Abstract
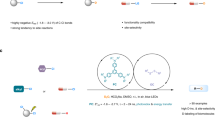
Herein, a facile and general electroreductive deuteration of unactivated alkyl halides (X = Cl, Br, I) or pseudo-halides (X = OMs) using D2O as the economical deuterium source was reported. In addition to primary and secondary alkyl halides, sterically hindered tertiary chlorides also work very well, affording the target deuterodehalogenated products with excellent efficiency and deuterium incorporation. More than 60 examples are provided, including late-stage dehalogenative deuteration of natural products, pharmaceuticals, and their derivatives, all with excellent deuterium incorporation (up to 99% D), demonstrating the potential utility of the developed method in organic synthesis. Furthermore, the method does not require external catalysts and tolerates high current, showing possible use in industrial applications.
Similar content being viewed by others
Introduction
Compounds with D-labeling play an important role in the realm of chemistry, mechanistic studies, and pharmaceutical science1,2,3,4,5,6,7,8,9. Dehalogenative deuteration from organic halides is a straightforward way to obtain deuterated target compounds. However, major studies focused on dehalogenative deuteration of aryl halides and activated alkyl halides10,11,12,13,14,15,16,17,18,19,20. In contrast, corresponding reports on unactivated alkyl halides, particularly on unactivated alkyl chlorides, have not been well elucidated. Several realizations have been reported but suffer from a number of problems, such as limiting to alkyl iodides using radical initiators21,22, the usage of a stoichiometric metal reductant23, long reaction time, and low reactivity of tertiary alkyl halides using photocatalysis24, etc. Therefore, the development of simple, efficient, and environmentally-friendly approaches to access deuterated compounds from unactivated alkyl halides with high D-incorporation is highly desirable.
Meanwhile, organic electrochemistry has become a powerful tool for sustainable synthesis by employing electron instead of stoichiometric amounts of redox reagents in the past decade25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44. Organic halides are important players in traditional organic chemistry as well as promising electrochemistry, owing to their versatile reactivity and readily available properties. Thus, impressive electrochemical progress of organic halides has been made, such as reductive bifunctionalization of alkenes45,46,47,48, reductive coupling with halides49,50,51,52,53,54,55,56, and others57,58,59,60,61,62,63,64,65,66,67,68. Hence, electrochemical deuteration of organic halides could be a straightforward and promising route for valuable deuterated compounds, which has gained increasing attention69,70,71. However, current research and success in electrochemical deuteration have been focused on aryl halides. In sharp contrast, electrochemically driven dehalogenative deuteration of unactivated halides has proven elusive (Fig. 1a). Direct electrochemical deuteration of unactivated alkyl halides is challenging due to their extremely negative reduction potential19,72,73,74, and likelihood of competitive undesired side reactions50.
Herein, we report our effort in developing this dehalogenative deuteration of unactivated alkyl halides, making it a general, economically and environmentally approach through electrochemistry (Fig. 1b). Notable features of this strategy include: (a) broad scope and excellent D-incorporation, including unsatisfactory tertiary chlorides in previous studies, (b) D2O as an economical deuterium source, (c) external catalysts-free and the use of electricity as an environmentally-friendly reductant, (d) catalytic amounts of electrolyte and no sacrificial anode, (e) tolerance with high current (up to 500 mA), making it potentially applicable in industrial production, (f) late-stage deuteration of complex natural products and drug derivatives.
Result
Optimization of electroreductive dehalogenation deuteration
Initially, we started our investigation using D2O as the economical deuterium source, unactivated alkyl bromide 1 as the substrate, and DMF as the solvent. After careful optimization, the corresponding product 2 was finally obtained in excellent yield and D-incorporation under 30 mA constant current with carbon felt (CF) and Pb as electrodes, in the presence of DIPEA as well as the catalytic amount of TBAI as the electrolyte in an undivided cell at room temperature for 10 hours (Table 1, entry 1). The control experiment revealed that the electricity was essential for the observed reactivity (entry 2). The reaction still worked well in the absence of DIPEA with 60% yield (entry 3), which showed that DMF could also be oxidized on anode. In addition, reducing the current to 20 mA led to lower yield and D-incorporation (entry 4). Surprisingly, the desired transformation proceeded smoothly with a higher current at 100 mA in 60 min (entry 5). It is notable that even under an extremely high current at 500 mA, the reaction still proceeded well to give the target product in only 15 min, without diminishing the yield and D-labeling level, showing potential usage in industrial amplification (entry 6). DMSO was proven to be a poor solvent for this transformation (entry 7), while MeCN gave a comparable yield but lower D-incorporation (entry 8). The reaction also worked well when nBu4NBF4 was used as the electrolyte, albeit in a slightly lower yield (entry 9). Finally, different electrodes were tested but none of the results surpassed entry 1 (entries 10-13).
