions. Especially for experimental conditions 2 and 4, the adsorption and desorption capacities were quite high and stable, and hence resin longevity seems to be good over five adsorption–desorption cycles.
Regression models
For each experimental set, a regression model was generated for the alkaline rinse, the acid rinse, and the total combined wash (alkaline + acid rinse) in order to predict the overall final regeneration effluent phosphorus concentration as a function of important operational parameters. The effluent concentration was chosen as the dependent variable since the main goal of the regression modeling was to gain insights into the effect of the operational parameters on the recovered fertilizer value.
The multiple regression process estimates coefficients for the effects of each of the factors and also takes into account factor interactions. The regression coefficients and their level of significance are presented in Supplementary Table 2 for experimental sets B–D (the more sustainable regeneration solutions). All calculated coefficients are displayed in this table, but only the most significant terms (p < 0.1) were retained in the final model formulation in order to increase the variability explained by the model and to remove extraneous terms. It should be remarked that it was not possible to produce any useful linear regression model for experimental set A. Upon data analysis, all of the linear models were inadequate for the data, yielding a negative coefficient of determination. Hence, these results are not presented. The source code is provided as Supplementary Methods 1 for reference.
Recovered nutrient solutions
The values for %P as P2O5 in all of the recovered nutrient solutions were very low, i.e., 0.02% in average for experimental set A and 0.01% in average for experimental sets B–D.
Discussion
Regarding resin characterization, the static beaker tests, yielding an adsorption capacity of 21.92 ± 4.13 mg PO4-P/g resin were determined to be valid after comparison with results generated by Nur et al.20 that claim an adsorption capacity of 19 mg PO4-P/g resin. In the operational continuous microcolumn setting, however, a much lower adsorption capacity, approximately 3 mg PO4-P/g resin, was calculated. This is likely due to the very high presence of sulfate (50 mg SO4/L vs. 2 mg PO4-P/L) in the synthetic wastewater sample. Indeed, as a major competitor for ion exchange sites, the sulfates likely bound to the ion exchange resin, thereby dampening the observed phosphorus adsorption capacity of the overall media24. The adsorption efficiency of the HIX-Nano resin reaches and remains around 100% up to 500 bed volumes of synthetic wastewater treated, after which breakthrough occurs, which is comparable to results obtained by Pan et al.24 and Nur et al.20.
In terms of phosphorus desorption, the regeneration chemistry used in experimental set A (KOH + H2SO4) was efficient in recovering phosphorus, as is evidenced by the high phosphorus concentrations and recovery efficiencies present in the alkaline rinse. The combined phosphorus concentration is higher in the samples that were regenerated using approximately 5 total bed volumes of regenerant solution [A1–A4] (average of 111.2 mg PO4-P/L), while the samples regenerated with 50 total bed volumes showed a weaker final concentration in the solution [A5–A8] (average of 27.8 mg PO4-P/L). This is expected, since a higher volume of regenerant solution used will dilute further the final solution. However, the experiments using a low volume experienced an average of only 46 ± 10% total phosphate recovery while the experiments using a high volume experienced an average of 99 ± 12%. This is likely related to the fact that there was simply not enough regenerant volume and hence OH− ions to adequately remove all of the phosphorus in solution out of the resin matrix in the low-volume method.
Further, for experimental set B (KOH + H2O), the wide range of values (concentrations and recovery efficiencies) observed was related to the effect of the number of bed volumes passed during desorption. In general, the higher bed volume trials showed a higher average P recovery efficiency from the column, but due to the larger volume, the concentration was lower. This is demonstrated, for example, by comparing the average P concentration of trials performed with 50 BV alkaline rinse (37.61 ± 14.12 mg PO4-P/L) [B5–B8] with that of trials performed with 5 BV alkaline rinse (183.44 ± 80.46 mg PO4-P/L) [B1–B4]. Similarly, in the samples with high bed volumes the phosphate was almost exclusively found in the alkaline rinse while in the samples with fewer bed volumes treated (5 BV), there was a significantly higher average concentration of phosphate found in both the alkaline (183.44 ± 80.46 mg PO4-P/L vs. 37.61 ± 14.12 mg PO4-P/L) and acidic rinse (40.53 ± 57.31 mg PO4-P/L vs. 1.42 ± 1.53 mg PO4-P/L) steps. This is an interesting finding, because according to the desires of the client for whom the system is to be installed, it may be pertinent to keep the regeneration phase effluents separate. However, using fewer bed volumes would likely require the combination of regenerant solutions of several alkaline rinsing steps in order to produce an adequate volume of fertilizer.
