Abstract
There are few medical issues that have generated as much controversy as screening for breast cancer. In science, controversy often stimulates innovation; however, the intensely divisive debate over mammographic screening has had the opposite effect and has stifled progress. The same two questions—whether it is better to screen annually or bi-annually, and whether women are best served by beginning screening at 40 or some later age—have been debated for 20 years, based on data generated three to four decades ago. The controversy has continued largely because our current approach to screening assumes all women have the same risk for the same type of breast cancer. In fact, we now know that cancers vary tremendously in terms of timing of onset, rate of growth, and probability of metastasis. In an era of personalized medicine, we have the opportunity to investigate tailored screening based on a woman’s specific risk for a specific tumor type, generating new data that can inform best practices rather than to continue the rancorous debate. It is time to move from debate to wisdom by asking new questions and generating new knowledge. The WISDOM Study (Women Informed to Screen Depending On Measures of risk) is a pragmatic, adaptive, randomized clinical trial comparing a comprehensive risk-based, or personalized approach to traditional annual breast cancer screening. The multicenter trial will enroll 100,000 women, powered for a primary endpoint of non-inferiority with respect to the number of late stage cancers detected. The trial will determine whether screening based on personalized risk is as safe, less morbid, preferred by women, will facilitate prevention for those most likely to benefit, and adapt as we learn who is at risk for what kind of cancer. Funded by the Patient Centered Outcomes Research Institute, WISDOM is the product of a multi-year stakeholder engagement process that has brought together consumers, advocates, primary care physicians, specialists, policy makers, technology companies and payers to help break the deadlock in this debate and advance towards a new, dynamic approach to breast cancer screening.
Similar content being viewed by others
Introduction
Annual screening mammography—the most common approach in the US today—has its roots in the large, randomized screening trials of the 1980s.1 The first trial of annual screening, the U.S. Health Insurance Plan of Greater New York, began in 1963 and included 31,000 women in each arm.2 At 18 years of follow-up, it showed a 25% reduction in mortality, although benefit to women in their forties accrued after they were 50. The overview of the Swedish trials of bi- or triennial screening showed a relative reduction in breast cancer mortality of 21%, with maximum benefit for women in their sixties.3 The degree and timing of benefit to younger women in particular has generated a great deal of controversy.4 Even a decade later, there remains a continuing debate over the methodologic flaws of each of these studies, the net effect of which has impeded consensus on public recommendations for breast screening.5,6,7
From the outset, translating these studies into population-based screening recommendations stirred controversy, with debate focused on the frequency and most appropriate age to begin screening. The January 1997 Consensus Development Panel convened by the National Institutes of Health recommended women aged 40–49 be informed of the benefits and risks of screening and decide for themselves.8 The National Cancer Institute (NCI) and American Cancer Society (ACS), however, recommended regular screening for women in their forties while disagreeing on screening frequency, with the former recommending every 1–2 years, the latter annually. Partly because of the controversy generated, the NCI later stopped issuing screening guidelines.
Now, 20 years later, we find ourselves in a familiar place—still reviewing and reanalyzing data from the same trials, debating the optimum age to begin and interval, with professional societies that set guidelines compelled to “take a side” in the debate. The controversy following the 2009 JAMA commentary “Rethinking Screening”9 and updates to USPSTF guidelines10 illustrates how entrenched both sides have become. Consensus on recommendations remains distant.