Substrate scope
With the optimized reaction conditions in hand, we probed the generality of this electroreductive deuteration reaction. Firstly, various alkyl bromides were screened (Fig. 2A). Alkyl bromides with different lengths of carbon chains worked well to afford the corresponding products with excellent D-incorporation (2-7). Substituents with electron-withdrawing or donating properties were compatible and showed little effect on the reaction (8-13). Besides, other aromatic rings, including naphthalene, indole, and dihydrobenzofuran (15-17), were all well tolerated. The success of this procedure could be confirmed by the excellent compatibility of a wide range of functionalities, such as terminal ether (18), ester (19), and thioether (20), unprotected lactam (21-23), Boc-protected amine (24), terminal olefin (25), tetrahydropyran (26), as well as internal ester (27). Interestingly, for substrates containing both Br and Cl, it was found that the C-Br bond was easier to cleave (28). Besides, alkyl iodides also worked smoothly in this protocol, affording the corresponding products with excellent deuterium incorporation (Fig. 2B, 29-31). We were pleased to find that OMs could be a suitable leaving group to give the target product in 64% yield and 99% D-incorporation (32).
Electrochemically driven dehalogenative deuteration of unactivated alkyl halides. A Scope of alkyl bromides, B scope of alkyl iodides, C scope of alkylchlorides, and D complex biologically important substrates. aReaction conditions: bromide, iodide or pseudo-halides (0.5 mmol), D2O (25 mmol), TBAI (20 mol %), DIPEA (1.5 mmol), DMF (5.0 mL) under 30 mA constant current in an undivided cell at room temperature for 10 h with carbon felt as anode and lead plate as cathode. Isolated yield of product. Deuterium incorporation determined by 1H NMR. bAlkyl chlorides (0.5 mmol) was conducted under 50 mA constant current with graphite felt as anode. cDeuterium incorporation determined by GC-MS.
Next, we turned our attention to a more challenging target, unactivated alkyl chlorides (Fig. 2C). To our delight, primary alkyl chlorides took part in the reaction with a broad range of functionalities and showed similar results to alkyl bromides, yielding the corresponding products in good efficiency and D-incorporation (33-45). Furthermore, other alkyl chlorides, especially tertiary substrates that did not work well in previous studies24, yielded the desired products unquestionably in satisfactory D-incorporation using this method (46-49). To further demonstrate the generality, and environmentally-friendly nature of the developed method, as well as its application in bioactive molecules and drug discovery, the late-stage deuteration was conducted using a series of natural products, pharmaceuticals, and their derivatives (Fig. 2D). The dehalogenative deuteration of pharmaceuticals including Estrone (50), Pregnenolone (52), DL-Menthol (53), Phytol (57), and Triclosan (58) was successfully achieved in 77–83% yield with 97-98% D-incorporation. Pharmaceutical intermediates ethynyl estradiol (51) and dihydro cuminyl alcohol (54) containing unsaturated bonds can also be used in this protocol. Ibuprofen (55) and naproxen (56), which are commonly used for the ease of pain, afforded the corresponding deuterated compounds in good yields (67%, 88%) with excellent deuterium incorporation (both 98% D). Furthermore, amino acid and glucose derivatives that widely exist in organisms can also be deuterated smoothly with high levels of D-incorporation (59−64, 97−99%). In addition, the tolerance of the reaction system to tertiary chlorides can be extended to complex substrates (65, 66).
