Abstract
Despite the consistent finding of an attenuated niacin-induced flush response in schizophrenia, its long-term stability and relationship to the membrane polyunsaturated fatty acid (PUFA) levels remain unknown. We conducted niacin skin tests and measured the membrane PUFAs using gas chromatography among 46 schizophrenia inpatients and 37 healthy controls at the baseline and the 2-month follow-up. Attenuated flush responses were persistently observed in schizophrenia patients in both acute and partial remission states, whereas an increased flush response was found in the controls. A persistent decrease in both dihomo-gamma-linolenic acid and docosahexaenoic acid and an increased turnover of arachidonic acid (ARA) via endogenous biosynthesis were found in schizophrenia patients. A composite niacin flush score by combining those with a control-to-case ratio of >1.4 (i.e., scores at 5 min of 0.1 M, 0.01 M, and 0.001 M + 10 min of 0.01 M and 0.001 M + 15 min of 0.001 M) at the baseline was correlated positively with ARA levels among controls but not among schizophrenia patients, whereas the flush score at the 2-month follow-up was correlated positively with ARA levels among patients. The 2-month persistence of attenuated niacin-induced flush response in schizophrenia patients implies that the niacin skin test might tap a long-term vulnerability to schizophrenia beyond acute exacerbation.
Similar content being viewed by others
Introduction
An attenuated niacin-induced flush response in schizophrenia has exhibited several characteristics that render it a potential endophenotype of schizophrenia1,2,3. The prevalence of the niacin response abnormality was higher in schizophrenia patients4,5,6, first-episode psychosis patients7, and ultra-high risk patients8 than in healthy controls. An attenuated niacin flush response has also been found in nonpsychotic first-degree relatives of schizophrenia patients9,10,11, and greater familial loading of schizophrenia was associated with more impairment in the flush response12. A genome-wide linkage scan of the niacin skin flush response among families of siblings co-affected with schizophrenia has led to some genetic linkage signals13. Nevertheless, whether the impairment in the niacin flush response is state-independent, another important characteristic for an endophenotype14, has received relatively less attention3. Repeated oral administration of nicotinic acid in healthy individuals exhibited tolerance within 2 days15. Only a few studies of small sample sizes examined whether the niacin response abnormality subsided after therapeutic intervention16,17,18. Thus, a prospective follow-up of niacin skin flush in patients with schizophrenia is warranted to clarify the stability of the niacin response abnormality.
The niacin-induced flush is exerted via a prostaglandin D2-related microvasodilation pathway19,20, triggered by its binding to a G-protein-coupled receptor on epidermal Langerhans cells21 to stimulate phospholipase A2 (PLA2) to release arachidonic acid (ARA) from cell membrane phospholipids22, which is further metabolized into prostaglandin molecules by cyclooxygenase-2 (COX-2)23. As postulated in the membrane hypothesis of schizophrenia, the loss of polyunsaturated fatty acids (PUFA), including ARA, results in membrane rigidity and can alter the conformation and functioning of proteins, receptors, and ion channels24,25. Dysfunctional ARA signaling, such as abnormal activities of PLA2 or COX-2 (refs. 17,26) and fatty acids composition of red blood cell (RBC) membranes27,28 have been implicated in schizophrenia patients. However, the relations of the genetic variants encoding the PLA2/COX-2 enzymes to the abnormal niacin response in schizophrenia is complex. When the distributions of the genetic variants of PLA2G6 (encoding calcium-independent PLA2 beta) were compared between schizophrenia patients and controls, one study in Brazilians29 found a significant difference but three studies in Chinese30 or Croatians31,32 did not. When similar comparisons were conducted for PLA2G4C (encoding cytosolic PLA2 gamma), two studies in Chinese found significant differences30,33 but one study in Croatians did not32, although the latter did find a significant difference in PLA2G6 and PLA2G4C genotype combination distributions32. Meanwhile, the niacin response in schizophrenia patients was associated not with the genetic variants of PLA2G6 and PLA2G4C34 but with the variants of PLA2G4A (encoding cytosolic PLA2) and PTGS2 (encoding COX-2)35. Two meta-analyses found the levels of docosapentaenoic (n-3) acid (DPA), docosahexaenoic (n-3) acid (DHA), and ARA of RBCs were decreased in medication-naïve patients, and a smaller reduction was also shown in medicated patients, though substantial heterogeneity among studies was noted36,37. Genetic studies found that desaturases, including delta-5 (D5D) and delta-6 (D6D) desaturase, play essential roles in the endogenous synthesis of ARA, whereas dietary intakes were the major source for DHA38.
The niacin response abnormality may reflect dysfunctional ARA-related signaling in schizophrenia patients. When schizophrenia patients took oral niacin, patients with the absence of a flush response showed reduced RBC levels of ARA and DHA16. Three studies applying topical niacin patches to schizophrenia patients failed to find any correlation between a niacin response abnormality and RBC membrane ARA levels35,39,40, though one of them did find such correlation in healthy controls40. Hence, the niacin response abnormality might be associated with a homeostatic imbalance in schizophrenia patients. Seldom examined is whether an acute state of schizophrenia could affect the relationship between the niacin-induced flush response and RBC membrane ARA levels.
To fill in these gaps in the literature, we conducted a follow-up study of schizophrenia patients at acute admission and partial remission 2 months later, as well as obtained similar measurements 2 months apart in a group of sex- and age-matched healthy controls. Specific aims of the study were to investigate the following: (1) whether the niacin flush response in the acute phase of schizophrenia at admission was attenuated compared to the response in healthy controls, and whether the attenuation would diminish at partial remission; (2) whether schizophrenia patients had altered RBC membrane levels of PUFAs or frequencies of the genetic variants of PLA2G6 for calcium-independent PLA2 or PLA2G4A for cytosolic PLA2, compared to healthy controls, and whether the altered levels persisted at partial remission; and (3) whether there were correlations between the niacin-induced flush response and RBC membrane levels of PUFAs among schizophrenia patients in the acute phase at admission and in partial remission, and, for comparison, among healthy controls.