The US Preventative Task Force systematic review concluded in 2015,11 much like it had in 2009,10 that mammographic screening benefits women over 50 and that biennial, not annual, screening was recommended for women ages 50–74. After weighing the balance of harms and benefits for women aged 40–49, screening was not recommended routinely for women in their forties. Instead, the USPSTF suggested an individualized approach taking patient risk and personal preference into account. In contrast, 2017 guidelines from the American College of Radiology and the Society of Breast Imaging currently recommend annual screening starting at age 40.12 The American Cancer Society has revised their guidelines and recommend annual mammograms for women over 45 of average risk, with women between the ages of 40–44 provided the opportunity to begin annual screening. Women over the age of 55 are recommended to receive biennial screening, although annual screening may be considered.13
Although the academic debate has progressed little in these 30 years, what has changed is public awareness of this issue. The vast press coverage of the Canadian National Breast Cancer Screening Study (CNBCSS) in 2014 brought the potential harms of screening into the public spotlight. Not only did the 25-year analysis of the CNBCSS show, as it had at both 10 and 15 years, that annual screening failed to reduce breast cancer mortality, it provided the first-ever estimates of overdiagnosis in a population-based annual screening program: half of all screen-detected non-palpable cancers were estimated to be indolent lesions that would otherwise never have come to clinical attention.14
For some, this result simply confirmed previous findings showing that up to 20% of all cancers (50% of screen-detected cancers) fall into the category of overdiagnosis15,16,17—meaning a woman has a greater chance of being over-diagnosed than of having her life saved by screening.18 Others pointed to studies demonstrating there is little, if any overdiagnosis in breast cancer,19, 20 labeling CNBCSS as a flawed analysis or simply the latest way to attack screening. The debate reached such an unhealthy tenor that published exchanges even included accusations of a scientific conspiracy to reduce access to mammography.21, 22
Like the screening debates that preceded it, the controversy surrounding overdiagnosis has now settled into a familiar pattern. It focuses on largely technical arguments over statistical assumptions, corrections for lead time bias and varied demographics23, 24 that create uncertainties in the data and ultimately have shifted the debate into the realm of opinion, rather than fact. Recent characterization of a molecular profile to define an ultralow risk biology may provide a tool to more objectively categorize ultralow risk breast cancers that have little systemic risk of progression. This is an important advance that may help us to improve our ability to treat the disease and further tailor individual screening recommendations.25
We must remember, however, that the data we are arguing over are from decades-old trials, from an era before most of the effective systemic therapies were available. That there is an impact of modern systemic therapies on reducing breast cancer mortality is undeniable—some estimate that systemic therapy accounts for 2/3 of the observed reduction in mortality.26, 27 The rise of endocrine therapy28, 29 may also mitigate the impact of finding some cancers later.
Whether one believes these figures or not, the takeaway is that we are stuck in an endless cycle of academic debate, arguing over data that have little context in the modern treatment setting. Breast cancer treatment continues to rapidly evolve towards a patient-centered, precision medicine approach that recognizes what is perhaps the most important lesson we have learned over the past two decades of research: that breast cancer is not a monolithic entity, but a spectrum of disease. From indolent lesions of epithelial origin (IDLE)9 requiring no treatment, to aggressive disease requiring equally aggressive treatment, it has resisted all our attempts to lump it into a single bin.
Yet we continue our one-size-fits-all approach to breast cancer screening. It is contrary to the very nature of the disease. We cannot continue to focus the entirety of our efforts on a screening approach that is based on an outdated understanding of breast cancer biology, expecting that the uncertainties and debate will finally be resolved. Instead, we must be willing to innovate and to entertain new paradigms of screening that incorporate our current understanding of breast cancer, its treatment and risk susceptibility by putting them to the test.
We may have little choice. Because the consequences of failing to do so may be to further alienate the very women screening is supposed to help.
What women want: better, not more screening
Even though generations of women educated in the benefits of screening mammography generally regard it positively, experience shows it is a fragile trust. A single false positive can cause psychological distress for up to 3 years and reduce adherence to subsequent screening by 37%.30,31,32,33,34
Considering the specificity of mammography is generally accepted to be ~ 90% (e.g., 1 in 10 are false positive), whereas the real breast cancer rate is ~ 5 in 1000 women, the majority of abnormal mammograms are, in fact, false positives. After 10 years of annual screening, over half of all women receive a false-positive recall and 7–9% have a false-positive biopsy.35
Furthermore, in the wake of the CNBCSS, information concerning overdiagnosis is increasingly available to women,36 undermining their confidence in screening. Women given controlled, qualitative, and quantitative education on the risks of overdiagnosis have less positive attitudes about screening and demonstrate reduced intent to screen.37 Similarly, primary care physicians, key influencers in a woman’s screening decisions, are far less willing to refer patients 40–49 for screening when fully educated about the potential risks/benefits of screening.38
Further, our conflicting recommendations have made this divisive debate a public one, sowing distrust and a deepening confusion for women over how to prevent the disease that scares them the most.39, 40
The question we need to be asking, therefore, is not whether we should screen more or less, earlier or later. It is how can we make screening better for women, reduce false-positive recalls and improve our ability to more accurately prevent and detect clinically significant cancers sufficiently early. This is, after all, is what women tell us they want41 and what we have observed to date in the WISDOM trial (described below).