Electrochemical dehalogenative deuteration of alkyl bromides with 500 mA current
As high current density has a key impact on the amplification of reaction scales, we further explored the practicability of some representative substrates at a current of 500 mA. To our delight, the reactions were complete during a much shorter reaction time (in only 15 minutes). Simple alkyl bromides with a variety of functional groups (2, 16, 23, and 26) as well as complex alkyl bromides derived from ibuprofen (55), L-phenylalanine (59), Diacetonefructose (61), and D-mannofuranose (63) were all amenable. level of D-incorporation has not changed at all (Fig. 3). Alkyl chloride was also well tolerated and the corresponding product 37 was obtained in 15 min. In addition, this electrochemical reduction protocol could be applied to the gram-scale preparation of deuterated product 8 under 500 mA constant current (Fig. 4a).
aReaction conditions: alkyl bromide (0.5 mmol), D2O (25.0 mmol), TBAI (20 mol %), DIPEA (1.5 mmol), DMF (5.0 mL) under 500 mA constant current in an undivided cell at room temperature for 15 minutes with carbon felt as anode and lead plate as cathode. Isolated yield of product. bAlkyl chlorides (0.5 mmol) was conducted with graphite felt as anode.
Mechanistic studies
Several mechanistic studies were conducted to gain insight into the reaction mechanism. Firstly, 2 equiv. of TEMPO was added to the reaction system and in the absence of D2O, the TEMPO adduct was obtained in 64% yield (67), together with hydrodehalogenated product 2-H in 28% yield (Fig. 4b, eq 1). Then, repeat the reaction with 2 equiv. of D2O resulted in the TEMPO adduct in 21% yield and deuterodehalogenated product 2 in 75% yield with 55% D-incorporation (Fig. 4b, eq 2). These results indicated that the alkyl radical might be involved in the electrochemical system.
Then, we sought to gain further insight into the reaction mechanism through cyclic voltammetry (CV) experiments. A reduction peak of alkyl bromides 1 at −3.40 V (vs Ag/Ag+ in CH3CN) was observed (Fig. 5a, the red line), and no obvious reduction peak of D2O was observed (Fig. 5a). At the same time, the oxidation potential of TBAI (Fig. 5b, pink line, 1.0 V) was very close to that of DIPEA (Fig. 5b, blue line, 1.1 V). Thus we inferred that both TBAI and DIPEA might act as the electron donors in this reaction. Notably, the anodic oxidation of alkyl bromide 1 was also indicated with higher potential than that of DIPEA (green line). This might explain the improved yield once external DIPEA was added by inhibiting the undesired oxidation or decomposition of the starting material (Table 1, entries 1 and 3). In addition, during the reaction system without TBAI, an increase in current can be clearly observed, indicating the electrolysis of DIPEA incorporated with formed bromo anion may further generate conductive substances (Fig. 5c).
Based on the mechanistic studies and previous literature5,48, a plausible mechanism is proposed (Fig. 6). The reaction was initiated by the anodic oxidation of DIPEA and/or TBAI or DMF and the direct reduction of alkyl halides on the cathode to form the corresponding alkyl radical I and halide anions. The radical I continued to be reduced to an alkyl anion II at the cathode. Finally, intermediate II reacted with deuterated water to provide the expected product III. At the same time, iminium ion intermediate IV generated from DIPEA on the anode might combine with the released halogen anions to form ammonium salt, a possible conductive species.
Discussion
In conclusion, we have developed a general dehalogenative deuteration of unactivated alkyl (pseudo)halides (X = Cl, Br, I, OMs) driven by electrochemical force with high reactivity, selectivity, and excellent D-incorporation. D2O was used as an efficient and economical deuterium source. A wide range of unactivated alkyl halides containing diverse functionalities are well tolerated, as well as the extension to late stage deuteration of natural products, pharmaceuticals, and their derivatives. We envisage that the native features, including tolerance of high current and environmentally-friendly conditions, will bring further opportunity in its application to organic synthesis, drug discovery and modification, and even industrial production. Further electrochemical transformations of unactivated alkyl halides are currently ongoing in our laboratory.
