Abstract
Current marine research primarily depends on weighty and invasive sensory equipment and telemetric network to understand the marine environment, including the diverse fauna it contains, as a function of animal behavior and size, as well as equipment longevity. To match animal morphology and activity within the surrounding marine environment, here we show a physically flexible and stretchable skin-like and waterproof autonomous multifunctional system, integrating Bluetooth, memory chip, and high performance physical sensors. The sensory tag is mounted on a swimming crab (Portunus pelagicus) and is capable of continuous logging of depth, temperature, and salinity within the harsh ocean environment. The fully packaged, ultra-lightweight (<2.4 g in water), and compliant “Marine Skin” system does not have any wired connection enabling safe and weightless cutting-edge approach to monitor and assess marine life and the ecosystem’s health to support conservation and management of marine ecosystems.
Similar content being viewed by others
Introduction
Marine ecosystems are experiencing worldwide anthropogenic-driven change, including extensive overfishing, run-off population, pollution, and increasing global warming.1,2 The ability to monitor and record various environmental and population parameters allows greater understanding of human impact, enhanced mitigation strategies, and the opportunity for systematic feedback to shape policy implementation. In that context, rapid advancements in electronic tagging and tracking tools have enabled the research community to remotely study a broad array of variables to monitor marine ecosystem health and how changes in the environment affect marine animals. Electronic tagging of marine life has provided information on animal behavior, environmental conditions, and geographical position.3,4,5,6,7 At the same time, marine tags should not weight more than 2% of the dry body weight of the tagged animal to maintain normal behavior, physiology, and survival of the tagged individual.8,9 Yet, most devices in the market are unsuitable for young specimens, invertebrates, or small species, because the tag exceeds this tenet.10,11 While many studies have focused on larger species, such as Cetaceans, Dolphins, and Sirenians, attachment methods are invasive. As an example, standard attachment techniques include using a shotgun or crossbow to insert tags into the animal’s tissue, or cutting tools and bolts to fix a tag to the dorsal fins.12 Such techniques often lead to infection of the area, or over sensitivity. For other animals, internal or external sensor attachment can be done through capture and short-term sedation.13 However, in both cases the invasiveness of the procedures could stress the tagged individuals and compromise the animal’s health. Several studies have been conducted analyzing the repercussions of marine tagging, where they showed that the extra carried weight and the design of the tag can affect diving patterns, mating, nesting behavior, and swimming drag.14,15,16,17 Therefore, the current size of CTD (conductivity–temperature–depth) sensors limit the diversity of species that could be studied, and further technological developments are required to provide more comfortable animal-friendly tagging devices that are not invasive and can conform to the animal’s morphology.
Despite the advances made on marine electronic tagging research,18,19,20 there are still major areas for improvement, including prolonged tag lifetime, increased sensor capabilities, better attachment techniques, and tag conformity to promote natural and unrestricted movement. Marine tag design should include animal comfort without compromising the performance, data validity, and endurance of the system. Also, sensor cost should not exceed current market standards to be considered commercially effective. Therefore, a compliant and stretchable marine tag should be lightweight, non-invasive, durable, biocompatible, and capable of monitoring the marine environment while retaining high performance and resolution standards.
With advances in state-of-the-art miniaturized electronics capitalizing on the emergence of flexible and stretchable form factors to integrate life, device, data, and processes through Internet of Everything21,22,23, here we develop a waterproof ultra-lightweight (<2.4 g), fully conforming (physically flexible and stretchable), standalone wireless multisensory (conductivity, temperature, depth) “Marine Skin” platform (55 mm×55 mm×0.3 mm). This ultra-lightweight sensor has non-invasive application, high performance multi-sensing, constant data logging with significantly lower cost. Integrated arrays of temperature, pressure, and conductivity sensors simultaneously monitor the animal’s diving patterns, and the surrounding environmental conditions (Fig. 1).