As opposed to experimental set A, the phosphorus recovery efficiency in the combined effluent is very low (24 ± 6.7% for the low-volume samples and 43 ± 13% for high-volume samples). This is most likely due to the fact that this particular regenerant solution does not target the ion exchange sites (no presence of competing sulfate ions), thus any phosphorus attached to the resin via ion exchange is not removed. This regenerant solution does not seem useful for industrial use.
Next, experimental C (KOH/K2SO4 + H2O) included the addition of potassium sulfate salt in order to desorb simultaneously the ion exchange sites and the adsorption sites on the resin. Similar to the experimental sets mentioned above, the volume of the regeneration solution was the most influencing factor. The average recovery efficiency (58.67 ± 22.78%) of this chemistry is lower than that of the control experiment, but higher than experimental set B. One would, however, expect a relatively high recovery with the intentional inclusion of the potassium salt for ion exchange site regeneration.
Figure 3a shows that the adsorption capacity dropped with each subsequent adsorption cycle. This is likely due to the inactivation of the ion exchange component of the resin by the free sulfate ions in solution. The FerrIX A33e ion exchange resin base is more selective to sulfates, particularly in high concentration, than phosphate. Thus, upon subsequent adsorption after a regeneration cycle, the phosphate ions may not be present in high enough concentration to dislodge the sulfate ions from the resin matrix, thereby significantly dampening the functionality of the nano-enhanced resin over time.
Finally, experimental set D showed removal and recovery comparable to the “control” experiments for the high bed volumes (75 ± 15 vs. 89 ± 13%, respectively). Moreover, the resin did not significantly lose efficiency across the five adsorption and desorption cycles conducted using this regeneration chemistry. This is likely related to the fact that the base used in this experimental set, i.e., 1% NH4OH, is less damaging for the resin than the potassium sulfate used in experimental set C and thus leads to less resin degradation and iron leaching. This is likely due to the lower base concentration, reducing the osmosis phenomenon explained earlier. Moreover, no free sulfate ions that can inhibit adsorption are present in experimental set D. This is promising for the implementation of this regeneration chemistry because it is the only one of the four that uses a low-cost by-product. As the alkaline effluent of an ammonia stripping–scrubbing process, this solution could both desorb phosphorus from the nano-enhanced adsorptive resin and provide ammonia nitrogen for fertilizer application downstream.
Regarding the linear regression models, no linear models could be developed for experimental set A. The available data did not provide sufficient information for a reliable model to be built. For all models shown in Table 4, the assumptions of normality were respected upon examination of the residual plots. Residual plots analysis did not show any particular trends and were distributed normally. Hence, the application of the models in industrial settings and for scale-up operations is pertinent to understand the effect of operational parameters on the phosphorus concentration in the recovered effluent solution.
Overall, the models showed a general trend of significance with respect to the bed volume factor, X1. Depending on the end user’s goals, the volume of the regenerant solution could be varied in order to reach the desired phosphorus concentration in the fertilizer effluent. However, when selecting the recovery efficiency as predictor variable for model development (results not reported), the most significant factor was again the bed volume, but the effect was reversed, i.e., higher bed volumes presented lower recovery efficiencies and vice versa. Hence, a compromise should be made between recovery efficiency and effluent P concentration when selecting the optimal number of regenerant bed volumes to be used.
The results obtained in this study do not suggest the necessity of a more concentrated regenerant solution nor a very high or very low rinse speed in the studied ranges. Hence, the cost of chemical products to be used for the regeneration can be minimized by minimizing the strength of the alkaline solution, while the pumping speed can be adjusted in order to obtain a certain residence time depending on the case.
The concentration of P in the recovered nutrient solutions is low due to the high volume of base used during regeneration as compared to the amount of P on the resin. Hence, the recovered solution is very diluted with an average N–P2O5–K2O composition of 0.67–0.015–0.4 (%w/w). With respect to some common fertilizers in the N–P2O5–K2O form for tomatoes (4–18–38), lettuce (8–15–36), and strawberries (8–12–32), the recovered nutrient solutions do not provide an adequate fertilizer composition for common crops25. Thus downstream concentration or processing, for example, through precipitation, is required in order to make these solutions feasible for fertilization processes. Combining these nutrient effluents with by-products from other nutrient recovery processes could improve product formulation, as will be explored by the authors in future research.
