Abstract
Tetraploid emmer wheat (Triticum turgidum ssp. dicoccon) is a progenitor of the world’s most widely grown crop, hexaploid bread wheat (Triticum aestivum), as well as the direct ancestor of tetraploid durum wheat (T. turgidum subsp. turgidum). Emmer was one of the first cereals to be domesticated in the old world; it was cultivated from around 9700 bc in the Levant1,2 and subsequently in south-western Asia, northern Africa and Europe with the spread of Neolithic agriculture3,4. Here, we report a whole-genome sequence from a museum specimen of Egyptian emmer wheat chaff, 14C dated to the New Kingdom, 1130–1000 bc. Its genome shares haplotypes with modern domesticated emmer at loci that are associated with shattering, seed size and germination, as well as within other putative domestication loci, suggesting that these traits share a common origin before the introduction of emmer to Egypt. Its genome is otherwise unusual, carrying haplotypes that are absent from modern emmer. Genetic similarity with modern Arabian and Indian emmer landraces connects ancient Egyptian emmer with early south-eastern dispersals, whereas inferred gene flow with wild emmer from the Southern Levant signals a later connection. Our results show the importance of museum collections as sources of genetic data to uncover the history and diversity of ancient cereals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Sequence data are deposited in the ENA with study accession number PRJEB31103. The genotype calls are also provided as the source data for Fig. 2. The database of archaeobotanical observations is provided as the source data for Extended Data Fig. 1. Source data are available for Figs. 2–4 and Extended Data Figs. 1–5.
References
Arranz-Otaegui, A., Colledge, S., Zapata, L., Teira-Mayolini, L. C. & Ibáñez, J. J. Regional diversity on the timing for the initial appearance of cereal cultivation and domestication in southwest Asia. Proc. Natl Acad. Sci. USA 113, 14001–14006 (2016).
Fuller, D. Q., Willcox, G. & Allaby, R. G. Early agricultural pathways: moving outside the ‘core area’ hypothesis in southwest Asia. J. Exp. Bot. 63, 617–633 (2012).
Fuller, D. Q. & Lucas, L. in Encyclopedia of Global Archaeology (Ed. Smith, C.) 7812–7817 (Springer, 2014).
McClatchie, M. et al. Neolithic farming in north-western Europe: archaeobotanical evidence from Ireland. J. Archaeol. Sci. 51, 206–215 (2014).
Mascher, M. et al. Genomic analysis of 6,000-year-old cultivated grain illuminates the domestication history of barley. Nat. Genet. 48, 1089–1093 (2016).
Ramos-Madrigal, J. et al. Genome sequence of a 5,310-year-old maize cob provides insights into the early stages of maize domestication. Curr. Biol. 26, 3195–3201 (2016).
Vallebueno-Estrada, M. et al. The earliest maize from San Marcos Tehuacán is a partial domesticate with genomic evidence of inbreeding. Proc. Natl Acad. Sci. USA 113, 14151–14156 (2016).
Kistler, L. et al. Multiproxy evidence highlights a complex evolutionary legacy of maize in South America. Science 362, 1309–1313 (2018).
Smith, O. et al. A domestication history of dynamic adaptation and genomic deterioration in sorghum. Nat. Plants 5, 369–379 (2018).
Palmer, S. A., Smith, O. & Allaby, R. G. The blossoming of plant archaeogenetics. Ann. Anat. 194, 146–156 (2012).
Bilgic, H., Hakki, E. E., Pandey, A., Khan, M. K. & Akkaya, M. S. Ancient DNA from 8400 year-old Çatalhöyük wheat: implications for the origin of neolithic agriculture. PLoS ONE 11, e0151974 (2016).
Purugganan, M. D. & Fuller, D. Q. The nature of selection during plant domestication. Nature 457, 843–848 (2009).
Avni, R. et al. Wild emmer genome architecture and diversity elucidate wheat evolution and domestication. Science 357, 93–97 (2017).