Marine Skin illustration. Marine species wearing Marine Skin system, for continuous tracking of the marine ecosystem. Outset digital photo depicts the reconfigurable net-like array design for geometric stretchability and flexibility of Marine Skin. The waterproofed system continuously logs temperature, pressure, and salinity data from the surrounding environment, to assess ocean health and track animal mobility using marine life as allies. Real-time data is then wirelessly transmitted upon resurfacing of the marine animal
Marine skin for temperature, depth, and salinity logging
Current CTD-like multisensory platforms allow data collection to predict changes in behavior, population size, and range/distribution of marine species. However, available designs disturb the natural and smooth underwater movement of animals through increased drag and turbulence in fluvial dynamics.24 Conductivity (i.e., salinity), temperature, and depth provide the most fundamental description of the ocean’s environment, allowing characterization of water masses and the niche used by marine animals (refer to section 1.1 of Supplementary Information (SI)). Hence, efforts in the development of marine sensors are mainly focused on these environmental parameters.25 Yet, one of the greatest challenges for effective monitoring and experimentation is the physical size of equipment, weight, and attachment invasiveness.
Biocompatible packaging materials
Effective marine sensors must survive the particularly corrosive saline aqueous environment (35-40 practical salinity unit, PSU)26,27 and maintain high sensitivity and repeatability. Therefore, the development of a “Marine skin” tag requires a compliant packaging material that takes into consideration biocompatibility, toxicology, cost, endurance in saline environments, temperature and pressure working ranges, and degradation with time. Additionally, the appropriate packaging material needs to comply with the soft elasticity of the hosting surface, minimizing any kind of discomfort caused when the tag is placed on the asymmetric surface (often soft) of marine animals, and supporting freedom of movement without any restrictions.
We chose to base the “Marine Skin” sensory tag developed here on polydimethylsiloxane (PDMS) Sylgard 184™ as our conformal packaging material among other flexible and stretchable polymeric counterparts (e.g., Ecoflex®). It is hydrophobic, non-toxic, and non-irritating to the skin, does not decompose under high heat or halogenation, and unlike Ecoflex which is biodegradable,28 it does not undergo major degradation or polymeric reduction when exposed to certain microorganisms found in sea water.29 Most importantly, PDMS is effective for the current cause due to its ease of flow integration and compatibility with CMOS processes using state-of-the-art industry equipment. However, it must be also considered that PDMS has low surface energy, which is increased due to its hydrophobicity, easing adhesion of particles from the aqueous environment. Hence, although hydrophobicity is desired to prevent water from running inside the devices, it is unknown how this affects biofouling. Therefore, future work on PDMS as a compliant encapsulation will involve long-term study of biofouling and possible anti-fouling coatings to prevent accretion of microbial films30,31
Compliant tag design and integration
“Marine Skin” uniqueness lies in its physical flexible and stretchable design, displaying multi-sensory capability with simultaneous sensing and selectivity. The array exhibits an ultra-thin net-shape construction (Fig. 1), easily integrated in an intimate fashion on curved animal surfaces. The wavy network pattern was designed for optimal two-dimensional expansion, with inherent elasticity and twisting capability, enabled through intrinsic PDMS elasticity and the geometry of thin metal routings. The packaged tag dimensions are 55 mm×55 mm×0.3 mm, although these can be easily downscaled by decreasing the array size. Marine tags are generally constrained by the bulky size of their systems and packaging. However, our employed packaged system is compact (21 mm×10 mm) and conformal (3.5 mm in height). The tag consists of large arrays of capacitive pressure sensors and resistive temperature detectors (RTD) incorporating individual salinity sensors based on electrodes separated by a 2 mm gap (Fig. 1).
Different designs can be implemented to achieve a variety of sizes and elastic deformation. The geometrical “net” architecture along with the intrinsic stretchable properties of PDMS, allow the system to conform to the body of the animal and stretch/contract with their movements, ensuring comfort and adhesion under any circumstance. The fabrication process is conducted using a low-cost CMOS compatible approach, allowing ease of scalability, batch fabrication, and precision. Detailed integration strategy of the “Marine Skin” tag is described in the process schematics of Fig. 2, showing compliant 3D integration of the SoC on top of the sensory array (Fig. 2(a)), accompanied with conformal encapsulation and release of the final flexible tag (Fig. 2(13–15)). Further information about the process flow and mechanical properties of materials can be found in section 1.2 of SI text.
