Abstract
The interplay between schizophrenia liability and environmental influences has been considered to be responsible for the development of schizophrenia. Recent neuroimaging studies have linked aberrant functional connectivity (FC) between the default-mode network (DMN) and the frontoparietal network (FPN) in the resting-state to the underlying neural mechanism of schizophrenia. By using schizotypy as the proxy for genetic-based liability to schizophrenia and methylation of brain-derived neurotrophic factor (BDNF) to represent environmental exposure, this study investigated the impact of the interaction between vulnerability and the environment on the neurobiological substrates of schizophrenia. Participants in this study included 101 healthy adults (HC) and 46 individuals with ultra-high risk for psychosis (UHR). All participants were tested at resting-state by functional magnetic resonance imaging, and group-independent component analysis was used to identify the DMN and the FPN. The Perceptual Aberration Scale (PAS) was used to evaluate the schizotypy level. The methylation status of BDNF was measured by pyrosequencing. For moderation analysis, the final sample consisted of 83 HC and 32 UHR individuals. UHR individuals showed reduced DMN-FPN network FC compared to healthy controls. PAS scores significantly moderated the relationship between the percentage of BDNF methylation and DMN-FPN network FC. The strength of the positive relationship between BDNF methylation and the network FC was reduced when the schizotypy level increased. These findings support the moderating role of schizotypy on the neurobiological mechanism of schizophrenia in conjunction with epigenetic changes.
Similar content being viewed by others
Introduction
Schizophrenia is a devastating mental illness that manifests early in life. The development trajectory of schizophrenia is heterogeneous but typically consists of a premorbid period with nonspecific developmental or behavioral deviances1, a prodromal phase with a subclinical presentation of illness from first noticeable symptoms to prominent psychotic symptoms2,3, and an active phase with the onset of a fully psychotic episode. Although the exact neuronal mechanisms of schizophrenia remain unknown, neuroimaging has found extensive altered functional connectivity (FC) patterns in resting-state brain networks4. Among disconnections across brain networks, reduced connectivity between the default-mode network [DMN; involved in internally oriented cognition and the self-related thought process5] and the frontoparietal network [FPN; involved in external task performance and goal-directed regulation6] has been consistently reported in individuals with schizophrenia [for meta-analysis, see Dong et al.7]. Furthermore, this aberrant coupling of DMN-FPN has also been demonstrated in individuals at ultra-high risk (UHR) for psychosis8,9 who are in the ‘putative’ prodromal phase of schizophrenia. Therefore, the abnormal coordination between DMN and FPN, which reflects dysregulation between a self-focused mode and stimulus-dependent attention10, may drive the path to schizophrenia, which is characterized by confusion between internal thoughts and external reality.
Schizophrenia liability and schizophrenia-like traits exist as part of continuum in the general population11,12. Schizotypy, a set of enduring traits that reflect the subclinical symptoms and signs of schizophrenia, has been proposed as a proxy for genetic-based liability to schizophrenia13,14,15 and is conjectured to be found in 10% of the general population14. Empirical work supports the commonality of schizotypy with schizophrenia in terms of shared genetics12 and abnormalities in cognition, including perceptual experiences across all sensory modalities16,17. Although the reports were highly heterogenous, resting-state functional neuroimaging studies showed comparable altered networks in individuals with high levels of schizotypy and schizophrenia18,19,20. Schizotypal symptoms have also been associated with FC in healthy adults20, individuals with schizotypal personality disorder21, and schizophrenic individuals22. Thereby, it is empirically supported that schizotypy may be a promising endophenotype for schizophrenia that, with the appropriate technology, is detectable along the pathway to disease.
Environmental influences in conjunction with a predisposing vulnerability is contemporary psychiatry’s common explanation for the development and evolvement of a psychiatric disorder23. Epigenetic changes, such as DNA methylation, provide a mechanism for environmental influences to alter disease risk24. Aberrant levels of brain-derived neurotrophic factor (BDNF), a key protein that regulates neuronal development25 and neuroplasticity26, were shown to be involved in the pathophysiology of stress-related psychiatric outcomes27. Varying effects of different stressors on the regulation of BDNF transcripts, including both upregulation and downregulation, have been found in animal models of stress [for review article, see ref. 28], and altered BDNF methylation was found in blood samples of stressed humans29. Epigenetic modification of BDNF was found in postmortem brains of schizophrenic individuals and in brains of mice with schizophrenia-like behavioral abnormalities born from prenatal stress mice30. Several studies have shown alterations in BDNF methylation in blood samples of schizophrenic individuals31,32, although the results have been mixed33,34. Given the role of BDNF in neuroplasticity26, changes in BDNF methylation have been suggested as an important mediator of environmental stress on psychopathology. Therefore, the association between BDNF methylation and suspected neural substrates in psychopathology have been studied. For example, higher BDNF methylation was recently reported to be associated with higher amygdala reactivity to negatively-valanced emotional face processing in healthy individuals35. Increased BDNF methylation was also associated with medial prefrontal cortex activity when watching stressful stimuli in violence-related samples36. However, there has been limited attention to the role of epigenetic reprogramming of the BDNF gene in relation to the resting-state brain functional network in individuals with schizophrenia.
