Abstract
Asthma is closely associated with inflammation. We evaluated the predictive and prognostic value of leptin status in asthma. We searched the electronic databases for articles that determined the leptin level in asthma cases through May 2020. We compared the differences of leptin level between asthma and non-asthma controls, as well as between severe and mild asthma cases. We also investigated the impact of age and gender on these differences by using meta-regression analysis. 59 studies were included in our pooled analysis. Asthma cases demonstrated significantly higher leptin level than that in non-asthma controls among overall populations (SMD:1.061, 95% CI: 0.784–1.338, p < 10−4), Caucasians (SMD:0.287, 95% CI: 0.125–0.448, p = 0.001), Asians (SMD:1.500, 95% CI: 1.064–1.936, p < 10−4) and Africans (SMD: 8.386, 95% CI: 6.519–10.253, p < 10−4). Severe asthma cases showed markedly higher leptin level than that in mild asthma cases among overall populations (SMD:1.638, 95% CI: 0.952–2.323, p < 10–4) and Asians (SMD:2.600, 95% CI: 1.854–3.345, p < 10–4). No significant difference of leptin level between severe and mild asthma was observed in Caucasians (SMD:−0.819, 95% CI: −1.998–0.360, p = 0.173). Cumulative analyses yielded similar results regarding the difference of leptin status between asthma and non-asthma controls, as well as between severe and mild asthma cases among overall populations. Age and male/ female ratio were not associated with the difference of leptin status between asthma and non-asthma controls (coefficient:−0.031, 95% CI: −0.123–0.061, p = 0.495; coefficient:0.172, 95% CI: −2.445–2.789, p = 0.895), as well as between severe and mild asthma cases among overall populations (coefficient:−0.072, 95% CI: −0.208–0.063, p = 0.279; coefficient: 2.373, 95% CI: −0.414–5.161, p = 0.090). Asthma demonstrated significantly higher level of leptin than that in non-asthma controls among overall populations, Caucasians, Asians and Africans. Severe asthma cases showed markedly higher leptin level than that in mild cases among overall populations and Asians. Leptin may be a risk predictor and prognostic marker of asthma. Early monitoring and intervention of leptin may be needed for asthma.
Similar content being viewed by others
Introduction
Asthma, a common respiratory tract disease, is likely to occur in both children and adults1. Frequent attacks of asthma may lead to irreversible airway obstruction, cardiac events, and even death2. In terms of the morbidity and mortality of asthma, early prevention and monitoring of asthma seems imperative. The past decades witnessed an increasing trend of asthma prevalence across the world due to many factors, such as environmental and lifestyle changes3. Allergy and inflammation are well-documented inducers of asthma, whereas the occurrence and progression of certain asthma cases remained unexplained4. Hence, an in-depth investigation of the potential risk factors for asthma susceptibility and progression is necessary.
Leptin, a hormone secreted by adipocyte, plays a main role in controlling body weight through influencing appetite and energy expenditure5. Obesity cases demonstrated higher level of leptin than that in normal controls, indicating that obesity may be a leptin resistance condition6. Meanwhile, obesity is closely associated with asthma susceptibility7. On the other hand, leptin plays a role in the pro-inflammatory activities, which is closely associated with asthma risk and progression8. Leptin secretion is associated with bronchial hyperresponsiveness and insulin resistance9. Leptin receptor is also expressed in the lung10. In this sense, we speculated that leptin may also be associated with asthma risk and progression.
In the past decades, many studies were performed to determine the leptin levels in asthma cases11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69. The results were not consistent among the studies. Some investigations yielded that leptin status was significantly higher in asthma cases than that in non-asthma controls, whereas some studies showed a null difference of leptin levels between asthma and controls. An improved understanding of this issue has important significance that early monitoring or intervention may lower the risk or progression of asthma. A previous pooled analysis showed that higher level of leptin was associated with asthma70. However, the association between leptin status and asthma progression was not studied.
With the accumulating evidence, we conducted this updated pooled analysis to investigate the predictive and prognostic value of leptin status in asthma, we also studied the influence of age, gender and ethnicities on the differences of leptin status among different groups with the aim of yielding a more robust finding on this issue.
