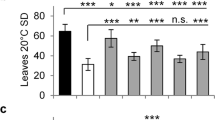
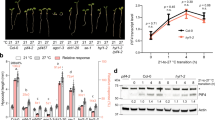
Abstract
Post-embryonic development and longevity of flowering plants are, for a large part, determined by the activity and maturation state of stem cell niches formed in the axils of leaves, the so-called axillary meristems (AMs)1,2. The genes that are associated with AM maturation and underlie the differences between monocarpic (reproduce once and die) annual and the longer-lived polycarpic (reproduce more than once) perennial plants are still largely unknown. Here we identify a new role for the Arabidopsis AT-HOOK MOTIF NUCLEAR LOCALIZED 15 (AHL15) gene as a suppressor of AM maturation. Loss of AHL15 function accelerates AM maturation, whereas ectopic expression of AHL15 suppresses AM maturation and promotes longevity in monocarpic Arabidopsis and tobacco. Accordingly, in Arabidopsis grown under longevity-promoting short-day conditions, or in polycarpic Arabidopsis lyrata, expression of AHL15 is upregulated in AMs. Together, our results indicate that AHL15 and other AHL clade-A genes play an important role, directly downstream of flowering genes (SOC1, FUL) and upstream of the flowering-promoting hormone gibberellic acid, in suppressing AM maturation and extending the plant’s lifespan.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All processed data are contained in the manuscript, the Extended Data or the Supplementary Information. Raw data and materials generated during this study are available upon reasonable request.
References
Grbić, V. & Bleecker, A. B. Axillary meristem development in Arabidopsis thaliana. Plant J. 21, 215–223 (2000).
Wang, B., Smith, S. M. & Li, J. Genetic regulation of shoot architecture. Ann. Rev. Plant Biol. 69, 437–468 (2018).
Park, S. J., Eshed, Y. & Lippman, Z. B. Meristem maturation and inflorescence architecture—lessons from the Solanaceae. Curr. Opin. Plant Biol. 17, 70–77 (2014).
Munné-Bosch, S. Do perennials really senesce? Trends Plant Sci. 13, 216–220 (2008).
Amasino, R. Floral induction and monocarpic versus polycarpic life histories. Genome Biol. 10, 228 (2009).
Melzer, S. et al. Flowering-time genes modulate meristem determinacy and growth form in Arabidopsis thaliana. Nat. Genet. 40, 1489–1492 (2008).
Kiefer, C. et al. Divergence of annual and perennial species in the Brassicaceae and the contribution of cis-acting variation at FLC orthologues. Mol. Ecol. 26, 3437–3457 (2017).
Zhao, J., Favero, D. S., Peng, H. & Neff, M. M. Arabidopsis thaliana AHL family modulates hypocotyl growth redundantly by interacting with each other via the PPC/DUF296 domain. Proc. Natl Acad. Sci. USA 110, E4688–E4697 (2013).
Street, I. H., Shah, P. K., Smith, A. M., Avery, N. & Neff, M. M. The AT-hook-containing proteins SOB3/AHL29 and ESC/AHL27 are negative modulators of hypocotyl growth in Arabidopsis. Plant J. 54, 1–14 (2008).
Xiao, C., Chen, F., Yu, X., Lin, C. & Fu, Y.-F. Over-expression of an AT-hook gene, AHL22, delays flowering and inhibits the elongation of the hypocotyl in Arabidopsis thaliana. Plant Mol. Biol. 71, 39–50 (2009).
Ng, K.-H., Yu, H. & Ito, T. AGAMOUS controls GIANT KILLER, a multifunctional chromatin modifier in reproductive organ patterning and differentiation. PLoS Biol. 7, e1000251 (2009).
Yun, J., Kim, Y.-S., Jung, J.-H., Seo, P. J. & Park, C.-M. The AT-hook motif-containing protein AHL22 regulates flowering initiation by modifying FLOWERING LOCUS T chromatin in Arabidopsis. J. Biol. Chem. 287, 15307–15316 (2012).
Andrés, F. & Coupland, G. The genetic basis of flowering responses to seasonal cues. Nat. Rev. Genet. 13, 627–639 (2012).
Ko, J., Han, K., Park, S. & Yang, J. Plant body weight-induced secondary growth in Arabidopsis and its transcription phenotype revealed by whole-transcriptome profiling. Plant Physiol. 135, 1069–1083 (2004).