To determine how schizophrenia emerges, it is crucial to investigate interactions between vulnerability and environmental influences on known altered brain networks related to schizophrenia. Because vulnerability exists as a continuum in the general population11, it is important for its analysis to encompass general and clinical individuals and to use psychometrically defined schizotypy as a proxy measure. Because UHR individuals, who are at ~30% risk for overt psychotic disorder within 2–3 years of follow-up37, are relatively free from the confounding effects of the chronicity of illness in schizophrenia38 and prolonged exposure to antipsychotic medications39, they were chosen for this study. Investigating schizotypy in UHR individuals could also provide evidence of schizotypy as an additional strategy to identify “true positive” individuals in the pre-psychotic phase. Thus, the objectives of this study were (1) to investigate changes in resting-state brain network FC in UHR individuals compared with healthy control (HC) individuals and (2) to evaluate the moderating effect of schizotypy on the relationship between BDNF methylation on network FC. Based on previous studies, we hypothesized that changes in network FC in UHR individuals would occur between the DMN and the FPN and that the level of schizotypy would influence the relationship between BDNF methylation and the aberrant network FC across HC and UHR individuals.
Results
Demographic and clinical characteristics
Participant characteristics are provided in Table 1. Age did not differ significantly between the HC and UHR groups. Compared to the HC group, the UHR group had a higher percentage of male participants (χ2 = 5.3, P = 0.022) and a lower level of education (χ2 = 11.9, P = 0.018). SIPS diagnoses of UHR individuals were as follows: Only APS (n = 31), only BIPS (n = 1), APS and GRDS (n = 10), APS and BIPS (n = 3), and APS, BIPS, and GRDS combined (n = 1). Fifteen UHR individuals were medicated with antipsychotics [chlorpromazine equivalent dose40, mean (SD) = 176.1 (115.4)].
PAS and BDNF DNA methylation
The UHR group had higher PAS scores than the HC group after controlling for age, sex, and level of education [n: HC = 93, UHR = 41; mean (SD): HC = 3.4 (3.7), UHR = 9.3 (7.5); F(1,132) = 26.2, P < 0.001; Fig. 1A]. When comparing HCs, UHR individuals not diagnosed with genetic risk and deterioration syndrome (n = 31), and UHR individuals diagnosed with genetic risk and deterioration syndrome (n = 10), UHR individuals with genetic risk showed significantly higher PAS scores (P < 0.001; Supplementary Fig. 3). Regarding BDNFm, UHR individuals showed lower percentages of BDNF methylation than HCs after controlling for age, sex, and level of education [n: HC = 83, UHR = 32; mean (SD): HC = 3.9 (0.7), UHR = 3.6 (0.7); F(1,113) = 4.6, P = 0.033; Fig. 1B].
DMN-FPN network FC
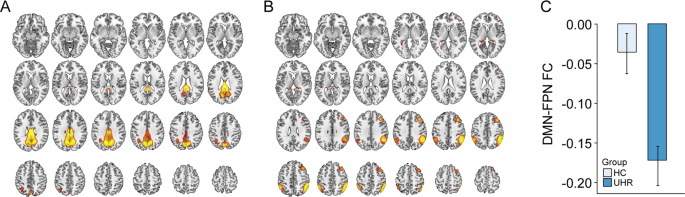
The DMN-FPN network FC (Fig. 2A, B) differed between the HC and UHR groups after controlling for age, sex, and level of education. Compared to HCs, UHR individuals showed reduced DMN-FPN network FC [F(1,145) = 9.4, P = 0.003; Fig. 2C]. After controlling for age, sex, educational level, and dose of medication in UHR individuals, the DMN-FPN network FC remained reduced in UHR individuals compared to HCs [F(1,145) = 5.2, P = 0.024].
Interaction between PAS and DNA Methylation on the DMN-FPN network FC across HC and UHR individuals
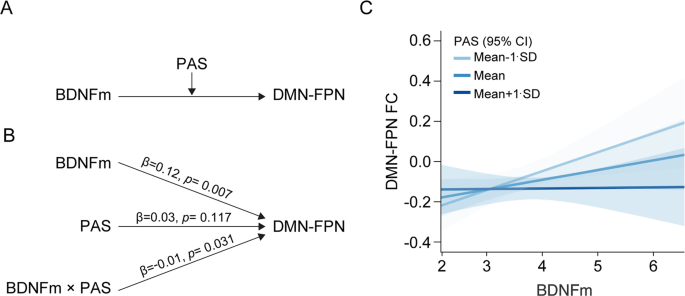
PAS scores moderated the relationship between BDNFm and the DMN-FPN network FC after controlling for age, sex, and level of education (P = 0.031, f2 = 0.047, LLCI/ULCI = −0.0193/−0.0009, Fig. 3). When the additional analysis was conducted, including the group as an independent variable in the model, the moderating effect of PAS scores remained (P = 0.046). BDNFm had an effect on the DMN-FPN FC when PAS scores were one SD below the mean (β = 0.11, P = 0.008) and at the mean (β = 0.09, P = 0.017), but not at one SD above the mean (β = 0.03, P = 0.319). Therefore, as the level of schizotypy (PAS score) increased, the strength of the positive relationship between BDNFm and the network FC decreased. When the analysis was repeated controlling for age, sex, level of education, and medication dosage, the effect of PAS on the relationship between BDNFm and the DMN-FPN network FC remained (P = 0.045).