Methods
General procedure of the electrochemical dehalogenative deuteration of alkyl bromide, iodide or pseudo-halides
The electrocatalysis was carried out in an undivided cell with a carbon felt anode (10 mm × 15 mm × 5 mm) and a lead cathode (10 mm × 15 mm × 0.3 mm). To a 15 mL pre‐dried undivided electrochemical cell (15 mL) equipped with a magnetic bar were added alkyl bromide, iodide or pseudo-halides (0.5 mmol, 1 equiv), TBAI (36.9 mg, 0.1 mmol, 20 mol%), and DMF (5.0 mL). Then D2O (25 mmol, 50 equiv) and DIPEA (1.5 mmol, 3.0 equiv) were added via a syringe. The electrocatalysis was performed at room temperature with a constant current of 30 mA maintained for 10 h. The carbon felt anode was washed with EtOAc (3 × 5 mL) in an ultrasonic bath. H2O (20 mL) was added to the system, and the resulting mixture was extracted with EtOAc (3 ×20 mL). The combined organic phase was dried with anhydrous Na2SO4, filtered, and concentrated in vacuo. The crude product was purified by column chromatography to furnish the desired product.
General procedure of the electrochemical dehalogenative deuteration of alkyl chlorides
The electrocatalysis was carried out in an undivided cell with a graphite felt anode (10 mm × 15 mm × 5 mm) and a lead cathode (10 mm × 15 mm × 0.3 mm). To a 15 mL pre‐dried undivided electrochemical cell (15 mL) equipped with a magnetic bar were added alkyl chloride (0.5 mmol, 1 equiv), TBAI (36.9 mg, 0.1 mmol, 20 mol%) and DMF (5.0 mL). Then D2O (25 mmol, 50 equiv) and DIPEA (1.5 mmol, 3.0 equiv) were added via a syringe. The electrocatalysis was performed at room temperature with a constant current of 50 mA maintained for 10 h. The carbon felt anode was washed with EtOAc (3 × 5 mL) in an ultrasonic bath. H2O (20 mL) was added to the system, and the resulting mixture was extracted with EtOAc (3 × 20 mL). The combined organic phase was dried with anhydrous Na2SO4, filtered, and concentrated in vacuo. The crude product was purified by column chromatography to furnish the desired product.
General procedure of the electrochemical dehalogenative deuteration of alkyl bromides and chloride with 500 mA current
The electrocatalysis was carried out in an undivided cell with a carbon felt anode (or graphite felt) (10 mm × 15 mm × 5 mm) and a lead cathode (10 mm × 15 mm × 0.3 mm). To a 15 mL pre‐dried undivided electrochemical cell (15 mL) equipped with a magnetic bar were added alkyl bromide or chloride (0.5 mmol, 1 equiv), TBAI (36.9 mg, 0.1 mmol, 20 mol%) and DMF (5.0 mL). Then D2O (25 mmol, 50 equiv) and DIPEA (1.5 mmol, 3.0 equiv) were added via a syringe. The electrocatalysis was performed at room temperature with a constant current of 500 mA maintained for 15 min (reaction system exothermic). The carbon felt anode was washed with EtOAc (3 × 5 mL) in an ultrasonic bath. H2O (20 mL) was added to the system, and the resulting mixture was extracted with EtOAc (3 × 20 mL). The combined organic phase was dried with anhydrous Na2SO4, filtered, and concentrated in vacuo. The crude product was purified by column chromatography to furnish the desired product.
Gram-scale synthesis of 8
To an undivided reaction flask (diameter: 40 mm, length: 130 mm, volume: 200 mL) equipped with a teflon-coated magnetic stirring bar and teflon cap, a carbon felt anode (25 mm × 50 mm × 5 mm), and a lead cathode (25 mm × 50 mm × 0.3 mm) were added (4-bromobutoxy)benzene (3.44 g, 15 mmol), TBAI (1.11 g, 3.0 mmol), DMF (100 mL), D2O (15 mL, 0.75 mol) and DIPEA (5.80 g, 45 mmol). The electrocatalysis was performed at room temperature with a constant current of 500 mA maintained for 4 h (the reaction system was exothermic). The carbon felt anode was washed with EtOAc (3 × 20 mL) in an ultrasonic bath. H2O (200 mL) was added to the system, and the resulting mixture was extracted with EtOAc (3 × 200 mL). The combined organic phase was dried with anhydrous Na2SO4, filtered, and concentrated in vacuo. The crude product was purified by column chromatography to furnish the desired product.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information files. Extra data are available from the author upon request.
References
Wiberga, K. B. The Deuterium Isotope Effect. Chem. Rev. 55, 713–743 (1995).