Process flow of the waterproof & stretchable multisensory Marine Skin. Low-cost CMOS compatible approach, allowing ease of scalability, batch fabrication and precision. Steps (a–c) are prepared on a separate wafer 1, to make flexible 10 µm sheets of polyimide (PI). The multisensory system is fabricated on wafer 2, through steps (1–12), illustrating the integration of arrays of capacitive pressure sensors, resistive temperature detectors, and salinity sensing capability. Step (13) displays the conformal 3D system integration, followed by (14) a compliant encapsulation of the system, and (15) final system release
Results and discussion
CTD logging in sea water
Effect of salts on electrode performance is critical for marine systems development. Therefore we conducted long-term experiments (over 20 days) to test the integrity of our encapsulation in Red Sea water (~ 40 PSU). Results, discussed in section 2.1 of SI and Fig. S1, highlight a promising and durable flexible packaging. Ultimately, “Marine Skin” is targeted for conformal placement on marine animals of irregular sizes and shapes. Hence, sensors’ functionalities need to be tested under diverse mechanical bending conditions (Fig. 3) to assess their performance and stability when employed on curved surfaces. All measurements were conducted in seawater (40 PSU), varying depth and water temperature (Fig. 3). Water temperature determination and depth calibration plots are shown in Fig. S2 and discussed in section 2.2 of SI, where correlation between underwater pressures and corresponding depth values generally varies depending on the salinity and density of the water, and hence on temperature.
Marine skin performance under concave bending conditions. a Marine Skin resistive response to water temperature variations. b Marine Skin capacitive response to increased underwater pressures/depth. c Marine Skin resistive response to salinity changes via KCl concentration increments. d Effect of temperature on the pressure sensor’s performance. e Dynamical resistance fluctuations obtained from different thermal conditions. Dash lines denote depth of immersions. f Effect of depth of immersion on the temperature sensor’s performance
Temperature sensor’s response under different bending radii shows that the sensor’s sensitivity stayed intact with an average sensitivity of Stemp, avg = 22.66 mΩ/°C and a temperature resolution of 0.03 °C, sufficient to detect desirable fluctuations of 0.1 °C in the ocean’s temperature (Fig. 3a). Minor strain-induced increase of 0.61% in absolute resistance value is observed under bending. Effect of strain would generally be greater, however, the implemented wavy design, along with the sensors’ placement in the neutral plane of the packaging, minimizes stress and strain propagation in the structures.
Similarly, Fig. 3b highlights the pressure sensor’s stability under different bending conditions, with an average linear sensitivity of Sdepth, avg = 0.71 nF/dBar = 6.93 pF/cm = 0.056 cm−1, a standard deviation of σ ± 8.3 × 10−4 cm−1 attributed to variations in platform curvature, and an estimated high depth resolution of 0.14 mm. Given the available means, our testing setup was limited to an 80 cm depth range, which is considered shallow for marine tagging applications. However, “Marine Skin” sensors display high resolution with great linearity, without any signs of signal saturation or attenuation. These findings show promise of continued high performance functionality under higher pressures, and can be used to predict sensory behavior for extended depths.
Response and recovery times of both temperature and pressure sensors are determined through real-time underwater logging. Conducted experiments, including variations in bending, are discussed in section 2.3 and section 2.4 of SI. Corresponding results in Fig. S3–5 highlight an almost instantaneous tag response and recovery to continuous changes in temperatures, as well as a fast response and recovery of tresponse = 321.9 ms and trecovery = 429.2 ms, respectively, equivalent to Δd = 10.5 cm changes in depth. Nevertheless, “Marine Skin” was characterized by an instantaneous response and recovery towards continuous transition in depth position, attributed to its thin conformal nature promoting superior thermal conductivity. This fast tracking and recovery is especially desired when animal behavior and response is to be evaluated dynamically under dangerous conditions (e.g., prey, competitors).
Salinity detection is analyzed in Fig. 3c, displaying the measured resistance generated from the produced ion channel versus salinity levels. For water temperature of 21 °C, salinity levels ranging from 20 PSU to 41 PSU are, respectively, translated into conductivities from 29.56 to 56.3 mS/cm, determined using an online CTD salinity to conductivity converter “Salinometry”, considering T = 21 °C and 10 dBar pressure. As salinity levels increase, the water solution becomes more conductive, and hence resistance decreases. We observe a fairly linear behavior with sensitivity of Ssalinity = 6.2 kΩ/PSU = 0.00466 /PSU and a high salinity resolution of 0.016 PSU, capable of distinguishing slight variations in ecologically-relevant changes in ocean’s salinity.32 Response and recovery times were then analyzed through continuous logging at T = 21 °C in incremental PSU solutions (Fig. S6a). For a fixed salinity of 20 PSU, we determine a total response time of tsalinity_response = 38 s and a total recovery time of tsalinity_recovery = 45 s. It is important to note that conductivity sensors are also sensitive to changes in water temperature and conditions, where thermal conductivity of water is kwater = 0.611 W/m K.33 Higher temperatures lead to faster ions movement in water, increasing the conductivity. However, since our tag also contains a temperature sensor, this information can be always accounted for and calibrations can be performed in order to retrieve temperature-corrected salinity values.