A wastewater treatment plant in Quebec is used as a case study to provide preliminary insight into the economic feasibility of HIX-Nano for phosphorus removal and recovery in small-scale wastewater treatment operations. The wastewater treatment plant under study currently has an effluent phosphorus discharge limit of 0.8 mg PO4-P/L, which will decrease to 0.3 mg PO4-P/L upon implementation of the new Pan-Canadian discharge regulations. Thus the application of HIX-Nano capable of removing phosphorus down to very low concentrations could present an interesting technological solution. The effluent flow from the wastewater treatment plant is 1633 m3/day and the phosphorus concentration averages 0.895 mg/L PO4-P. Assuming that the plant uses ammonia stripping and scrubbing to recover nitrogen, the plant would have a continuous supply of ammonium hydroxide to regenerate the column. Purolite Inc. claims a cost of approximately $17.66 CAD per liter of FerrIX A33e resin. Thus daily operation with daily regeneration of the column would cost $13.30 CAD per day. The amount of phosphorus recovered per day (1.46 kg P/day × 75% efficiency = 1.095 kg P/day) would yield $0.11 CAD per day, based off the most recent United States Geological Survey data (2019)26. The value of the recovered nitrogen, assuming that 50 bed volumes of 1% NH4OH are used to regenerate the column per day, would yield $89.48 CAD, given the current market price of $573 CAD/ton nitrogen27. This, not including the cost for labor, electricity, transportation, and downstream upgrading of the recovered nutrient solution, may yield a profit of $76.29 CAD per day. Given the fact that the wastewater treatment plant will be obliged to meet the new stringent discharge levels and currently pays high chemical costs (aluminum salts) to reduce the phosphorus concentrations down to 1 mg PO4-P/L only (!), these preliminary economic numbers provide indication that nano-enhanced adsorptive media may become of interest as phosphorus removal and eventually recovery technology to achieve stringent discharge levels at small wastewater treatment plants. However, a profound economic analysis based on pilot-scale results has to be assessed, which will be the aspect of further research by the authors.
Overall, future work should further investigate the use of low-cost regenerant solutions with special emphasis on by-products derived from nutrient recovery processes such as the weak NH4OH solution followed by a tap water rinse tested in this study. Following an initial screening as performed in this study, in-depth experimentation with the addition of central points into the experimental plan should be performed in order to allow for an analysis of variance. Attention should be paid to resin degradation and the concentration of the recovered nutrient solutions.
Resin improvement to further enhance the selectivity for phosphorus upon adsorption and desorption will also be aspect of further research by the authors. For example, Ferrix A33e includes a strong base anion exchange resin. However, if the nanoparticles would be integrated in a weak base anion exchange resin, no base would be required to regenerate the resin, only NaCl or K2SO4. Hence, the recovered nutrient solution would be less diluted.
Finally, future research will look into the upstream concentration or processing of the recovered nutrient solutions in order to valorize these streams as a fertilizer or a precursor for other phosphorus products. Pilot-scale experiments will also be performed for model validation and to perform a more profound economic assessment.
Methods
Experimental planning and selection of regeneration chemistries
Four sets of regenerant solutions (experimental sets A–D) composed of both an alkaline rinse and an acid rinse were depicted as shown in Table 1.
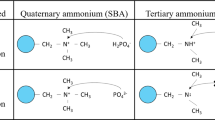
The first set of experiments (A) completed was used as the “control” set of experiments, using both a standard synthetic strong base and acid, i.e., KOH and H2SO4. KOH was used instead of the conventional NaOH because it contains the valuable macronutrient K, whereas H2SO4 was chosen because of its common use in industry and the presence of the valuable macronutrient S.
Because the PZC of the iron oxide is around 7.9, tap water (pH 7.3) is both low cost and acidic enough to change the surface chemistry of the iron oxide particles. Hence, the second experimental set (B) was conducted according to the same experimental plan as the control experiments using KOH as the alkaline rinse, but with tap water as the acid rinse.