A Conceptual and B statistical models and C the plot showing the simple effects with standard errors of the estimates to visualize the association between BDNF methylation and the DMN-FPN network FC moderated by PAS. Regression coefficients were calculated in a moderation analysis model including age, sex, and years of education as covariates. Abbreviations: BDNFm the percentage of BDNF methylation, DMN default-mode network, FC functional connectivity, FPN frontoparietal network, HC healthy control, PAS perceptual aberration scale, UHR ultra-high risk for psychosis.
Discussion
UHR individuals showed reduced DMN-FPN network FC compared to HCs. Moreover, the DMN-FPN network FC was affected by the interaction between the level of schizotypy and the percentage of BDNF methylation across HC and UHR individuals. To the best of our knowledge, this is the first study to investigate the effect of schizotypy, a proxy of schizophrenia liability, in conjunction with BDNF methylation, which represents environmental exposure, on vantage points of the stress-vulnerability model. Because the schizotypy-BDNF methylation interaction was associated with altered network FC, which was significantly different between HC and UHR individuals, our findings support the widely accepted explanation of the development of schizophrenia by environmental influences in conjunction with predisposing vulnerability.
Consistent with previous studies on schizophrenia [for meta-analysis, see7] and UHR individuals8,9, the current study demonstrated aberrant coordination between the DMN and the FPN in UHR individuals. The disconnection model with regards to the onset of schizophrenia has been previously reviewed7,41. According to this model, there is a disturbance in the maintenance of the integrated self, which may be a core phenomenon of schizophrenia42 and may be associated with further enriched risk of overt psychosis conversion in UHR individuals43, that is tightly linked to dysconnectivity between the large-scale brain systems responsible for internal thought (DMN), goal-directed regulation (FPN), salience processing (ventral attention network), emotion processing (affective network), and gating information (thalamus network)7. The reduced network FC finding in our UHR group supports the disconnection model and confirms the hypo-connectivity between the DMN and the FPN, which reflects an imbalanced coordination between self-related activity and externally focused cognitive activity. Although all the brain networks listed above were not analyzed in this study, the significant difference that we found in the DMN-FPN network FC between the UHR and HC groups supports the rationale to measure the DMN-FPN network FC when evaluating the neurobiological substrates of schizophrenia.
In regards to the DMN-FPN network FC, the novel challenge of this study was to examine the statistical interaction between the level of schizotypy and BDNF methylation to determine whether vulnerability and environmental influence have a joint impact on the underlying neurobiological mechanism of schizophrenia. The vast majority of studies on the interplay between genetic risk and environmental exposure driving schizophrenia have used indirect measures44. Likewise, this study used the level of schizotypy as a proxy for the genetic-based liability to schizophrenia and the percentage of methylation of the BDNF promoter to represent environmental influence. As revealed by examining the moderating effect of schizotypy on the association between BDNF methylation and the DMN-FPN network FC in this study, the positive relationship between BDNF methylation and the network FC decreased when the level of schizotypy was high. The additional analysis, including the group as an independent variable in the model, showed that the association was not driven by group differences. The remaining moderating effect of schizotypy after additionally controlling for chlorpromazine equivalent dose eliminated the confounding effects of antipsychotic medications. A remaining question is how epigenetically inhibited BDNF transcription could enhance network FC. One possible explanation is that downregulated BDNF may directly modulate synaptic strength to alter environmental sensitivity45. Another possibility is the effect of BDNF on other neurotransmitter systems46,47, which in turn modifies brain reactivity. Previous studies on the effect of peripheral DNA methylation on FC changes in schizophrenia have reported inconclusive results across heterogenous target genes [for review article, see ref. 48]. In the case of BDNF methylation on brain activity related to schizophrenia, one study49 found that BDNF methylation in healthy individuals was associated with prefrontal cortex functional activity and working memory accuracy, which was used as an index of phenotypes relevant to schizophrenia. Our findings and previous research49 suggest that BDNF methylation can bridge the environmental exposures to the endophenotypes of schizophrenia; however, caution is required when interpreting hypothetical linkages between BDNF methylation and brain network changes in schizophrenia due to the scarcity research. With this precaution, the moderation effect of schizotypy on the positive association between BDNF methylation and network FC indicates that dampening of this positive association in individuals with high levels of schizophrenia liability may result in reduced DMN-FPN network FC in individuals at high risk for schizophrenia who show elevated levels of schizotypy.
At present, despite extensive studies on BDNF methylation and the development of various psychiatric disorders, changes in methylation have not been used as a specific biomarker for certain psychiatric phenotypes. In addition, most of the changes in BDNF methylation have been very small between clinical samples and healthy controls, a 1.3% difference in individuals with schizophrenia as an example50. However, a 9–15% change in BDNF methylation has been suggested as necessary to induce alterations in BDNF transcripts50. A recent study also reported no significant difference in mean methylation of BDNF among first-episode psychosis individuals, their unaffected siblings, and healthy controls; however, the study found higher levels of methylation in individuals with childhood trauma independent of diagnosis34. The nonspecific association of BDNF methylation with psychiatric disorders and the discordance between the level of BDNF methylation and its protein may be due to other regulators of expression. BDNF methylation itself may reflect regulation of genetic expression instead of direct influence51, and stressors may interrupt this gene expression balance28. Recent studies on the epigenetic inheritance of DNA methylation52,53 also have suggested a novel underlying mechanism of epigenetic regulation. Therefore, a comprehensive analysis of encompassing factors that impact BDNF expression is needed to determine environmental influence both on the level of BDNF methylation and its expression.