Bergonzi, S. et al. Mechanisms of age-dependent response to winter temperature in perennial flowering of Arabis alpina. Science 340, 1094–1097 (2013).
Zhou, C.-M. et al. Molecular basis of age-dependent vernalization in Cardamine flexuosa. Science 340, 1097–1100 (2013).
Lee, J. & Lee, I. Regulation and function of SOC1, a flowering pathway integrator. J. Exp. Bot. 61, 2247–2254 (2010).
Immink, R. G. H. et al. Characterization of SOC1’s central role in flowering by the identification of its upstream and downstream regulators. Plant Physiol. 160, 433–449 (2012).
Tao, Z. et al. Genome-wide identification of SOC1 and SVP targets during the floral transition in Arabidopsis. Plant J. 70, 549–561 (2012).
Matsoukas, I. G., Massiah, A. J. & Thomas, B. Florigenic and antiflorigenic signaling in plants. Plant Cell Physiol. 53, 1827–1842 (2012).
Turnbull, C. Long-distance regulation of flowering time. J. Exp. Bot. 62, 4399–43413 (2011).
Song, Y. H., Ito, S. & Imaizumi, T. Flowering time regulation: photoperiod- and temperature-sensing in leaves. Trends Plant Sci. 18, 575–583 (2013).
Andrés, F. et al. SHORT VEGETATIVE PHASE reduces gibberellin biosynthesis at the Arabidopsis shoot apex to regulate the floral transition. Proc. Natl Acad. Sci. USA 111, E2760–E2679 (2014).
Yu, S. et al. Gibberellin regulates the Arabidopsis floral transition through miR156-targeted SQUAMOSA promoter binding-like transcription factors. Plant Cell 24, 3320–3332 (2012).
Wang, J. W. Regulation of flowering time by the miR156-mediated age pathway. J. Exp. Bot. 5, 4723–4730 (2014).
Matsushita, A., Furumoto, T., Ishida, S. & Takahashi, Y. AGF1, an AT-hook protein, is necessary for the negative feedback of AtGA3ox1 encoding GA 3-oxidase. Plant Physiol. 143, 1152–1162 (2007).
Huang, S. et al. Overexpression of 20-Oxidase confers a gibberellin-overproduction phenotype in Arabidopsis. Plant Physiol. 118, 773–781 (1998).
Rieu, I. et al. The gibberellin biosynthetic genes AtGA20ox1 and AtGA20ox2 act, partially redundantly, to promote growth and development throughout the Arabidopsis life cycle. Plant J. 53, 488–504 (2008).
Tilmes, V. et al. Gibberellins act downstream of Arabis PERPETUAL FLOWERING1 to accelerate floral induction during vernalization. Plant Physiol. 180, 1549–1563 (2019).
Zhao, J., Favero, D. S., Qiu, J., Roalson, E. H. & Neff, M. M. Insights into the evolution and diversification of the AT-hook Motif Nuclear Localized gene family in land plants. BMC Plant Biol. 14, 266 (2014).
Remington, D. L., Figueroa, J. & Rane, M. Timing of shoot development transitions affects degree of perenniality in Arabidopsis lyrata (Brassicaceae). BMC Plant Biol. 15, 1–13 (2015).
Lim, P. O. et al. Overexpression of a chromatin architecture-controlling AT-hook protein extends leaf longevity and increases the post-harvest storage life of plants. Plant J. 52, 1140–1153 (2007).
Dekker, J., Marti-Renom, M. A. & Mirny, L. A. Exploring the three-dimensional organization of genomes: interpreting chromatin interaction data. Nat. Rev. Genet. 14, 390–403 (2013).
Wang, J.-W., Czech, B. & Weigel, D. miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138, 738–749 (2009).
Karimi, M., Depicker, A. & Hilson, P. Recombinational cloning with plant gateway vectors. Plant Physiol. 145, 1144–1154 (2007).
Passarinho, P. et al. BABY BOOM target genes provide diverse entry points into cell proliferation and cell growth pathways. Plant Mol. Biol. 68, 225–237 (2008).
Becker, D., Kemper, E., Schell, J. & Masterson, R. New plant binary vectors with selectable markers located proximal to the left T-DNA border. Plant Mol. Biol. 20, 1195–1197 (1992).
Den Dulk-Ras, A. & Hooykaas J. P. Electroporation of Agrobacterium tumefaciens. Methods Mol. Biol. 55, 63–72 (1995).