Results
Sample characteristics
Among 48 people with schizophrenia and 37 age- and sex-matched healthy volunteers recruited for this study, two patients were excluded from final analysis because their fatty acid analysis was not available. For the two SNPs genotyped in this study, PLA2G6 rs4375 passed the quality control whereas PLA2G4A rs10798059 did not and hence only the former is reported here. Both the schizophrenia patients (n = 46) and healthy controls (n = 37) were similar in the distribution of sex, age, length of follow-up, and the genotypes of PLA2G6 rs4375, but the patients had a higher prevalence of tobacco smoking than the controls (Table 1). Among the schizophrenia patients recruited, only four were at first onset with minimal treatment with antipsychotics while the others had varying histories of treatment for the illness. Since the two groups were frequency-matched with age and sex, we adjusted for potential confounders in comparing patients with controls using a propensity score consisting of age, sex, and smoking. From the baseline at admission to the follow-up at 8 weeks later or on the first out-patient follow-up if the patient had been discharged by 8 weeks, the Positive and Negative Syndrome Scale (PANSS) total score as well as its subscales scores of schizophrenia patients showed significant reduction. Hence, for simplicity the two time points were referred to as the baseline (acute) and approximately 2-month follow-up (partial remission) of schizophrenia patients, respectively, in the subsequent text.
Longitudinal pattern of niacin-induced flush
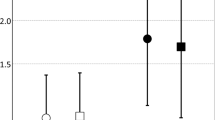
Schizophrenia patients had lower mean niacin flush scores at a variety of time–concentration combinations than healthy controls at both baseline and the 2-month follow-up (Supplementary Table S1). In comparing the niacin flush score at the two time points, we summed up the rating over the three concentrations for different time points. As shown in Fig. 1, the healthy controls showed a significant increase in flush scores from baseline to the 2-month follow-up for the summed ratings at 5 and 10 min, but not at 15 min, after correction for multiple testing. In contrast, schizophrenia patients did not show any increase in flush score from admission to discharge 2 months later. Furthermore, the patients’ flush scores were significantly lower than the controls’ scores for all summed scores across concentrations at 5, 10, and 15 min.
Mean niacin flush score that was summed over the three concentrations for ratings at 5, 10, and 15 min, respectively, at different time points in a healthy controls and b schizophrenia patients, with error bars representing standard error. Time 1 = baseline, time 2 = week 2, time 3 = week 4, and time 4 = week 8 or on the first outpatient follow-up if the patient had been discharged by 8 weeks. Solid lines represent the scores of all participants who had flush scores at both time 1 and time 4 (n = 37 for controls and 46 for schizophrenia) and dashed lines the scores of a subgroup of participants who had flush scores at all four time points (n = 22 for controls and 35 for schizophrenia). For those summed niacin flush scores that changed significantly with time in a mixed-effects model were denoted with p values.
For participants who had additional niacin ratings at weeks 2 and 4, displayed as the dotted line in Fig. 1, the niacin ratings on four occasions (week 0, week 2, week 4, and week 8) of controls (n = 22) exhibit an increasing trend for ratings at 5 and 10 min, whereas those of schizophrenia patients (n = 35) did not. The results of the mixed-effects regression analysis confirmed that there was a significant group effect plus a significant interaction between group and time for ratings at 5 and 10 min, respectively.
To derive a composite niacin score that maximized the contrast between schizophrenia patients and healthy controls at baseline, we calculated the ratio of niacin score at each time–concentration point (Supplementary Table S2). We combined the ratings of those time–concentration points that have a ratio of >1.40, i.e., scores at (5 min of 0.1 M, 0.01 M, and 0.001 M) + (10 min of 0.01 M and 0.001 M) + (15 min of 0.001 M), to construct the composite niacin flush score.
We then examined whether the composite niacin flush score at baseline was associated with PLA2G6 genotype (rs4375), which was grouped into the presence of C (i.e., CC or CT) and absence of C (i.e., TT), separately for schizophrenia patients and healthy controls. Comparing the mean of two genotype groups with adjustment using a propensity score consisting of age, sex, and smoking, neither schizophrenia patients (6.55 [SD = 1.02] with n = 22 vs. 6.13 [SD = 0.95] with n = 23, p = 0.79) nor healthy controls (9.94 [SD = 0.76] with n = 17 vs. 10.21 [SD = 0.82] with n = 19, p = 0.27) reached statistical significance.
Membrane PUFAs and schizophrenia
The levels of 24 fatty acids in the RBC membrane are listed in Supplementary Table S3 for controls and Supplementary Table S4 for schizophrenia patients. Since this study was focused on PUFAs, only this part of the profile is reported in Table 2. We calculated the average of controls’ measurements at baseline and the 2-month follow-up as the pooled reference. Compared to the pooled reference, schizophrenia patients at admission had increased levels of gamma-linoleic acid (GLA; effective size = 0.6) and adrenic acid (effective size = 0.8), but had decreased levels of dihomo-gamma-linolenic acid (DGLA; effective size = −0.7) and docosahexaenoic acid (DHA; effective size = −0.6) after correction for multiple testing (Table 2). At the 2-month follow-up, schizophrenia patients had decreased levels of DGLA (effective size = −0.6), DHA (effective size = −0.8), and sum of n-3 fatty acids (effective size = −0.7) after correction for multiple testing.
Since the level of ARA (20:4n-6) might be influenced by endogenous biosynthesis, i.e., being converted to adrenic acid (22:4n-6) via elongation and being replenished from DGLA (C20:3n-6) via desaturation, we then examined the relationship of the levels of ARA to that of its product (adrenic acid) as well as its precursor (DGLA) (Supplementary Fig. S1). For controls at baseline, levels of ARA were correlated neither with adrenic acid nor with DGLA. For schizophrenia patients, ARA was correlated positively with adrenic acid both in the acute state (r = 0.487) and at partial remission (r = 0.574), and was correlated negatively with DGLA at partial remission (r = −0.606) but not in the acute state. When Spearman correlation was used for these analyses, the results remain the same (Supplementary Table S5).
To further explore the mechanisms underpinning the altered levels of fatty acids among schizophrenia patients, we estimated desaturase activities by means of product/precursor ratios of PUFA, following a previous study41. The results revealed that schizophrenia patients at both baseline and the 2-month follow-up had higher D5D and D6D activities than controls (Table 3).
PUFAs and the niacin flush response
Then, we examined the relations of ARA, DGLA, and adrenic acid to the composite niacin flush score (Supplementary Table S6). Both ARA and DGLA exhibited correlations with the niacin composite score of controls at baseline, and DGLA exhibited correlations with the niacin composite score of schizophrenia patients at partial remission; meanwhile, adrenic acid did not have any correlation with the composite niacin flush score.
Figure 2 plots the niacin composite score with ARA and DGLA, respectively. For controls at baseline, ARA levels were positively correlated with the composite niacin score (r = 0.317, p = 0.028), while DGLA levels were negatively correlated with the composite niacin score (r = −0.361, p = 0.014). In contrast, for schizophrenia patients at baseline, neither ARA nor DGLA exhibited correlations with the composite niacin flush score. However, for schizophrenia patients at the 2-month follow-up, the composite niacin flush score had a borderline positive correlation with ARA (r = 0.239, p = 0.059) and a negative correlation with DGLA (r = −0.372, p = 0.006). When Spearman correlation was used for these analyses, the results remain the same (Supplementary Table S7).