The UHR construct has enabled researchers to identify individuals at high risk for the transition to schizophrenia. Because of “false positive” cases in UHR individuals, researchers have tried to use additional strategies to identify “true positive” individuals in the pre-psychotic phase. It remains unclear how schizotypy is positioned in the high-risk research paradigm. However, previous research on schizotypy in the general population54,55, genetically high risk individuals56,57,58, and clinically high risk individuals59,60,61 has suggested that schizotypy associates with risk of development of psychosis [for a review, see ref. 62]. Our results provide evidence for schizotypy as a proxy for genetic-based vulnerability to schizophrenia, by showing the differences of PAS scores among HC, UHR individuals without genetic risks, and UHR individuals with genetic risks. Thus, this study suggests that measuring schizotypy could identify individuals with ‘true’ susceptibility for aberrant network FC when combined with epigenetic changes.
This study has several limitations. First, the cross-sectional data limits understanding of any causal relationships. The medium sample size of UHR individuals did not allow for generalization of our findings, such as the range of percentages of methylation of the BDNF promoter. It was also restricted to analyses of the relationships between the variables and psychotic transition. Therefore, further prospective studies with larger numbers of UHR individuals, who are thoroughly followed-up to observe clinical outcomes and assessed by using interviewer-rated anomalous self-experience measures, such as the Examination of Anomalous Self-Experience63, are needed in the near future. BDNF methylation was measured to reflect BDNF gene regulation or epigenetic mechanisms as directly as possible. However, given the discordance between the level of BDNF methylation and its protein51, analyzing the mRNA or protein levels of BDNF could provide detailed information about BDNF gene expression and its relationship with FC in further studies. Lastly, some UHR participants were taking antipsychotic medications. Although the use of covariates in the analysis cannot completely rule out potential confounding effects, this study yielded identical results when the analyses were repeated controlling for dosage of antipsychotic medications in addition to age, sex, and level of education. Thus, the effect of antipsychotic medications is unlikely to change the overall findings in this study.
In conclusion, the main findings of this study support the hypothesis that the impact of schizotypy on the candidate network FC, the putative underlying neural substrate of schizophrenia, is due to interactions with epigenetic changes that reflect environmental influence. By demonstrating the moderating role of schizotypy on the association between BDNF methylation and changes in the DMN-FPN network FC, this study provides pivotal neurobiological data substantiating the stress-vulnerability model of developing schizophrenia.
Methods
Participants
Participants initially included 101 healthy young adults and 46 UHR individuals. Participants were excluded from some analyses if they had missing or incomplete data. Healthy individuals were recruited via online advertisements, and UHR individuals were enrolled from a specialized UHR clinic as part of the Green Program for Recognition and Prevention of Early Psychosis (GRAPE) project at Severance Hospital in Seoul, Republic of Korea. All participants were screened for psychiatric illnesses using the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders, Fourth edition64,65, and individuals with any current or past history of major psychiatric disorders were excluded from the HC group. UHR participants were diagnosed using the Structured Interview for Prodromal Syndromes [SIPS; ref. 66]; therefore, each UHR participant met one or more of the three diagnoses: attenuated positive symptom syndrome (APS), brief intermittent psychotic symptom syndrome (BIPS), or genetic risk and deterioration syndrome (GRDS). Further details on the GRAPE project have been described previously67,68. This study was reviewed and approved by the Institutional Review Board at Severance Hospital, and the study was performed in accordance with the Declaration of Helsinki. All of the participants and the guardians of the participants (if younger than 18 years old) provided written informed consent. A study flow chart summarizing the number of individuals in the analyses is presented in Supplementary Fig. 1.
Psychometric measure of schizotypy
Because previous researchers (Rado, 1960; Meehl, 1964) described a key component of schizotypic phenotypes as an aberrant awareness of one’s body, a well-known psychometric tool, the Perceptual Aberration Scale [PAS; ref. 69] has been used to index schizophrenia liability. The PAS contains 35 items to assess psychotic-like perceptual experiences, which includes bodily discontinuities and unusual scenery experiences (e.g., “I have felt that something outside my body was a part of my body”). PAS-identified persons with high levels of schizotypy were found to be similar to schizophrenics with regards to various psychological, cognitive, and physiological factors15. Moreover, genomic association of the PAS score with a single nucleotide polymorphism of interest to schizophrenia research70 provided genetic evidence that the etiology of schizophrenia involves perceptual aberrations. Perceptual aberrations also associated with simpler psychological processes, such as exteroceptive and proprioceptive tasks71, rather than complex constructs; therefore, reducing heterogeneity by focusing on PAS could reveal underlying biological states15. Thus, to evaluate their levels of schizotypy, all participants were asked to complete the PAS; however, PAS data for 8 HC and 5 UHR individuals were missing.