Sensors selectivity
The selectivity of the sensors towards changes in the marine environment is a fundamental component of the sensor’s performance. Plots in Fig. 3d–f show the effect of temperature variations on pressure sensitivity and effect of depth on water temperature sensitivity. Supporting cross-sensitivity plots can be found in Fig. S7 and S8 and details about the findings are discussed in section 2.5 of SI. Nevertheless, “Marine Skin” tag integrates sensory multifunctionality, with pressure and temperature values continuously being recorded and dependent on one another. Each sensor is highly selective to its targeted stimuli and no significant performance change was observed from cross-sensitivity via water temperature and depth variations. Ultimately, the observed disparities and strain-induced changes follow well defined trends that can be easily compensated for.
System integration and field deployment
Lightweight and conformal system interface
In marine tag developments, the system which comprises of a microcontroller, interface circuitry, communication unit, storage, memory, and power management unit, seems to be the bottleneck that transforms the whole-sensing system into heavy and bulky devices, unsuitable for tagging relatively small animals. To achieve the goal of a truly lightweight and conformal tag, the system components need to be simplified as much as possible, to reduce weight and footprint while maintaining good performance. We developed a wireless marine system using low power Bluetooth technology (BLE), capable of reading both capacitive (pressure) and resistive (temperature and salinity) sensors. The system is comprised of a Programmable System-on-Chip (PSoC) with integrated Bluetooth transceiver. This PSoC, including passive components and antenna, is mounted on a 10 mm×10 mm printed circuit board (Fig. 2(a)), and a schematic of the interface is shown in Fig. 4a. Data is continuously logged at 1 Hz in the PSoC’s internal flash memory (256 KB), and only when the animal emerges out of water, data is wirelessly collected via BLE as depicted in Fig. 1. Further technicalities about how we achieved the low power interface are discussed in section 3 of SI.
Autonomous and conformal wireless marine system. a System schematics and block diagrams of the Marine skin sensory system integrating a BLE chip and a battery. b Waterproofed system integrity with performance testing in sea water under 70 cm deep tank. c Zoomed-in depth response collected from the sensory system as it is pushed down to 70 cm
This efficient lightweight system interface made it possible to perform underwater experiments for longer periods of time without hindering the animal’s usual movement. The fully packaged Marine Skin tag weights an incomparable <6 g in air and <2.4 g in water, and has a battery lifetime of up to 1 year assuming a logging rate of 2 s. Tests of system integrity in seawater are shown in Fig. 4, displaying continuous and repeatable response (please refer to section 4 of SI and Fig. S9 for calibration plots). Data retrieval when animals surface, is appropriate for air-breathing animals (marine mammals and reptiles) and also can be used with a pop-up device to retrieve data from continuously submerged animals.34
Testing on marine animals—crustaceans
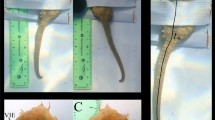
In order to test the feasibility of this approach, we conducted field tests on crustaceans—specifically on a swimming crab (Portunus pelagicus) captured along the east coast of the central Red Sea. Marine Skin tag was conformally and non-invasively attached on the crab’s shell using superglue (Fig. 5a). For testing on a wider range of marine animals, especially mammals (e.g., dolphins), biocompatible adhesives should be used as an alternative to superglue for a sturdy and non-invasive attachment method (please refer to section 5 of SI). We then monitored the crab’s behavior in sea water using a logging frequency of 1 Hz. The depth pattern logged in Fig. 5b depicts continuous and active tracking of the crab’s diving and resurfacing patterns for 6 min. Using a 1 Hz logging frequency, the system consumes low power, yielding an operation life of 5 months. Battery life can be further extended by optimizing the operating conditions and reducing sampling intervals. As for temperature logging, the current system can detect changes of 0.2 mV, which translates into 0.5 °C resolution. The portable system can be further optimized to detect changes of 0.437 µV, and hence improve the detection resolution up to 0.001 °C, but this would translate into higher power consumption and the need for a bigger and heavier battery.