Discussion
In this prospective study, healthy controls exhibited enhanced niacin-induced flush at the 2-month follow-up, whereas schizophrenia patients did not have significant changes in their flush response from the baseline to the 2-month partial remission, remaining attenuated at both time points. Both groups did not differ in the genotype distribution of PLA2G6 rs4375, and participants’ niacin responses were not associated with this gene’s variants within each group. Lower RBC membrane levels of DGLA and DHA in patients as compared to the controls were consistently observed at both time points. In addition, increased levels of the precursor (DGLA) and product (adrenic acid) of the endogenous biosynthesis of ARA were observed in schizophrenia patients only at admission. A composite score of the niacin flush response was positively correlated with ARA and negatively correlated with DGLA levels in the controls at baseline, but failed to show such correlations in schizophrenia patients. Nevertheless, at the partial remission, the niacin flush response in schizophrenia patients became weakly correlated with ARA and negatively correlated with DGLA levels. These findings provide support for elevated turnover of ARA signaling as the pathophysiology underpinning the attenuated flush response to niacin in schizophrenia and its dynamic relationship to membrane PUFAs.
A surprising finding is that healthy controls had increased niacin-induced flush response to the repeated niacin skin tests at the 2-month follow-up. This is different from the finding of a previous study of healthy volunteers, in which the flush response to oral niacin dosing diminished within 2 days15. In contrast, schizophrenia patients’ flush response to the repeated niacin skin tests remained attenuated and unchanged during the 2-month period, even when patients already had partial remission in symptoms. Previous stability studies in schizophrenia patients used a binary rating of the flush after oral niacin intake18 or application of a topical niacin patch17. Despite the differences in the method of administration and scoring, approximately 70–80% of the nonflushers among schizophrenia patients persisted to be so 2 (ref. 17) or 3 months18 later. Since the dermal niacin receptor that binds niacin is located in Langerhans cells21, there may be immune memory by these immune cells or other intermediates in the cascade after the initial activation by the niacin skin patch in healthy controls. Meanwhile, schizophrenia patients might have certain immunological aberrations that are related to the persistence of attenuated niacin-induced flush response. Increasing evidence indicates that there are immune system disturbances in schizophrenia42,43, e.g., autoantibodies to neurotransmitter receptors44,45,46 or inflammation47,48. Whether these immune system disturbances are associated with the aberrations in niacin-induced flush response in schizophrenia warrants future investigation.
One potential contributor to the niacin-induced flush abnormality in schizophrenia examined in this study is the genetic variants of PLA2G6 (rs4375). We found that its genotype distributions were not different between schizophrenia patients and healthy controls, which is different from the association found in Brazilians29 but consistent with studies in Chinese30 or Croatians31,32. Furthermore, we also found the niacin-induced flush response was not associated with genotypes of PLA2G6, regardless of schizophrenia patients or healthy controls. This is consistent with a previous study in Croatian patients with schizophrenia34. Nevertheless, there are other PLA2 genes that were found to be associated with the risk of schizophrenia, e.g., PLA2G4C29,30,33, or the niacin-induced flush response in schizophrenia patients, e.g., PLA2G4A and PTGS2 (ref. 35). In addition, when the plasma levels of different forms of PLA2 were measured, the levels of calcium-independent PLA2 were higher in both schizophrenia patients49 and ultra-high risk individuals50 than in controls, whereas the level of cytosolic PLA2 were higher only in individuals with ultra-high risk as compared to controls50. Whether the niacin-induced flush abnormality could be attributed to the genetic polymorphisms of other PLA2 genes or the altered levels of PLA2 warrants future investigation.
In comparing the fatty acid composition between groups or time points, we reported in the form of percentage of total fatty acids rather than the quantification of the actual amount (weight) of each fatty acid that is present in the RBC membrane. Regarding the RBC membrane’s PUFAs, schizophrenia patients had persistent decrease at both baseline and the 2-month partial remission in the levels of DHA, a n-3 PUFA with main source from dietary intake, and DGLA, a n-6 PUFA and precursor of the biosynthesis of ARA. The lowered level of DHA is consistent with the findings from two meta-analyses36,37. Unlike ARA, which is released from membrane preferentially by cytosolic PLA2 and has prooxidant and proinflammatory properties, DHA is predominantly released by calcium-independent PLA2 and has antioxidant and anti-proinflammatory properties51,52. In addition to lower dietary intake of PUFAs53, increased calcium-independent PLA2 levels might also contribute to the lowered level of RBC membrane’s DHA49.
Meanwhile, the persistent decrease in the level of DGLA in schizophrenia patients should be understood in the context of the endogenous biosynthesis of ARA38. Under this circumstance, the endogenous synthesis of ARA was not apparent in healthy controls, indicated by their lack of ARA’s correlation with its precursor, but was active in schizophrenia, with ARAs having a negative correlation with DGLA. One reason that schizophrenia patients had active endogenous synthesis of ARA is due to an increased turnover of membrane ARA as a result of increased cytosolic PLA2 activity. Because of this replenishment of ARA via endogenous synthesis, we did not find a decrease in the membrane levels of ARA in schizophrenia patients, which was also reported in some previous studies40,54. Another explanation is the possible normalization effect of long-term anti-psychotic treatment55. Since the majority of our patients were not in their first episode, we could not rule out this possibility.
Furthermore, the D5D desaturase activity, which turns DGLA into ARA, was found to be higher in schizophrenia patients than in controls at both time points in this study. Meanwhile, the membrane level of adrenic acid in schizophrenia patients was elevated in the acute state but not at the 2-month partial remission. Consequently, our findings indicate that the temporal availability of membrane ARA in schizophrenia patients might be more at the partial remission than in the acute state. A previous study of small sample size (20 schizophrenia patients and 20 controls) also reported a tendency for schizophrenia patients to have an increased membrane levels of adrenic acid40. Intriguingly, our results showed that patients’ DGLA levels had a negative correlation with that of ARA only at the 2-month partial remission, when a continuous replenishment of ARA from DGLA was in synchronization with a slowdown in the turnover of ARA into adrenic acid at this time point.
The positive correlation between the niacin-induced flush response with the membrane levels of ARA in heathy controls in this study was consistent with a previous study showing a positive correlation between Doppler-measured blood flow and levels of ARA40. However, the relations of the niacin-induced flush response to ARA’s precursor or product varied across studies. The niacin-induced flush response in this study was correlated with DGLA levels but not with adrenic acid levels in healthy controls at baseline, whereas the Doppler-measured blood flow in the previous study40 showed trends towards correlation with adrenic acid levels but not with DGLA levels in healthy controls. The discrepancies might be due to varying enzymatic activities of D5D in the endogenous biosynthesis of ARA for patients in different clinical stages.
