Abstract
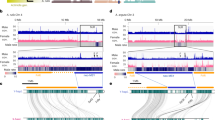
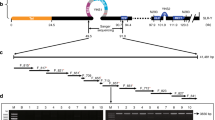
Dioecy, the presence of male and female individuals, has evolved independently in multiple flowering plant lineages1,2,3. Although theoretical models for the evolution of dioecy, such as the ‘two-mutations’ model, are well established4,5, little is known about the specific genes determining sex and their evolutionary history3. Kiwifruit, a major tree crop consumed worldwide, is a dioecious species. In kiwifruit we previously identified a Y-encoded sex-determinant candidate gene acting as the suppressor of feminization (SuF), named Shy Girl (SyGI)6. Here, we identify a second Y-encoded sex-determinant that we named Friendly Boy (FrBy), which exhibits strong expression in tapetal cells. Gene-editing and complementation analyses in Arabidopsis thaliana and Nicotiana tabacum indicated that FrBy acts for the maintenance of male (M) functions, independently of SyGI, and that these functions are conserved across angiosperm species. We further characterized the genomic architecture of the small (<1 megabase pairs (Mb)) male-specific region of the Y chromosome (MSY), which harbours only two genes expressed extensively in developing gynoecia and androecia, respectively: SyGI and FrBy. Re-sequencing of the genome of a natural hermaphrodite kiwifruit revealed that this individual is genetically male but carries deletion(s) of parts of the Y chromosome, including SyGI. Additionally, expression of FrBy in female kiwifruit resulted in hermaphrodite plants. These results clearly indicate that Y-encoded SyGI and FrBy act independently as the SuF and M factors in kiwifruit, respectively, and provide insight into not only the evolutionary path leading to a two-factor sex-determination system, but also a new breeding approach for dioecious species.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request. All sequence data generated in the context of this manuscript have been deposited in the appropriate DNA Database of Japan: Illumina reads for gDNA-seq and mRNA-seq in the Short Read Archives (SRA) database (SRA Submission ID: DRA008474, Run IDs: DRR180225–180236), and the genomic contig sets constructed with 10X Genomics reads were submitted to Genbank (IDs LC482704–482713).
References
Ming, R., Bendahmane, A. & Renner, S. S. Sex chromosomes in land plants. Ann. Rev. Plant Biol. 62, 485–514 (2011).
Renner, S. S. The relative and absolute frequencies of angiosperm sexual systems: dioecy, monoecy, gynodioecy, and an updated online database. Am. J. Bot. 101, 1588–1596 (2014).
Henry, I. M., Akagi, T., Tao, R. & Comai, L. One hundred ways to invent the sexes: theoretical and observed paths to dioecy in plants. Annu. Rev. Plant Biol. 69, 553–575 (2018).
Westergaard, M. The mechanism of sex determination in dioecious flowering plants. Adv. Genet. 9, 217–281 (1958).
Charlesworth, B. & Charlesworth, D. A model for the evolution of dioecy and gynodioecy. Am. Nat. 112, 975–997 (1978).
Akagi, T. et al. A Y-encoded suppressor of feminization arose via lineage-specific duplication of a cytokinin response regulator in kiwifruit. Plant Cell 30, 780–795 (2018).
Charlesworth, D. Plant sex chromosome evolution. J. Exp. Bot. 64, 405–420 (2013).
Charlesworth, D. Plant contributions to our understanding of sex chromosome evolution. New Phytol. 208, 52–65 (2015).
Kazama, Y. et al. A new physical mapping approach refines the sex-determining gene positions on the Silene latifolia Y-chromosome. Sci. Rep. 6, 18917 (2016).
Liu, Z. et al. A primitive Y chromosome in papaya marks incipient sex chromosome evolution. Nature 427, 348–352 (2004).
Wang, J. et al. Sequencing papaya X and Yh chromosomes reveals molecular basis of incipient sex chromosome evolution. Proc. Natl Acad. Sci. USA 109, 13710–13715 (2012).
Akagi, T., Henry, I. M., Tao, R. & Comai, L. A Y-chromosome-encoded small RNA acts as a sex determinant in persimmons. Science 346, 646–650 (2014).
Harkess, A. et al. The asparagus genome sheds light on the origin and evolution of a young Y chromosome. Nat. Commun. 8, 1279 (2017).