Epigenotyping procedures
Genomic DNA was extracted from 83 HC and 32 UHR individuals [peripheral whole blood (n; HC = 76, UHR = 25); saliva (n; HC = 7, UHR = 7)]. A CpG-rich region of the BDNF promoter, including seven CpG sites between −694 and −577 relative to the transcriptional start site, was analyzed [GenBank accession number: JX84862072; Supplementary Fig. 1]. This region was studied in Korean individuals72,73 and differential methylation of the analogous region in rat BDNF was associated with BDNF mRNA expression74. DNA methylation status was determined by Macrogen, Inc. (Seoul, Republic of Korea) using standard procedures (for the detailed protocol, see Supplementary Methods). Average values of the percent methylation at the seven CpG sites of the BDNF promoter (BDNFm) were used in the analyses.
Magnetic resonance imaging (MRI) data acquisition
MRI data were acquired using a 3T scanner (Intera Achieva, Philips Medical Systems, Best, The Netherlands). Participants were instructed to rest in the scanner with their eyes closed and to stay still and quiet without sleeping or focusing on any specific thought. T2*-weighted gradient echo-planar imaging was used: repetition time (TR) = 2000 ms, echo time (TE) = 30 ms, flip angle = 90°, 31 interleaved slices, matrix size = 80 × 80, voxel size = 2.75 × 2.75 × 4 mm3, field-of-view (FOV) = 220 mm. High-resolution structural T1-weighted images were acquired using the turbo field echo sequence with the following settings: TR = 9.7 ms, TE = 4.6 ms, flip angle = 8°, 180 slices, matrix = 256 × 256, voxel size = 0.859 × 0.859 × 1.2 mm3, FOV = 220 mm.
Image preprocessing
Functional MRI (fMRI) data were analyzed using Statistical Parametric Mapping 12 software (Wellcome Trust Centre for Neuroimaging, London, UK) and Functional Connectivity toolbox version 19b (CONN; McGovern Institute for Brain Research, Massachusetts Institute of Technology, Cambridge, MA, USA)75 in MATLAB (Mathworks Inc., Natick, MA, USA). The first five volumes of the functional datasets from each participant were discarded to ensure signal stabilization. Using CONN’s default preprocessing pipeline, the remaining images underwent standard preprocessing steps: functional realignment and unwarp, functional and structural center to (0, 0, 0) coordinates, slice-timing correction, identification of outliers (global mean z-threshold < 3, motion threshold < 1 mm) with the Artifact Detection Tool, segmentation, normalization to the standard Montreal Neurological Institute space, and functional smoothing (spatial convolution with a 6 mm full-width Gaussian kernel at half-maximum). The images were resampled to 2 × 2 × 2 mm3 voxels. After preprocessing, fMRI data were denoised using CONN’s default denoising pipeline: linear regression of potential confounding effects (motion-related artifacts, noise components from white matter and cerebrospinal areas, scrubbing), temporal band-pass filtering (0.008–0.09 Hz), and linear detrending.
Group-independent component analysis
Group-independent component analysis [ICA; ref. 76] was performed to identify the intrinsic functional organization of the brain as follows: group-level dimensionality reduction using principal component analysis, estimation of spatially-independent components (ICs) using the FastICA algorithm, and back-reconstruction of individual-level ICs using dual regression. The current study constrained the number of ICs to 30, which is suitable for a low-dimensional ICA decomposition77. Group-averaged ICs were inspected by two experienced investigators (SKA and HYP) to identify networks involving the DMN and FPN. ICs were labelled based upon agreement between experts. Moreover, to better characterize each network, a meta-analytic decoding of ICs was also performed using NeuroSynth (http://neurosynth.org78) (Supplementary Table S1). Calculation of the correlation coefficient between the time courses of each spatial component estimated the network FC between the DMN and the FPN. FC values for the DMN-FPN of each participant were extracted for further analysis.
Statistical analysis
Demographic variables were compared using the independent t-test and chi-square test. For comparing PAS scores and BDNFm between groups, we performed analyses of covariance (ANCOVA) using the age, sex, and level of education as covariates. Square root transformation was applied to the PAS scores to produce a normal distribution of responses; therefore, all variables had skewness levels less than 1.0, which was acceptable for ANCOVA.
When determining a statistically significant difference in the FC of the DMN-FPN between HC and UHR individuals, ANCOVA was conducted controlling for age, sex, and level of education.
To determine whether schizotypy moderated the relationship between BDNF methylation and the DMN-FPN FC network, the effects of PAS scores and BDNFm on the DMN-FPN FC were analyzed using the simple moderation model in PROCESS macro in SPSS79 with age, sex, and level of education as covariates. Because the results might be driven by group differences, the additional analysis was conducted, including using the group as an independent variable in the model. To control for possible confounding effects of antipsychotic medications in UHR individuals, the analysis was also repeated by controlling for the chlorpromazine equivalent dose, as well as age, sex, and level of education. For all analyses, P < 0.05 was considered statistically significant.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Liu, C. H., Keshavan, M. S., Tronick, E. & Seidman, L. J. Perinatal risks and childhood premorbid indicators of later psychosis: Next steps for early psychosocial interventions. Schizophrenia Bull. 41, 801–816 (2015).