Despite that schizophrenia patients had similar levels of ARA in both the acute and partial remission states, the correlation between ARA level and the composite niacin-induced flush score was lost in the acute state but regained weakly at the 2-month partial remission. We postulated that only when the temporal availability of ARA increased as a result of balance between replenishment of ARA from DGLA with the turnover of ARA into adrenic acid, the niacin-induced flush response had a positive correlation with ARA levels and a negative correlation with DGLA levels. Future studies are warranted to investigate whether the synchronization in the turnover of both DGLA and ARA on the RBC membranes is an indicator of partial remission.
Alongside the increased turnover of ARA, a n-6 PUFA, via endogenous biosynthesis, schizophrenia patients exhibited a lowered membrane level of DHA, a n-3 PUFA relying mainly on dietary intake, at both the baseline and the 2-month partial remission. This might reflect an increased turnover of DHA due to genetic predisposition56, lowered dietary intake57, or elevated oxidative stress55,58,59 in schizophrenia patients. Regardless of the causes, supplementation with DHA might have a beneficial effect on patients’ improvement in psychotic symptoms. Indeed, supplementation of antipsychotics with n-3 PUFAs has been shown to improve symptoms in schizophrenia patients52,60,61,62. A recent Mendelian randomization analysis revealed that schizophrenia patients might have difficulty converting short-chain PUFA into long-chain PUFAs63.
A related question is whether the attenuated niacin-induced flush response existed in individuals at increased risk for schizophrenia. Previous family studies indicated that individuals at familial risk for schizophrenia did have increased risk of attenuated niacin-induced flush response9,10,11,12. However, the results in individuals at ultra-high risk for psychosis remain conflicting8,64. Furthermore, several randomized trials of large sample sizes65,66,67,68 failed to replicate an earlier report that supplementation of n-3 PUFAs could lower the risk of conversion to psychosis among individuals at ultra-high risk for psychosis69. Hence, the relationship between membrane levels of PUFAs and the symptoms of attenuated psychosis syndrome warrants further investigation.
Our results have implications for the niacin-induced flush abnormality as a biomarker for schizophrenia and help shed light on its underlying pathophysiology. First, the 2-month persistence of attenuated flush response in schizophrenia patients implies that the niacin skin test might tap a long-term vulnerability to schizophrenia beyond acute exacerbation. When schizophrenia patients underwent membrane lipid compositional changes from acute phase at baseline to partial remission at the 2-month follow-up, e.g., an increased level of adrenic acid, resumption of a negative correlation in levels between DGLA and ARA, and thus resulting in a correlation of the composite flush score with ARA (positively) and DGLA (negatively) as healthy controls at baseline. Second, the 2-month attenuated niacin-induced flush response in schizophrenia patients is accompanied by the persistent lowering of RBC membrane’s DGLA and DHA. DGLA is a n-6 PUFA that can be converted from GLA by elongation and, in addition to be converted into ARA by D5D desaturase, can be metabolized by COX-1 into prostaglandin E1, free radicals, and reactive oxygen species70. Meanwhile, DHA is a n-3 essential fatty acid that is released from membrane by iPLA2 and can be metabolized by lipoxygenases to antioxidative molecules, such as docosanoids, resolvins, and neuroprotectins52. Thus, a long-lasting vulnerability underlying schizophrenia tapped by the niacin skin test might be dysfunction in antioxidative and anti-proinflammatory mechanisms in schizophrenia patients47,71,72. Third, if that is the case, it stands to reason that once the levels of DGLA and DHA return to normal, which might render the niacin-induced flush response un-attenuated, a patient with schizophrenia might be in a state free of relapse in a relatively long period. At that time, the levels of prostaglandin E1, resolvins, and protectins, might also be back to normal. Thus, it is of importance to follow-up the niacin flush response in schizophrenia patients for longer terms to assess whether an increase of flush response is parallel to an increase in RBC membrane’s levels of DGLA and DHA as well as antioxidative and anti-inflmmatory molecules. Finally, in contrast to the complexity and high-cost in the measurement of RBC membrane’s PUFAs, the niacin skin test appears to be a simple, cheap, and fast test that taps the disturbance in membrane’s lipid homeostasis in schizophrenia and can be easily applied in clinical settings.
There were limitations in this study. First, our use of a 4-point Likert scale in measuring niacin-induced flush has relatively low resolution compared to Doppler-based blood flow. Nevertheless, our method allowed for testing in multiple concentrations and calculating a composite flush score to help enhance the discrimination of schizophrenia patients from healthy controls. Second, we did not have detailed food intake history of the participants and could not exclude the influence of food intake on the RBC membrane levels of PUFAs. There have been reports that schizophrenia patients had lower dietary intakes of PUFAs than controls53,57,73,74, though some studies reported an opposite direction75. Third, despite our statistical adjustment for tobacco smoking in comparing the niacin-induced skin flushing between schizophrenia patients and healthy controls, there might still be residual confounding since the former had a much higher prevalence of smoking than the latter. Cigarette smoke may directly contribute to oxidative stress and decreases in antioxidants76,77, and result in nicotine-induced inhibition of neuronal PLA2 activity78,79. Finally, a mixture of schizophrenia patients with varying numbers of psychotic episodes make it difficult to assess the influence of illness duration and antipsychotic treatment. Finally, the lack of associations between the levels of certain PUFAs and flush scores might be due to the small sample size of this study. A future study of larger sample size is warranted to validate the clues observed in this study.
In conclusion, this 2-month follow-up study found that unlike healthy controls’ enhanced flush response to the repeated niacin skin tests, schizophrenia patients’ flush response remained attenuated and unchanged at the 2-month follow-up. The difference in the niacin response could not attributed to the distribution of PLA2G6 variants. In a comprehensive measurement of RBC membrane’s PUFAs, there were aberrations in the levels of both n-3 and n-6 PUFAs in schizophrenia patients, particularly a persistent decrease in DHA and an increased turnover of ARA via endogeneous biosynthesis. Furthermore, the niacin-induced flush response in schizophrenia had a dynamic relationship with the aberrant ARA signaling from the acute state to the 2-month partial remission. These findings warrant future investigation on how the dysfunctional regulation of membrane phospholipids can be predicted or corrected in schizophrenia patients at an early stage.