Gómez-Gallego, M. & Sierra, M. A. Kinetic Isotope Effects in the Study of Organometallic Reaction Mechanisms. Chem. Rev. 111, 4857–4963 (2011).
Kushner, D. J., Baker, A., & Dunstall, T. G. C. Pharmacological Uses and Perspectives of Heavy Water and Deuterated Compounds. J. Physiol. Pharmacol. 77, 79−88 (1999).
Pirali, T., Serafini, M., Cargnin, S. & Genazzani, A. A. Applications of Deuterium in Medicinal Chemistry. J. Med. Chem. 62, 5276–5297 (2019).
Liu, X., Liu, R., Qiu, J., Cheng, X. & Li, G. Chemical-Reductant-Free Electrochemical Deuteration Reaction using Deuterium Oxide. Angew. Chem. Int. Ed. 59, 13962–13967 (2020).
Li, W. et al. Scalable and Selective Deuteration of (Hetero)arenes. Nat. Chem. 14, 334–341 (2022).
Norcott, P. L. Current electrochemical approaches to selective deuteration. Chem. Commun. 58, 2944–2953 (2022).
Liao, L.-L. et al. Electrochemical Ring-Opening Dicarboxylation of Strained Carbon–Carbon Single Bonds with CO2: Facile Synthesis of Diacids and Derivatization into Polyesters. J. Am. Chem. Soc. 144, 2062–2068 (2022).
Kopf, S. et al. Recent Developments for the Deuterium and Tritium Labeling of Organic Molecules. Chem. Rev. 122, 6634–6718 (2022).
Cockrell, J. R. & Murray, R. W. Deuterium labeling by electrochemical reactions. J. Electrochem. Soc. 119, 849–851 (1972).
Renaud, R. N. Electrochemical Synthesis of Deuterio Organic Compounds. I. Electrochemical Reduction of 1-Halonaphthalenes and Synthesis of 1-Chloro-4-methylnaphthalene. Can. J. Chem. 52, 376–380 (1974).
Grimshaw, J. & Trocha-Grimshaw, J. Electrochemical reactions. Part XVIII. Reductive cleavage of aromatic carbon–halogen bonds in the presence of deuterium oxide. J. Chem. Soc. Perkin Trans. 2, 215–218 (1975).
Tashiro, M., Iwasaki, A. & Fukata, G. Studies on Selective Preparation of Aromatic Compounds. 14. An Attempt to Prepare All Possible Deuterated Phenols by the Reductive Dehalogenation of the Corresponding Halophenols with Raney Alloys in an Alkaline Deuterium Oxide Solution. J. Org. Chem. 43, 196–199 (1978).
Janni, M. & Peruncheralathan, S. Catalytic Selective Deuteration of Halo(hetero)Arenes. Org. Biomol. Chem. 14, 3091–3097 (2016).
Kuriyama, M. et al. Deuterodechlorination of Aryl/Heteroaryl Chlorides Catalyzed by a Palladium/Unsymmetrical NHC System. J. Org. Chem. 81, 8934–8946 (2016).
Li, W. et al. Copper-catalysed Low-temperature Water–gas Shift Reaction for Selective Deuteration of Aryl Halides. Chem. Sci. 12, 14033–14038 (2012).
Wang, X. et al. General and Practical Potassium Methoxide/Disilane-Mediated Dehalogenative Deuteration of (Hetero)Arylhalides. J. Am. Chem. Soc. 140, 10970–10974 (2018).
Liu, C. et al. Controllable Deuteration of Halogenated Compounds by Photocatalytic D2O splitting. Nat. Commun. 9, 80 (2018).
Zhou, Z.-Z., Zhao, J.-H., Gou, X.-Y., Chen, X.-M. & Liang, Y.-M. Visible-Light-Mediated Hydrodehalogenation and Br/D Exchange of Inactivated Aryl and Alkyl Halides with a Palladium Complex. Org. Chem. Front. 6, 1649–1654 (2019).
Ling, X. et al. A Visible-light-photocatalytic Water-splitting Strategy for Sustainable Hydrogenation/deuteration of Aryl Chlorides. Sci. China Chem. 63, 386–392 (2020).
Soulard, V., Villa, G., Vollmar, D. P. & Renaud, P. Radical Deuteration with D2O: Catalysis and Mechanistic Insights. J. Am. Chem. Soc. 140, 155–158 (2018).
