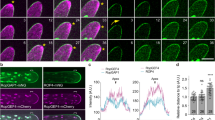
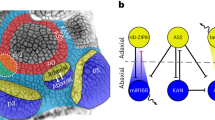
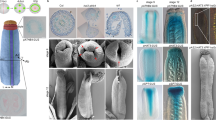
Abstract
Multicellular development requires coordinated cell polarization relative to body axes, and translation to oriented cell division1,2,3. In plants, it is unknown how cell polarities are connected to organismal axes and translated to division. Here, we identify Arabidopsis SOSEKI proteins that integrate apical–basal and radial organismal axes to localize to polar cell edges. Localization does not depend on tissue context, requires cell wall integrity and is defined by a transferrable, protein-specific motif. A Domain of Unknown Function in SOSEKI proteins resembles the DIX oligomerization domain in the animal Dishevelled polarity regulator. The DIX-like domain self-interacts and is required for edge localization and for influencing division orientation, together with a second domain that defines the polar membrane domain. Our work shows that SOSEKI proteins locally interpret global polarity cues and can influence cell division orientation. Furthermore, this work reveals that, despite fundamental differences, cell polarity mechanisms in plants and animals converge on a similar protein domain.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Butler, M. T. & Wallingford, J. B. Planar cell polarity in development and disease. Nat. Rev. Mol. Cell Biol. 18, 375–388 (2017).
Nakamura, M., Kiefer, C. S. & Grebe, M. Planar polarity, tissue polarity and planar morphogenesis in plants. Curr. Opin. Plant. Biol. 15, 593–600 (2012).
St Johnston, D. & Ahringer, J. Cell polarity in eggs and epithelia: parallels and diversity. Cell 141, 757–774 (2010).
Chiou, J.-G., Balasubramanian, M. K. & Lew, D. J. Cell polarity in yeast. Annu. Rev. Cell. Dev. Biol. 33, 77–101 (2017).
St. Johnston, D. Establishing and transducing cell polarity: common themes and variations. Curr. Opin. Cell Biol. 51, 33–41 (2018).
Yoshida, S. et al. Genetic control of plant development by overriding a geometric division rule. Dev. Cell. 29, 75–87 (2014).
De Rybel, B. et al. A bHLH complex controls embryonic vascular tissue establishment and indeterminate growth in Arabidopsis. Dev. Cell. 24, 426–437 (2013).
Gälweiler, L. et al. Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science 282, 2226–2230 (1998).
Müller, A. et al. AtPIN2 defines a locus of Arabidopsis for root gravitropism control. EMBO J. 17, 6903–6911 (1998).
Friml, J., Wiŝniewska, J., Benková, E., Mendgen, K. & Palme, K. Lateral relocation of auxin efflux regulator PIN3 mediates tropism in Arabidopsis. Nature 415, 806–809 (2002).
Friml, J. et al. Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature 426, 147–153 (2003).
Langowski, L. et al. Cellular mechanisms for cargo delivery and polarity maintenance at different polar domains in plant cells. Cell Discov. 2, 16018 (2016).
Dong, J., MacAlister, C. A. & Bergmann, D. C. BASL controls asymmetric cell division in Arabidopsis.Cell 137, 1320–1330 (2009).
Takano, J. et al. Polar localization and degradation of Arabidopsis boron transporters through distinct trafficking pathways. Proc. Natl Acad. Sci. 107, 5220–5225 (2010).
Pillitteri, L. J., Peterson, K. M., Horst, R. J. & Torii, K. U. Molecular profiling of stomatal meristemoids reveals new component of asymmetric cell division and commonalities among stem cell populations in Arabidopsis. Plant Cell 23, 3260–3275 (2011).
Kania, U., Fendrych, M. & Friml, J. Polar delivery in plants; commonalities and differences to animal epithelial cells. Open Biol. 4, 140017 (2014).
Moller, B. & Weijers, D. Auxin control of embryo patterning. Cold Spring Harb. Perspect. Biol. 1, a001545 (2009).
Hardtke, C. S. & Berleth, T. The Arabidopsis gene MONOPTEROS encodes a transcription factor mediating embryo axis formation and vascular development. EMBO J. 17, 1405–1411 (1998).
Berleth, T. & Jürgens, G. The role of the monopteros gene in organising the basal body region of the Arabidopsis embryo. Development 118, 575–587 (1993).
Möller, B. K. et al. Auxin response cell-autonomously controls ground tissue initiation in the early Arabidopsis embryo. Proc. Natl Acad. Sci. USA 114, E2533–E2539 (2017).
Schlereth, A. et al. MONOPTEROS controls embryonic root initiation by regulating a mobile transcription factor. Nature 464, 913–916 (2010).
Weijers, D. et al. An arabidopsis minute-like phenotype caused by a semi-dominant mutation in a RIBOSOMAL PROTEIN S5 gene. Development 128, 4289–4299 (2001).
Scheres, B. et al. Mutations affecting the radial organisation of the Arabidopsis root display specific defects throughout the embryonic axis. Development 121, 53–62 (1995).
Benfey, P. N. et al. Root development in Arabidopsis: four mutants with dramatically altered root morphogenesis. Development 119, 57–70 (1993).
Radoeva, T., ten Hove, C. A., Saiga, S. & Weijers, D. Molecular characterization of Arabidopsis GAL4/UAS enhancer trap lines identifies novel cell type-specific promoters. Plant Physiol. 171, 1169–1181 (2016).
Alassimone, J.., Naseer, S. & Geldner, N. . A developmental framework for endodermal differentiation and polarity. Proc. Natl Acad. Sci. USA 107, 5214–5219 (2010).