Methods
Participants
Our participants were from the Lipid Biology of Schizophrenia and Schizotypal Traits (LIBISS) project conducted at the Department of Psychiatry, National Taiwan University Hospital, NTUH Yun-Lin Branch, and Taipei City Hospital Song-De Branch, as described previously80. Briefly, a total of 48 people with schizophrenia (aged 23–66) and 37 age- and sex-matched healthy volunteers made up of students and the general population were recruited between August 2009 and July 2012. Patients meeting the DSM-IV criteria of schizophrenia were recruited for interviewing using the Chinese translation81 of the Diagnostic Interview for Genetic Studies (DIGS)82. Healthy volunteers were screened using the DIGS to confirm the absence of a current or previous psychiatric history. Exclusion criteria for the study were severe neurological abnormality, prominent substance use problem, or mental retardation. The study was approved by the Research Ethics Committee of National Taiwan University Hospital (200812089R). Each participant provided written informed consent after being given a complete description of the study.
Measurements
For schizophrenia patients, the severity of their symptoms was assessed by their attending psychiatrist using the Positive and Negative Syndrome Scale (PANSS)83 at the baseline as well as 8 weeks later or on the first outpatient follow-up if the patient had been discharged by 8 weeks (referred to as the 2-month follow-up for simplicity). For both schizophrenia patients and healthy controls, we performed fasting blood sample collection and a niacin skin test at the baseline as well as the 2-month follow-up.
The PANSS consists of 30 items, each with a 7-point rating scale, and is classified into the positive symptom subscale (7 items), negative symptom subscale (7 items), and general psychopathology subscale (16 items), and its translation into a Mandarin Chinese version was shown to have good interrater reliability84, with intraclass correlation coefficients ranging from 0.64 to 0.96) in a later study85. All the participating psychiatrists had received relevant training on the use of the PANSS.
Niacin skin test and visual rating scale
The niacin skin test was modified from Ward et al.86. We used laboratory filters saturated with aqueous methyl nicotinate (AMN) as patches to apply on each subject’s forearm using four patches: three contained 50 μl of different concentrations (0.001, 0.01, and 0.1 M) of AMN and the other one was a blank negative control. After 5 min exposure to AMN, the patches were removed. The flush reaction was rated at 5, 10, and 15 min following the application with a four-point scale (0 = no erythema, 1 = incomplete erythema, 2 = complete erythema, and 3 = erythema plus edema). For quality control, a photograph was taken at each rating for later cross-evaluation. To evaluate the trend in the niacin-induced flush over time, some of the participants (35 schizophrenia patients and 22 controls) underwent additional niacin skin tests at weeks 2 and 4, along with the baseline and week 8 (or on the first outpatient follow-up if the patient had been discharged by 8 weeks) tests.
The interrater reliability of the four-point scale in rating the niacin-induced skin flush was shown to be excellent between two psychiatrists in a preliminary study of 50 subjects (34 schizophrenic patients, 4 bipolar affective patients, and 12 normal controls) with the intraclass correlation coefficient ranging from 0.85 to 0.94 for different concentrations of niacin87. In a later study of 50 subjects (25 schizophrenic patients and 25 controls), five research assistants trained by the two psychiatrists performed the rating independently and their interrater reliability was shown to be good, with the intraclass correlation coefficient ranging from 0.69 to 0.76 (ref. 11). The research assistants of this study received similar training before they conducted the test independently.
Fatty acid analysis
We used the method described by Moser et al.88 to prepare total lipid fatty acid methyl esters of plasma and RBCs. More details are provided in Supplementary Methods. Plasma layer and RBCs were first separated and stored at −80 °C until analysis. For the RBCs, after an overnight extraction, the lower layer was transferred to a clean tube. We then dried the sample under nitrogen. Then, 1 ml of methanol:dichloromethane (3/1, v/v) were added into 200 μl of the RBCs preparation. Mixed with 0.2 ml acetyl chloride, we heated the tube in a 75 °C oven for 1 h. We then used Agilent 7820 (Agilent Technology, Santa Clara, CA) for gas chromatography with flame ionization detector. The fatty acids peaks were determined by means of comparison of the retention times with those of a standard mixture of 37 FAME, PUFA2, and PUFA3 (Supelco/Sigma-Aldrich, Bellefonte, PA). The fatty acid composition was expressed as the percentage of the total fatty acid (% total fatty acids). The fatty acid analysis was not available for two patients, and hence the final sample size for this study was 46 patients and 37 controls.
Genotyping
Genomic DNA samples extracted from venous blood using the QIAamp DNA Blood Mini Kit (Qiagen, Hilden, Germany) were subjected to genotyping using an ABI TaqMan 7900 system (Applied Biosystems, Foster City, CA) for two polymorphisms: (1) the C/T (AvrII polymorphic site; rs4375) in the fourth intron of PLA2G6 and (2) the A/G (BanI polymorphic site; rs10798059) in the first intron of PLA2G4A29. The results of rs10798059 failed to distinguish genotypes clearly and were not included for analysis.
Data analysis
The sample size of this study for detecting group differences in niacin-induced flush response between schizophrenia patients and healthy controls was estimated under the following assumptions: power = 0.8, α = 0.05, equal number in each group, the proportion of subjects with absent flush response being 10% for healthy controls and 25% for schizophrenia patients, and each participant receiving repeated measurements of four times with the correlation structure being first-order autoregressive of ρ. Then the sample size was 72 if ρ = 0.2 and 94 if ρ = 0.4.
Group comparisons were performed using the χ2 test for categorical variables and t-test for continuous variables, if indicated. The mixed-effects models using the procedure of PROC MIXED, SAS Version 9.4 (SAS Institute, Cary, NC) were applied to examine the influence of time, dose, and group on the flush scores, adjusting for other covariates using the propensity score method.
Multivariable linear regression analyses were used to compare fatty acid levels between two groups with adjustment using the propensity score. We corrected for multiple testing using false discovery rate. Effect size was calculated as the mean difference between two groups divided by the SD of the whole sample.
Within each group of controls and patients, Pearson correlation coefficients were used to examine the correlation between flush scores and fatty acid composition. Since the primary hypothesis was directional40, i.e., higher AA or adrenic acid levels would correlate with more niacin response whereas higher DGLA levels would correlate with less niacin response, the significance testing of the correlation was one-tailed. For sensitivity analysis, we used Spearman correlation to repeat this part of the analysis. A p value less than 0.05 was considered significant.
Data availability
The datasets used and analyzed in the current study are not publicly available due to conditions on participant consent and other ethical restrictions. However, the data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
The SAS code that supports the findings of this study are available from the corresponding author upon reasonable request.
References
Smesny, S. et al. Endophenotype properties of niacin sensitivity as marker of impaired prostaglandin signalling in schizophrenia. Prostaglandins Leukot. Essent. Fatty Acids 77, 79–85 (2007).