The answer is simply that we must move on. We must begin developing and testing new and better approaches that respond to women’s needs. Fortunately, in this respect, there is one thing upon which we all agree—women must have the opportunity to make informed screening choices.
Individualized, informed choice
Overwhelmingly, women want information about their personal risk of breast cancer.42, 43 Currently, only 10% have accurate perceptions of their personal risk and 40% have never discussed their personal breast cancer risk with a doctor.44 Yet, a realistic view of their risk is prerequisite to making informed screening decisions.
We have the tools to better inform women of their personal risk, through well characterized models that incorporate family history and breast density, endocrine exposures, gene mutations, and atypia,45,46,47,48,49 along with a number of common gene variants.50, 51 They teach us that not all women whom we classify for screening purposes as “average-risk,” actually have the same lifetime risk of breast cancer. Armed with a better understanding of their individual risk, such women will expect—and demand—screening recommendations commensurate with their personal risk.
Unless we are prepared to ignore the modern tools available to us, we are therefore compelled to shepherd breast cancer screening into the era of precision medicine. Now is the time to begin evaluating a patient-centric model, focusing on individually tailored recommendations on when to start, when to stop, and how often to screen, depending upon a woman’s personal risk. Only through clinical testing can we establish the evidence that tells us how best to apply risk.
The idea of risk-based screening is not revolutionary. In fact, we already do it, although in a crude fashion. It is standard of practice for high risk women with mutations in the BRCA genes and first degree relatives from high risk families to begin screening at a much earlier age, and to do so more frequently (annual mammogram alternating with annual MRI).52 But our understanding of breast cancer risk goes much further than our current screening recommendations reflect. Our failure to incorporate our current understanding of personal risk into our screening recommendations means we may be asking some women to accept risk/benefit ratios they might not be comfortable with if they were fully informed.
Within the context of well-designed, randomized, controlled clinical trials, we have the ability to investigate new screening models in a safe, systematic manner, beginning with conservative estimates that minimize the chances of misclassification of risk and avoid underscreening. If we are successful, it could help establish a new baseline for cancer screening, reduce confusion and anxiety for women over conflicting recommendations, improve women’s perception of their true risk of breast cancer, improve adherence,53 and reinforce confidence in providers. Perhaps most importantly, it allows us to learn who is at risk for what kind of cancer, and establish a cycle of continuous improvement in breast cancer screening.
Risk-based screening may or may not be the answer to all of screening’s shortcomings, but it is perhaps an answer to the current deadlock in which we find ourselves. In the words of philosopher David Hume, it is time “we start spilling our sweat, and not our blood.”
