Abstract
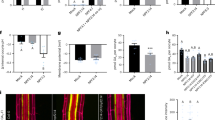
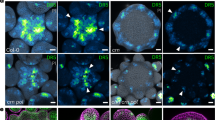
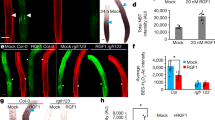
The phytohormone gibberellin (GA) is a key regulator of plant growth and development. Although the upstream regulation and downstream responses to GA vary across cells and tissues, developmental stages and environmental conditions, the spatiotemporal distribution of GA in vivo remains unclear. Using a combinatorial screen in yeast, we engineered an optogenetic biosensor, GIBBERELLIN PERCEPTION SENSOR 1 (GPS1), that senses nanomolar levels of bioactive GAs. Arabidopsis thaliana plants expressing a nuclear localized GPS1 report on GAs at the cellular level. GA gradients were correlated with gradients of cell length in rapidly elongating roots and dark-grown hypocotyls. In roots, accumulation of exogenously applied GA also correlated with cell length, intimating that a root GA gradient can be established independently of GA biosynthesis. In hypocotyls, GA levels were reduced in a phytochrome interacting factor (pif) quadruple mutant in the dark and increased in a phytochrome double mutant in the light, indicating that PIFs elevate GA in the dark and that phytochrome inhibition of PIFs could lower GA in the light. As GA signalling directs hypocotyl elongation largely through promoting PIF activity, PIF promotion of GA accumulation represents a positive feedback loop within the molecular framework driving rapid hypocotyl growth.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sun, T. P. Gibberellin metabolism, perception and signaling pathways in Arabidopsis. Arabidopsis Book 6, e0103 (2008).
Stowe, B. B. & Yamaki, T. The history and physiology of the gibberellins. Annu. Rev. Plant Physiol. 1957, 181–216 (1957).
Hu, J. et al. Potential sites of bioactive gibberellin production during reproductive growth in Arabidopsis. Plant Cell 20, 320–336 (2008).
Mitchum, M. G. et al. Distinct and overlapping roles of two gibberellin 3-oxidases in Arabidopsis development. Plant J. 45, 804–818 (2006).
Plackett, A. R. et al. Analysis of the developmental roles of the Arabidopsis gibberellin 20-oxidases demonstrates that GA20ox1, -2, and -3 are the dominant paralogs. Plant Cell 24, 941–960 (2012).
Sun, N. et al. Arabidopsis SAURs are critical for differential light regulation of the development of various organs. Proc. Natl Acad. Sci. USA 113, 6071–6076 (2016).
Okamoto, M. et al. Measurement of abscisic acid and gibberellins by gas chromatography/mass spectrometry. Methods Mol. Biol. 495, 53–60 (2009).
Achard, P. et al. Gibberellin signaling controls cell proliferation rate in Arabidopsis. Curr. Biol. 19, 1188–1193 (2009).
Band, L. R. et al. Growth-induced hormone dilution can explain the dynamics of plant root cell elongation. Proc. Natl Acad. Sci. USA 109, 7577–7582 (2012).
Bassel, G. W. et al. Mechanical constraints imposed by 3D cellular geometry and arrangement modulate growth patterns in the Arabidopsis embryo. Proc. Natl Acad. Sci. USA 111, 8685–8690 (2014).
Ubeda-Tomas, S. et al. Root growth in Arabidopsis requires gibberellin/DELLA signalling in the endodermis. Nat. Cell Biol. 10, 625–628 (2008).
Ubeda-Tomas, S. et al. Gibberellin signaling in the endodermis controls Arabidopsis root meristem size. Curr. Biol. 19, 1194–1199 (2009).
Shani, E. et al. Gibberellins accumulate in the elongating endodermal cells of Arabidopsis root. Proc. Natl Acad. Sci. USA 110, 4834–4839 (2013).
Bentsink, L. & Koornneef, M. Seed dormancy and germination. Arabidopsis Book 6, e0119 (2008).
Jiang, Z. et al. Phytochrome B and REVEILLE1/2-mediated signalling controls seed dormancy and germination in Arabidopsis. Nat. Commun. 7, 12377 (2016).
Hirose, F. et al. Cryptochrome and phytochrome cooperatively but independently reduce active gibberellin content in rice seedlings under light irradiation. Plant Cell Physiol. 53, 1570–1582 (2012).
Reid, J. B. et al. Control of gibberellin levels and gene expression during de-etiolation in pea. Plant Physiol. 128, 734–741 (2002).