Methods
Search strategy
We searched the papers that tested the leptin levels in asthma cases through May 2020 by using PubMed, Embase, Cochrane and Chinese WanFang databases. No restriction was imposed on the searched language. The used terms were as follows: (1) leptin, adipocyte, adiponectin; and (2) asthma, bronchial asthma, respiratory tract disease. We searched the associated papers by combining these terms. We also reviewed the references of extracted papers. If the same participants were recruited in more than one study, we chose the study with the complete analysis. The participants data were extracted from the public publications, hence the consent was waived. Ethics approval: This study was approved by the institutional review board of Shanghai Sixth People’s Hospital (No: 2018–106).
Inclusion and exclusion criteria
Inclusion criteria: (1) case-control, cohort, prospective or observational study; and (2) asthma as the cases; and (3) leptin status (mean and standard deviation or data to calculate them) available.
Exclusion criteria: (1) case reports, reviews and editorials; (2) levels of other factors in asthma; and (3) detailed leptin level was not available and multiple publications of the same data.
Data extraction and synthesis
We extracted the characteristics from each recruited study. The data were recorded as the following: first author’s family name, publication year, ethnicity of participants, study design, gender, number of asthma cases and controls, leptin levels, and adjustment for covariates. The criteria for the definition of severe and mild asthma was not totally same among the recruited studies. Severe asthma was defined as the continuous use of inhaled steroids and bronchodilators, and mild asthma as the intermittent use of inhaled steroids or bronchodilators in the majority of enrolled studies. On the other hand, controlled and uncontrolled asthma were defined as severe and mild asthma, respectively. In a word, the severity of asthma depends on the treatment response and dependence across the included studies. We also evaluated the quality of each recruited study using Newcastle-Ottawa Quality Assessment Scale, which included the assessment for participants selection, exposure and comparability. A study can be awarded a maximum of one score for each numbered item within the selection and exposure categories. A maximum of two scores can be given for comparability71. Two authors conducted the literature search independently, study selection, quality assessment and data extraction with any disagreements resolved by discussion.
Statistical analysis
Standard mean difference (SMD) was used to measure the differences of leptin levels between asthma and non-asthma controls, as well as severe and mild asthma cases across the recruited studies. Heterogeneity of SMDs across the studies was tested by using the Q statistic (significance level at p < 0.05). The I2 statistic, a quantitative measure of inconsistency across studies, was also calculated. The combined SMDs were calculated using a fixed-effects model, or, in the presence of heterogeneity, random-effects model. In addition, 95% confidence intervals (CIs) were also calculated. We evaluated the influence of a single study on the pooled SMDs by excluding one study in each turn. Subgroup analyses were conducted according to the ethnicity. Meta-regression analyses were performed to investigate the influence of age and gender on the SMDs between asthma and controls, and as well as between severe and mild asthma. Potential publication bias was assessed by Egger’s test and Begg rank correlation test at the p < 0.05 level of significance. All analyses were performed using STATA version 12.0 (Stata Corp, College Station, TX). P < 0.05 was considered statistically significant, except where otherwise specified.
Results
Literature search
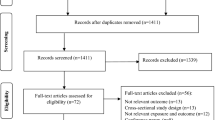
We initially extracted 417 relevant publications from the PubMed, Embase, Cochrane and Chinese WanFang databases. Of these, 358 studies were excluded according to the inclusion and exclusion criteria, 59 articles11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69 were included in our final meta- analysis (Fig. 1). The retrieved data were recorded as follows: first author’s surname, publication year, ethnicity, study design, gender (male/female ratio), age, the number of severe asthma, mild asthma, and non-asthma controls. A flow chart showing the study selection is presented in Fig. 1.
Characteristics for included studies
51 studies were identified for the analysis of the differences of leptin levels between asthma and non-asthma controls. 25 studies were performed for the analysis of the differences of leptin levels between severe and mild asthma. These studies were published between 2004 and 2019. Twenty-one studies were conducted in Caucasians, thirty-seven in Asians, and one in Africans. Forty-nine studies were case-control design, six for cross-sectional design, and four for cohort. A total of 1044 severe asthma, 2536 mild asthma and 7176 non-asthma controls. The number of awarded scores of included studies ranged from 4 to 6. Thirty-one studies were awarded for four scores, twenty-five for five scores and three for six scores. As shown in Table 1.