Nalam, V. J., Vales, M. I., Watson, C. J. W., Kianian, S. F. & Riera-Lizarazu, O. Map-based analysis of genes affecting the brittle rachis character in tetraploid wheat (Triticum turgidum L.). Theor. Appl. Genet. 112, 373–381 (2006).
Pourkheirandish, M. et al. Evolution of the grain dispersal system in barley. Cell 162, 527–539 (2015).
Fuller, D. Q. Contrasting patterns in crop domestication and domestication rates: recent archaeobotanical insights from the old world. Ann. Bot. 100, 903–924 (2007).
Harlan, J. R., de Wet, J. M. J. & Price, E. G. Comparative evolution of cereals. Evolution 27, 311–325 (1973).
Salamini, F., Özkan, H., Brandolini, A., Schäfer-Pregl, R. & Martin, W. Genetics and geography of wild cereal domestication in the near east. Nat. Rev. Genet. 3, 429–441 (2002).
Horovitz, A. The soil seed bank of wild emmer. In Proc. International Symposium on In situ Conservation of Plant Genetic Diversity (eds Zencirci, N. et al.) 185–188 (Central Research Institute for Field Crops, 1998).
Nave, M., Avni, R., Ben-Zvi, B., Hale, I. & Distelfeld, A. QTLs for uniform grain dimensions and germination selected during wheat domestication are co-located on chromosome 4B. Theor. Appl. Genet. 129, 1303–1315 (2016).
Allaby, R. G., Stevens, C., Lucas, L., Maeda, O. & Fuller, D. Q. Geographic mosaics and changing rates of cereal domestication. Proc. R. Soc. B 372, 20160429 (2017).
Crawford, D. J. Food: tradition and change in Hellenistic Egypt. World Archaeol. 11, 136–146 (1979).
Caton-Thompson, G. & Gardner, E. W. The Desert Fayum (Royal Anthropological Institute of Great Britain and Ireland, 1934).
Nesbitt, M. & Samuel, D. From stable crop to extinction? The archaeology and history of the hulled wheats. In Proc. First International Workshop on Hulled Wheats (eds Padulosi, S. et al.) 41–100 (International Plant Genetic Resources Institute, 1996).
Wetterstrom, W. in The Archaeology of Africa (eds Andah, B., Okpoko, A., Shaw, T. & Sinclair, P.) 165–226 (Routledge, 1993).
Zaharieva, M., Ayana, N. G., Al Hakimi, A., Misra, S. C. & Monneveux, P. Cultivated emmer wheat (Triticum dicoccon Schrank), an old crop with promising future: a review. Genet. Resour. Crop Evol. 57, 937–962 (2010).
Brunton, G. & Caton-Thompson, G. The Badarian Civilization and Predynastic Remains Near Badari (British School of Archaeology in Egypt, 1928).
Günther, T. & Nettelblad, C. The presence and impact of reference bias on population genomic studies of prehistoric human populations. PLoS Genet. 15, 1008302 (2019).
Jónsson, H., Ginolhac, A., Schubert, M., Johnson, P. L. F. & Orlando, L. MapDamage2.0: fast approximate bayesian estimates of ancient DNA damage parameters. Bioinformatics 29, 1682–1684 (2013).
IWGSC. Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 361, eaar7191 (2018).
Golenberg, E. M. Outcrossing rates and their relationship to phenology in Triticum dicoccoides. Theor. Appl. Genet. 75, 937–944 (1988).
Fuller, D. Q. Agricultural origins and frontiers in south Asia: a working synthesis. J. World Prehist. 20, 1–86 (2006).
Stevens, C. J. et al. Between China and south Asia: a middle Asian corridor of crop dispersal and agricultural innovation in the bronze age. Holocene 26, 1541–1555 (2016).