Clough, S. J. & Bent, aF. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Baltes, N. J., Gil-Humanes, J., Cermak, T., Atkins, P. & Voytas, D. F. DNA replicons for plant genome engineering. Plant Cell 26, 151163 (2014).
Murashige, T. & Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 15, 473–497 (1962).
Doyle, J. J. Isolation of plant DNA from fresh tissue. Focus 12, 13–15 (1990).
Anandalakshmi, R. et al. A viral suppressor of gene silencing in plants. Proc. Natl Acad. Sci. USA 95, 13079–13084 (1998).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 29, e45 (2001).
Mourik, H. Van, Muiño, J. M., Pajoro, A., Angenent, G. C. & Kaufmann, K. Characterization of in vivo DNA-binding eents of plant transcription factors by ChIP–seq: experimental potocol and computational analysis. Methods Mol. Biol 1284, 93–121 (2015).
Balanzà, V. et al. Genetic control of meristem arrest and life span in Arabidopsis by a FRUITFULL-APETALA2 pathway. Nat. Commun. 9, 565 (2018).
Bemer, M. et al. FRUITFULL controls SAUR10 expression and regulates Arabidopsis growth and architecture. J. Exp. Bot. 68, 3391–3403 (2017).
Altschup, S. F., Gish, W., Pennsylvania, T. & Park, U. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Katoh, K., Misawa, K., Kuma, K. & Miyata, T. MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 30, 3059–3066 (2002).
Capella-gutiérrez, S., Silla-martínez, J. M. & Gabaldón, T. TrimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 25, 1972–1973 (2009).
Eddy, S. R. Accelerated profile HMM searches. PLoS Comp. Biol. 7, e1002195 (2011).
Guindon, S. & Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 5, 696–704 (2003).
Ronquist, F. & Huelsenbeck, J. P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19, 1572–1574 (2003).
Liebert, M. A., Chen, K., Durand, D., Farach-colton, M. & Al, C. E. T. NOTUNG: a program for dating gene duplications. J. Comp. Biol. 7, 429–447 (2000).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Harmon, L. J. et al. GEIGER: investigating evolutionary radiations. Bioinformatics 24, 129–131 (2008).
Acknowledgements
We thank K. Boutilier and T. Greb for critical comments on the manuscript. O.K. was financially supported by a grant from the Iran Ministry of Science, Research and Technology (no. 89100156), and subsidies were provided by Generade and the Building Blocks of Life research programme (no. 737.016.013, to R.O.), which is (partly) financed by the Dutch Research Council (NWO). M.K. was financially supported by a fellowship of the Institute of Biotechnology & Genetic Engineering at the Agricultural University of Peshawar with financial support by the Higher Education Commission of Pakistan. R.R.H. was financially supported by the KU Leuven Research Fund. M.B. was supported by grant no. ALWOP.199, which is (partly) financed by NWO. P.M. and M.C. were supported in part by Innovation Subsidy Collaborative Projects (no. IS054064) from the Dutch Ministry of Economic Affairs.
Author information
Authors and Affiliations
Contributions
O.K. and R.O. conceived and supervised the project. All authors designed the experiments and analysed and interpreted the results. O.K. and A.R. performed the majority of the Arabidopsis experiments, with contributions from M.B., P.M. and M.C. M.K. generated and analysed the tobacco lines. R.R.H. and V.N. analysed the AHL gene families in mono- and polycarpic plant species. O.K. and R.O. wrote the manuscript. All authors read and commented on versions of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Plants thanks Richard Amasino and the other, anonymous, reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Expression of a dominant negative AHL15-ΔG mutant protein.
Expression of a dominant negative AHL15-ΔG mutant protein in the Arabidopsis ahl15 mutant background causes early flowering and impairs seed development. a. The schematic domain structure of AHL15 and the dominant negative AHL15-ΔG version, in which six-conserved amino-acids (Gly-Arg-Phe-Glu-Ile-Leu, red box) are deleted from the C-terminal PPC domain. b. Wild-type seed development in pAHL15:AHL15-ΔG siliques. c. Aberrant seed development (arrowheads) in ahl15/+ pAHL15:AHL15-ΔG siliques (observed in 3 independent pAHL15:AHL15-ΔG lines crossed with the ahl15 mutant). Similar results were obtained in three independent experiments. d, e. The number of rosette leaves produced by the SAM in wild-type, ahl15, pAHL15:AHL15-ΔG and ahl15/+ pAHL15:AHL15-ΔG plants grown in long day (LD, d) or short day (SD, e) conditions. Two independent transgenic lines (1 and 2) were used in each experiment. Dots in d and e indicate the rosette leaf number per plant (n=15 biologically independent plants), horizontal lines the mean and error bars the s.e.m. Letters (a, b, c) indicate statistically significant differences (P < 0.01), as determined by a one-way ANOVA with a Tukey’s HSD post hoc test. The P values can be found in the Supplementary Tables 6 and 7.