Song, Z. et al. Water Compatible Hypophosphites-d2 Reagents: Deuteration Reaction via Deutero-deiodination in Aqueous Solution. Org. Lett. 22, 1736–1741 (2020).
Xia, A., Xie, X., Hu, X., Xu, W. & Liu, Y. Dehalogenative Deuteration of Unactivated Alkyl Halides Using D2O as the Deuterium Source. J. Org. Chem. 84, 13841–13857 (2019).
Li, Y. et al. Organophotocatalytic Selective Deuterodehalogenation of Aryl or Alkyl Chlorides. Nat. Commun. 12, 2894 (2021).
Yoshida, J., Kataoka, K., Horcajada, R. & Nagaki, A. Modern Strategies in Electroorganic Synthesis. Chem. Rev. 108, 2265–2299 (2008).
Francke, R. & Little, R. D. Redox Catalysis in Organic Electrosynthesis: Basic Principles and Recent Developments. Chem. Soc. Rev. 43, 2492–2521 (2014).
Yan, M., Kawamata, Y. & Baran, P. S. Synthetic Organic Electrochemical Methods Since 2000: On the Verge of a Renaissance. Chem. Rev. 117, 13230–13319 (2017).
Sauermann, N., Meyer, T. H., Qiu, Y. & Ackermann, L. Electrocatalytic C−H Activation. ACS Catal. 8, 7086–7103 (2018).
Yuan, Y. & Lei, A. Electrochemical Oxidative Cross-Coupling with Hydrogen Evolution Reactions. Acc. Chem. Res. 52, 3309–3324 (2019).
Röckl, J. L., Pollok, D., Franke, R. & Waldvogel, S. R. A Decade of Electrochemical Dehydrogenative C, C-Coupling of Aryls. Acc. Chem. Res. 53, 45–61 (2020).
Jiao, K.-J., Xing, Y.-K., Yang, Q.-L., Qiu, H. & Mei, T.-S. Site-Selective C–H Functionalization via Synergistic Use of Electrochemistry and Transition Metal Catalysis. Acc. Chem. Res. 53, 300–310 (2020).
Qiu, Y. et al. Rhodaelectro-Catalyzed C–H and C–C Activation. CCS Chem. 2, 1529–1552 (2020).
Novaes, L. F. T. et al. Electrocatalysis as an Enabling Technology for Organic Synthesis. Chem. Soc. Rev. 50, 7941–8002 (2021).
Cheng, X., et al. Recent Applications of Homogeneous Catalysis in Electrochemical Organic Synthesis. CCS Chem. https://doi.org/10.31635/ccschem.021.202101451 (2022).
Bäckvall, J.-E., Gogoll, A. Palladium-Hydroquinone Catalysed Electrochemical 1,4-Oxidation of Conjugated Dienes. J. Chem. Soc. Chem. Commun. 1236–1238 (1987).
Horn, E. J. et al. Scalable and Sustainable Electrochemical Allylic C–H oxidation. Nature 533, 77–81 (2016).
Qiu, Y., Tian, C., Massignan, L., Rogge, T. & Ackermann, L. Electrooxidative Ruthenium-Catalyzed C−H/O−H Annulation by Weak O-Coordination. Angew. Chem. Int. Ed. 57, 5818–5822 (2018).
Tang, S., Liu, Y. & Lei, A. Electrochemical Oxidative Cross-coupling with Hydrogen Evolution: A Green and Sustainable Way for Bond Formation. Chem 4, 27–45 (2018).
Shrestha, A., Lee, M., Dunn, A. L. & Sanford, M. S. Palladium-Catalyzed C−H Bond Acetoxylation via Electrochemical Oxidation. Org. Lett. 20, 204–207 (2018).
Xiang, J. et al. Hindered Dialkyl Ether Synthesis with Electrogenerated Carbocations. Nature 573, 398–402 (2019).
Shen, T. & Lambert, T. H. Electrophotocatalytic Diamination of Vicinal C–H Bonds. Science 371, 620–626 (2021).
Liang, Y. et al. Electrochemically Induced Nickel Catalysis for Oxygenation Reactions with Water. Nat. Catal. 4, 116–123 (2021).