For the third experimental set (C), a 50%/50% by weight combination of potassium hydroxide and potassium sulfate was used as alkaline rinse; the 1% KOH/K2SO4 solution had a pH of 12.7, and the 6% solution had a pH of 13.1. This ratio was used because it was previously used in the literature and was proven effective for conventional regeneration using NaOH and NaCl21. The reason for using the salt K2SO4 is related to the strong affinity of sulfates to the resin ion exchange sites, which can hence maximize the recovery of phosphorus bound to these sites23. Moreover, when using high amounts of KOH, the resin may release OH− ions during the next adsorption step, thereby potentially inhibiting adsorption since the pH could rise above the PZC18. Tap water was again used for the acid rinse.
Finally, the fourth experimental set (D) aimed at using a by-product from an established resource recovery process in wastewater treatment plants as the alkaline rinse. As such, the alkaline rinse was a 1% w/w% NH4OH solution (pH 11.3) as may be recovered through ammonia stripping and scrubbing with water28,29. Similar to the previous experimental sets, the acid rinse used in this experimental set was tap water.
Table 2 shows the 23 full-factorial experimental plan that was carried out for experimental sets A–C. The factors under study are the number of bed volumes passed (5 or 50 for both the alkaline and acid rinse), the rinse speed (2 or 20 mL/min), and the base strength (1 or 6 wt%), which are three major operational parameters influencing the operational costs of an adsorption–desorption system. A center point was not taken due to experimental restrictions. The factor ranges were depicted based on previous operational experience obtained within the team and guidelines of the resin provider. The operational parameters chosen are those generally studied in other adsorption research. For experimental set D, the base strength was removed as a factor since the alkaline rinse concerns a diluted by-product from ammonia stripping and scrubbing at fixed concentration as described above. Hence, the experiment constituted a 22 full-factorial experimental design, as shown in Table 3.
Resin characterization
Before starting the experiments, the resin was characterized to provide a baseline understanding of its removal potential. The first step was a static beaker test. The goal of this test was to determine the maximum adsorption capacity of the resin in milligrams of phosphorus removed per wet gram of resin at complete resin saturation. Samples were also taken at time intervals in order to register the relationship between time and adsorption. For these tests, 1 g of the FerrIX A33e hybridized ion exchange resin was added to each of three 250 mL Erlenmeyer flasks. A solution containing 1000 mg P/L was prepared using phosphoric acid and 100 mL of this solution was added to each flask. Thus each flask contained approximately 100 mg P. The solution was neutralized with sodium hydroxide until it reached pH 7. The three flasks were then shaken on an agitation table at 200 RPM, and samples were taken at hours 1, 3, 24, 48, 72, and 122 throughout the span of 1 week. The samples were analyzed using HACH Total Phosphorus Testing Kits (TNT 845—molybdovanadate method) and spectrophotometry using the HACH DR 5000 spectrophotometer.
Following the static beaker tests, a continuous adsorption test was performed in a microcolumn containing 1.1 g of wet resin (Fig. 4) to determine the operating adsorption capacity of the resin. Microcolumn experiments were made because adsorption is known as a scalable process and microcolumns are widely used in adsorption research17,23. In addition, this technique is recommended for pH-sensitive processes, including adsorption23. For this test, synthetic wastewater with a composition similar to the concentration of phosphorus and sulfate in tertiary treatment effluent at a full-scale wastewater treatment plant in Quebec, i.e., 2.05 mg PO4-P/L and 50 mg SO4−2/L, was pumped over the column at a speed of 1 mL/min. This test was designed to determine the expected adsorption capacity during continuous adsorption cycles, thereby also considering the inhibitory effect of sulfate on the resin performance.
Desorption and regeneration experiments
The main experiments studied the effect of different experimental conditions, or factors, on the phosphate desorption and regeneration of the FerrIX A33e ion exchange resin. Microcolumns of approximately 2 mL volume were loaded with the FerrIX A33e resin and saturated with phosphorus using a synthetic solution of approximately 50 mg PO4-P/L. The synthetic wastewater samples were passed through the resin bed at a speed of 2 mL/min providing an EBCT of 1 min. The volume and initial and final phosphorus concentrations of the solution before and after treatment were recorded, and the amount of phosphorus adsorbed on the resin in each column in milligrams was calculated prior to regeneration. The columns were then regenerated according to the experimental plan described in Tables 2 and 3. The alkaline and acid rinses were collected in a separate beaker and measured for phosphorus concentration. The concentrations of N and K were calculated using mass balances at the end of each regeneration cycle, considering that these elements do not adsorb on the resin. From these measurements, an N–P–K fertilizer composition was calculated.