Differences of leptin levels between asthma and controls
Asthma cases demonstrated significantly higher leptin level than that in non-asthma controls among overall populations (SMD: 1.061, 95% CI: 0.784–1.338, p < 10–4), Caucasians (SMD: 0.287, 95% CI: 0.125–0.448, p = 0.001), Asians (SMD: 1.500, 95% CI: 1.064–1.936, p < 10−4) and Africans (SMD: 8.386, 95% CI: 6.519–10.253, p < 10−4) (Table 2, Fig. 2). Significant heterogeneity was observed using Q and I2 statistic for overall populations (p < 10−4, I2 = 94.1%), Caucasians (p = 0.005, I2 = 54.7%) and Asians (p < 10−4, I2 = 95.2%). Exclusion of any single study did not change the overall SMDs for overall populations (95% CI: 0.727–1.455), Caucasians (95% CI: 0.085–0.502) and Asians (95% CI: 0.829–2.014) (Table 2). Cumulative analysis indicated that leptin status was significantly higher in asthma cases than that in non-asthma controls among overall populations (Fig. 3).
Differences of leptin levels between severe asthma and mild asthma
Severe asthma cases showed markedly high leptin level than that in mild asthma cases among overall populations (SMD: 1.638, 95% CI: 0.952–2.323, p < 10−4) and Asians (SMD: 2.600, 95% CI: 1.854–3.345, p < 10−4) (Table 2, Fig. 4). No significant difference of leptin level between severe and mild asthma was observed in Caucasians (SMD: −0.819, 95% CI: −1.998–0.360, p = 0.173) (Table 2, Fig. 4). Significant heterogeneity was observed using Q and I2 statistic for overall populations (p < 10−4, I2 = 96.7%), Caucasians (p < 10−4, I2 = 96.5%) and Asians (p < 10−4, I2 = 95.7%). Exclusion of any single study did not change the overall SMDs for overall populations (95% CI: 0.682–2.568), Caucasians (95% CI: −2.572–0.692) and Asians (95% CI: 1.548–3.528) (Table 2). Cumulative analysis indicated that leptin status was significantly higher in severe asthma cases than that in mild asthma cases among overall populations (Fig. 5).
Meta-regression analysis of the age/gender in the association between leptin status and asthma risk/progression
Age and male/female ratio were not associated with the differences of leptin status between asthma and non-asthma controls among overall populations (coefficient: −0.031, 95% CI: −0.123 to 0.061, p = 0.495; coefficient: 0.172, 95% CI: −2.445 to 2.789, p = 0.895) (Table 3). Age and male/female ratio were not associated with the differences of leptin status between severe and mild asthma cases among overall populations (coefficient: −0.072, 95% CI: −0.208 to 0.063, p = 0.279; coefficient: 2.373, 95% CI: −0.414 to 5.161, p = 0.090) (Table 3).
Publication bias
The Begg rank correlation test and Egger linear regression test indicated no significant publication bias among Caucasians in the difference of leptin status between asthma and non-asthma controls (Begg, p = 0.65; Egger, p = 0.994). The Begg rank correlation test and Egger linear regression test showed marked publication bias among Asians in the difference of leptin status between asthma and non-asthma controls (Begg, p < 10−4; Egger, p < 10−4). The Begg rank correlation test and Egger linear regression test indicated no marked publication bias among Caucasians in the difference of leptin status between severe and mild asthma cases (Begg, p = 0.230; Egger, p = 0.054). The Begg rank correlation test and Egger linear regression test showed marked publication bias among Asians in the difference of leptin status between severe and mild asthma cases (Begg, p = 0.002; Egger, p = 0.003).
Discussion
Increasing attention has been paid to the potential role of leptin in the development and progression of asthma. Our pooled analysis showed that asthma cases had markedly higher leptin level than that in non-asthma controls among overall populations, Caucasians, Asians and Africans, and severe asthma cases had significantly higher leptin level than that in mild asthma cases among overall populations and Asians. Age and gender did not influence the association between leptin level and asthma risk/progression. Our results indicated that leptin dysregulation may be associated with asthma risk/progression, frequent monitoring and early intervention of leptin status may be helpful for asthma prevention and therapy.