Extended Data Fig. 2 AHL15 and other clade A AHL genes represses AM maturation in Arabidopsis.
a, b. The number of rosette leaves produced per rosette AM of wild-type, ahl15, ahl15 pAHL15:AHL15, pAHL15:AHL15-ΔG and ahl15/+ pAHL15:AHL15-ΔG plants 5 (a), 9 (b), or 10 weeks (c) after germination in long day (LD, a,b) or short day (SD, c) conditions. d, e. The number of cauline leaves produced by rosette AMs (d) or by aerial AMs (e) of 7-week-old wild-type, ahl15, ahl15 pAHL15:AHL15, pAHL15:AHL15-ΔG and ahl15/+ pAHL15:AHL15-ΔG plants. Dots in a-e indicate rosette or cauline leaf number per AM per plant (n=15 biologically independent plants), horizontal lines the mean, and error bars the s.e.m. Letters (a, b, c) indicate statistically significant differences (P < 0.01), as determined by a one-way ANOVA with a Tukey’s HSD post hoc test. The P values are provided in Supplementary Tables 8–12. f. A lateral inflorescence with cauline leaves formed on the first inflorescence node of a wild-type (top) or ahl15/+ pAHL15:AHL15-ΔG plant (bottom). Similar results were obtained in three independent experiments.
Extended Data Fig. 3 Arabidopsis AHL genes enhance the vegetative activity and suppress the floral transition of rosette AMs.
a. Schematic representation of the vegetative activity of rosette AMs of six-week-old wild-type, ahl15 and ahl15/+ pAHL15:AHL15-ΔG-1 plants. Each row represents a single plant, and each square represents an individual AM in a cotyledon axil (C1 and C2) or in a rosette leaf axils (L1 to L12). The numbers within a square represent the number of rosette leaves produced by a rosette AM. A green square indicates a leaf axil with an active AM, as indicated by bud outgrowth or leaf development, and a white square indicates a leaf axil without an (active) AM. b. Developmental phase of the rosette AMs of six-week-old wild-type, ahl15 and ahl15/+ pAHL15:AHL15-ΔG-1 plants. White, yellow, green or red squares indicate axils without (active) AM, or rosette AMs with at least one visible leaf primordium, producing rosette leaves (vegetative) or producing cauline leaves or flowers (reproductive), respectively. Plants in a and b were grown in LD conditions.
Extended Data Fig. 4 AHL15 overexpression delays floral transition of the SAM and represses AM maturation.
a. Shoot phenotype of a flowering 7-week-old 35S:AHL15-GR plant that was DEX-treated upon bolting (5 weeks old). Similar results were obtained in four independent experiments. b, c. Number of rosette leaves (b) or cauline leaves (c) produced by rosette AMs of 7-week-old mock-treated wild-type, DEX-treated wild-type, mock-treated 35S:AHL15-GR and DEX-treated 35S:AHL15-GR plants. Plants were DEX-treated upon bolting (5 weeks old) and scored 2 weeks later. d. The number of rosette leaves produced by the SAM in mock-treated wild-type, DEX-treated wild-type, mock-treated 35S:AHL15-GR and DEX-treated 35S:AHL15-GR plants. Non-flowering (3-week-old) plants were treated and the SAM-produced rosette leaves were counted after bolting. Dots in b-d indicate number of leaves (per AM or SAM) per plant (n=15 biologically independent plants), horizontal lines the mean, and error bars the s.e.m. Letters (a, b, c) indicate statistically significant differences (P < 0.01), as determined by a one-way ANOVA with a Tukey’s HSD post hoc test. The P values are provided in Supplementary Tables 13–15. Plants were grown in LD conditions.
Extended Data Fig. 5 Overexpression of Arabidopsis AHL15 paralogs or putative orthologs represses AM maturation in Arabidopsis.