The WISDOM study: overcoming challenges
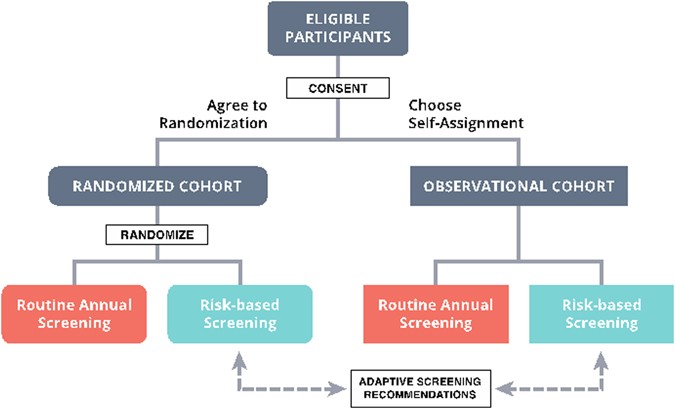
We have recently been awarded a grant from the Patient Centered Outcomes Research Institute (PCORI) to evaluate a risk-based screening approach within a pragmatic, controlled trial. The “WIDSOM” study (Women Informed to Screen Depending On Measures of risk) is a multicenter trial comparing risk-based screening to annual screening in 100,000 women aged 40–74, initially opening in the Athena Breast Health Network in California and the Midwest (Table 1). The study is a “preference tolerant design” (Fig. 1) that encourages women to be randomized (n~ 65,000) but also allows self-assignment for those with strong personal preference for either annual or risk-based screening (a pilot study was conducted in 2015 in which 74% of women agreed to randomization). Importantly, WISDOM is an adaptive design, allowing us to learn and adjust, continuing to improve the risk-assessment and screening recommendation models over the course of the trial.
An essential aspect of developing WISDOM has been the engagement of all stakeholders, including consumers, policy makers/guideline organizations and multiple specialties, and payers, to agree upfront on metrics for success. This ensures the trial remains relevant to the needs of the end-user and sets the stage for rapid adoption should it prove successful.
Patients and advocates in particular, through the Athena Consumer and Community Advisory Committee, have been key partners in WISDOM since its conception. The preference-tolerant design that allows all women to participate regardless if they have strong personal reservations about being randomized grew from vigorous discussions with this group. The consumer voice is deeply embedded in WISDOM, with influence in all aspects of study design and planning, including enrollment strategies, consent processes, primary care physician outreach and education, risk notification, and participant retention.
The buy-in of health care payers is essential to enable rapid dissemination once results are presented. Modeling shows a risk-based strategy will be more cost effective in terms of screening, but requires an initial outlay of resources for one-time genetic testing and comprehensive risk assessments. After almost 2 years of discussion and negotiation, WISDOM’s “Payer Working Group”, led by Blue Shield of California and including all insurers in California, has reached an agreement to implement a “Coverage with Evidence Development” model to cover clinical costs not funded through PCORI.54 This model allows innovative treatment approaches to be tested transparently. The use of a coverage model that fosters the development of evidence, using a coverage with trial participation approach, allows agreement on metrics for adoption and should shorten the timeline for adoption. By engaging payers early, should the study prove successful, we will have laid the foundations to address future challenges in implementation related to standard coverage. We are in the process of engaging other payers.
The most formidable challenge in terms of stakeholders in developing a trial of risk-based screening, given the voracity of the academic debate, lies within the academic community. Among the highest priorities has been to define acceptable parameters of risk assessment, stratification and screening recommendations. We have also invested considerable effort to reach consensus regarding what constitutes success. WISDOM’s ‘Risk Thresholds Group’ and ‘Primary Care Physician Working Group,” consisting of primary care teams, representatives of the radiology community and others have shared these tasks.
Risk assessments and recommendations
The Breast Cancer Surveillance Consortium (BCSC) model was selected as the foundation of individual risk assessments for WISDOM, based on its accuracy, ease of implementation, its large (>1 million women) multiethnic target population, and incorporation of ethnicity and breast density as risk factors.55, 56 Additional assessments also include polygenic risk based on nearly 200 SNPs, as well as a 9 high-penetrance gene mutation panel.