Several mechanisms may explain the association between leptin status and asthma risk/progression. First, asthma was essentially the breathing problems induced by airway narrowing and obstruction, which was exacerbated by the inflammation72. Inflammation was positively associated with the severity of asthma. Systemic inflammation acted as a mechanism linking insulin resistance with asthma73. Leptin showed pro-inflammatory actions, stimulating the production of inflammatory cytokines in bronchial and alveolar cells74. Persistent stimulation of inflammation may induce the injury and fibrosis of airway, increasing the susceptibility and progress of asthma. Meanwhile, leptin played a role in the regulation of T cell proliferation and activation, monocytes/macrophages recruitment, exerting effects in airway inflammation, respiratory diseases and immune system75. In this sense, leptin increased the inflammatory response through various ways, leptin may increase the risk and severity of asthma through activating the inflammation. Second, obesity was a risk factor for asthma susceptibility, and some immune changes present in asthma cases were augmented in obese asthmatics76. Meanwhile, obesity was closely associated with an obstructive pattern induced by disproportionated growth between lung parenchyma size and airway caliber, which led to a reduced lung function. Weight loss may lead to an improvement in lung function, airway reactivity and asthma control. Leptin, an adipocyte-derived hormone produced by white fat tissue in the conditions of excessive caloric intake, played a role in controlling body weight by influencing appetite and energy expenditure77. Leptin level was higher in obese than that in the normal weight cases, which means that obesity may be a leptin resistance condition. In terms of the close relationship between leptin and obesity, it was reasonable to predict that high level of leptin may increase the risk and severity of asthma through its interaction with obesity. Regrettably, the lack of detailed data of obesity and BMI made it unfeasible to study the influence of obesity/BMI on the association between leptin status and asthma. Further studies should be performed on this issue. Finally, leptin is also expressed in the lung and produced by the lung fibroblasts during alveolar differentiation, promoting the synthesis of surfactant protein78. Leptin plays a direct role in the lung development and remodeling, indicating that leptin disorder may affect the lung pulmonary homeostasis79. Lepin may influence the lung function, Which was consistent with our findings that leptin staus was higher in the asthma cases compared with non-asthma controls, as well as in severe asthma compared with mild asthma cases. In this sense, it is reasonable to predict that the pulmonary function may be influenced by leptin dysregulation.
Our findings for the association between leptin levels and asthma risk/progression were consistent with the above-mentioned evidence. It indicated that leptin may be a risk predictor and prognostic marker of asthma independent of age and gender. Asthma showed significantly higher leptin level than that in non-asthma controls, which might be due to the effects of leptin in the inflammation, obesity and lung development. Notably, we found that no marked difference of leptin level was observed between severe and mild asthma among Caucasians, indicating that leptin was not associated with asthma progression among Caucasians. We speculated that it may be due to the facts that Caucasians were more prone to be obese than other populations, and obesity may be associated with high level of leptin. It may lead to the comparatively similar leptin level between severe and mild asthma. On the other hand, only seven studies were recruited for the analysis of the difference of leptin level between severe and mild asthma among Caucasians, which may reduce the statistical power. Further larger number of participants should be involved in the future studies to verify our findings. Nevertheless, no marked publication bias was observed in the studies regarding the difference of leptin level between severe and mild asthma among Caucasians, which indicated that our finding was comparatively robust. Interestingly, we found that age and gender did not affect the differences of leptin levels between asthma and non-asthma, as well as severe and mild asthma, which indicated that leptin status was associated with asthma risk/progression independent of age and gender. Early monitoring and intervention of leptin level may be of great clinical implications.
Our study has obvious strengths. For example, the enrolled subjects were from different regions and the quality of the included studies was comparatively high, which increased the statistical power and promoted the generalization of our conclusions, which made the risk prediction for asthma susceptibility and progression possible. On the other hand, the analysis of the potential role of age and gender in the association between leptin status and asthma also provided a comparatively robust conclusion. Meanwhile, several limitations merited attention in our pooled analysis. First, the heterogeneities among included studies might affect the results of our investigation, although a random-effects model had been performed. Publication bias was also observed. Nevertheless, the sensitivity analyses did not change the overall results, cumulative analyses also showed a similar trend to our results and meta- regression also excluded the possibility of the influence of age and gender in our results, which proved that our conclusions were comparatively solid. Second, the study design of recruited paper were mainly case-control, which may lead to the recall bias, the disease course and medications may also affect the results. Due to the limit of available data, the in-depth analysis was not performed. Hence, further larger number, prospective studies with controlling confounding factors should be performed in the future. Third, obesity and BMI may influence the leptin level, higher leptin level was usually observed in obesity and high-BMI cases. Many asthma cases were obese than non-asthma controls, and obesity was also a risk factor for asthma susceptibility and progress. We also found that asthma cases had higher level of BMI in some of the included studies, while there were no differences of obesity ratio and BMI between asthma and controls in some of enrolled participants. The unavailable detailed data of BMI and obesity made it not possible to perform the in-depth influence of obesity and BMI on the association between leptin level and asthma. Nevertheless, our findings still had important implications that leptin level may be an auxiliary indicator for asthma susceptibility and progress due to the facts the some severe asthma cases were not obese and comprehensive analysis of multiple factors may be a better choice. Meanwhile, further multiple regression analysis involving multiple risk factors for asthma susceptibility and progress may needed in the future.