For all phosphorus measurements, the HACH DR5000 spectrophotometer was used for colorimetric detection in combination with the HACH TNT-843 (molybdate + ascorbic acid) reactive phosphorus low-range testing kits and the TNT-845 (molybdovanadate) reactive phosphorus high-range testing kits. The effluent solutions were analyzed after each washing step (alkaline and acid) and then the results were combined (alkaline + acid) to provide a picture of the final solution should the alkaline rinse and acid rinse step effluents be collected in the same receptacle.
For experimental sets C and D, the adsorption–regeneration cycle was repeated five times using the same resin in order to study the functionality of the resin over five cycles.
Model development
Regression models were developed and analyzed to test their significance and the goodness of fit for each of the experimental phases (alkaline rinse, acid rinse, combined rinse). The models were generated using multiple linear regression considering the main factors (bed volumes, flow rate, base strength) and their interaction terms (Table 4) and response variable ([P]).
Significance of each factor and interaction term was determined using the R software, and the final model was generated by retaining the significant factors and removing those that were insignificant. The goodness of fit was determined using analysis of the p value of the F-statistic of the model at the 95% confidence level provided by R’s linear regression software package. The models’ validity was assessed using residual analysis and the adjusted coefficient of determination, represented by the adjusted R2 value. Randomized experimental order assured that the data was independent and assumed to be linear and the homoscedasticity (variance of residuals) and normality of residuals were checked visually using residual plots and normality plots were generated for each model using the R software.
Valorization potential of recovered nutrient solutions
In order to evaluate the potential of each recovered nutrient solution as a fertilizer, the concentrations of P, N, and K were converted to a standard form (%N–%P2O5–%K2O) (%w/w) as reported in an N–P–K fertilizer formula. Each of the regeneration effluent solutions was characterized in phosphorus concentration using spectrophotometry as described above, and the potassium and nitrogen concentrations were determined according to mass balances.
Data availability
All treated data that support the findings of this study are included in the present article. Raw datasets can be obtained from the corresponding author upon request.
Code availability
The codes that support the findings of this study are included as Supplementary Information in the present article.
Change history
17 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41545-021-00104-7
References
Wong, P. Y., Cheng, K. Y., Kaksonen, A. H., Sutton, D. C. & Ginige, M. P. A novel post denitrification configuration for phosphorus recovery using polyphosphate accumulating organisms. Water Res. 47, 6488–6495 (2013).
Carmichael, W. The toxins of Cyanobacteria. Sci. Am. 270, 78–86 (1994).
Sutton, M. A. et al. Our Nutrient World: The Challenge to Produce More Food and Energy with Less Pollution (Centre for Ecology & Hydrology on behalf of the Global Partnership on Nutrient Management and the International Nitrogen Initiative, Edinburgh, 2013).
Khan, F. A. & Ansari, A. A. Eutrophication: an ecological vision. Botanical Rev. 71, 449–482 (2005).
Environmental Protection Agency. Clean Water Act, CWA 303(d), 33 U.S.C § 1251(a) (Environmental Protection Agency, 1994).
South Florida Water Management District. State of Florida Industrial Wastewater Facility Permit, FL0778451 (South Florida Water Management District, 2017).
Canadian Water Network. Canada’s Challenges & Opportunities to Address Contaminants in Wastewater (Canadian Water Network, 2018).
Tchobanoglous, G. Wastewater Engineering: Treatment and Reuse 4th edn (McGraw-Hill, Boston, 2001).
Vaneeckhaute, C., Belia, E., Meers, E., Tack, F. M. G. & Vanrolleghem, P. A. Nutrient recovery from digested waste: towards a generic roadmap for setting up an optimal treatment train. Waste Manag. 78, 385–392 (2018).
International Institute for Sustainable Development. Nutrient Recovery and Reuse in Canada (International Institute for Sustainable Development, 2018).
Kabbe, C. Nutrient Recovery 2.0 (WaterWorld, 2016).
Martin, B. D., Parsons, S. A. & Jefferson, B. Removal and recovery of phosphate from municipal wastewaters using a polymeric anion exchanger bound with hydrated ferric oxide nanoparticles. Water Sci. Technol. 60, 2637–2645 (2009).
Hallas, J., Mackowiak, C., Wilkie, A. & Harris, W. Struvite phosphorus recovery from aerobically digested municipal wastewater. Sustainability 11, 376 (2019).