Yao, J. K. et al. Prevalence and specificity of the abnormal niacin response: a potential endophenotype marker in schizophrenia. Schizophr. Bull. 42, 369–376 (2016).
Messamore, E. The niacin response biomarker as a schizophrenia endophenotype: a status update. Prostaglandins Leukot. Essent. Fatty Acids 136, 95–97 (2018).
Messamore, E., Hoffman, W. F. & Janowsky, A. The niacin skin flush abnormality in schizophrenia: a quantitative dose-response study. Schizophr. Res. 62, 251–258 (2003).
Buretic-Tomljanovic, A. et al. Phospholipid membrane abnormalities and reduced niacin skin flush response in schizophrenia. Psychiatr. Danub. 20, 372–383 (2008).
Sun, L. et al. Identification of the niacin-blunted subgroup of schizophrenia patients from mood disorders and healthy individuals in Chinese population. Schizophr. Bull. 44, 896–907 (2018).
Hu, Y. et al. A potential objective marker in first-episode schizophrenia based on abnormal niacin response. Schizophr. Res. https://doi.org/10.1016/j.schres.2021.06.028 (2021).
Langbein, K. et al. State marker properties of niacin skin sensitivity in ultra-high risk groups for psychosis—an optical reflection spectroscopy study. Schizophr. Res. 192, 377–384 (2018).
Waldo, M. C. Co-distribution of sensory gating and impaired niacin flush response in the parents of schizophrenics. Schizophr. Res. 40, 49–53 (1999).
Shah, S. H., Vankar, G. K., Peet, M. & Ramchand, C. N. Unmedicated schizophrenic patients have a reduced skin flush in response to topical niacin. Schizophr. Res. 43, 163–164 (2000).
Lin, S.-H. et al. Familial aggregation in skin flush response to niacin patch among schizophrenic patients and their nonpsychotic relatives. Schizophr. Bull. 33, 174–182 (2007).
Chang, S.-S. et al. Impaired flush response to niacin skin patch among schizophrenia patients and their nonpsychotic relatives: the effect of genetic loading. Schizophr. Bull. 35, 213–221 (2009).
Lien, Y.-J. et al. A genome-wide quantitative linkage scan of niacin skin flush response in families with schizophrenia. Schizophr. Bull. 39, 68–76 (2013).
Gottesman, I. I. & Gould, T. D. The endophenotype concept in psychiatry: etymology and strategic intentions. Am. J. Psychiatry 160, 636–645 (2003).
Stern, R. H., Spence, J. D., Freeman, D. J. & Parbtani, A. Tolerance to nicotinic acid flushing. Clin. Pharmacol. Ther. 50, 66–70 (1991).
Glen, A. I. M. et al. Membrane fatty acids, niacin flushing and clinical parameters. Prostaglandins Leukot. Essent. Fatty Acids 55, 9–15 (1996).
Tavares, H., Yacubian, J., Talib, L. L., Barbosa, N. R. & Gattaz, W. F. Increased phospholipase A2 activity in schizophrenia with absent response to niacin. Schizophr. Res. 61, 1–6 (2003).
Nilsson, B. M., Hultman, C. M. & Ekselius, L. Test-retest stability of the oral niacin test and electrodermal activity in patients with schizophrenia. Prostaglandins Leukot. Essent. Fatty Acids 81, 367–372 (2009).
Kamanna, V. S., Ganji, S. H. & Kashyap, M. L. The mechanism and mitigation of niacin-induced flushing. Int. J. Clin. Pract. 63, 1369–1377 (2009).
Andersson, R. G., Aberg, G., Brattsand, R., Ericsson, E. & Lundholm, L. Studies on the mechanism of flush induced by nicotinic acid. Acta Pharmacol. Toxicol. 41, 1–10 (1977).
Benyó, Z., Gille, A., Bennett, C. L., Clausen, B. E. & Offermanns, S. Nicotinic acid-induced flushing is mediated by activation of epidermal Langerhans cells. Mol. Pharmacol. 70, 1844–1849 (2006).
Tang, Y. et al. Enhancement of arachidonic acid signaling pathway by nicotinic acid receptor HM74A. Biochem. Biophys. Res. Commun. 345, 29–37 (2006).
Korbecki, J., Baranowska-Bosiacka, I., Gutowska, I. & Chlubek, D. The effect of reactive oxygen species on the synthesis of prostanoids from arachidonic acid. J. Physiol. Pharmacol. 64, 409–421 (2013).
Horrobin, D. F., Glen, A. I. & Vaddadi, K. The membrane hypothesis of schizophrenia. Schizophr. Res. 13, 195–207 (1994).
Horrobin, D. F. The membrane phospholipid hypothesis as a biochemical basis for the neurodevelopmental concept of schizophrenia. Schizophr. Res. 30, 193–208 (1998).
Nadalin, S., Buretic-Tomljanovic, A., Rubesa, G., Tomljanovic, D. & Gudelj, L. Niacin skin flush test: a research tool for studying schizophrenia. Psychiatr. Danub. 22, 14–27 (2010).
Skosnik, P. D. & Yao, J. K. From membrane phospholipid defects to altered neurotransmission: is arachidonic acid a nexus in the pathophysiology of schizophrenia? Prostaglandins Leukot. Essent. Fatty. Acids 69, 367–384 (2003).
Law, M. H., Cotton, R. G. H. & Berger, G. E. The role of phospholipases A2 in schizophrenia. Mol. Psychiatry 11, 547–556 (2006).
Junqueira, R., Cordeiro, Q., Meira-Lima, I., Gattaz, W. F. & Vallada, H. Allelic association analysis of phospholipase A2 genes with schizophrenia. Psychiatr. Genet. 14, 157–160 (2004).
Yu, Y. et al. A genetic study of two calcium-independent cytosolic PLA2 genes in schizophrenia. Prostaglandins Leukot. Essent. Fat. Acids 73, 351–354 (2005).
Nadalin, S. et al. BanI polymorphism of cytosolic phospholipase A2 gene is associated with age at onset in male patients with schizophrenia and schizoaffective disorder. Prostaglandins Leukot. Essent. Fatty Acids 78, 351–360 (2008).
Nadalin, S. & Buretić-Tomljanović, A. An association between PLA2G6 and PLA2G4C gene polymorphisms and schizophrenia risk and illness severity in a Croatian population. Prostaglandins Leukot. Essent. Fatty Acids 121, 57–59 (2017).
Tao, R. et al. Cytosolic PLA2 genes possibly contribute to the etiology of schizophrenia. Am. J. Med. Genet. B Neuropsychiatr. Genet. 137b, 56–58 (2005).