In translating individual risk to screening recommendations, the primary consideration of the working groups was to develop guidelines that were sufficiently conservative to minimize risk of potential harm from underscreening, yet progressive enough to minimize potential harm from overdiagnosis, while permitting outcome measures with sufficient study power. The consensus risk stratification and related screening recommendations to be employed within WISDOM are shown in Table 2, and include more frequent screening for those at highest risk or those at risk for faster growing (e.g., hormone negative) cancer. In the risk-based assessment arm, no woman will receive a recommendation for less screening than current USPSTF guidelines—individual risk ≥ 1.3% over 5 years initiates screening. Because the uptake of risk-reducing interventions has been very poor despite level 1 evidence of benefit, we will use a stringent threshold (top 2.5 percentile of risk for breast cancer by age group, or lifetime risk in the range of 30% or higher) for identifying participants to target and counsel about endocrine risk-reducing therapy. The gene-based tests also inform the risk for hormone positive or negative breast cancer and impact screening and prevention recommendations. Additional details on the rationale and evidence used to develop this model are published elsewhere.57, 58
A shared decision-making (e-prognosis) tool based on recent modeling of comorbidity and impact of screening59 will be used to identify women unlikely to benefit from screening due to limited life expectancy. These rules will inform our risk assignments, age to start, age to stop, frequency, and appropriate modality of screening. The trial is designed to adapt over time, and refine categorization and screening frequency based on the actual cancer rate and biology of tumors that develop.
Defining success
If, after completing WISDOM, we are to avoid simply adding additional fuel to the fire of the screening debate, the scientific questions we ask must be well defined and the answers definitive. This is particularly challenging given the nature of the current debate and is further complicated by statistical requirements, population size limitations and the 5-year follow-up limitation of the funding. Such deliberations within WISDOM working groups strengthened the study significantly, emphasized safety as the overriding priority and established a series of outcomes with achievable and highly relevant goals.
WISDOM’s primary endpoints are, first, to determine whether risk-based screening is non-inferior to annual screening for late-stage cancers detected. The outcome is the number of Stage IIB or higher cancers found using personalized vs. annual screening. The study has been powered assuming annual incidence rates of 95 Stage IIB or higher cancers per 100,000 women in each arm.60 Over 65,000 randomized patients, this provides 90% power to detect a difference lower than 0.05% in risk of being diagnosed with Stage IIB or higher in the personalized vs. annual arm in a given year (83% for a difference <0.035%).58
Second, we will compare the morbidity of personalized vs. annual screening on the basis of the number of biopsies performed. Assuming 16% of first time mammograms and 8% of subsequent screens lead to false positive recalls,61 65,000 patients equally randomized between annual and personalized screening offers 90% power to detect a difference as small as 1.1% (22 vs. 20.9%).
Additional secondary objectives will further our understanding of the impacts of personalized screening and include measures of morbidity (e.g., rates of systemic therapy, rates of DCIS, chemoprevention) and the comparative attitudes and acceptance of each screening modality by women enrolled in the trial (e.g., adherence, measures of anxiety, decision regret). Finally, we will determine whether an understanding of personalized risk, especially the ability to predict hormone positive breast cancer, will provide better motivation for and uptake of endocrine risk reducing therapies and lifestyle changes.
Since opening in September 2016, Over 4000 women have enrolled in WISDOM. About two-thirds have agreed to randomization. The other one-third opted to self-select their screening approach in the observation arm: 85% have elected for personalized screening. Although preliminary, our experience to date provides critical insight into the comfort women feel with the concept of individualized, risk-based screening.
Conclusions
The United States is the only country where annual screening starting at age 40 is standard practice, yet our breast cancer mortality rate is no better than countries that screen less.62 Clearly, there is room for improvement. Progress will only come by investigating other possibilities. The WISDOM study will evaluate one such possibility—screening based on a woman’s individual risk—opening its first site in August 2016, expanding to other sites nationally in 2017. It is certainly unlikely that all women benefit equally from screening. Investing in pragmatic studies like WISDOM allows us to learn who is at risk for what kind of breast cancer, tailor screening accordingly and build a new framework for continuous improvement.
References
Nyström, L. et al. Breast cancer screening with mammography: overview of Swedish randomised trials. Lancet 341, 973–978 (1993).
Shapiro, S. Periodic screening for breast cancer: the HIP Randomized Controlled Trial. Health Insurance Plan. JNCI Monogr. 1997, 27–30 (1997).
Nyström, L. et al. Long-term effects of mammography screening: updated overview of the Swedish randomised trials. Lancet 359, 909–919 (2002).
