Abstract
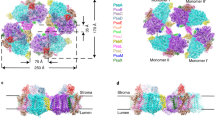
Photosystem I (PSI) is able to form different oligomeric states across various species. To reveal the structural basis for PSI dimerization and tetramerization, we structurally investigated PSI from the cyanobacterium Anabaena. This revealed a disrupted trimerization domain due to lack of the terminal residues of PsaL in the lumen, which resulted in PSI dimers with loose connections between monomers and weaker energy-coupled chlorophylls than in the trimer. At the dimer surface, specific phospholipids, cofactors and interactions in combination facilitated recruitment of another dimer to form a tetramer. Taken together, the relaxed luminal connections and lipid specificity at the dimer interface account for membrane curvature. PSI tetramer assembly appears to increase the surface area of the thylakoid membrane, which would contribute to PSI crowding.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and the Extended Data figure information. The cryo-EM maps have been deposited in the Electron Microscopy Data Bank with accession code no. EMD-10461. The atomic model has been deposited in the Protein Data Bank under accession code no. 6TCL.
References
Boekema, E. J. et al. Evidence for a trimeric organization of the photosystem I complex from the thermophilic cyanobacterium Synechococcus sp. FEBS Lett. 217, 283–286 (1987).
Amunts, A., Drory, O. & Nelson, N. The structure of a plant photosystem I supercomplex at 3.4 Å resolution. Nature 447, 58–63 (2007).
Gardian, Z. et al. Organisation of photosystem I and photosystem II in red alga Cyanidium caldarium: encounter of cyanobacterial and higher plant concepts. Biochim. Biophys. Acta Bioenergy 1767, 725–731 (2007).
Suga, M. et al. Structure of the green algal photosystem I supercomplex with a decameric light-harvesting complex I. Nat. Plants 5, 626–636 (2019).
Su, X. et al. Antenna arrangement and energy transfer pathways of a green algal photosystem-I–LHCI supercomplex. Nat. Plants 5, 273–281 (2019).
Qin, X. et al. Structure of a green algal photosystem I in complex with a large number of light-harvesting complex I subunits. Nat. Plants 5, 263–272 (2019).
Amunts, A. & Nelson, N. Plant photosystem I design in the light of evolution. Structure 17, 637–650 (2009).
Watanabe, M., Kubota, H., Wada, H., Narikawa, R. & Ikeuchi, M. Novel supercomplex organization of photosystem I in Anabaena and Cyanophora paradoxa. Plant Cell Physiol. 52, 162–168 (2011).
Li, M., Semchonok, D. A., Boekema, E. J. & Bruce, B. D. Characterization and evolution of tetrameric photosystem I from the thermophilic cyanobacterium Chroococcidiopsis sp. TS-821. Plant Cell 26, 1230–1245 (2014).
Semchonok, D. A., Li, M., Bruce, B. D., Oostergetel, G. T. & Boekema, E. J. Cryo-EM structure of a tetrameric cyanobacterial photosystem I complex reveals novel subunit interactions. Biochim. Biophys. Acta Bioenergy 1857, 1619–1626 (2016).
Shelaev, I. V. et al. Comparisons of electron transfer reactions in a cyanobacterial tetrameric and trimeric photosystem I complexes. Photochem. Photobiol. 94, 564–569 (2018).
Jordan, P. et al. Three-dimensional structure of cyanobacterial photosystem I at 2.5 Å resolution. Nature 411, 909–917 (2001).
Zehr, J. P. Nitrogen fixation by marine cyanobacteria. Trends Microbiol. 19, 162–173 (2011).
Halfmann, C., Gu, L. & Zhou, R. Engineering cyanobacteria for the production of a cyclic hydrocarbon fuel from CO2 and H2O. Green Chem. 16, 3175–3185 (2014).