Overexpression of Arabidopsis AHL15 paralogs or putative orthologs represses AM maturation in Arabidopsis. (a and b) Wild-type (Col-0) or transgenic 7-week-old Arabidopsis plants overexpressing Arabidopsis AHL19, AHL20, AHL27 and AHL29 (a), or the putative AHL15 orthologs from Brassica oleracea (BoAHL15) or Medicago trunculata (MtAHL15) (b). For a and b similar results were obtained in two independent experiments. Plants were grown in LD conditions. For presentation purposes, the original background of the images was replaced by a homogeneous white background.
Extended Data Fig. 6 AHL15 enhances the longevity of short day-grown Arabidopsis plants.
Phenotype of 5-month-old wild-type (Col-0, left), ahl15 (middle) and ahl15 pAHL15:AHL15 (right) plants. The plants were grown in SD conditions. Similar results were obtained in two independent experiments. For presentation purposes, the original background of the image in was replaced by a homogeneous white background.
Extended Data Fig. 7 AHL15 expression is day length sensitive.
Expression of the pAHL15:GUS reporter in the axil of a cauline leaf (left, arrowheads), Young lateral inflorescence (middle) and rosette base (right) of a 9-week-old plant grown in LD conditions (top) or a 4-month-old plant grown under SD conditions (bottom). Similar results were obtained in two independent experiments.
Extended Data Fig. 8 AHL15 overexpression in the rosette base and leaf axils delays AM maturation in Arabidopsis.
a. Expression of pMYB58:GUS and pMYB103:GUS reporters in the rosette base (top) or leaf axils (bottom) of Arabidopsis plants respectively one or three weeks after flowering, as monitored by histochemical GUS staining. Similar results were obtained in two independent experiments. b, c. The number of cauline leaves produced by rosette AMs (b) or aerial AMs (c) of 6-week-old (b) or 7-week-old (c) wild-type, pMYB85:AHL15 or pMYB103:AHL15 plants grown in LD conditions. Dots in b and c indicate number of cauline leaves produced per AM per plant (n=15 biologically independent plants), horizontal lines indicate the mean, and error bars the s.e.m. Letters (a, b, c) indicate statistically significant differences (P < 0.01), as determined by a one-way ANOVA with a Tukey’s HSD post hoc test. The P values can be found in Supplementary Tables 16 and 17.
Extended Data Fig. 9 AHL15 overexpression promotes longevity in Arabidopsis and tobacco.
a. Renewed vegetative growth on aerial branches of a 5-month-old Arabidopsis 35S:AHL15-GR plant, 4 weeks after spraying with 20 μM DEX. Similar results were obtained in three independent experiments. b. Efficient production of leaves and inflorescences in a 3-year-old 35S::AHL15-GR tobacco plant, 3 weeks after treatment with 30 μM DEX, following 6 previous cycles of DEX-induced seed production. Similar results were obtained in two independent experiments. Plants in a and b were grown in LD conditions.
Extended Data Fig. 10 Expression of clade-A AHL genes in seedlings or in the rosette base of flowering Arabidopsis or A. lyrata plants.
a. Shoot phenotype of a 3-month-old Arabidopsis (upper panel) or a 4-month-old A. lyrata (lower panel) plant grown in LD conditions. Similar results were obtained in two independent experiments. b, c. qPCR analysis of the expression of clade-A AHL genes in 2-week-old seedlings or in the rosette base of 2-month-old flowering plants of A. thaliana (b) or A. lyrata (c). Dots in b and c indicate relative expression levels per experiment (n=3 biologically independent replicates), bars indicate the mean, and error bars indicate the s.e.m. Asterisks indicate significant differences from mock-treated plants (* p<0.05, ** p<0.01, *** p<0.001), as determined by a two-sided Student’s t-test.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, and Tables 1 and 2.
Supplementary Tables 3–19.
Statistics corresponding to figures.
Rights and permissions
About this article
Cite this article
Karami, O., Rahimi, A., Khan, M. et al. A suppressor of axillary meristem maturation promotes longevity in flowering plants. Nat. Plants 6, 368–376 (2020). https://doi.org/10.1038/s41477-020-0637-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-020-0637-z
This article is cited by
-
ASAP: a platform for gene functional analysis in Angelica sinensis
BMC Genomics (2024)
-
Widely conserved AHL transcription factors are essential for NCR gene expression and nodule development in Medicago
Nature Plants (2023)
-
Natural variation of Dt2 determines branching in soybean
Nature Communications (2022)
-
An Arabidopsis AT-hook motif nuclear protein mediates somatic embryogenesis and coinciding genome duplication
Nature Communications (2021)