Tabár, L., Duffy, S. W. & Chen, H. H. Re: Quantitative interpretation of age-specific mortality reductions from the Swedish breast cancer-screening trials. JNCI J. 88, 52–55 (1996).
Nelson, H. D. et al. Harms of breast cancer screening: systematic review to update the 2009 U.S. preventive services task force recommendation. Ann. Intern. Med. 164, 256 (2016).
Gøtzsche, P. C. & Jørgensen, K. J. Screening for breast cancer with mammography. Cochrane Database Syst. Rev. 156, 193 (1996).
Shieh, Y. et al. Population-based screening for cancer: hope and hype. Nat. Rev. Clin. Oncol. 13, 550–565 (2016).
National Institutes of Health Consensus Development Conference Statement. Breast cancer screening for women ages 40–49. Natl. Inst. Health Consens. Dev. Panel 89, 1015–1026 (1997).
Esserman, L., Shieh, Y. & Thompson, I. Rethinking screening for breast cancer and prostate cancer. JAMA 302, 1685–1692 (2009).
Nelson, H. D. et al. Screening for breast cancer: an update for the U.S. Preventive Services Task Force. Ann. Intern. Med. 151, 727–37 (2009).
Siu, A. L. On behalf of the U.S. Preventive services task force. Screening for breast cancer: U.S. Preventive services task force recommendation statement. Ann. Intern. Med. 164, 279 (2016).
Mainiero, M. B. et al. ACR appropriateness criteria breast cancer screening. J. Am. Coll. Radiol. 13, R45–R49 (2016).
Oeffinger, K. C. et al. Breast cancer screening for women at average risk: 2015 guideline update from the American Cancer Society. JAMA 314, 1599–1614 (2015).
Miller, A. B. et al. Twenty five year follow-up for breast cancer incidence and mortality of the Canadian National Breast Screening Study: randomised screening trial. BMJ 348, g366 (2014).
Esserman, L. J. et al. Impact of mammographic screening on the detection of good and poor prognosis breast cancers. Breast Cancer Res. Treat. 130, 725–734 (2011).
Welch, H. G. & Black, W. C. Overdiagnosis in cancer. JNCI J. 102, 605–613 (2010).
Marmot, M. G. et al. The benefits and harms of breast cancer screening: an independent review. Br. J. Cancer 108, 2205–2240 (2013).
Pace, L. E. & Keating, N. L. A systematic assessment of benefits and risks to guide breast cancer screening decisions. JAMA 311, 1327–1335 (2014).
Duffy, S. W. et al. Overdiagnosis and overtreatment of breast cancer: estimates of overdiagnosis from two trials of mammographic screening for breast cancer. Breast Cancer Res. 7, 258–265 (2005).
Paci, E. & Duffy, S. Overdiagnosis and overtreatment of breast cancer: overdiagnosis and overtreatment in service screening. Breast Cancer Res. 7, 266 (2005).
Kopans, D. B. Arguments against mammography screening continue to be based on faulty science. Oncologist 19, 107–112 (2014).
Bleyer, A. Were our estimates of overdiagnosis with mammography screening * in the united states “based on faulty science”? Oncologist 19, 113–126 (2014).
Kaniklidis, C. Beyond the mammography debate: a moderate perspective. Curr. Oncol. 22, 220 (2015).
Yaffe, M. J. Response to: ‘Beyond the mammography debate: a moderate perspective’. Curr. Oncol. 22, e401–3 (2015).
Esserman, L. J., Yau, C., Thompson, C. K., van t Veer, L. J., Borowsky, A. D., Hoadley, K. A., et al. Use of Molecular Tools to Identify Patients With Indolent Breast Cancers With Ultralow Risk Over 2 Decades. JAMA Oncology. http://doi.org/10.1001/jamaoncol.2017.1261 (2017).
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 365, 1687–1717 (2005).
Berry, D. A. et al. Effect of screening and adjuvant therapy on mortality from breast cancer. N. Engl. J. Med. 353, 1784–1792 (2005).