Bolton, L., Joseph, S. D. & Greenway, M. Phosphorus adsorption onto an enriched biochar substrate in constructed wetlands treating wastewater. Ecol. Eng. 1, 100005 (2019).
Huggins, T., Haeger, A., Biffinger, J. & Ren, Z. Granular biochar compared with activated carbon for wastewater treatment and resource recovery. Water Res. 94, 225–232 (2016).
Kocatürk‐Schumacher, N. P., Bruun, S., Zwart, K. & Jensen, L. S. Nutrient recovery from the liquid fraction of digestate by clinoptilolite. CLEAN Soil Air Water 45, 150–153 (2017).
Liberti, L., Petruzzelli, D. & De Florio, L. REM NUT ion exchange plus struvite precipitation process. Environ. Technol. 22, 1313–1324 (2001).
Padungthon, S. Development of Hybrid Polymeric/inorganic Ion Exchanger: Preparation, Characterization, and Environmental Applications. PhD thesis, Lehigh Univ. (2013).
Blaney, L., Cinar, S. & Sengupta, A. Hybrid anion exchanger for trace phosphate removal from water and wastewater. Water Res. 41, 1603–1613 (2007).
Nur, T. et al. Phosphate removal from water using an iron oxide impregnated strong base anion exchange resin. J. Ind. Eng. Chem. 20, 1301–1307 (2014).
Sposito, G. On points of zero charge. Environ. Sci. Technol. 32, 2815–2819 (1998).
Kosmulski, M. PH-dependent surface charging and points of zero charge. IV. Update and new approach. J. Colloid Interface Sci. 337, 439–448 (2009).
SenGupta, A. K. Ion exchange in Environmental Processes: Fundamentals, Applications and Sustainable Technology (Wiley, 2017).
Pan, B. et al. Development of polymer-based nanosized hydrated ferric oxides (HFOs) for enhanced phosphate removal from waste effluents. Water Res. 43, 4421–4429 (2009).
Chem-Gro. Chem-Gro fertilizer compositions. Hydro-gardens. https://hydro-gardens.com/product-category/fertilizers/chemgro/ (2019).
United States Geological Survey. Mineral industry surveys: marketable phosphate rock. https://www.usgs.gov/centers/nmic/phosphate-rock-statistics-and-information (2019).
Knorr, B. Fertilizer outlook - ammonia holds line in bearish fertilizer market. https://beef2live.com/story-fertilizer-outlook-ammonia-holds-line-bearish-fertilizer-market-22-196414 (2019).
Clark, C. Ammonia Removal Utilizing an Ammonia Scrubber (Gulf Coast Environmental Systems, 2017).
Jamaludin, Z., Rollings-Scattergood, S., Lutes, K. & Vaneeckhaute, C. Evaluation of sustainable scrubbing agents for ammonia recovery from anaerobic digestate. Bioresour. Technol. 270, 596–602 (2018).
Acknowledgements
Funding for this project was provided in part by the Fulbright Canada Commission through an MSc scholarship awarded to the first author, as well as granting from the Natural Science and Engineering Research Council of Canada through the award of an NSERC Discovery Grant (RGPIN-2017-04838) to C.V. C.V. holds the Canada Research Chair in Resource Recovery and Bioproducts Engineering.
Author information
Authors and Affiliations
Contributions
C.V. and M.O. conceptualized and planned the experiments. M.O. and D.-A.D. did the experimental work. M.O. performed data analysis. C.V., M.O., and D.-A.D. interpreted the data. M.O. drafted the initial version of the manuscript. C.V. and D.-A.D. substantially revised the manuscript. All authors contributed to and approved the final version of the manuscript. All authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work were appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ownby, M., Desrosiers, DA. & Vaneeckhaute, C. Phosphorus removal and recovery from wastewater via hybrid ion exchange nanotechnology: a study on sustainable regeneration chemistries. npj Clean Water 4, 6 (2021). https://doi.org/10.1038/s41545-020-00097-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41545-020-00097-9
This article is cited by
-
Combined advanced oxidation dye-wastewater treatment plant: design and development with data-driven predictive performance modeling
npj Clean Water (2024)
-
A comprehensive review on zinc(II) sequestration from wastewater using various natural/modified low-cost agro-waste sorbents
Biomass Conversion and Biorefinery (2023)