Field testing on marine animals—crustaceans. a Digital photos of the marine skin system seamlessly attached on the crab using superglue. b Monitoring crab (Portunus pelagicus) movement in its natural habitat—Depth pattern recognition and animal behavior was actively and continuously recorded in real time with Δt = 1 s logging interval
Comparative analysis
To put in perspective the advancements and possibilities created through Marine Skin sensory tag, the benchmark table (Fig. 6) compares the most notable developments in marine tags, including commercial35,36,37,38,39,40,41 and academic projects.18,19,20 We compare tags that exhibit similar functionalities to our platform, and we focus our comparison based on form factor, weight, and battery lifetime (hereafter referred to as Tag Deployment Lifetime “TDL”), which was normalized according to the respective sampling rate of each project. Although Marine Skin tag presents promising sensory results, at this stage of our study, sensors were reliably tested only down to 80 cm underwater. Therefore, we have no rigid grounds for performance and resolution comparison with commercial sensors, which show tests down hundreds and thousands of meters. Nevertheless, performance does not seem to be an issue with sensing based projects, since current technology advancements are used to come up with the best sensing solution. However, improving TDL and reducing form factor are the major challenges that require focus. Form factor has a significant effect on the underwater behavior and stress of tagged animals. Devices extruding-off of animal skin create drag, which forces marine animals to make extra effort in order to move, altering their natural behavior.10,1517,42 The extra carried weight and tag design affect diving patterns, mating, nesting behavior, swimming drag, movement capacity, and performance ability of marine animals.34 Tagged marine animals are samples of a bigger population and the ecosystem as a whole, therefore the incorporation of telemetric devices should not alter their natural performance, behavior, physiology, or survival. Among all advances listed in Fig. 6, Marine Skin tag overcomes this concern owing to its extremely lightweight design and compliant form factor made specifically to adapt to different animal sizes and shapes, while maintaining a long lifetime. In contrast, currently available solutions have proven to be unsuitable for tagging young specimens, invertebrates, or small species due to their rigid design, heavy weight and bulky form factor. But also, can be economically impractical with short shelf life given their expensive price tag or limited functionality.
Conclusion
As sensors and tracking tools for marine animals continue to evolve with advances in technology and research, so will its application for understanding, and responding to the ecological and conservation implications of marine animal behavior. Increasing global temperatures, non-source pollution, and extensive overfishing are leading to the degradation of the oceans and the many services marine ecosystems provide to society,1 driving the increased need to monitor the marine environment and the behavior of marine animals. Therefore, compliant sensory tags that are non-invasively attached to marine animals can enhance the quality of aquatic life while advancing scientific exploration. Here we show, a waterproof, flexible, stretchable, Bluetooth-enabled standalone Marine Skin tag capable of operating under vast pressure, temperature, and salinity regimes. It is easily adaptable to a diversity of animals of any size and shape, focused on maintaining animal comfort and movement through a compliant and cost-effective design. Unlike anything else, Marine Skin tag is non-invasive and lightweight (<2.4 g), exhibiting a long deployment lifetime without compromising performance and resolution.
Methods
Fabrication process of sensors
We start with a Si (100) wafer (Wafer 1) on top of which we spin coat a 10-µm thick polyimide film (PI-2611). We perform a soft bake at T = 90 °C for 90 s, followed by a second bake at T = 150 °C for 90 s. Final curing is performed by ramping the hot plate temperature from 150 to 350 °C at a rate of 240°/h, and leaving the wafer to cure for 30 min at T = 350 °C. These steps are shown in Fig. 2(a–c).