Herrero, A., Stavans, J. & Flores, E. The multicellular nature of filamentous heterocyst-forming cyanobacteria. FEMS Microbiol. Rev. 40, 831–854 (2016).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Malavath, T., Caspy, I., Netzer-El, S. Y., Klaiman, D. & Nelson, N. Structure and function of wild-type and subunit-depleted photosystem I in Synechocystis. Biochim. Biophys. Acta Bioenergy 1859, 645–654 (2018).
Mazor, Y., Nataf, D., Toporik, H. & Nelson, N. Crystal structures of virus-like photosystem I complexes from the mesophilic cyanobacterium Synechocystis PCC 6803. eLife 3, e01496 (2014).
Şener, M. K. et al. Excitation migration in trimeric cyanobacterial photosystem I. J. Chem. Phys. 120, 11183–11195 (2004).
Van Eerden, F. J., Melo, M. N., Frederix, P. W. J. M. & Marrink, S. J. Prediction of thylakoid lipid binding sites on photosystem II. Biophys. J. 113, 2669–2681 (2017).
Thallmair, S., Vainikka, P. A. & Marrink, S. J. Lipid fingerprints and cofactor dynamics of light-harvesting complex II in different membranes. Biophys. J. 116, 1446–1455 (2019).
Gu, J. et al. Cryo-EM structure of the mammalian ATP synthase tetramer bound with inhibitory protein IF1. Science 364, 1068–1075 (2019).
Corradi, V. et al. Emerging diversity in lipid-protein interactions. Chem. Rev. 119, 5775–5848 (2019).
Sakurai, I., Mizusawa, N., Ohashi, S., Kobayashi, M. & Wada, H. Effects of the lack of phosphatidylglycerol on the donor side of photosystem II. Plant Physiol. 144, 1336–1346 (2007).
Zouni, A. et al. Crystal structure of photosystem II from Synechococcus elongatus at 3.8 Å resolution. Nature 409, 739–743 (2001).
Sheng, X., Liu, X., Cao, P., Li, M. & Liu, Z. Structural roles of lipid molecules in the assembly of plant PSII–LHCII supercomplex. Biophys. Rep. 4, 189–203 (2018).
Guo, R., Zong, S., Wu, M., Gu, J. & Yang, M. Architecture of human mitochondrial respiratory megacomplex I2III2IV2. Cell 170, 1247–1257.e12 (2017).
Kovacs, T. et al. Photosystem I oligomerization affects lipid composition in Synechocystis sp. PCC 6803. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1864, 1384–1395 (2019).
Popova, A. V. & Andreeva, A. S. in Advances in Planar Lipid Bilayers and Liposomes Vol. 17 (eds Iglič, A. & Genova, J.) 215–236 (Academic Press, 2013).
Toporik, H., Li, J., Williams, D., Chiu, P.-L. & Mazor, Y. The structure of the stress-induced photosystem I–IsiA antenna supercomplex. Nat. Struct. Mol. Biol. 26, 443–449 (2019).
Casella, S. et al. Dissecting the native architecture and dynamics of cyanobacterial photosynthetic machinery. Mol. Plant. 10, 1434–1448 (2017).
Nozue, S., Katayama, M., Terazima, M. & Kumazaki, S. Comparative study of thylakoid membranes in terminal heterocysts and vegetative cells from two cyanobacteria, Rivularia M-261 and Anabaena variabilis, by fluorescence and absorption spectral microscopy. Biochim. Biophys. Acta Bioenergy 1858, 742–749 (2017).
Magnuson, A. & Cardona, T. Thylakoid membrane function in heterocysts. Biochim. Biophys. Acta Bioenergy 1857, 309–319 (2016).
Pernil, R. & Schleiff, E. Metalloproteins in the biology of heterocysts. Life 9, 32 (2019).
Pan, X. et al. Structure of the maize photosystem I supercomplex with light-harvesting complexes I and II. Science 360, 1109–1113 (2018).
Zheng, L. et al. Structural and functional insights into the tetrameric photosystem I from heterocyst-forming cyanobacteria. Nat. Plants 5, 1087–1097 (2019).