Nadalin, S., Radović, I. & Buretić-Tomljanović, A. Polymorphisms in PLA2G6 and PLA2G4C genes for calcium-independent phospholipase A2 do not contribute to attenuated niacin skin flush response in schizophrenia patients. Prostaglandins Leukot. Essent. Fatty Acids 100, 29–32 (2015).
Nadalin, S., Giacometti, J., Jonovska, S., Tomljanovic, D. & Buretic-Tomljanovic, A. The impact of PLA2G4A and PTGS2 gene polymorphisms, and red blood cell PUFAs deficit on niacin skin flush response in schizophrenia patients. Prostaglandins Leukot. Essent. Fatty Acids 88, 185–190 (2013).
van der Kemp, W. J. M., Klomp, D. W. J., Kahn, R. S., Luijten, P. R. & Hulshoff Pol, H. E. A meta-analysis of the polyunsaturated fatty acid composition of erythrocyte membranes in schizophrenia. Schizophr. Res. 141, 153–161 (2012).
Hoen, W. P. et al. Red blood cell polyunsaturated fatty acids measured in red blood cells and schizophrenia: a meta-analysis. Psychiatry Res. 207, 1–12 (2013).
Lattka, E., Illig, T., Koletzko, B. & Heinrich, J. Genetic variants of the FADS1 FADS2 gene cluster as related to essential fatty acid metabolism. Curr. Opin. Lipidol. 21, 64–69 (2010).
Maclean, R., Ward, P. E., Glen, I., Roberts, S. J. & Ross, B. M. On the relationship between methylnicotinate-induced skin flush and fatty acids levels in acute psychosis. Prog. Neuropsychopharmacol. Biol. Psychiatry 27, 927–933 (2003).
Messamore, E., Hoffman, W. F. & Yao, J. K. Niacin sensitivity and the arachidonic acid pathway in schizophrenia. Schizophr. Res. 122, 248–256 (2010).
Bentsen, H. et al. Bimodal distribution of polyunsaturated fatty acids in schizophrenia suggests two endophenotypes of the disorder. Biol. Psychiatry 70, 97–105 (2011).
Horváth, S. & Mirnics, K. Immune system disturbances in schizophrenia. Biol. Psychiatry 75, 316–323 (2014).
Bergink, V., Gibney, S. M. & Drexhage, H. A. Autoimmunity, inflammation, and psychosis: a search for peripheral markers. Biol. Psychiatry 75, 324–331 (2014).
Coutinho, E., Harrison, P. & Vincent, A. Do neuronal autoantibodies cause psychosis? A neuroimmunological perspective. Biol. Psychiatry 75, 269–275 (2014).
Pathmanandavel, K. et al. Antibodies to surface dopamine-2 receptor and N-methyl-D-aspartate receptor in the first episode of acute psychosis in children. Biol. Psychiatry 77, 537–547 (2015).
Jézéquel, J., Johansson, E. M., Leboyer, M. & Groc, L. Pathogenicity of antibodies against NMDA receptor: molecular insights into autoimmune psychosis. Trends Neurosci. 41, 502–511 (2018).
Carter, C. S., Bullmore, E. T. & Harrison, P. Is there a flame in the brain in psychosis? Biol. Psychiatry 75, 258–259 (2014).
Leza, J. C. et al. Inflammation in schizophrenia: a question of balance. Neurosci. Biobehav. Rev. 55, 612–626 (2015).
Xu, C. et al. An investigation of calcium-independent phospholipase A2 (iPLA2) and cytosolic phospholipase A2 (cPLA2) in schizophrenia. Psychiatry Res. 273, 782–787 (2019).
Talib, L. L. et al. Increased PLA(2) activity in individuals at ultra-high risk for psychosis. Eur. Arch. Psychiatry Clin. Neurosci. 271, 1593–1599 (2021).
Sanchez-Mejia, R. O. & Mucke, L. Phospholipase A2 and arachidonic acid in Alzheimer’s disease. Biochim. Biophys. Acta 1801, 784–790 (2010).
Sun, G. Y. et al. Docosahexaenoic acid (DHA): an essential nutrient and a nutraceutical for brain health and diseases. Prostaglandins Leukot. Essent. Fat. Acids 136, 3–13 (2018).
Dipasquale, S. et al. The dietary pattern of patients with schizophrenia: a systematic review. J. Psychiatr. Res. 47, 197–207 (2013).
Peet, M., Shah, S., Selvam, K. & Ramchand, C. N. Polyunsaturated fatty acid levels in red cell membranes of unmedicated schizophrenic patients. World J. Biol. Psychiatry 5, 92–99 (2004).
Arvindakshan, M. et al. Essential polyunsaturated fatty acid and lipid peroxide levels in never-medicated and medicated schizophrenia patients. Biol. Psychiatry 53, 56–64 (2003).
Messamore, E. & Yao, J. K. Phospholipid, arachidonate and eicosanoid signaling in schizophrenia. Oilseeds Fats Crops Lipids 23, D112 (2016).
Kim, E. J. et al. Low dietary intake of n-3 fatty acids, niacin, folate, and vitamin C in Korean patients with schizophrenia and the development of dietary guidelines for schizophrenia. Nutr. Res. 45, 10–18 (2017).
Yao, J. K. & Reddy, R. Oxidative stress in schizophrenia: pathogenetic and therapeutic implications. Antioxid. Redox Signal. 15, 1999–2002 (2011).
Cadenhead, K. et al. Omega-3 fatty acid versus placebo in a clinical high-risk sample from the North American Prodrome Longitudinal Studies (NAPLS) Consortium. Schizophr. Bull. 43, S16–S16 (2017).
Berger, G. E. et al. Ethyl-eicosapentaenoic acid in first-episode psychosis: a randomized, placebo-controlled trial. J. Clin. Psychiatry 68, 1867–1875 (2007).
Pawelczyk, T., Grancow-Grabka, M., Kotlicka-Antczak, M., Trafalska, E. & Pawełczyk, A. A randomized controlled study of the efficacy of six-month supplementation with concentrated fish oil rich in omega-3 polyunsaturated fatty acids in first episode schizophrenia. J. Psychiatr. Res. 73, 34–44 (2016).
Hsu, M. C. & Ouyang, W. C. A systematic review of effectiveness of omega-3 fatty acid supplementation on symptoms, social functions, and neurobiological variables in schizophrenia. Biol. Res. Nurs. 23, 723–737 (2021).
Jones, H. J. et al. Associations between plasma fatty acid concentrations and schizophrenia: a two-sample Mendelian randomisation study. Lancet Psychiatry 8, 1062–1070 (2021).