Akagi, T. et al. Epigenetic regulation of the sex determination gene MeGI in polyploid persimmon. Plant Cell 28, 2905–2915 (2016).
Datson, P. M. & Ferguson, A. R. Wild Crop Relatives: Genomic and Breeding Resources. Tropical and Subtropical Fruits (ed. Kole, C.) 1–20 (Springer, 2011).
Fraser, L. G. et al. A gene-rich linkage map in the dioecious species Actinidia chinensis (kiwifruit) reveals putative X/Y sex-determining chromosomes. BMC Genom. 10, 102 (2009).
Zhang, Q. et al. High-density interspecific genetic maps of kiwifruit and the identification of sex-specific markers. DNA Res. 22, 367–375 (2015).
Messina, R. Microsporogenesis in male-fertile cv. Matua and male-sterile cv. Hayward of Actinidia deliciosa var. deliciosa (Kiwifruit). Adv. Hort. Sci. 7, 77–81 (1993).
Falasca, G. et al. Tapetum and middle layer control male fertility in Actinidia deliciosa. Ann. Bot. 112, 1045–1055 (2013).
Tan, H., Liang, W., Hu, J. & Zhang, D. MTR1 encodes a secretory fasciclin glycoprotein required for male reproductive development in rice. Dev. Cell 22, 1127–1137 (2012).
Pilkington, S. M. et al. A manually annotated Actinidia chinensis var. chinensis (kiwifruit) genome highlights the challenges associated with draft genomes and gene prediction in plants. BMC Genom. 19, 257 (2018).
Zhang, D. & Yang, L. Specification of tapetum and microsporocyte cells within the anther. Curr. Opin. Plant Biol. 17, 49–55 (2014).
Ye, J. et al. Proteomic and phosphoproteomic analyses reveal extensive phosphorylation of regulatory proteins in developing rice anthers. Plant J. 84, 527–544 (2015).
Ye, J. et al. Abundant protein phosphorylation potentially regulates Arabidopsis anther development. J. Exp. Bot. 67, 4993–5008 (2016).
Tsugama, D. et al. A putative MYB35 ortholog is a candidate for the sex-determining genes in Asparagus officinalis. Sci. Rep. 7, 41497 (2017).
Murase, K. et al. MYB transcription factor gene involved in sex determination in Asparagus officinalis. Genes Cells 22, 115–123 (2017).
Huang, S. et al. Draft genome of the kiwifruit Actinidia chinensis. Nat. Comm. 4, 2640 (2013).
Weisenfeld, N. I. et al. Direct determination of diploid genome sequences. Genome Res. 27, 757–767 (2017).
Ben-Dor, A., Chor, B. & Pelleg, D. RHO - radiation hybrid ordering. Genome Res. 10, 365–378 (2000).
Schäffer, A. A., Rice, E. S., Cook, W. & Agarwala, R. rh_tsp_map 3.0: end-to-end radiation hybrid mapping with improved speed and quality control. Bioinformatics 23, 1156–1158 (2007).
Bachtrog, D. et al. Sex determination: why so many ways of doing it? PLoS Biol. 12, e1001899 (2014).
Stanke, M., Steinkamp, R., Waack, S. & Morgenstern, B. AUGUSTUS: a web server for gene finding in eukaryotes. Nucleic Acids Res. 32, 309–312 (2004).
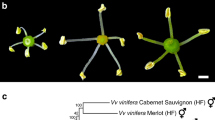
McNeilage, M. A. & Steinhagen, S. Flower and fruit characters in a kiwifruit hermaphrodite. Euphytica 101, 69–72 (1998).
Varkonyi-Gasic, E. et al. Mutagenesis of kiwifruit CENTRORADIALIS‐like genes transforms a climbing woody perennial with long juvenility and axillary flowering into a compact plant with rapid terminal flowering. Plant Biotech. J. 17, 869–880 (2019).
Mayer, S. S. & Charlesworth, D. Cryptic dioecy in flowering plants. Trends Ecol. Evol. 6, 320–325 (1991).
Torres, M. et al. Genus-wide sequencing supports a two-locus model for sex-determination in Phoenix. Nat. Comm. 9, 3969 (2018).
Yang, H.-W., Akagi, T., Kawakatsu, T. & Tao, R. Gene networks orchestrated by MeGI: a single‐factor mechanism underlying sex determination in persimmon. Plant J. 98, 97–111 (2019).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Genome Biol. 11, R106 (2010).