Kato, K. et al. Structure of a cyanobacterial photosystem I tetramer revealed by cryo-electron microscopy. Nat. Commun. 19, 4929 (2019).
Allen, M. M. Simple conditions for growth of unicellular blue-green algae on plates. J. Phycol. 4, 1–4 (1968).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Scheres, S. H. W., Núñez-Ramírez, R., Sorzano, C. O. S., Carazo, J. M. & Marabini, R. Image processing for electron microscopy single-particle analysis using XMIPP. Nat. Protoc. 3, 977–990 (2008).
Kimanius, D., Forsberg, B. O., Scheres, S. H. W. & Lindahl, E. Accelerated cryo-EM structure determination with parallelisation using GPUs in RELION-2. eLife 5, e18722 (2016).
De la Rosa-Trevín, J. M. et al. Scipion: a software framework toward integration, reproducibility and validation in 3D electron microscopy. J. Struct. Biol. 195, 93–99 (2016).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Pettersen, E. F. et al. UCSF Chimera-A visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Kobayashi, K., Endo, K. & Wada, H. Specific distribution of phosphatidylglycerol to photosystem complexes in the thylakoid membrane. Front. Plant Sci. 8, 1991 (2017).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. D 75, 861–877 (2019).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
DeLano, W. L. Pymol: an open-source molecular graphics tool. CCP4 Newslett. Prot. Crystallogr. 40, 11 (2002).
Goddard, T. D. et al. UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci. 27, 14–25 (2018).
Sievers, F. et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 7, 539 (2011).
Acknowledgements
We thank Y. Yin from Plant Science Facility of the Institute of Botany CAS for excellent technical assistance with 77 K low-temperature fluorescence analysis, and members of Amunts laboratory for active discussions throughout the project. Data were collected at the SciLifeLab cryo-EM facility funded by the foundations Knut and Alice Wallenberg, Family Erling Persson and Kempe. We acknowledge support from SciLifeLab proteogenomics core facility. This work was supported by the National Natural Science Foundation of China (grant nos. 21506113 and 31470229), the Swedish Foundation for Strategic Research (grant no. FFL15:0325), the Ragnar Söderberg Foundation (grant no. M44/16), the Swedish Research Council (grant no. NT_2015–04107), Cancerfonden (grant no. 2017/1041), the European Research Council (grant no. ERC-2018-StG-805230) and the Knut and Alice Wallenberg Foundation (grant no. 2018.0080). A.A. is supported by the EMBO Young Investigator Programme.
Author information
Authors and Affiliations
Contributions
M.C. and A.A. designed the project and planned the experiments. M.C., A.P.-B. and L.Z. performed protein purification and cryo-EM. A.P.-B. built and refined the model. M.C., A.P.-B. and A.A. analysed the data. A.A. wrote the manuscript with contributions from M.C. and A.P.-B. All authors discussed and commented on the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Plants thanks Christopher Gisriel, Michael Hippler and Mei Li for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Optimization of the sample preparation.
a, Comparison of PSI extraction with digitonin and β-DDM. Both detergents were set to the final concentration of 1% (w/v) and incubated for 30 min on ice. The relative intensity of the digitonin-solubilized material have been quantified: tetramer: dimer:monomer 4.9:1.5:3.6. At least three independent experiments were repeated with the similar BN-PAGE results. MW marker units are in kDa. b, Stability assay. Concentrations of 0.5%, 1% and 1.5% (w/v) were tested for extraction of PSI. For each concentration, an incubation time of 0.5 h, 1.0 h, 2.0 h was used. The experiment suggests that using 1% digitonin results most PSI tetramer, and no difference was detected for the different time points. Three independents experiments were repeated with the similar results. c, Purification of PSI tetramer. Different amounts of sample were loaded on to three sucrose gradients, labeled 1 to 3. The PSI fractions were analyzed by BN-PAGE, prior to size exclusion chromatography, for which the 280 nm absorption is shown. The eluted peak was assessed for purity on BN-PAGE and subjected to cryo-EM. At least six independent experiments were repeated with the similar results.
