Abstract
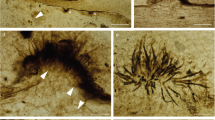
The colonization of land by vascular plants is an extremely important phase in Earth’s life history. This key evolutionary process is thought to have begun during the Middle Cambrian1 period and culminated in the Silurian/Early Devonian period (interval about 509–393 million years ago (Ma)), and is documented primarily by microfossils (that is, by dispersed spores, phytodebris including fragments of algae, tissues, sporangia and cuticles), tubes and rare megafossils2. A newly recognized fossil cooksonioid plant with in situ spores from the Barrandian area, Czech Republic, is of the highest importance because it represents extremely ancient megafossil evidence of land plant diploid generation: sporophytes (~432 Ma). The robust size of this plant places it among the largest known early polysporangiate land plants and it is probable that it attained adequate size for both aeration and effective photosynthetic competence. This would mean not only that sporophytes were photosynthetically autonomous but also that the they might have been able to sustain a relatively gametophyte-independent existence.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Morris, J. L. et al. The timescale of early land plant evolution. Proc. Natl Acad. Sci. USA 115, 2274–2283 (2018).
Wellman, C. H. & Strother, P. The terrestrial biota prior to the origin of land plants (embryophytes): a review of the evidence. Palaeontology 58, 601–627 (2015).
Rubinstein, C. V., Gerrienne, P., de la Punte, G. S., Astini, R. A. & Steemans, P. Early Middle Ordovician evidence for land plants in Argentina (eastern Gondwana). New Phytol. 188, 365–369 (2010).
Wellman, C. H. & Gray, J. The microfossil record of early land plants. Phil. Trans. R. Soc. Lond. B 355, 717–731 (2000).
Edwards, D., Morris, J. L., Richardson, J. B. & Kenrick, P. Cryptospores and cryptophytes reveal hidden diversity in early land floras. New Phytol. 202, 50–78 (2014).
Morris, J. L., Edwards, D., Richardson, J. B., Axe, L. & Davies, K. L. New plant taxa from the Lower Devonian (Lochkovian) of the Welsh Borderland, with a hypothesis on the relationship between hilate and trilete spore producers. Rev. Palaeobot. Palynol. 167, 51–81 (2011).
Edwards, D., Feehan, J. & Smith, D. G. A late Wenlock flora from Co. Tipperary, Ireland. Bot. J. Linn. Soc. 86, 19–36 (1983).
Taylor, T. N., Taylor, E. L. & Krings, M. Palaeobotany, the Biology and Evolution of Fossil Plants 2nd edn (Elsevier, Amsterdam, 2009).
Boyce, C. K. How green was Cooksonia? The importance of size in understanding the early evolution of physiology in the vascular plant lineage. Paleobiology 34, 179–194 (2008).
Edwards, D. & Kenrick, P. The early evolution of land plants, from fossils to genomics: a commentary on Lang (1937) ‘on the plant-remains from the Downtonian of England and Wales’. Phil. Trans. R. Soc. B 370, 20140343 (2015).
Edwards, D., Banks, H. P., Ciurca, S. J. & Laub, S. R. New Silurian cooksonias from dolostones of north-eastern North America. Bot. J. Linn. Soc. 146, 399–413 (2004).
Daber, R. Cooksonia—one of the most ancient psilophytes—widely distributed, but rare. Botanique (Nagpur) 2, 35–40 (1971).
Yurina, A. L. The Devonian flora of central Kazakhstan. Mat. Geol. Centr. Kazakh. 8, 14–19 (1969).
Gonez, P. & Gerrienne, P. A new definition and a lectotypification of the genus Cooksonia Lang 1937. Int. J. Plant Sci. 171, 199–215 (2010).
Edwards, D. Cells and tissues in the vegetative sporophytes of early land plants. New Phytol. 125, 225–247 (1993).
Lang, W. H. On the plant-remains from the Dowtonian of England and Wales. Phil. Trans. R. Soc. Lond. B 227, 245–291 (1937).
Gonez, P. & Gerrienne, P. Aberlemnia caledonica gen. et comb. nov., a new name for Cooksonia caledonica Edwards 1970. Rev. Palaeobot. Palynol. 163, 64–72 (2010).
Puttick, M. N. et al. The interrelationships of land plants and the nature of the ancestral embryophyte. Curr. Biol. 28, 733–745 (2018).
Cox, C. J., Li, B., Foster, P. G., Embley, T. M. & Civáň, P. Conflicting phylogenies for early land plants are caused by composition biases among synonymous substitutions. Syst. Biol. 63, 272–279 (2014).
Wickett, N. J. et al. Phylotranscriptomic analysis of the origin and early diversification of land plants. Proc. Natl Acad. Sci. USA 111, 4859–4868 (2014).
Harrison, J. Development and genetics in the evolution of land plant body plans. Phil. Trans. R. Soc. B 372, 20150490 (2016).
Thomas, R. J., Stanton, D. S., Longendorfer, D. H. & Farr, M. E. Physiological evaluation of the nutritional autonomy of a hornwort sporophyte. Bot. Gaz. 139, 306–311 (1978).
Proctor, M. C. F. 14C experiments on the nutrition of liverwort sporophytes: Pellia epiphylla, Cephalozia bicuspidata and Lophocolea heterophylla. J. Bryol. 12, 279–285 (1982).
Li, F. W., Villarreal, J. C. & Szövényi, P. Hornworts: an overlooked window into carbon-concentrating mechanisms. Trends Plant Sci. 22, 275–277 (2017).
Čelakovský, L. Ueber die verschiedenen Formen und die Bedeutung des Generationswechsels der Pflanzen. Sitz. Königl. Böhm. Ges. Wiss. Prag. 1874, 21–61 (1874).
Bower, F. O. On antithetic as distinct from homologous alternation of generations in plants. Ann. Bot. 4, 374–440 (1890).
Stebbins, G. L. & Hill, G. J. C. Did multicellular plants invade the land? Am. Nat. 115, 342–353 (1980).
Kenrick, P. Changing expressions: a hypothesis for the origin of the vascular plant life cycle. Phil. Trans. R. Soc. B 373, 20170149 (2018).
Acknowledgements
We acknowledge financial support from the Grant Agency of Czech Republic, First vascular land plants and palynomorphs from the Silurian–Lower Devonian of the Barrandian area, Czech Republic (GAČR 17-102333S). Part of this contribution was also supported by the Research Program of the Institute of Geology AS CR, v.v.i. (RVO67985831). Work in this field by V.Ž. was supported by the EU-PLANTORIGINS-ITN project 238640. Special thanks to D. Edwards, School of Earth and Ocean Sciences, Cardiff University, UK, for valuable discussions.
Author information
Authors and Affiliations
Contributions
M.L., J.K., V.Ž. and J.B. conceived the study. M.L., J.B. and P.Š. performed the laboratory work and analysed the data. J.K., M.L., V.Ž. and J.B. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Material, Supplementary References, Supplementary Figures 1–4 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Libertín, M., Kvaček, J., Bek, J. et al. Sporophytes of polysporangiate land plants from the early Silurian period may have been photosynthetically autonomous. Nature Plants 4, 269–271 (2018). https://doi.org/10.1038/s41477-018-0140-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-018-0140-y
This article is cited by
-
Fossils and plant evolution: structural fingerprints and modularity in the evo-devo paradigm
EvoDevo (2022)
-
The origin of a land flora
Nature Plants (2022)
-
Low atmospheric CO2 levels before the rise of forested ecosystems
Nature Communications (2022)
-
The impact of vegetation on meandering rivers
Nature Reviews Earth & Environment (2022)