Liao, C. Y. & Weijers, D. A toolkit for studying cellular reorganization during early embryogenesis in Arabidopsis thaliana. Plant J. 93, 963–976 (2018).
Traverso, J. A. et al. Roles of N-terminal fatty acid acylations in membrane compartment partitioning: Arabidopsis h-type thioredoxins as a case study. Plant Cell 25, 1056–1077 (2013).
Bienz, M. Signalosome assembly by domains undergoing dynamic head-to-tail polymerization. Trends. Biochem. Sci. 10, 487–495 (2014).
Ehebauer, M. T. & Arias, A. M. The structural and functional determinants of the Axin and Dishevelled DIX domains. BMC Struct. Biol. 9, 70 (2009).
Schwarz-Romond, T. et al. The DIX domain of Dishevelled confers Wnt signaling by dynamic polymerization. Nat. Struct. Mol. Biol. 14, 484–492 (2007).
Mansfield, C. et al. Ectopic BASL reveals tissue cell polarity throughout leaf development in Arabidopsis thaliana. Curr. Biol. 28, 2638–2646 (2018).
Fukaki, H. et al. Genetic evidence that the endodermis is essential for shoot gravitropism in Arabidopsis thaliana. Plant J. 14, 425–430 (1998).
Helariutta, Y. et al. The SHORT-ROOT gene controls radial patterning of the Arabidopsis root through radial signaling. Cell 101, 555–567 (2000).
De Rybel, B. et al. A versatile set of ligation-independent cloning vectors for functional studies in plants. Plant Physiol. 156, 1292–1299 (2011).
Weijers, D., Van Hamburg, J. P., Van Rijn, E., Hooykaas, P. J. & Offringa, R. Diphtheria toxin-mediated cell ablation reveals interregional communication during Arabidopsis seed development. Plant Physiol. 133, 1882–1892 (2003).
Llavata-Peris, C., Lokerse, A., Moller, B., De Rybel, B. & Weijers, D. Imaging of phenotypes, gene expression, and protein localization during embryonic root formation in Arabidopsis. Methods Mol. Biol. 959, 137–148 (2013).
Marhavý, P. et al. Targeted cell elimination reveals an auxin-guided biphasic mode of lateral root initiation. Genes Dev. 30, 471–483 (2016).
von Wangenheim, D. et al. Live tracking of moving samples in confocal microscopy for vertically grown roots. eLife 6, e26792 (2017).
de Reuille, P. B. et al. MorphoGraphX: A platform for quantifying morphogenesis in 4D. eLife 4, 05864 (2015).
Rios, A. F., Radoeva, T., De Rybel, B., Weijers, D. & Borst, J. W. FRET-FLIM for visualizing and quantifying protein interactions in live plant cells. Methods Mol. Biol. 1497, 135–146 (2017).
Acknowledgements
We thank D. Hagemans and V. Mol for support with experiments and E. Benkova for supporting P.M. This work was supported by the European Research Council under the European Union’s Seventh Framework Programme (FP7/2007-2013) under REA grant agreement number 291734, and a Marie Curie Fellowships (contract 753138) to S.Y. by a European Research Council grant (ERC-StG CELLPATTERN; contract 281573) and ALW Open Competition grant (820.02.019) and an ALW-VIDI grant (864.06.012) from the Netherlands Organization for Scientific Research (NWO) to D.W., an ALW-VENI grant (863.21.010) from the Netherlands Organization for Scientific Research (NWO) to C.A.t.H., a grant (831.13.001) from the Netherlands Organization for Scientific Research (NWO) to M.v.D. and a FEBS long-term fellowship to P.M.
Author information
Authors and Affiliations
Contributions
D.W. conceived the study. S.Y. performed most of the experiments under the supervision of D.W. and with help from A.v.d.S., L.v.G. and S.S. M.v.D. carried out the BiFC and FRET-FLIM experiments, SOK1 localization in ground tissue and in shr and scr mutants (together with C.A.t.H.) and SOK1/SOK5 swaps. B.M. initiated the project and identified the SOK1 gene. M.A. and R.S. performed 3D SOK1 localization analysis. P.M. helped to perform live imaging under J.F.’s supervision. S.Y. and D.W. wrote the manuscript with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review Information Nature Plants thanks Marie Barberon, Dominique Bergmann, Juan Dong and Markus Grebe for their contribution to the peer review of this work
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–9, Supplementary Tables 1–3 and Supplementary Video Legends.
Supplementary Video 1
Localization of SOK1-YFP in heart stage embryo. Embryo was stained with Renaissance RS2200 (white), observed by confocal microscopy and 3D reconstructed.
Supplementary Video 2
A time-lapse series of SOK1-YFP during lateral root initiation. Root was stained with propidium iodide and observed by confocal microscopy. Time (hours) is indicated. The experiment was repeated in seven lateral roots with similar results.
Supplementary Video 3
A time-lapse series of SOK1-YFP during root growth. Root was stained with propidium iodide and observed by vertical confocal microscopy. Time (hours) is indicated. The experiment was repeated in three roots with similar results.
Rights and permissions
About this article
Cite this article
Yoshida, S., van der Schuren, A., van Dop, M. et al. A SOSEKI-based coordinate system interprets global polarity cues in Arabidopsis. Nature Plants 5, 160–166 (2019). https://doi.org/10.1038/s41477-019-0363-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-019-0363-6
This article is cited by
-
Plant cell polarity as the nexus of tissue mechanics and morphogenesis
Nature Plants (2021)
-
Conserved assembly of polarity complexes
Nature Reviews Molecular Cell Biology (2020)
-
Intracellular dynamics and transcriptional regulations in plant zygotes: a case study of Arabidopsis
Plant Reproduction (2020)