Finally, although a total of 59 studies were included in our studies, the number of studies regarding the difference of leptin level between severe and mild asthma among Caucasians was relatively small, which may decrease the statistical power. Larger number of participants with different ethnicities should be involved in the further studies to verify our findings.
In terms of our findings, further investigations may be performed to focus on the following issues: (1) elucidation of the detailed mechanism behind leptin and asthma risk/progression, (2) in-depth analysis of the association of disease course and medications with leptin status, (3) long-term, continuous observation of the changes of leptin status in asthma with a favorable study design.
Conclusion
Our study indicated that asthma had significantly higher level of leptin than that in non-asthma controls among overall populations, Caucasians, Asians and Africans. Severe asthma cases showed markedly higher leptin level than that in mild cases among overall populations and Asians. Our findings were of great implications that leptin may be a risk predictor and prognostic marker of asthma. Early monitoring and intervention of leptin may be needed for asthma.
Data availability
The data was extracted from the public databases, including PubMed, Embase, Cochrane and Chinese WanFang databases. The readers can obtain the data from these public databases. The extracted references articles were all in the reference list of the manuscript. Furthermore, the data will also be shared by the corresponding author upon the scientific request of the readers.
References
Boulet, L. P. & Boulay, M. È. Asthma-related comorbidities. Expert Rev. Respir. Med. 5, 377–393 (2011).
Boulet, L. P. et al. Asthma with irreversible airway obstruction in smokers and nonsmokers: Links between airway inflammation and structural changes. Respiration. 8, 1–11 (2020).
Huang, S. et al. Home environmental and lifestyle factors associated with asthma, rhinitis and wheeze in children in Beijing, China. Environ. Pollut. 256, 113426 (2020).
Russell, R. J. & Brightling, C. Pathogenesis of asthma: Implications for precision medicine. Clin. Sci. (Lond) 131, 1723–1735 (2017).
Olea-Flores, M. et al. 12. New actors driving the epithelial-mesenchymal transition in cancer: The role of leptin. Biomolecules. 10, 1676 (2020).
Kumar, R. et al. Association of leptin with obesity and insulin resistance. Cureus. 12, e12178 (2020).
Peters, U., Dixon, A. E. & Forno, E. Obesity and asthma. J. Allergy Clin. Immunol. 141, 1169–1179 (2018).
Mims, J. W. Asthma: Definitions and pathophysiology. Int. Forum Allergy Rhinol. 5, S2–S6 (2015).
Baek, H. S., Choi, J. H., Oh, J. W. & Lee, H. B. Leptin and urinary leukotriene E4 and 9alpha, 11beta- prostaglandin F2 release after exercise challenge. Ann. Allergy Asthma Immunol. 111, 112–117 (2013).
Jutant, E. M., Tu, L., Humbert, M., Guignabert, C. & Huertas, A. The Thousand Faces of Leptin in the Lung. Chest. 159, 239–248 (2021).
Zhu, H. M., Tang, H. P., Liu, J. & Lu, W. X. The association between serum interleukin-17 and leptin in obese asthma patients. Contemporary Med. 17, 10–11 (2013).
Bian, F. F., Zhang, C. L., Zhen, Q. & Qu, C. X. Correlation of leptin and IL-17 with asthma predicting index in infants with wheeze. J. Clin. Pulm. Med. 21, 1853–1855 (2016).
Zhang, H. P., Pang, S. J., Li, Y. X., Feng, N. & Wang, C. F. Correlation of serum leptin, simple obesity and childhood asthma. Anhui Med. J. 33, 293–295 (2012).
Peng, F., Wu, S. Y. & Liu, Z. M. Influence of leptin on Th1/Th2 balance in obese children with asthma. Chinese J. Immunol. 7, 968–971 (2014).
Li, Y. X. & Ji, X. Associations of leptin and leptin receptor mRNA expression to asthma and obesity. J. Clin. Pulm. Med. 7, 1175–1179 (2014).
Gao, W. W. et al. Relationships between leptin lvels and the degree of asthma control. J. Chengde Med. College 33, 367–369 (2016).
Li, J., Weng, Y. Q., Zeng, L. H. & Zeng, F. R. Research on leptin in the pathogenesis of obese adult asthma. China Med. Herald 15, 55–58 (2018).