We then take a second Si (100) wafer (Wafer 2), on top of which we sputter a thin layer of Ti/Au. Au film is used since it has low bonding energy with PDMS, and hence will act as a great intermediate layer to ease the final release process. We then spin coat 100-µm thick PDMS (Sylgard 184™) and cure it at T = 90 °C for 30 min, which will act as the bottom encapsulation layer. This is then followed by an O2 plasma treatment for 2 min, which will temporarily make PDMS hydrophilic to improve its adhesion to the subsequent layer. We then peel of the 4 inch PI film from Wafer 1 and carefully transfer it on top of the treated PDMS of Wafer 2 (steps 1 to 4 in Fig. 2). In order to pattern thick PI film, an Aluminum (Al) hard mask was used for its selectivity to PI etching gases. First, PI film was treated with oxygen plasma at low power (30 W) for 2 min. This step is necessary to improve adhesion of metal films on top of PI and avoid delamination issues. This is followed by 200 nm sputtering of Al and patterning using mask #1 (Fig. 2, step 7). We then proceed by making another oxygen plasma on top of patterned PI and sputter bottom contact 10 nm/180 nm Titanium/Gold (Ti/Au), which is then patterned using mask #2 (Fig. 2, step 10). This first metal layer consists of the bottom contacts of the pressure/depth sensors. We then deposit the pressure sensitive rubber of the depth sensors, via 50-µm thick PDMS cured at T = 75 °C for 75 min (Fig. 2, step 10). A second metal film of Ti/Au (10 nm/180 nm) is then deposited by repeating the same exact steps taken to create the first metal layer. These steps begin with a second transfer of another PI film, all the way to the end by sputtering and patterning the metal film, reflected through steps 4 to 10 in Fig. 2. This second metal film consists of the top contacts of the depth sensors, the temperature sensors array, as well as the conductivity/salinity sensors.
Finally, system-on-chip integration is performed (Fig. 2 step 13) and final tag encapsulation is performed by spin coating a 150-µm thick PDMS and curing it at T = 90 °C for 30 min (Fig. 2 step 14), which will also enable the metal routings of our structure to be in the neutral plane. Finally, the completely packaged Marine Skin Tag is release from the Si wafer through simple release using a tweezer (Fig. 2 step 15).
Compliant system on chip integration
The PSoC used along with the coin cell battery are placed on top of the Marine Skin sensors (before top encapsulation). Conformal connections between the system and the sensors are created via thin layers of silver epoxy or paint, which preserves the compliant form factor required along with mechanical robustness.
Salinity solutions preparation
Conductivity allows determination of effective salinity. Seawater is a solution of 86% by mass NaCl, with supplemental Magnesium, Calcium, Potassium, and Strontium cations.27 To simulate water solutions with fixed salinity levels, we used defined concentrations of KCl mixed in tap water, to achieve salinities ranging from 20 to 41 PSU. PSU, which is based on the properties of seawater conductivity, and is equivalent to: 1 PSU = 1 g of salt/ 1 kg of seawater = 1 mg KCl/ 1 L of water (in the presented work). Salinity sensor is then dipped into the distinct saline solutions, and a corresponding electrical conduit is produced between the sensor’s electrodes, specific to the water conductivity.
Individual testing of sensors
Electrical characterization of each of the sensors was performed using Keithley semiconductor analyzer 4200-SCS and real-time measurements (both resistive and capacitive) were conducted at a sampling rate of 100 ms. Underwater saline environments were prepared by filling an acrylic tank with ~80 cm full of water directly from the Red Sea. Water temperatures were varied by using a hot plate below the tank, and a magnetic stirrer to allow uniform heating profile in the water. Temperature calibrations were then retrieved using a commercial thermocouple from Fluke 289.
Bio ethics statement
No animal was harmed during the experiment and all experiments were conducted under the guidance of experienced animal handler. The KAUST Bio Ethics Committee approved this testing in accordance with best international standards for animal safety and preservation requirements.
Data availability
The data that support the findings of this study are available from the authors on reasonable request. The authors declare that the data supporting the findings of this study are available within the article and the corresponding Supplementary Information File.
References
Duarte, C. M. Global change and the future ocean: a grand challenge for marine sciences. Front. Mar. Sci. 1, 63 (2014).
Halpern, B. S. et al. A global map of human impact on marine ecosystems. Science 319, 948–952 (2008).
Block, B. A. et al. Tracking apex marine predator movements in a dynamic ocean. Nature 475, 86–90 (2011).