Alexander, D. H., Novembre, J. & Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 19, 1655–1664 (2009).
van der Veen, M. Consumption, Trade and Innovation: exploring the Botanical Remains from the Roman and Islamic Ports at Quseir al-Qadim, Egypt (Africa Magna Verlag, 2011).
Murray, M. A. in Ancient Egyptian Materials and Technology (eds Nicholson, P. T. & Shaw, I.) 505–536 (Cambridge Univ. Press, 2000).
Patterson, N. et al. Ancient admixture in human history. Genetics 192, 1065–1093 (2012).
Marcussen, T. et al. Ancient hybridizations among the ancestral genomes of bread wheat. Science 345, 1250092 (2014).
Olsen, K. M. et al. Selection under domestication: evidence for a sweep in the rice waxy genomic region. Genetics 173, 975–983 (2006).
Walsh, B. & Lynch, M. Evolution and Selection of Quantitative Traits (Oxford Univ. Press, 2018).
Fuller, D. Q., Lucas, L., Gonzalez Carretero, L. & Stevens, C. From intermediate economies to agriculture: trends in wild food use, domestication and cultivation among early villages in southwest Asia. Paleorient 44, 59–74 (2018).
Badaeva, E. D. et al. Chromosomal passports provide new insights into diffusion of emmer wheat. PLoS ONE 10, e0128556 (2015).
Luo, M. C. et al. The structure of wild and domesticated emmer wheat populations, gene flow between them, and the site of emmer domestication. Theor. Appl. Genet. 114, 947–959 (2007).
Wengrow, D., Dee, M., Foster, S., Stevenson, A. & Ramsey, C. B. Cultural convergence in the neolithic of the Nile Valley: a prehistoric perspective on Egypt’s place in Africa. Antiquity 88, 95–111 (2014).
Fuller, D. & Hildebrand, E. in The Oxford Handbook of African Archaeology (eds Mitchell, P. & Lane, P.) 507–525 (Oxford Univ. Press, 2013).
Hasel, M. G. Domination and Resistance: Egyptian Military Activity in the Southern Levant, ca. 1300–1185 B.C. (Brill, 1998).
Civáň, P., Ivaničová, Z. & Brown, T. A. Reticulated origin of domesticated emmer wheat supports a dynamic model for the emergence of agriculture in the fertile crescent. PLoS ONE 8, e81955 (2013).
He, F. et al. Exome sequencing highlights the role of wild-relative introgression in shaping the adaptive landscape of the wheat genome. Nat. Genet. 51, 896–904 (2019).
Di Donato, A., Filippone, E., Ercolano, M. R. & Frusciante, L. Genome sequencing of ancient plant remains: findings, uses and potential applications for the study and improvement of modern crops. Front. Plant Sci. 9, 441 (2018).
Zohary, D., Hopf, M. & Weiss, E. Domestication of Plants in the Old World (Oxford Univ. Press, 2012).
Bronk Ramsey, C. Bayesian analysis of radiocarbon dates. Radiocarbon 51, 337–360 (2009).
Reimer, P. J. et al. IntCal13 and Marine13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 55, 1869–1887 (2013).
Meyer, M. & Kircher, M. Illumina sequencing library preparation for highly multiplexed target capture and sequencing. Cold Spring Harb. Protoc. 6, pdb.prot5448 (2010).
Rohland, N., Harney, E., Mallick, S., Nordenfelt, S. & Reich, D. Partial uracil–DNA–glycosylase treatment for screening of ancient DNA. Proc. R. Soc. B 370, 20130624 (2015).
Schubert, M., Lindgreen, S. & Orlando, L. AdapterRemoval v2: rapid adapter trimming, identification, and read merging. BMC Res. Notes 9, 88 (2016).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Meyer, M. et al. A high coverage genome sequence from an archaic Denisovan individual. Science 338, 222–226 (2013).
Briggs, A. W. et al. Patterns of damage in genomic DNA sequences from a Neandertal. Proc. Natl Acad. Sci. USA 104, 14616–14621 (2007).