Kalager, M., Zelen, M., Langmark, F. & Adami, H.-O. Effect of Screening mammography on breast-cancer mortality in Norway. N. Engl. J. Med. 363, 1203–1210 (2010).
Dowsett, M. et al. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J. Clin. Oncol. 28, 509–518 (2010).
Bond, M. et al. Systematic review of the psychological consequences of false-positive screening mammograms. Health Technol. Assess. 17, 1–170 (2013).
Klompenhouwer, E. G. et al. Re-attendance at biennial screening mammography following a repeated false positive recall. Breast Cancer Res. Treat. 145, 429–437 (2014).
McCann, J., Stockton, D. & Godward, S. Impact of false-positive mammography on subsequent screening attendance and risk of cancer. Breast Cancer Res. 4, 954 (2002).
DeFrank, J. T. et al. Influence of false-positive mammography results on subsequent screening: do physician recommendations buffer negative effects? J. Med. Screen. 19, 35–41 (2012).
Dabbous, F. M. et al. Impact of a false-positive screening mammogram on subsequent screening behavior and stage at breast cancer diagnosis. Cancer Epidemiol. Biomark. Prev. 26, 397–403 (2017).
Elmore, J. G. et al. Ten-year risk of false positive screening mammograms and clinical breast examinations. N. Engl. J. Med. 338, 1089–1096 (1998).
Ghanouni, A. et al. Information on ‘overdiagnosis’ in breast cancer screening on prominent united kingdom- and Australia-oriented health websites. PLoS ONE 11, e0152279 (2016).
Hersch, J. et al. Use of a decision aid including information on overdetection to support informed choice about breast cancer screening: a randomised controlled trial. Lancet 385, 1642–1652 (2015).
Bryan, T., Castiglioni, A., Estrada, C. & Snyder, E. Impact of an educational intervention on provider knowledge, attitudes, and comfort level regarding counseling women ages 40–49 about breast cancer screening. JMDH 8, 209–216 (2015).
USPSTF. Breast Cancer Screening Recommendations Could Endanger Women. http://www.acr.org/About-Us/Media-Center/Press-Releases/2016-Press-Releases/20160111-USPSTF-Breast-Cancer-Screening-Recommendations-Could-Endanger-Women. Accessed 10 May 2016 (2016).
Squiers, L. B. et al. The Public’s Response to the U.S. Preventive services task force’s 2009 recommendations on mammography screening. Am. J. Prev. Med. 40, 497–504 (2011).
The Society for Women’s Health Research. What Women Want: Expectations and Experiences in Breast Cancer Screening. http://swhr.org/science/swhr-mammography-survey/. Accessed 10 May 2016 (2014).
Fisher, B. A., Wilkinson, L. & Valencia, A. Women’s interest in a personal breast cancer risk assessment and lifestyle advice at NHS mammography screening. J. Public. Health. 39, 1–9 (2016).
Sphingotec, L. L. C. Survey: Majority of Women Do Not Discuss Breast Cancer Risks or Mammography Screening Limitations with Healthcare Providers. http://www.marketwired.com/press-release/survey-majority-women-do-not-discuss-breast-cancer-risks-mammography-screening-limitations-2016448.htm. Accessed 15 June 2016 (2015).
Printz, C. Most women have an inaccurate perception of their breast cancer risk. Cancer 120, 314–315 (2014).
Costantino, J. P. et al. Validation studies for models projecting the risk of invasive and total breast cancer incidence. JNCI J. 91, 1541–1548 (1999).
Parmigiani, G., Berry, D. & Aguilar, O. Determining carrier probabilities for breast cancer-susceptibility genes BRCA1 and BRCA2. Am. J. Hum. Genet. 62, 145–158 (1998).
Tyrer, J., Duffy, S. W. & Cuzick, J. A breast cancer prediction model incorporating familial and personal risk factors. Stat. Med. 23, 1111–1130 (2004).
Claus, E. B. Risk models used to counsel women for breast and ovarian cancer: a guide for clinicians. Fam. Cancer 1, 197–206 (2001).