Ma, C. et al. Recent advances in organic electrosynthesis employing transition metal complexes as electrocatalysts. Sci. Bull. 66, 2412–2429 (2021).
Zhang, B., et al. Ni-Electrocatalytic C(sp3)–C(sp3) Doubly Decarboxylative Coupling. Nature, https://doi.org/10.1038/s41586-022-04691-4. (2022).
Kurono, N., Honda, E., Komatsu, F., Orito, K. & Tokuda, M. Regioselective Synthesis of Substituted 1-Indanols, 2,3-Dihydrobenzo-furans and 2,3-Dihydroindoles by Electrochemical Radical Cyclization Using an Arene Mediator. Tetrahedron 60, 1791–1801 (2004).
Mitsudo, K. et al. Electro-Reductive Cyclization of Aryl Halides Promoted by Fluorene Derivatives. Electrochim. Acta 82, 444–449 (2012).
Zhang, W. & Lin, S. electroreductive Carbofunctionalization of Alkenes with Alkyl Bromides via a Radical-Polar Crossover Mechanism. J. Am. Chem. Soc. 142, 20661–20670 (2020).
Dong, X., Roeckl, J. L., Waldvogel, S. R. & Morandi, B. Merging Shuttle Reactions and Paired Electrolysis for Reversible Vicinal Dihalogenations. Science 371, 507–514 (2021).
Kuroboshi, M., Shiba, T. & Tanaka, H. Viologen as Catalytic Organic Reductant: Electro-Reductive Dimerization of Aryl Bromides in a Pd/Viologen Double Mediatory System. Tetrahedron Lett. 54, 3666–3668 (2013).
Jiao, K. et al. Nickel-Catalyzed Electrochemical Reductive Reley Cross-Coupling of Alkyl Halides to Aryl Halides. Angew. Chem. Int. Ed. 59, 6520–6524 (2020).
Qiu, H. et al. Enantioselective Ni-Catalyzed Electrochemical Synthesis of Biaryl Atropisomers. J. Am. Chem. Soc. 142, 9872–9878 (2020).
Li, Z.-J. et al. Electrochemically Enabled, Nickel-Catalyzed Dehydroxylative Cross-Coupling of Alcohols with Aryl Halides. J. Am. Chem. Soc. 143, 3536–3543 (2021).
Kumar, G. S. et al. Nickel-Catalyzed Chain-Walking Cross-Electrophile Coupling of Alkyl and Aryl Halides and Olefin Hydroarylation Enabled by Electrochemical Reduction. Angew. Chem. Int. Ed. 59, 6513–6519 (2020).
Truesdell, B. L., Hamby, T. B. & Sevov, C. S. General C(sp2)−C(sp3) Cross-Electrophile Coupling Reactions Enabled by Overcharge Protection of Homogeneous Electrocatalysts. J. Am. Chem. Soc. 142, 5884–5893 (2020).
Zhang, W., et al. Electrochemically Driven Cross-Electrophile Coupling of Alkyl Halides. See, ChemRxiv preprint https://doi.org/10.26434/chemrxiv-2021-c2hd6-v2 (2021).
Zhang, W. et al. Electrochemically driven cross-electrophile coupling of alkyl halides. Nature 604, 292–297 (2022).
Kim, H., Kim, H., Lambert, T. H. & Lin, S. Reductive Electrophotocatalysis: Merging Electricity and Light to Achieve Extreme Reduction Potentials. J. Am. Chem. Soc. 142, 2087–2092 (2020).
Sun, L., Sahloul, K. & Mellah, M. Use of Electrochemistry to Provide Efficient SmI2 Catalytic System for Coupling Reactions. ACS Catal. 3, 2568–2573 (2013).
Gao, Y. et al. Electrochemical Nozaki−Hiyama−Kishi Coupling: Scope, Applications, and Mechanism. J. Am. Chem. Soc. 143, 9478–9488 (2021).
Liu, D., Ma, H.-X., Fang, P. & Mei, T.-S. Nickel-Catalyzed Thiolation of Aryl Halides and Heteroaryl Halides through Electrochemistry. Angew. Chem. Int. Ed. 58, 5033–5037 (2019).
Kawamata, Y. et al. Electrochemically Driven, Ni-Catalyzed Aryl Amination: Scope, Mechanism, and Applications. J. Am. Chem. Soc. 141, 6392–6402 (2019).