Costa, D. P., Breed, G. A. & Robinson, P. W. New insights into pelagic migrations: implications for ecology and conservation. Annu. Rev. Ecol. Evol. Syst. 43, 73–96 (2012).
Cooke, S. J. et al. Developing a mechanistic understanding of fish migrations by linking telemetry with physiology, behavior, genomics and experimental biology: an interdisciplinary case study on adult Fraser River sockeye salmon. Fisheries 33, 321–339 (2008).
Humphries, N. E., Weimerskirch, H., Queiroz, N., Southall, E. J. & Sims, D. W. Foraging success of biological Lévy flights recorded in situ. Proc. Natl Acad. Sci. USA 109, 7169–7174 (2012).
Miller, K. M. et al. Genomic signatures predict migration and spawning failure in wild Canadian salmon. Science 331, 214–217 (2011).
Bridger, C. J. & Booth, R. K. The effects of biotelemetry transmitter presence and attachment procedures on fish physiology and behavior. Rev. Fish. Sci. 11, 13–34 (2003).
Jepsen, N., Schreck, C., Clements, S. & Thorstad, E. Aquatic telemetry: advances and applications. In: Proc. Fifth Conference on Fish Telemetry held in Europe 255–259 (COISPA Technology and Research and Food and Agriculture Organization of the United Nations, Rome, 2005).
Cooke, S. J. et al. Biotelemetry: a mechanistic approach to ecology. Trends Ecol. Evol. 19, 334–343 (2004).
Brown, R. S., Cooke, S. J., Anderson, W. G. & McKinley, R. S. Evidence to challenge the “2% rule” for biotelemetry. North Am. J. Fish. Manag. 19, 867–871 (1999).
Walker, K. A., Trites, A. W., Haulena, M. & Weary, D. M. A review of the effects of different marking and tagging techniques on marine mammals. Wildl. Res. 39, 15–30 (2012).
Geraldi, N. R. & Powers, S. P. Subtle changes in prey foraging behavior have cascading effects in a shallow estuary. Mar. Ecol. Prog. Ser. 427, 51–58 (2011).
Broderick, A. C. & Godley, B. J. Effect of tagging marine turtles on nesting behaviour and reproductive success. Anim. Behav. 58, 587–591 (1999).
Alex Shorter, K., Murray, M. M., Johnson, M., Moore, M. & Howle, L. E. Drag of suction cup tags on swimming animals: modeling and measurement. Mar. Mammal. Sci. 30, 726–746 (2014).
Hammerschlag, N., Cooke, S. J., Gallagher, A. J. & Godley, B. J. Considering the fate of electronic tags: interactions with stakeholders and user responsibility when encountering tagged aquatic animals. Methods Ecol. Evol. 5, 1147–1153 (2014).
Todd Jones, T. et al. Calculating the ecological impacts of animal‐borne instruments on aquatic organisms. Methods Ecol. Evol. 4, 1178–1186 (2013).
Broadbent, H. A., Ketterl, T. P., Reid, C. S., & Dlutowski, J. A Low-Cost, Miniature CTD for Animal-Borne Ocean Measurements 1–7 (Oceans 2010 MTS/IEEE, Seattle, 2010).
Broadbent, H. A CTD biotag for mid-sized marine predators. PhD thesis, Univ. South Florida, (2012).
SMRU-Instrumentation. CTD oceanography SRDL (Argos) http://www.smru.st-and.ac.uk/Instrumentation/CTD/ (2017).
Nassar, J. M., Rojas, J. P., Hussain, A. M. & Hussain, M. M. From stretchable to reconfigurable inorganic electronics. Extrem. Mech. Lett. 9, 245–268 (2016).
Shaikh, S. F. et al. Freeform compliant CMOS electronic systems for internet of everything applications. IEEE Trans. Electron Devices 64, 1894–1905 (2017).
Hussain, A. M. & Hussain, M. M. CMOS‐technology‐enabled flexible and stretchable electronics for internet of everything applications. Adv. Mater. 28, 4219-4249 (2015).
Hussey, N. E. et al. Aquatic animal telemetry: a panoramic window into the underwater world. Science 348, 1255642 (2015).
Hooker, S. K. & Boyd, I. L. Salinity sensors on seals: use of marine predators to carry CTD data loggers. Deep Sea Res. Part I Oceanogr. Res. Pap. 50, 927–939 (2003).