Yung, A. R. & McGorry, P. D. The prodromal phase of first-episode psychosis: Past and current conceptualizations. Schizophrenia Bull. 22, 353–370 (1996).
Fusar-Poli, P. et al. Predicting psychosis: Meta-analysis of transition outcomes in individuals at high clinical risk. Archives Gen. Psychiatry 69, 220–229 (2012).
Northoff, G. & Duncan, N. W. How do abnormalities in the brain’s spontaneous activity translate into symptoms in schizophrenia? From an overview of resting state activity findings to a proposed spatiotemporal psychopathology. Prog. Neurobiol. 145, 26–45 (2016).
Buckner, R. L., Andrews-Hanna, J. R. & Schacter, D. L. The brain’s default network: Anatomy, function, and relevance to disease. Ann. N Y Acad. Sci. 1124, 1–38 (2008).
Seeley, W. W. et al. Dissociable intrinsic connectivity networks for salience processing and executive control. J. Neurosci. 27, 2349–2356 (2007).
Dong, D., Wang, Y., Chang, X., Luo, C. & Yao, D. Dysfunction of large-scale brain networks in schizophrenia: A meta-analysis of resting-state functional connectivity. Schizophrenia Bull. 44, 168–181 (2018).
Shim, G. et al. Altered resting-state connectivity in subjects at ultra-high risk for psychosis: An fMRI study. Behavioral Brain Funct.: BBF 6, 58 (2010).
Wotruba, D. et al. Aberrant coupling within and across the default mode, task-positive, and salience network in subjects at risk for psychosis. Schizophr Bull. 40, 1095–1104 (2014).
Fox, M. D. et al. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc. Natl Acad. Sci. USA 102, 9673–9678 (2005).
Johns, L. C. & Van, Os, J. The continuity of psychotic experiences in the general population. Clin. Psychol. Rev. 21, 1125–1141 (2001).
Ettinger, U., Meyhöfer, I., Steffens, M., Wagner, M. & Koutsouleris, N. Genetics, cognition, and neurobiology of schizotypal personality: A review of the overlap with schizophrenia. Front. Psychiatry 5, 18 (2014).
Meehl, P. E. Toward an integrated theory of schizotaxia, schizotypy, and schizophrenia. J. Personality Disorders 4, 1–99 (1990).
Lenzenweger, M. F. & Korfine, L. Confirming the latent structure and base rate of schizotypy: A taxometric analysis. J. Abnorm. Psychol. 101, 567 (1992).
Lenzenweger, M. F. Schizotypy: An organizing framework for schizophrenia research. Curr. Directions Psychol. Sci. 15, 162–166 (2006).
Ettinger, U. et al. Cognition and brain function in schizotypy: A selective review. Schizophr Bull. 41 (Suppl. 2), S417–426 (2015).
Chan, R. C. et al. Neurological soft signs precede the onset of schizophrenia: a study of individuals with schizotypy, ultra-high-risk individuals, and first-onset schizophrenia. Eur. Archives Psychiatry Clin. Neurosci. 268, 49–56 (2018).
Lagioia, A., Van De Ville, D., Debbané, M., Lazeyras, F. & Eliez, S. Adolescent resting state networks and their associations with schizotypal trait expression. Front. Syst. Neurosci. https://doi.org/10.3389/fnsys.2010.00035 (2010).
Wang, Y. et al. Altered corticostriatal functional connectivity in individuals with high social anhedonia. Psychol. Med. 46, 125–135 (2016).
Wang, Y., Ettinger, U., Meindl, T. & Chan, R. C. K. Association of schizotypy with striatocortical functional connectivity and its asymmetry in healthy adults. Human Brain Mapping 39, 288–299 (2018).
Zhu, Y. et al. Reduced functional connectivity between bilateral precuneus and contralateral parahippocampus in schizotypal personality disorder. BMC Psychiatry 17, 48 (2017).
Wang, Y. M. et al. Altered functional connectivity of the default mode network in patients with schizo-obsessive comorbidity: A comparison between schizophrenia and obsessive-compulsive disorder. Schizophr Bull. 45, 199–210 (2019).
Ingram, R. E. & Luxton, D. D. Vulnerability-Stress Models 32–46 (Sage Publications, 2005).
Guintivano, J. & Kaminsky, Z. A. Role of epigenetic factors in the development of mental illness throughout life. Neurosci. Res. 102, 56–66 (2016).
Binder, D. K. & Scharfman, H. E. Brain-derived neurotrophic factor. Growth Factors 22, 123–131 (2004).
Autry, A. E. & Monteggia, L. M. Brain-derived neurotrophic factor and neuropsychiatric disorders. Pharmacol. Rev. 64, 238–258 (2012).
Cattaneo, A., Cattane, N., Begni, V., Pariante, C. M. & Riva, M. A. The human BDNF gene: Peripheral gene expression and protein levels as biomarkers for psychiatric disorders. Transl. Psychiatry 6, e958–e958 (2016).
Miao, Z., Wang, Y. & Sun, Z. The relationships between stress, mental disorders, and epigenetic regulation of BDNF. Int. J. Mol. Sci. 21, 1375 (2020).
Peng, H. et al. Childhood trauma, DNA methylation of stress-related genes, and depression: Findings from two monozygotic twin studies. Psychosomatic Med. 80, 599–608 (2018).