Zhao, X. et al. A study of gibberellin homeostasis and cryptochrome-mediated blue light inhibition of hypocotyl elongation. Plant Physiol. 145, 106–118 (2007).
Oh, E. et al. Light activates the degradation of PIL5 protein to promote seed germination through gibberellin in Arabidopsis. Plant J. 47, 124–139 (2006).
Ast, C. et al. Ratiometric Matryoshka biosensors from a nested cassette of green- and orange-emitting fluorescent proteins. Nat. Commun. 8, 431 (2017).
Monshausen, G. B., Messerli, M. A. & Gilroy, S. Imaging of the Yellow Cameleon 3.6 indicator reveals that elevations in cytosolic Ca2+ follow oscillating increases in growth in root hairs of Arabidopsis. Plant Physiol. 147, 1690–1698 (2008).
Deuschle, K. et al. Rapid metabolism of glucose detected with FRET glucose nanosensors in epidermal cells and intact roots of Arabidopsis RNA-silencing mutants. Plant Cell 18, 2314–2325 (2006).
Grossmann, G. et al. The RootChip: an integrated microfluidic chip for plant science. Plant Cell 23, 4234–4240 (2011).
Jones, A. M. et al. Abscisic acid dynamics in roots detected with genetically encoded FRET sensors. eLife 3, e01741 (2014).
Waadt, R. et al. FRET-based reporters for the direct visualization of abscisic acid concentration changes and distribution in Arabidopsis. eLife 3, e01739 (2014).
Okumoto, S., Jones, A. & Frommer, W. B. Quantitative imaging with fluorescent biosensors. Annu. Rev. Plant Biol. 63, 663–706 (2012).
Ueguchi-Tanaka, M. et al. Molecular interactions of a soluble gibberellin receptor, GID1, with a rice DELLA protein, SLR1, and gibberellin. Plant Cell 19, 2140–2155 (2007).
Sun, T. P. Gibberellin-GID1-DELLA: a pivotal regulatory module for plant growth and development. Plant Physiol. 154, 567–570 (2010).
Reed, J. W. et al. Phytochrome A and Phytochrome B have overlapping but distinct functions in Arabidopsis development. Plant Physiol. 104, 1139–1149 (1994).
Jones, E. W. Tackling the protease problem in Saccharomyces cerevisiae. Methods Enzymol. 194, 428–453 (1991).
Murase, K. et al. Gibberellin-induced DELLA recognition by the gibberellin receptor GID1. Nature 456, 459–463 (2008).
Nakajima, M. et al. Identification and characterization of Arabidopsis gibberellin receptors. Plant J. 46, 880–889 (2006).
Schuster, C. et al. A regulatory framework for shoot stem cell control integrating metabolic, transcriptional, and phytohormone signals. Dev. Cell 28, 438–449 (2014).
Palmer, A. E. et al. Ca2+ indicators based on computationally redesigned calmodulin-peptide pairs. Chem. Biol. 13, 521–530 (2006).
Silverstone, A. L. et al. Repressing a repressor: gibberellin-induced rapid reduction of the RGA protein in Arabidopsis. Plant Cell 13, 1555–1566 (2001).
Kato, J. Nonpolar transport of gibberellin through pea stem and a method for its determination. Science 128, 1008–1009 (1958).
Dayan, J. et al. Leaf-induced gibberellin signaling is essential for internode elongation, cambial activity, and fiber differentiation in tobacco stems. Plant Cell 24, 66–79 (2012).
Eriksson, S. et al. GA4 is the active gibberellin in the regulation of LEAFY transcription and Arabidopsis floral initiation. Plant Cell 18, 2172–2181 (2006).
King, R. W. et al. Long-day induction of flowering in Lolium temulentum involves sequential increases in specific gibberellins at the shoot apex. Plant Physiol. 127, 624–632 (2001).
Ragni, L. et al. Mobile gibberellin directly stimulates Arabidopsis hypocotyl xylem expansion. Plant Cell 23, 1322–1336 (2011).
Cheng, H. et al. Gibberellin regulates Arabidopsis floral development via suppression of DELLA protein function. Development 131, 1055–1064 (2004).
Cowling, R. J. & Harberd, N. P. Gibberellins control Arabidopsis hypocotyl growth via regulation of cellular elongation. J. Exp. Bot. 50, 1351–1357 (1999).
Gendreau, E. et al. Cellular basis of hypocotyl growth in Arabidopsis thaliana. Plant Physiol. 114, 295–305 (1997).