Kawamoto, T. Use of a new adhesive film for the preparation of multi-purpose fresh-frozen sections from hard tissues, whole-animals, insects and plants. Arch. Histol. Cytol. 66, 123–143 (2003).
Tamura, K. et al. MEGA5: molecular evolution genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739 (2011).
Suyama, M., Torrents, D. & Bork, P. PAL2NAL: robust conversion of protein sequence alignments into the corresponding codon alignments. Nucleic Acids Res. 34, W609–W612 (2006).
Librado, P. & Rozas, J. DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25, 1451–1452 (2009).
Yang, Z. PAML: a program package for phylogenetic analysis by maximum likelihood. Comput. Appl. Biosci. 13, 555–556 (1997).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotech. 29, 24–26 (2011).
Tsutsui, H. & Higashiyama, T. pKAMA-ITACHI vectors for highly efficient CRISPR/Cas9-mediated gene knockout in Arabidopsis thaliana. Plant Cell Physiol. 58, 46–56 (2017).
Voogd, C., Wang, T. & Varkonyi-Gasic, E. Functional and expression analyses of kiwifruit SOC1-like genes suggest that they may not have a role in the transition to flowering but may affect the duration of dormancy. J. Exp. Bot. 66, 4699–4710 (2015).
Wang, T. et al. Transformation of Actinidia eriantha: a potential species for functional genomics studies in Actinidia. Plant Cell Rep. 25, 425 (2006).
Acknowledgements
We thank L. Comai (UC Davis Department Plant Biology and Genome Center) for technical advice and bioinformatics support; Y. Kazama and K. Ishii (Riken Institute) for technical support in using the DelMapper programme; and N. Nieuwenhuizen and J. (L.) Zhang (Plant and Food Research) for vector construction. The KE population was originally provided by Kagawa Prefectural Agricultural Experiment Station. Some of this work was performed at the Vincent J. Coates Genomics Sequencing Laboratory at UC Berkeley, supported by NIH Instrumentation (grant No. S10 OD018174). This work was supported by PRESTO (grant Nos. JPMJPR15Q1 (to T.A.) and JPMJPR15Q6 (to S.S.S.)) from the Japan Science and Technology Agency (JST), by a Grant-in-Aid for Scientific Research on Innovative Areas (No. 19H04862 to T.A.) from JSPS and by the National Science Foundation (NSF) IOS award (grant No. 1457230 to I.M.H.).
Author information
Authors and Affiliations
Contributions
T.A., I.K. and R.T. conceived the study. T.A. designed the experiments. T.A., S.M.P., E.V.-G., S.S.S., M.S., A.F., M.J.D., T.W., R.R. and C.V. conducted the experiments. T.A., S.M.P., E.V.-G., S.S.S., M.S., I.M.H. and A.F. analysed the data. S.M.P., M.A.M., P.D., A.C.A., K.B. and I.K. initiated/bred and maintained the plant materials. T.A., S.M.P., E.V. and I.M.H. drafted the manuscript. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Plants thanks Roberta Bergero, Susanne Renner and Qi Zhou for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Figs. 1–14 and Supplementary Tables 1–12.
Rights and permissions
About this article
Cite this article
Akagi, T., Pilkington, S.M., Varkonyi-Gasic, E. et al. Two Y-chromosome-encoded genes determine sex in kiwifruit. Nat. Plants 5, 801–809 (2019). https://doi.org/10.1038/s41477-019-0489-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-019-0489-6
This article is cited by
-
Agrobacterium rhizogenes-mediated marker-free transformation and gene editing system revealed that AeCBL3 mediates the formation of calcium oxalate crystal in kiwifruit
Molecular Horticulture (2024)
-
Evolution of a plant sex chromosome driven by expanding pericentromeric recombination suppression
Scientific Reports (2024)
-
Genomic profiling of dioecious Amaranthus species provides novel insights into species relatedness and sex genes
BMC Biology (2023)
-
Comparative transcriptomic analysis of male and females in the dioecious weeds Amaranthus palmeri and Amaranthus tuberculatus
BMC Plant Biology (2023)
-
Development of sex-linked markers for gender identification of Actinidia arguta
Scientific Reports (2023)