Dong, E. et al. Brain-derived neurotrophic factor epigenetic modifications associated with schizophrenia-like phenotype induced by prenatal stress in mice. Biol. Psychiatry 77, 589–596 (2015).
Kordi-Tamandani, D. M., Dahmardeh, N. & Torkamanzehi, A. Evaluation of hypermethylation and expression pattern of GMR2, GMR5, GMR8, and GRIA3 in patients with schizophrenia. Gene 515, 163–166 (2013).
Ikegame, T. et al. DNA methylation analysis of BDNF gene promoters in peripheral blood cells of schizophrenia patients. Neurosci. Res. 77, 208–214 (2013).
Çöpoğlu, Ü. S. et al. DNA methylation of BDNF gene in schizophrenia. Med. Sci. Monitor: Int. Med. J. Exp. Clin. Res. 22, 397 (2016).
Fachim, H. A. et al. Early-life stress effects on BDNF DNA methylation in first-episode psychosis and in rats reared in isolation. Progress Neuro-Psychopharmacol. Biol. Psychiatry 108, 110188 (2021).
Redlich, R. et al. The role of BDNF methylation and Val66Met in amygdala reactivity during emotion processing. Human Brain Mapping 41, 594–604 (2020).
Moser, D. A. et al. BDNF methylation and maternal brain activity in a violence-related sample. PLoS One 10, e0143427 (2015).
Fusar-Poli, P. et al. The psychosis high-risk state: A comprehensive state-of-the-art review. JAMA Psychiatry 70, 107–120 (2013).
Li, T. et al. Brain-wide analysis of functional connectivity in first-episode and chronic stages of schizophrenia. Schizophr Bull. 43, 436–448 (2017).
Bolding, M. S. et al. Antipsychotic drugs alter functional connectivity between the medial frontal cortex, hippocampus, and nucleus accumbens as measured by H215O PET. Front. Psychiatry 3, 105 (2012).
Kroken, R. A., Johnsen, E., Ruud, T., Wentzel-Larsen, T. & Jørgensen, H. A. Treatment of schizophrenia with antipsychotics in Norwegian emergency wards, a cross-sectional national study. BMC Psychiatry 9, 1–9 (2009).
Mishara, A. et al. Neurobiological models of self-disorders in early schizophrenia. Schizophrenia Bull. 42, 874–880 (2015).
Parnas, J. et al. Self-experience in the early phases of schizophrenia: 5-year follow-up of the Copenhagen Prodromal Study. World Psychiatry 10, 200 (2011).
Nelson, B., Thompson, A. & Yung, A. R. Basic self-disturbance predicts psychosis onset in the ultra high risk for psychosis “prodromal” population. Schizophrenia Bull. 38, 1277–1287 (2012).
Van Os, J., Kenis, G. & Rutten, B. P. The environment and schizophrenia. Nature 468, 203–212 (2010).
Ehrlich, D. & Josselyn, S. Plasticity‐related genes in brain development and amygdala‐dependent learning. Genes, Brain Behav. 15, 125–143 (2016).
Matsumoto, T. et al. Brain-derived neurotrophic factor-induced potentiation of glutamate and GABA release: Different dependency on signaling pathways and neuronal activity. Mol. Cell. Neurosci. 31, 70–84 (2006).
Barnea, A. & Roberts, J. Induction of functional and morphological expression of neuropeptide Y (NPY) in cortical cultures by brain-derived neurotrophic factor (BDNF): evidence for a requirement for extracellular-regulated kinase (ERK)-dependent and ERK-independent mechanisms. Brain Res. 919, 57–69 (2001).
Wheater, E. N. et al. DNA methylation and brain structure and function across the life course: A systematic review. Neurosci. Biobehav. Rev. 113, 133–156 (2020).
Ursini, G. et al. BDNF rs6265 methylation and genotype interact on risk for schizophrenia. Epigenetics 11, 11–23 (2016).
Zheleznyakova, G. Y., Cao, H. & Schiöth, H. B. BDNF DNA methylation changes as a biomarker of psychiatric disorders: literature review and open access database analysis. Behavioral Brain Funct. 12, 17 (2016).
Schübeler, D. Epigenetic islands in a genetic ocean. Science 338, 756–757 (2012).
Joubert, B. R. et al. Maternal smoking and DNA methylation in newborns: In utero effect or epigenetic inheritance? Cancer Epidemiol. Prevention Biomarkers 23, 1007–1017 (2014).
Stroud, L. R. et al. Maternal smoking during pregnancy and infant stress response: Test of a prenatal programming hypothesis. Psychoneuroendocrinology 48, 29–40 (2014).
Kwapil, T. R., Gross, G. M., Silvia, P. J. & Barrantes-Vidal, N. Prediction of psychopathology and functional impairment by positive and negative schizotypy in the Chapmans’ ten-year longitudinal study. J. Abnormal Psychol. 122, 807 (2013).
Bogren, M. et al. Predictors of psychosis: A 50-year follow-up of the Lundby population. European Archives Psychiatry Clin. Neurosci. 260, 113–125 (2010).
Carter, J., Parnas, J., Cannon, T., Schulsinger, F. & Mednick, S. MMPI variables predictive of schizophrenia in the Copenhagen High‐Risk Project: A 25‐year follow‐up. Acta Psychiatrica Scand. 99, 432–440 (1999).