Leivar, P. et al. The Arabidopsis phytochrome-interacting factor PIF7, together with PIF3 and PIF4, regulates responses to prolonged red light by modulating phyB levels. Plant Cell 20, 337–352 (2008).
Shin, J. et al. Phytochromes promote seedling light responses by inhibiting four negatively-acting phytochrome-interacting factors. Proc. Natl Acad. Sci. USA 106, 7660–7665 (2009).
Castillon, A., Shen, H. & Huq, E. Phytochrome Interacting Factors: central players in phytochrome-mediated light signaling networks. Trends Plant Sci. 12, 514–521 (2007).
de Lucas, M. et al. A molecular framework for light and gibberellin control of cell elongation. Nature 451, 480–484 (2008).
Feng, S. et al. Coordinated regulation of Arabidopsis thaliana development by light and gibberellins. Nature 451, 475–479 (2008).
Zhang, Y. et al. A quartet of PIF bHLH factors provides a transcriptionally centered signaling hub that regulates seedling morphogenesis through differential expression-patterning of shared target genes in Arabidopsis. PLoS Genet. 9, e1003244 (2013).
Jones, A. M. et al. In vivo biochemistry: applications for small molecule biosensors in plant biology. Curr. Opin. Plant Biol. 16, 389–395 (2013).
De Michele, R. et al. Fluorescent sensors reporting the activity of ammonium transceptors in live cells. eLife 2, e00800 (2013).
Deuschle, K. et al. Genetically encoded sensors for metabolites. Cytometry A 64, 3–9 (2005).
Kalderon, D. et al. A short amino acid sequence able to specify nuclear location. Cell 39, 499–509 (1984). (3 Pt 2).
Grossmann, G. et al. Time-lapse fluorescence imaging of Arabidopsis root growth with rapid manipulation of the root environment using the RootChip. J Vis. Exp. 65, e4290 (2012).
Rekas, A. et al. Crystal structure of venus, a yellow fluorescent protein with improved maturation and reduced environmental sensitivity. J. Biol. Chem. 277, 50573–50578 (2002).
Lelimousin, M. et al. Intrinsic dynamics in ECFP and Cerulean control fluorescence quantum yield. Biochemistry 48, 10038–10046 (2009).
Acknowledgements
We gratefully acknowledge S. Manoj-Kumar for technical assistance and B. DeMeo for development of XT Mean Intensity Ratio. We thank the Gatsby Charitable Foundation for funding support of A.R., A.W. and A.M.J.
Author information
Authors and Affiliations
Contributions
A.R., A.W., V.L. and A.M.J. ran the experiments. A.R., A.W., V.L. and A.M.J. analysed the results. A.R. and A.M.J. wrote the manuscript, and W.B.F. and A.M.J. designed and supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–11, Supplementary Table 1.
Life sciences reporting summary
Life sciences reporting summary
Supplementary Video 1
Time-course of nlsGPS1 emission ratios of an Arabidopsis root tip growing in the RootChip16. Time between frames was 5 minutes. Perfusion environment was as follows: 15 minutes mock solution, 20 minutes 100 nM GA4, 120 minutes mock solution, 20 minutes 100 nM GA4, 80 minutes mock solution.Time-course of nlsGPS1 emission ratios of an root tip growing in the RootChip16. Time between frames was 5 minutes. Perfusion environment was as follows: 15 minutes mock solution, 20 minutes 100 nM GA, 120 minutes mock solution, 20 minutes 100 nM GA, 80 minutes mock solution.
Rights and permissions
About this article
Cite this article
Rizza, A., Walia, A., Lanquar, V. et al. In vivo gibberellin gradients visualized in rapidly elongating tissues. Nature Plants 3, 803–813 (2017). https://doi.org/10.1038/s41477-017-0021-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-017-0021-9
This article is cited by
-
Next-generation ABACUS biosensors reveal cellular ABA dynamics driving root growth at low aerial humidity
Nature Plants (2023)
-
Seeing is understanding
Nature Plants (2023)
-
Elucidation of Heat Shock Transcription Factor Family (HSFs) Postulates Significant Insights for the Identification of Their Putative Roles in Root Development and Hormonal Regulation in Tomato
Journal of Plant Growth Regulation (2023)
-
Leaf explant based direct and indirect regeneration and SCoT marker assisted genetic fidelity analysis of endemic taxon Corynandra chelidonii var. pallae
Vegetos (2023)
-
Plant hormone regulation of abiotic stress responses
Nature Reviews Molecular Cell Biology (2022)