McKenna, A. et al. The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Jordan, K. W. et al. A haplotype map of allohexaploid wheat reveals distinct patterns of selection on homoeologous genomes. Genome Biol. 16, 48 (2015).
Li, H. A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 27, 2987–2993 (2011).
Danecek, P. et al. The variant call format and VCFtools. Bioinformatics 27, 2156–2158 (2011).
Vavilov, N. I. Origin and Geography of Cultivated Plants (Cambridge Univ. Press, 1989).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2017).
Lee, T. H., Guo, H., Wang, X., Kim, C. & Paterson, A. H. SNPhylo: a pipeline to construct a phylogenetic tree from huge SNP data. BMC Genom. 15, 162 (2014).
Camacho, C. et al. BLAST+: architecture and applications. BMC Bioinform. 10, 421 (2009).
Kaplan, N. L., Hudson, R. R. & Langley, C. H. The hitchhiking effect revisited. Genetics 123, 887–899 (1989).
Haldane, J. B. S. The combination of linkage values and the calculation of distances between the loci of linked factors. J. Genet. 8, 299–309 (1919).
Cabrera, C. P. et al. Uncovering networks from genome-wide association studies via circular genomic permutation. G3 2, 1067–1075 (2012).
Acknowledgements
We thank Y. Diekmann, D. O’Rourke and A. Garnett for helpful discussions. M.F.S. and R.M. are supported by RCUK BBSRC grant BB/M011585/1. R.M. is also supported by RCUK BBSRC grant BB/P024726/1; L.R.B. by the Spanish Ministry of Economy and Competitiveness Severo Ochoa Programme for Centres of Excellence in R&D 2016-2019 (SEV-2015-0533) and CERCA Programme, Generalitat de Catalunya; M.G.T. and S.B. by a Wellcome Trust Senior Research Fellowship, grant 100719/Z/12/Z. D.Q.F. and C.S. are supported by the ERC ComPag project, grant number 323842. V.E.M. and S.B. are partially supported by the RCUK NERC grant NE/P012574/1. UCL computing infrastructure was supported by BBSRC grant BB/R01356X/1.
Author information
Authors and Affiliations
Contributions
L.R.B., M.F.S., R.M., D.Q.F., C.S., A.S. and M.G.T. designed and coordinated the study. M.F.S. designed and performed data analysis. L.R.B., S.B. and V.E.M. performed experiments. C.S. obtained image data. M.F.S. and R.M. coordinated the sequencing. D.Q.F. coordinated the carbon dating. M.G.T. supervised access to the ancient DNA laboratory. D.Q.F., A.S. and C.S. collated archaeobotanical data. M.F.S. and R.M. wrote the manuscript. All of the authors edited and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer Review Information Nature Plants thanks Thomas Gilbert, James Breen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
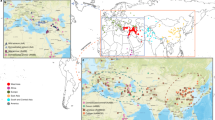
Extended Data Fig. 1 Map showing archaeobotanical observations of emmer wheat from 9000 – 1000 BCE in Northeast Africa and South West Asia.
Map showing archaeobotanical observations of emmer wheat from 9000 – 1000 BCE in Northeast Africa and South West Asia. The collection location for the sequenced accession (UC10164) is labelled.
Extended Data Fig. 2 Summary of sequencing data from UC10164 samples S1 and S2.
Summary of sequencing data from UC10164 samples S1 and S2. Panel A shows the C to T and G to A misincorporations of alignments against the emmer wheat reference genome as output by MapDamage. Both samples show a small excess of these misincorporations in the 2 bp at each fragment end, as expected for partially UDG treated aDNA libraries. Panel B shows the distribution of fragment sizes after the overlapping paired-end reads were collapsed and adapter sequence was removed using AdapterRemoval. Panel C shows coverage and subgenome representation for different minimum mapping quality scores after these fragments were aligned to the hexaploid bread wheat reference genome. Panel D shows the coverage obtained from alignment to the emmer wheat reference genome, split by minimum mapping quality filters. The exonic SNP sites (at which genotypes are called) have a much lower percentage of ambiguous alignments with low mapping quality scores.