Shah, J. et al. Multivariate prediction of emerging psychosis in adolescents at high risk for schizophrenia. Schizophrenia Res. 141, 189–196 (2012).
Johnstone, E. C., Ebmeier, K. P., Miller, P., Owens, D. G. & Lawrie, S. M. Predicting schizophrenia: Findings from the Edinburgh high-risk study. Br. J. Psychiatry 186, 18–25 (2005).
Salokangas, R. et al. Prediction of psychosis in clinical high-risk patients by the Schizotypal Personality Questionnaire. Results of the EPOS project. Eur. Psychiatry 28, 469–475 (2013).
Mason, O. et al. Risk factors for transition to first episode psychosis among individuals with ‘at-risk mental states’. Schizophrenia Res. 71, 227–237 (2004).
Bang, M. et al. Psychotic conversion of individuals at ultra-high risk for psychosis: The potential roles of schizotypy and basic symptoms. Early Intervention Psychiatry 13, 546–554 (2019).
Debbané, M. et al. Developing psychosis and its risk states through the lens of schizotypy. Schizophrenia Bull. 41, S396–S407 (2014).
Parnas, J. et al. EASE: Examination of anomalous self-experience. Psychopathology 38, 236 (2005).
First, M. B, Spitzer, R. L, Gibbon, M & Williams, J. B. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Patient Edition (SCID-I/P New York, 2002).
Spitzer, M., Robert, L., Gibbon, M. & Williams, J. Structured Clinical Interview for DSM-IV-TR Axis I Disorders, Research Version, Non-Patient Edition (SCID-I/NP). (Biometrics Research, New York State Psychiatric Institute, 2002).
Miller, T. J. et al. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: Predictive validity, interrater reliability, and training to reliability. Schizophrenia Bull. 29, 703 (2003).
An, S. K. et al. Attribution bias in ultra-high risk for psychosis and first-episode Schizophrenia. Schizophrenia Res. 118, 54–61 (2010).
Kang, J. I. et al. Reduced binding potential of GABA-A/benzodiazepine receptors in individuals at ultra-high risk for psychosis: An [18F]-fluoroflumazenil positron emission tomography study. Schizophrenia Bull. 40, 548–557 (2014).
Chapman, L. J., Chapman, J. P. & Raulin, M. L. Body-image aberration in Schizophrenia. J. Abnormal Psychol. 87, 399 (1978).
Lin, H.-F. et al. Neuregulin 1 gene and variations in perceptual aberration of schizotypal personality in adolescents. Psychol. Med. 35, 1589–1598 (2005).
Chang, B. P. & Lenzenweger, M. F. Somatosensory processing and schizophrenia liability: Proprioception, exteroceptive sensitivity, and graphesthesia performance in the biological relatives of schizophrenia patients. J. Abnormal Psychol. 114, 85 (2005).
Kang, H.-J. et al. BDNF promoter methylation and suicidal behavior in depressive patients. J. Affective Disorders 151, 679–685 (2013).
Kang, H.-J. et al. Longitudinal associations between BDNF promoter methylation and late-life depression. Neurobiology Aging 36, e1761–1764. e1767 (2015).
Roth, T. L., Lubin, F. D., Funk, A. J. & Sweatt, J. D. Lasting epigenetic influence of early-life adversity on the BDNF gene. Biol. Psychiatry 65, 760–769 (2009).
Whitfield-Gabrieli, S. & Nieto-Castanon, A. Conn: A functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connectivity 2, 125–141 (2012).
Calhoun, V. D., Adali, T., Pearlson, G. D. & Pekar, J. J. A method for making group inferences from functional MRI data using independent component analysis. Human Brain Mapping 14, 140–151 (2001).
Smith, S. M. et al. Functional connectomics from resting-state fMRI. Trends Cogn. Sci. 17, 666–682 (2013).
Yarkoni, T., Poldrack, R. A., Nichols, T. E., Van Essen, D. C. & Wager, T. D. Large-scale automated synthesis of human functional neuroimaging data. Nature Methods 8, 665–670 (2011).
Hayes, A. F. Introduction to Mediation, Moderation, and Conditional Process Analysis: A Regression-Based Approach (Guilford publications, 2017).
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning, Republic of Korea (grant number 2017R1A2B3008214, 2022R1A2B5B03002611). The authors have declared that there are no conflicts of interest in relation to the subject of this study.
Author information
Authors and Affiliations
Contributions
H.Y.P. and S.K.A. are responsible for study concept and design. H.Y.P. drafted the manuscript. H.Y.P. took responsibility for acquisition, analysis, or interpretation of data. M.B., E.S., S.J.K., and E.L. contributed to the discussion. M.B., E.S., S.J.K., E.L., and S.K.L. reviewed and edited the manuscript. A.S.K. is the guarantor of this work and as such, had full access to all study data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Park, H.Y., Bang, M., Seo, E. et al. The stress-vulnerability model on the path to schizophrenia: Interaction between BDNF methylation and schizotypy on the resting-state brain network. Schizophr 8, 49 (2022). https://doi.org/10.1038/s41537-022-00258-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-022-00258-4