Zhao, J. H., Hong, F. & Kong, W. Clinical significances of serum leptin and IL-17 status in children with asthma. Guangxi Med. J. 9, 1309–1310 (2014).
Xu, Q. L. et al. The significance of peripheral blood CD4+ T cell-derived leptin in asthmatic children. J. Clin. Pediatr. 2, 122–125 (2014).
Xiao, C. Y., Hou, J. H. & Chen, J. J. Detection and analysis of leptin level in asthma children. ShenZhen J. Integr. Traditional Chinese Western Med. 19, 158–159 (2009).
Li, Y. C. et al. Association between BMI and leptin level in asthma. Heilongjiang Med. Pharm. 37, 63–64 (2014).
Zhang, H. P., Li, Y. X., Pang, S. J., Feng, N. & Wang, C. F. The expression and clinical significance of the serum leptin、eotaxin and TIgE in asthmatic infant and young children. Shananxi Med. J. 42, 99–101 (2013).
Pan, J. W. Effect of long-term inhalation of glucocorticoids on the level of leptin, IL-13 and IL-2 in bronchial asthmatic children. J. Radioimmunol. 24, 369–372 (2011).
Zhang, J., Chen, S. Z., Lou, S. Z., Xu, C. H. & Wang, C. Y. The Clinical significances of serum leptin in the acute phase of asthma in children. Chinese J. Crit. Care Med. 3, 205–206 (2014).
Yang, M. & Chen, Y. P. Association between serum leptin level and disease severity of asthma in children. J. Clin. Emerg. 1, 27–29 (2014).
Liang, Y. et al. Analysis of correlative factors of serum leptin levels in asthmatic patients. Natl. Med. J. China 96, 2889–2992 (2016).
He, S. W., Zhu, S. Y., Liu, P. L. & Zheng, Y. L. Relationship between serum leptin levels in patients with severe asthma and asthma severity. Shandong Med. J. 52, 20–22 (2012).
Baek, H. S. et al. Serum leptin and adiponectin levels correlate with exercise-induced bronchoconstriction in children with asthma. Ann. Allergy Asthma Immunol. 107, 14–21 (2011).
Li, Z. et al. Role of Leptin in the Association Between Body Adiposity and Persistent Asthma: A Longitudinal Study. Obesity (Silver Spring) 27, 894–898 (2019).
Bodini, A. et al. Serum and exhaled breath condensate leptin levels in asthmatic and obesity children: a pilot study. J. Breath Res. 11, 046005 (2017).
Nasiri Kalmarzi, R. et al. Serum levels of adiponectin and leptin in asthmatic patients and its relation with asthma severity, lung function and BMI. Allergol. Immunopathol. (Madr) 45, 258–264 (2017).
Al-Ayed, M. et al. Obesity and childhood asthma in male schoolchildren in Saudi Arabia: Is there a role for leptin, interleukin-4, interleukin-5, and interleukin-21? Ann. Saudi Med. 39, 295–301 (2019).
Sood, A., Ford, E. S. & Camargo, C. A. Jr Association between leptin and asthma in adults. Thorax. 61, 300–305 (2006).
Szczepankiewicz, D. et al. Leptin gene polymorphism affects leptin level in childhood asthma. World J. Pediatr. 14, 601–606 (2018).
Huang, F. et al. Adipokines, asymmetrical dimethylarginine, and pulmonary function in adolescents with asthma and obesity. J. Asthma 54, 153–161 (2017).
Coffey, M. J., Torretti, B. & Mancuso, P. Adipokines and Cysteinyl Leukotrienes in the Pathogenesis of Asthma. J. Allergy (Cairo) 2015, 157919 (2015).
Morishita, R. et al. Body mass index, adipokines and insulin resistance in asthmatic children and adolescents. J. Asthma 53, 478–484 (2016).
van Huisstede, A. et al. Effect of bariatric surgery on asthma control, lung function and bronchial and systemic inflammation in morbidly obese subjects with asthma. Thorax 70, 659–667 (2015).
Rastogi, D. et al. Inflammation, metabolic dysregulation, and pulmonary function among obese urban adolescents with asthma. Am. J. Respir. Crit. Care Med. 191, 149–160 (2015).
Haidari, F., Mohammadshahi, M., Borsi, S. H., Haghighizadeh, M.-H. & Malgard, S. Comparison of essential fatty acid intakes and serum levels of inflammatory factors between asthmatic and healthy adults: A case-control study. Iran J. Allerg. Asthma Immunol. 13, 335–342 (2014).