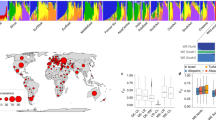
Extended Data Fig. 3 (a) phylogeny of modern emmer wheat accessions and (b) source population proportions inferred by the ADMIXTURE model for various number of source populations (K parameter).
(a) phylogeny of modern emmer wheat accessions and (b) Source population proportions inferred by the ADMIXTURE model for various number of source populations (K parameter). The maximum likelihood phylogeny in (a) was constructed from the full set of SNPs called among modern accessions, excluding UC10164. Bootstrap support for each node is shown where it is less than 100 (based on 1000 bootstraps). In (b), the majority of the ancestry proportion inferred for the ancient Egyptian accession (UC10164) across K values is from the same population that predominates among the “Indian Ocean” subgroup (green accession names). Furthermore, the ancient Egyptian accession has a relatively high proportion of ancestry inferred to come from population groups that are common among modern wild Southern Levant emmer wheats (light blue accession names).
Extended Data Fig. 4 Heatmap of the genotypic similarity between each pair of accessions across 86,594 SNP sites that were called in the ancient Egyptian accession (UC10164).
Heatmap of the genotypic similarity between each pair of accessions across 86,594 SNP sites that were called in the ancient Egyptian accession (UC10164). Below the diagonal, we plot similarity using identity by state. Above the diagonal, we plot haplotypic similarity, which is defined as the fraction of sliding windows of 50 SNP sites that are more than 95% concordant.
Extended Data Fig. 5 Differences between each modern accession and the ancient Egyptian accession (UC10164) across the genome.
Differences between each modern accession and the ancient Egyptian accession (UC10164) across the genome. The fraction of genotypic differences from UC10164 is calculated within sliding windows of 50 SNPs (moved in intervals of 25 SNPs). Each modern accession is plotted as a line and coloured according to its subgroup membership. Below zero we plot the ancestry mosaic inferred for each SNP site called in UC10164. The dissimilarity within sliding windows is plotted against the median physical map position of the SNPs in the sliding window. Each chromosome has a large region around the centromere that has low exonic SNP density and low recombination rate, resulting in long haplotypes.
Supplementary information
Supplementary Tables 1–5
Supplementary Table 1: table of alignment statistics for S1 and S2 after various filters have been applied. Supplementary Table 2: concordances between UC10164 and modern accessions, split by chromosome and accession subgroup. Supplementary Table 3: D statistics calculated for the phylogeny (outgroup, (Southern Levant Accession, (Indian Ocean, UC10164))). Supplementary Table 4: raw radiocarbon dating results. Supplementary Table 5: at SNP genotype sites called in UC10164, the proportion of substitutions that are C to T and G to A, relative to the outgroup genotype, and the ratio of transitions to transversions.
Rights and permissions
About this article
Cite this article
Scott, M.F., Botigué, L.R., Brace, S. et al. A 3,000-year-old Egyptian emmer wheat genome reveals dispersal and domestication history. Nat. Plants 5, 1120–1128 (2019). https://doi.org/10.1038/s41477-019-0534-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-019-0534-5
This article is cited by
-
Are cereal grasses a single genetic system?
Nature Plants (2024)
-
Genetic basis of geographical differentiation and breeding selection for wheat plant architecture traits
Genome Biology (2023)
-
Population genomics unravels the Holocene history of bread wheat and its relatives
Nature Plants (2023)
-
Genomic analysis of emmer wheat shows a complex history with two distinct domestic groups and evidence of differential hybridization with wild emmer from the western Fertile Crescent
Vegetation History and Archaeobotany (2023)
-
Nobel adjacency
Nature Plants (2022)