Ozanne, E. M., Howe, R., Omer, Z. & Esserman, L. J. Development of a personalized decision aid for breast cancer risk reduction and management. BMC Med. Inform. Decis. Mak. 14, 4 (2014).
Darabi, H. et al. Breast cancer risk prediction and individualised screening based on common genetic variation and breast density measurement. Breast Cancer Res. 14, R25 (2012).
Mavaddat, N. et al. Prediction of breast cancer risk based on profiling with common genetic variants. JNCI 107, djv036–djv036 (2015).
Daly, M. B. et al. Genetic/Familial High-Risk Assessment: Breast and Ovarian, Version 1.2017. NCCN Clinical Practice Guidelines in Oncology (NCCN, 2016).
Edwards, A., Unigwe, S., Elwyn, G. & Hood, K. Effects of communicating individual risks in screening programmes: cochrane systematic review. BMJ 327, 703–709 (2003).
Rosenberg-Wohl, S., Thygeson, M., Fiscalini, A. S. et al. Private payer participation in coverage with evidence development: a case study. Health Affairs, March 14 (2017).
Tice, J. A. et al. Using clinical factors and mammographic breast density to estimate breast cancer risk: development and validation of a new predictive model. Ann. Intern. Med. 148, 337–347 (2008).
Vachon, C. M. et al. The contributions of breast density and common genetic variation to breast cancer risk. JNCI J. 107, 397–400 (2015).
Shieh, Y., Eklund, M., Madlensky, L., Sawyer, S. D., Thompson, C. K., Stover Fiscalini, A., et al. Breast Cancer Screening in the Precision Medicine Era: Risk-Based Screening in a Population-Based Trial. JNCI J. 109, djw290 (2017).
Eklund, M., Broglio, C., Yau, C., Conner, J., Esserman, L. The WISDOM Breast Cancer Personalized Screening Trial: Design by System Analysis (submitted to JNCI).
Lee, S. J., Smith, A. S., Widera, E. Cancer screening. ePrognosis (online). https://eprognosis.ucsf.edu. Accessed 30 March 2017.
Esserman, L. J. et al. Biologic markers determine both the risk and the timing of recurrence in breast cancer. Breast Cancer Res. Treat. 129, 607–616 (2011).
Kerlikowske, K. et al. Outcomes of screening mammography by frequency, breast density, and postmenopausal hormone therapy. JAMA Intern. Med. 173, 807–816 (2013).
Jemal, A., Center, M. M., DeSantis, C. & Ward, E. M. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol. Biomark. Prev. 19, 1893–1907 (2010).
Author information
Authors and Affiliations
Author notes
Members of the Wisdom Study and Athena Investigators are listed before references.
Consortia
Contributions
The ideas that are expressed in this commentary are the result of a multiyear collaboration of the Athena Breast Health Network and resulted in the submission of a grant proposal to the Patient Centered Research Outcomes group. The first author summarized the work and the rationale for the proposed WISDOM study in the form of this commentary. All of the wisdom study and Athena investigators have read the manuscript and provided comments and have certified that the commentary captures the scientific basis underlying and the rationale for opening the WISDOM study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Esserman, L.J., the WISDOM Study and Athena Investigators. The WISDOM Study: breaking the deadlock in the breast cancer screening debate. npj Breast Cancer 3, 34 (2017). https://doi.org/10.1038/s41523-017-0035-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-017-0035-5
This article is cited by
-
Recent advances in polygenic scores: translation, equitability, methods and FAIR tools
Genome Medicine (2024)
-
The risk-based breast screening (RIBBS) study protocol: a personalized screening model for young women
La radiologia medica (2024)
-
Artificial intelligence in the treatment of cancer: Changing patterns, constraints, and prospects
Health and Technology (2024)
-
Impact of a risk based breast screening decision aid on understanding, acceptance and decision making
npj Breast Cancer (2023)
-
Polygenic scores in cancer
Nature Reviews Cancer (2023)