Wahab, A. A., Maarafiya, M. M., Soliman, A., Younes, N. B. M. & Chandra, P. Serum Leptin and Adiponectin Levels in Obese and Nonobese Asthmatic School Children in relation to Asthma Control. J. Allerg. (Cairo) 2013, 654104 (2013).
Youssef, D. M., Elbehidy, R. M., Shokry, D. M. & Elbehidy, E. M. The influence of leptin on Th1/Th2 balance in obese children with asthma. J. Bras. Pneumol. 39, 562–568 (2013).
Abd El-Kader, M. S., Al-Jiffri, O. & Ashmawy, E. M. Impact of weight loss on markers of systemic inflammation in obese Saudi children with asthma. Afr. Health Sci. 13, 682–688 (2013).
Cobanoglu, N., Galip, N., Dalkan, C. & Bahceciler, N. N. Leptin, ghrelin and calprotectin: inflammatory markers in childhood asthma? Multidiscip. Respir. Med. 8, 62 (2013).
Doniec, Z., Pierzchala-Koziec, K., Tomalak, W. & RyszardKurzawa, R. Serum level of leptin and neuropeptide Y in children with mild asthma. Pneumonol. Alergol. Pol. 72, 9–13 (2004).
Liu, W., Ji, X., Zhang, Z. Z., Wang, F. H. & Zhang, W. Y.Association between polymorphism of leptin rreceptor gene and serum leptin in asthma. J. Chin. Clin. Med. 29, 1147–1150 (2013).
Muc, M., Todo-Bom, A., Mota-Pinto, A., Vale-Pereira, S. & Loureiro, C. Leptin and resistin in overweight patients with and without asthma. Allergol. Immunopathol. (Madr) 42, 415–421 (2014).
Li, M. A., Si, L. Y., Wu, Hb. & Ma, P. Expression of leptin and Foxp3 in peripheral blood mononuclear cells in children with bronchial asthma. J. Mod. Lab. Med. 31, 58–64 (2016).
Tsaroucha, A. et al. Leptin, adiponectin, and ghrelin levels in female patients with asthma during stable and exacerbation periods. J. Asthma 50, 188–197 (2013).
Berthon, B. S., Macdonald-Wicks, L. K., Gibson, P. G. & Wood, L. G. Investigation of the association between dietary intake, disease severity and airway inflammation in asthma. Respirology. 18, 447–454 (2013).
Sideleva, O. et al. Obesity and asthma: an inflammatory disease of adipose tissue not the airway. Am. J. Respir. Crit. Care Med. 186, 598–605 (2012).
Sutherland, E. R. et al. Cluster analysis of obesity and asthma phenotypes. PLoS One 7, e36631 (2012).
Yuksel, H., Sogut, A., Yilmaz, O., Onur, E. & Dinc, G. Role of adipokines and hormones of obesity in childhood asthma. Allergy Asthma Immunol. Res. 4, 98–103 (2012).
Dajani, R., Al-Haj Ali, E. & Dajani, B. Macrophage colony stimulating factor and monocyte chemoattractant protein 2 are elevated in intrinsic asthmatics. Cytokine. 56, 641–647 (2011).
Jang, A.-S. et al. Association of serum leptin and adiponectin with obesity in asthmatics. J. Asthma 46, 59–63 (2009).
Leivo-Korpela, S. et al. Adipokine resistin predicts anti- inflammatory effect of glucocorticoids in asthma. J. Inflamm. (Lond) 8, 12 (2011).
Holguin, F., Rojas, M., Brown, L. A. & Fitzpatrick, A. M. Airway and plasma leptin and adiponectin in lean and obese asthmatics and controls. J. Asthma 48, 217–223 (2011).
Quek, Y.-W. et al. Associations of serum leptin with atopic asthma and allergic rhinitis in children. Am. J. Rhinol. Allerg. 24, 354–358 (2010).
Giouleka, P. et al. Body mass index is associated with leukotriene inflammation in asthmatics. Eur. J. Clin. Invest. 41, 30–38 (2011).
Arshi, M., Cardinal, J., Hill, R. J., Davies, P. S. W. & Wainwright, C. Asthma and insulin resistance in children. Respirology. 15, 779–784 (2010).
Chen, Z. G. et al. The role of serum leptin in infants with wheezing after respiratory syncytial virus infected. Chinese J. Experimental Clin. Virol. 23, 38–40 (2009).