Berger, G. E. et al. Niacin skin sensitivity is increased in adolescents at ultra-high risk for psychosis. PLoS ONE 11, e0148429 (2016).
Morrison, A. P. et al. Early detection and intervention evaluation for people at risk of psychosis: multisite randomised controlled trial. BMJ 344, e2233 (2012).
McFarlane, W. R. et al. Clinical and functional outcomes after 2 years in the early detection and intervention for the prevention of psychosis multisite effectiveness trial. Schizophr. Bull. 41, 30–43 (2015).
McGorry, P. D. et al. Effect of ω-3 polyunsaturated fatty acids in young people at ultrahigh risk for psychotic disorders: the NEURAPRO randomized clinical trial. JAMA Psychiatry 74, 19–27 (2017).
Davies, C. et al. Lack of evidence to favor specific preventive interventions in psychosis: a network meta-analysis. World Psychiatry 17, 196–209 (2018).
Amminger, G. P. et al. Long-chain ω-3 fatty acids for indicated prevention of psychotic disorders: a randomized, placebo-controlled trial. Arch. Gen. Psychiatry 67, 146–154 (2010).
Wang, X., Lin, H. & Gu, Y. Multiple roles of dihomo-γ-linolenic acid against proliferation diseases. Lipids Health Dis. 11, 25–25 (2012).
Aasheim, E. T., Hofsø, D., Hjelmesœth, J., Birkeland, K. I. & Bøhmer, T. Vitamin status in morbidly obese patients: a cross-sectional study. Am. J. Clin. Nutr. 87, 362–369 (2008).
Fraguas, D. et al. Oxidative stress and inflammation in first-episode psychosis: a systematic review and meta-analysis. Schizophr. Bull. 45, 742–751 (2019).
Henderson, D. C. et al. Dietary intake profile of patients with schizophrenia. Ann. Clin. Psychiatry 18, 99–105 (2006).
Amani, R. Is dietary pattern of schizophrenia patients different from healthy subjects? BMC Psychiatry 7, 15 (2007).
Strassnig, M., Brar, J. S. & Ganguli, R. Dietary fatty acid and antioxidant intake in community-dwelling patients suffering from schizophrenia. Schizophr. Res. 76, 343–351 (2005).
Pryor, W. A. & Stone, K. Oxidants in cigarette smoke: radicals, hydrogen peroxide, peroxynitrate, and peroxynitrite. Ann. NY Acad. Sci. 686, 12–27 (1993).
Zhang, X. Y. et al. Nicotine dependence, symptoms and oxidative stress in male patients with schizophrenia. Neuropsychopharmacology 32, 2020–2024 (2007).
Marin, P., Hamon, B., Glowinski, J. & Prémont, J. Nicotine-induced inhibition of neuronal phospholipase A2. J. Pharmacol. Exp. Ther. 280, 1277 (1997).
Sastry, B. V. R. & Hemontolor, M. E. Influence of nicotine and cotinine on retinal phospholipase A2 and its significance to macular function. J. Ocul. Pharmacol. Ther. 14, 447–458 (1998).
Lai, C.-Y. et al. Aberrant expression of microRNAs as biomarker for schizophrenia: from acute state to partial remission, and from peripheral blood to cortical tissue. Transl. Psychiatry 6, e717 (2016).
Chen, W. J. et al. Sustained attention deficit and schizotypal personality features in nonpsychotic relatives of schizophrenic patients. Am. J. Psychiatry 155, 1214–1220 (1998).
Nurnberger, J. I. Jr. et al. Diagnostic interview for genetic studies: rationale, unique features, and training. Arch. Gen. Psychiatry 51, 849–859 (1994).
Kay, S. R. Positive and Negative Syndromes in Schizophrenia: Assessment and Research (Brunner/Mazel, 1991).
Cheng, J. J., Ho, H., Chang, C. J., Lan, S. Y. & Hwu, H. G. Positive and Negative Syndrome Scale (PANSS): establishment and reliability study of a Mandarin Chinese language version. Chinese Psychiatry 10, 251–258 (1996).
Liu, S. K., Hwu, H. G. & Chen, W. J. Clinical symptom dimensions and deficits on the Continuous Performance Test in schizophrenia. Schizophr. Res. 25, 211–219 (1997).
Ward, P. E., Sutherland, J., Glen, E. M. T. & Glen, A. I. M. Niacin skin flush in schizophrenia: a preliminary report. Schizophr. Res. 29, 269–274 (1998).
Liu, C.-M. et al. Absent response to niacin skin patch is specific to schizophrenia and independent of smoking. Psychiatry Res. 152, 181–187 (2007).
Moser, A. B., Jones, D. S., Raymond, G. V. & Moser, H. W. Plasma and red blood cell fatty acids in peroxisomal disorders. Neurochem. Res. 24, 187–197 (1999).
Acknowledgements
The authors thank Ms. Ching-Ing Tseng for her help in handling the blood specimen. This study was supported by grants from the National Science Council of Taiwan (NSC98-2314-B-002-125-MY3 to W.J.C.) and Ministry of Education, Taiwan (“Aim for the Top University Project” to National Taiwan University, 2011-2017 to W.J.C.). None of the funding sources played any role in the study design; collection, analysis, or interpretation of data; in the writing of the report; or in the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
W.J.C. designed the study. Y.-H.Y. managed participant recruitment and data acquisition, compiled the database, and conducted data preprocessing. H.-M.S. and Y.-H.Y. undertook the fatty acid analysis. Y.-H.Y., S.-H.L., and P.-C.H. undertook the statistical analysis. Y.-T.L., C.-M.L., T.-J.H., M.H.H., C.-C.L., Y.-L.C., C.-J.K., and H.-G.H. managed participant recruitment and data acquisition. Y.-H.Y. and W.J.C. implemented the literature reviews and data interpretation. Y.-H.Y. wrote the first draft of the manuscript, and W.J.C. provided critical revisions and supervised the manuscript. All authors contributed to and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, YH., Su, HM., Lin, SH. et al. Niacin skin flush and membrane polyunsaturated fatty acids in schizophrenia from the acute state to partial remission: a dynamic relationship. Schizophr 8, 38 (2022). https://doi.org/10.1038/s41537-022-00252-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-022-00252-w
This article is cited by
-
Patients with first-episode psychosis in northern Taiwan: neurocognitive performance and niacin response profile in comparison with schizophrenia patients of different familial loadings and relationship with clinical features
BMC Psychiatry (2024)
-
Attenuated niacin response is associated with a subtype of first-episode drug-naïve psychosis characterized as serious negative symptoms
European Archives of Psychiatry and Clinical Neuroscience (2023)