Zhang, H.-J. et al. Chemoselective, Scalable Nickel-Electrocatalytic O-Arylation of Alcohols. J. Am. Chem. Soc. 143, 9478–9488 (2021).
Liu, D. et al. Nickel-Catalyzed N-Arylation of NH-Sulfoximines with Aryl Halides via Paired Electrolysis. Angew. Chem. Int. Ed. 60, 9444–9449 (2021).
Senboku, H., Kanaya, H., Fujimura, Y. & Tokuda, M. Stereo-chemical Study on Electrochemical Carboxylation of Vinyl Triflates. J. Electroanal. Chem. 507, 82–88 (2001).
Ang, N. W. J., Oliveira, J. C. A. & Ackermann, L. Electroreductive Cobalt-Catalyzed Carboxylation: Cross-Electrophile Electrocoupling with Atmospheric CO2. Angew. Chem. Int. Ed. 59, 12842–12847 (2020).
Sun, G.-Q. et al. Nickel-catalyzed electrochemical carboxylation of unactivated aryl and alkyl halides with CO2. Nat. Commun. 12, 7086 (2021).
Parrish, J. D. & Little, R. D. Electrochemical Formation of Glycals in THF. Tetrahedron Lett. 42, 7371–7374 (2001).
Huang, J.-M., Wang, X.-X. & Dong, Y. Electrochemical Allylation Reactions of Simple Imines in Aqueous Solution Mediated by Nanoscale Zinc Architectures. Angew. Chem., Int. Ed. 50, 924–927 (2001).
Mitsudo, K., Okada, T., Shimohara, S., Mandai, H. & Suga, S. Electro-reductive Halogen-Deuterium Exchange and Methylation of Aryl Halides in Acetonitrile. Electrochem 81, 362–364 (2013).
Liu, C., Han, S., Li, M., Chong, X. & Zhang, B. Electrocatalytic Deuteration of Halides with D2O as the Deuterium Source over a Copper Nanowire Arrays Cathode. Angew. Chem. Int. Ed. 59, 18527–18531 (2020).
Lu, L., Li, H., Zheng, Y., Bu, F. & Lei, A. Facile and Economical Electrochemical Dehalogenative Deuteration of (Hetero)Aryl Halides. CCS Chem. 3, 2669–2675 (2020).
Lambert, F. L. & Ingall, G. B. Voltammetry of organic halogen compounds. IV. Reduct. Org. chlorides vitreous (Glass.) carbon electrode Tetrahedron Lett. 15, 3231–3234 (1974).
Cybularczyk-Cecotka, M., Szczepanik, J. & Giedyk, M. Photocatalytic strategies for the activation of organic chlorides. Nat. Catal. 3, 872–886 (2020).
Wang, B. et al. Electrochemical Borylation of Alkyl Halides: Fast, Scalable Access to Alkyl Boronic Esters. J. Am. Chem. Soc. 143, 12985–12991 (2021).
Acknowledgements
Financial support from the Fundamental Research Funds for the Central Universities (No. 63213063), Frontiers Science Center for New Organic Matter, Nankai University (Grant No. 63181206) and Nankai University are gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
Y.Q. and P.L. conceived and designed the study and wrote the manuscript. P.L, C.C.G, S.W, D.M, T.F, Y.W, Y.Q. performed the experiments, mechanistic studies and revised the manuscript. All authors contributed to the analysis and interpretation of the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interest.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, P., Guo, C., Wang, S. et al. Facile and general electrochemical deuteration of unactivated alkyl halides. Nat Commun 13, 3774 (2022). https://doi.org/10.1038/s41467-022-31435-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-31435-9
This article is cited by
-
Electroreduction of unactivated alkenes using water as hydrogen source
Nature Communications (2024)
-
Electrochemical hydrogenation and oxidation of organic species involving water
Nature Reviews Chemistry (2024)
-
Nickel-electrocatalysed C(sp3)–C(sp3) cross-coupling of unactivated alkyl halides
Nature Catalysis (2024)
-
Electrochemical radical-polar crossover: a radical approach to polar chemistry
Science China Chemistry (2024)
-
Electrochemical halogen-atom transfer alkylation via α-aminoalkyl radical activation of alkyl iodides
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.