Bruno, A. et al. Leptin and leptin receptor expression in asthma. J. Allergy Clin. Immunol. 124, 230–237 (2009).
Erel, F. et al. Serum leptin levels and lipid profiles in patients with allergic rhinitis and mild asthma. Allergol. Immunopathol. (Madr) 35, 232–238 (2007).
Guler, N. et al. Leptin: does it have any role in childhood asthma? J. Allergy Clin. Immunol. 114, 254–259 (2004).
Tanju, A. et al. Association between clinical severity of childhood asthma and serum leptin levels. Indian J. Pediatr. 78, 291–295 (2011).
Canöz, M., Erdenen, F., Uzun, H., Müderrisoglu, C. & Aydin, S. The relationship of inflammatory cytokines with asthma and obesity. Clin. Invest. Med. 31, E373–E379 (2008).
Gurkan, F. et al. Serum leptin levels in asthmatic children treated with an inhaled corticosteroid. Ann. Allerg. Asthma Immunol. 93, 277–280 (2004).
Kim, K. W. et al. Relationship between adipokines and manifestations of childhood asthma. Pediatr. Allerg. Immunol. 19, 535–540 (2008).
da Silva, P. L. et al. Interdisciplinary therapy improves biomarkers profile and lung function in asthmatic obese adolescents. Pediatr. Pulmonol. 47, 8–17 (2012).
Zhang, L., Yin, Y., Zhang, H., Zhong, W. & Zhang, J. Association of asthma diagnosis with leptin and adiponectin: A systematic review and meta-analysis. J. Investig. Med. 65, 57–64 (2017).
Zeng, X. et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J. Evid. Based Med. 8, 2–10 (2015).
Banasiak, H. & Pawliczak, R. Clinical profile of chronic bronchial asthma patients in Poland: results of the PROKSAL study. Postepy Dermatol. Alergol. 37, 879–889 (2020).
Rastogi, D. & Holguin, F. Metabolic Dysregulation, Systemic Inflammation, and Pediatric Obesity-related Asthma. Ann. Am. Thorac. Soc. 14, S363–S367 (2017).
Liang, R., Zhang, W. & Song, Y. M. Levels of leptin and IL-6 in lungs and blood are associated with the severity of chronic obstructive pulmonary disease in patients and rat models. Mol. Med. Rep. 7, 1470–1476 (2013).
Gerriets, V. A. et al. Leptin directly promotes T-cell glycolytic metabolism to drive effector T-cell differentiation in a mouse model of autoimmunity. Eur. J. Immunol. 46, 1970–1983 (2016).
Hur, J., Kang, J. Y., Kim, Y. K., Lee, S. Y. & Lee, H. Y. Glucagon-like peptide 1 receptor (GLP-1R) agonist relieved asthmatic airway inflammation via suppression of NLRP3 inflammasome activation in obese asthma mice model. Pulm. Pharmacol. Ther. 12, 102003 (2021).
Perakakis, N., Farr, O. M. & Mantzoros, C. S. Leptin in Leanness and Obesity: JACC State-of- the-Art Review. J. Am. Coll. Cardiol. 77, 745–760 (2021).
Torday, J. S. & Rehan, V. K. Stretch-stimulated surfactant synthesis is coordinated by the paracrine actions of PTHrP and leptin. Am. J. Physiol. Lung Cell Mol. Physiol. 283, L130–L135 (2002).
Jutant, E. M., Tu, L., Humbert, M., Guignabert, C. & Huertas, A. The Thousand Faces of Leptin in the Lung. Chest. 159, 239–248 (2021).
Acknowledgements
This study was supported by Grant from the National Natural Science Foundation of China (grant number 81600578), weak discipline construction of Shanghai health and family planning commission (grant number 2016ZB0102-03) and grant from China Hospital Development Institute of Shanghai Jiao Tong University (grant number CHDI-2018-B-06) for the study design, data collection and analysis.
Author information
Authors and Affiliations
Contributions
J.W., S.M., and W.S. participated in the conception and design of the study. S.M. and R.Z. participated in the extraction and analysis of data. S.M. participated in the interpretation of data and writing of the paper. R.Z. participated in the English editing of the manuscript. All authors approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, J., Zhu, R., Shi, W. et al. Predictive and prognostic value of leptin status in asthma. npj Prim. Care Respir. Med. 33, 10 (2023). https://doi.org/10.1038/s41533-023-00332-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41533-023-00332-z