Hariri, K., Gladstone, W. & Facey, C. R. State of the marine environment - Report for the Red Sea and Gulf of Aden: 2006. (PERSGA, Jeddah, Kingdom of Saudi Arabia, 2014).
Balistrieri, L. S. & Murray, J. W. The influence of the major ions of seawater on the adsorption of simple organic acids by goethite. Geochim. Cosmochim. Acta 51, 1151–1160 (1987).
Bagheri, A. R., Laforsch, C., Greiner, A. & Agarwal, S. Fate of so‐called biodegradable polymers in seawater and freshwater. Glob. Chall. 1, 1700048 (2017).
Shastri, V. P. Non-degradable biocompatible polymers in medicine: past, present and future. Curr. Pharm. Biotechnol. 4, 331–337 (2003).
Zhang, H. & Chiao, M. Anti-fouling coatings of poly (dimethylsiloxane) devices for biological and biomedical applications. J. Med. Biol. Eng. 35, 143–155 (2015).
Krishnan, S., Weinman, C. J. & Ober, C. K. Advances in polymers for anti-biofouling surfaces. J. Mater. Chem. 18, 3405–3413 (2008).
Talley, L. D. Salinity patterns in the ocean. Earth Syst. 1, 629–640 (2002).
ChemWorx, A., Archives, C. & Photonics, A. Polymer data handbook. J. Am. Chem. Soc. 131, 16330–16330 (2009).
Hoolihan, J. P. et al. Evaluating post-release behaviour modification in large pelagic fish deployed with pop-up satellite archival tags. ICES J. Mar. Sci. 68, 880–889 (2011).
Cefas Technology. Data storage tags - G5 http://www.cefastechnology.co.uk/products/data-storage-tags/g5/ (2017).
LLC, D. S. S. SeaTag-MOD http://www.desertstar.com/page/seatag-mod (2017).
Star-Oddi. Data storage tags (DSTs) http://star-oddi.com/resources/Files/DST-baelingur-breyting-sept-NET.pdf (2017).
BioTrack. LAT 1000 temperature depth recorders http://www.biotrack.co.uk/lat1000-tdrs.php#s3 (2017).
MicroDaq. HOBO water level data logger https://www.microdaq.com/onset-hobo-u20-water-level-data-loggers.php (2017).
Wildlife Computers. MiniPat http://wildlifecomputers.com/our-tags/minipat/ (2017).
Microwave Telemetry. PTT-100 archival pop-up tag http://www.microwavetelemetry.com/fish/archival/specifications.cfm (2017).
Wilmers, C. C. et al. The golden age of bio‐logging: how animal‐borne sensors are advancing the frontiers of ecology. Ecology 96, 1741–1753 (2015).
Acknowledgements
This publication is based upon work supported by the King Abdullah University of Science and Technology (KAUST) Office of Sponsored Research (OSR) under Award No. Sensor Innovation Initiative OSR-2015-Sensors-2707.
Author information
Authors and Affiliations
Contributions
M.M.H. conceptualized the idea and directed the study. J.M.N. fabricated, characterized, and analyzed the data. S.M.K. developed the circuits and systems. S.J.V. assisted in fabrication and developed the packaging process. A.D.G. assisted in fabrication and system characterization. S.F.S. fabricated the integrated system. N.R.G. assisted in animal testing. G.A.T. assisted in tag development. C.M.D. advised on system requirement, environmental condition, and animal testing. All authors contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nassar, J.M., Khan, S.M., Velling, S.J. et al. Compliant lightweight non-invasive standalone “Marine Skin” tagging system. npj Flex Electron 2, 13 (2018). https://doi.org/10.1038/s41528-018-0025-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41528-018-0025-1
This article is cited by
-
Bioadhesive interface for marine sensors on diverse soft fragile species
Nature Communications (2024)
-
An underwater vest containing an antioxidant MXene hydrogel for sensitive recognition of fish locomotion
Microsystems & Nanoengineering (2024)
-
Printed temperature sensor array for high-resolution thermal mapping
Scientific Reports (2022)
-
A Review: Marine Bio-logging of Animal Behaviour and Ocean Environments
Ocean Science Journal (2021)
-
Reversible conductivity recovery of highly sensitive flexible devices by water vapor
npj Flexible Electronics (2018)