Extended Data Fig. 2
Cryo-EM image processing of PSI particles.
Extended Data Fig. 3 Resolution of cryo-EM reconstruction.
a, Map colored by local resolution, viewed from the surface and cut-through. b, FSC curve, showing overall resolution of the cryo-EM map. (c) Map to model FSC.
Extended Data Fig. 4 Conservation of subunit PsaL.
a, Sideview of the superimposed subunit PsaL from Anabaena, Synechocystis and T. Elongatus. C-terminus boxed. b, Corresponding sequence alignment. Both illustrate missing C-terminal residues in the lumen in PsaL from Anabaena. C-terminus boxed.
Extended Data Fig. 5 77 K fluorescence absorption spectra.
a, Sucrose gradient showing the monomer, dimer and tetramer of PSI. At least three independent experiments were repeated with similar sucrose gradients results. b, The emission spectra between 600 and 800 nm after excitation at 435 nm. As revealed in Fig. 2, dimer. Five independent experiments were repeated with similar results. c, Zoom into the peaks, showing a slight shift of 2.8 nm from PSI monomer to PSI dimer.
Extended Data Fig. 6 Localization of lipids.
Elements in the interphases found in the tetramer, but not in the trimer structures (PDBID: 1JB0 and 5OY0) are shown as circles12,17. Lipids are represented as orange circles and digitonin molecules in locations, where no lipids are found in the trimer are shown as purple circles and carotenoid as red ovals.
Extended Data Fig. 7 Nearest protein coordinates between two dimers.
a, Overview of the inter-dimer region. b, The nearest residues are within of 4.1–4.8 Å range, and no protein-protein interaction observed.
Extended Data Fig. 8 Digitonin molecules at the dimer-dimer interface.
a, Three digitonin molecules modeled into elongated densities (mesh). Residues coordinating the binding in the membrane plane are shown as described in the text, showing interactions and experimental density (contour levels used are 0.491 and 0.477 respectively). b, Environment of the digitonin molecules from a, in schematic representation, specific elements contributing to the binding are shown. c, Density for lipids found in the dimer-dimer interphase (contour levels used are 0.528, 0.422, 0.550, 0.488 and 0.422 respectively).
Extended Data Fig. 9 Carotenoid molecule at the dimer-dimer interface.
A carotenoid molecule BCR-855 is found in the PsaB region with its benzene rings in proximity to chlorophylls a816 and a821.
Extended Data Fig. 10 Model of PSI tetramer arrangement.
PSI trimer crowding model on the left is derived from the model obtained by atomic force and confocal fluorescence microscopy (MacGregor-Chatwin et al. 2017)51. PSI tetramer crowding model is adopted on the same principle, and the additional membrane surface would appear due to the curvature between the dimers as described in the text.
Supplementary information
Supplementary Information
Supplementary Tables 1–3 and Figs. 1 and 2.
Rights and permissions
About this article
Cite this article
Chen, M., Perez-Boerema, A., Zhang, L. et al. Distinct structural modulation of photosystem I and lipid environment stabilizes its tetrameric assembly. Nat. Plants 6, 314–320 (2020). https://doi.org/10.1038/s41477-020-0610-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-020-0610-x
This article is cited by
-
Structural insights into a unique PSI–LHCI–LHCII–Lhcb9 supercomplex from moss Physcomitrium patens
Nature Plants (2023)
-
Structural basis of mitochondrial membrane bending by the I–II–III2–IV2 supercomplex
Nature (2023)
-
The Ycf48 accessory factor occupies the site of the oxygen-evolving manganese cluster during photosystem II biogenesis
Nature Communications (2023)
-
Structure of a monomeric photosystem I core associated with iron-stress-induced-A proteins from Anabaena sp. PCC 7120
Nature Communications (2023)
-
Spectral diversity of photosystem I from flowering plants
Photosynthesis Research (2023)