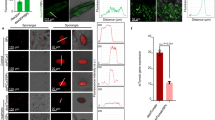
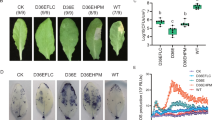
Abstract
The root-knot nematode Meloidogyne chitwoodi is a pest that affects potato production in the Pacific Northwest of the United States. Here, to develop new strategies against M. chitwoodi infection of potato, we engineered Bacillus subtilis to secrete the plant-defence elicitor peptide StPep1. Pre-treatment of potato roots with the bacteria secreting StPep1 substantially reduced root galling, indicating that a bacterial secretion of a plant elicitor is an effective strategy for plant protection.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated in the study are either included in this publication or available from the corresponding author on reasonable request.
References
European and Mediterranean Plant Protection Organization. Meloidogyne chitwoodi and Meloidogyne fallax. EPPO Bull. 39, 5–17 (2009).
Santo, G. S., O’Bannon, J. H., Finley, A. M. & Golden, A. M. Occurrence and host range of a new root-knot nematode (Meoidogyne chitwoodi) in the Pacific Northwest. Plant Dis. 64, 951–952 (1980).
Mojtahedi, H., Brown, C. R., Riga, E. & Zhang, L. H. A new pathotype of Meloidogyne chitwoodi race 1 from Washington state. Plant Dis. 91, 1051 (2007).
Quintana-Rodriguez, E., Duran-Flores, D., Heil, M. & Camacho-Coronel, X. Damage-associated molecular patterns (DAMPs) as future plant vaccines that protect crops from pests. Sci. Hort. 237, 207–220 (2018).
Huffaker, A., Dafoe, N. J. & Schmelz, E. A. ZmPep1, an ortholog of Arabidopsis elicitor peptide 1, regulates maize innate immunity and enhances disease resistance. Plant Physiol. 155, 1325–1338 (2011).
Huffaker, A., Pearce, G. & Ryan, C. A. An endogenous peptide signal in Arabidopsis activates components of the innate immune response. Proc. Natl Acad. Sci. USA 103, 10098–10103 (2006).
Huffaker, A. et al. Plant elicitor peptides are conserved signals regulating direct and indirect antiherbivore defense. Proc. Natl Acad. Sci. USA 110, 5707–5712 (2013).
Liu, Z. et al. BIK1 interacts with PEPRs to mediate ethylene-induced immunity. Proc. Natl Acad. Sci. USA 110, 6205–6210 (2013).
Lori, M. et al. Evolutionary divergence of the plant elicitor peptides (Peps) and their receptors: interfamily incompatibility of perception but compatibility of downstream signalling. J. Exp. Bot. 66, 5315–5325 (2015).
Tintor, N. et al. Layered pattern receptor signaling via ethylene and endogenous elicitor peptides during Arabidopsis immunity tobacterial infection. Proc. Natl Acad. Sci. USA 110, 6211–6216 (2013).
Huffaker, A. & Ryan, C. A. Endogenous peptide defense signals in Arabidopsis differentially amplify signaling for the innate immune response. Proc. Natl Acad. Sci. USA 104, 10732–10736 (2007).
Lee, M. W. et al. Plant elicitor peptides promote plant defences against nematodes in soybean. Mol. Plant Pathol. 19, 858–869 (2018).
Warnock, N. D. et al. Nematode neuropeptides as transgenic nematicides. PLoS Pathol. 13, e1006237 (2017).
Karasov, T. L., Chae, E., Herman, J. J. & Bergelson, J. Mechanisms to mitigate the trade-off between growth and defense. Plant Cell 29, 666 (2017).
Holmes, D. R., Grubb, L. E. & Monaghan, J. The jasmonate receptor COI1 is required for AtPep1-induced immune responses in Arabidopsis thaliana. BMC Res. Notes 11, 555 (2018).
Wiesel, L. et al. A transcriptional reference map of defence hormone responses in potato. Sci. Rep. 5, 15229 (2015).
Poncini, L. et al. In roots of Arabidopsis thaliana, the damage-associated molecular pattern AtPep1 is a stronger elicitor of immune signalling than flg22 or the chitin heptamer. PLoS ONE 2017, e0185808 (2017).
Halim, V. A. et al. PAMP-induced defense responses in potato require both salicylic acid and jasmonic acid. Plant J. 57, 230–242 (2009).
Bartels, S. & Boller, T. Quo vadis, Pep? Plant elicitor peptides at the crossroads of immunity, stress, and development. J. Exp. Bot. 66, 5183–5193 (2015).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408 (2001).
Acknowledgements
We acknowledge support by Washington State University and by the United States Department of Agriculture National Institute of Food and Agriculture, Hatch umbrella project no. 1015621. We thank S. Rosahl for the StCOI1‐RNAi lines; R. Navarre for the NahG potato plants; C. Brown for the M. chitwoodi Roza pathotype; C. Xia and Q. Sun for help with the RNA-seq analyses; L. Thomashow and M. Yang for help with B. subtilis extraction and enumeration; S. Izaguirre and M. De Leon for their help with growing potato plants and for their assistance in the laboratory; and Z. Dubois for illustration services.
Author information
Authors and Affiliations
Contributions
L.Z. designed and performed experiments. L.Z. and C.G. analysed data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Plants thanks Shahid Siddique and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods and Figs. 1–10.
Supplementary Tables
Supplementary Table 1: list of differentially regulated genes identified in the RNA-seq analysis in this study (6 h/0 h). Supplementary Table 2: list of primers used in this study. Supplementary Table 3: statistics and reproducibility—exact P values for Figs. 1 and 2, and Supplementary Figs. 1, 2, 5 and 8–10.
Rights and permissions
About this article
Cite this article
Zhang, L., Gleason, C. Enhancing potato resistance against root-knot nematodes using a plant-defence elicitor delivered by bacteria. Nat. Plants 6, 625–629 (2020). https://doi.org/10.1038/s41477-020-0689-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-020-0689-0
This article is cited by
-
Small secreted peptides (SSPs) in tomato and their potential roles in drought stress response
Molecular Horticulture (2023)
-
Root-Knot Nematodes (Meloidogyne spp.): Biology, Plant-Nematode Interactions and Their Environmentally Benign Management Strategies
Gesunde Pflanzen (2023)
-
Plant immunity inducers: from discovery to agricultural application
Stress Biology (2022)
-
New allies to fight worms
Nature Plants (2020)