Abstract
Life in the clouds of Venus, if present in sufficiently high abundance, must be affecting the atmospheric chemistry. It has been proposed that abundant Venusian life could obtain energy from its environment using three possible sulfur energy-metabolisms. These metabolisms raise the possibility of Venus’s enigmatic cloud-layer SO2-depletion being caused by life. We here couple each proposed energy-metabolism to a photochemical-kinetics code and self-consistently predict the composition of Venus’s atmosphere under the scenario that life produces the observed SO2-depletion. Using this photo-bio-chemical kinetics code, we show that all three metabolisms can produce SO2-depletions, but do so by violating other observational constraints on Venus’s atmospheric chemistry. We calculate the maximum possible biomass density of sulfur-metabolising life in the clouds, before violating observational constraints, to be ~10−5 − 10−3 mg m−3. The methods employed are equally applicable to aerial biospheres on Venus-like exoplanets, planets that are optimally poised for atmospheric characterisation in the near future.
Similar content being viewed by others
Introduction
Sulfur dioxide, SO2, is observed to be depleted in the cloud layer of Venus by orders of magnitude compared to its below-cloud abundance1. Gas-phase sulfur chemistry at the cloud top is dominated by the photochemical formation of sulfuric acid, where one photon of light, one H2O molecule, and up to two SO2 molecules, are consumed in the production of one H2SO4 molecule, which then condenses and forms the bulk of the mass loading in the clouds. According to this reaction scheme, the maximum depletion of SO2 that could possibly be achieved is within a factor of 2 of the H2O mixing ratio, however this is inconsistent with the observations of ~150 ppm below-cloud SO2 and ~10 ppb above-cloud SO2, yet only ~30 ppm below-cloud H2O—a factor of five lower than the SO2 mixing ratio. Thus, Venus’s observed SO2 depletion represents a key gap in our understanding of the planet’s atmosphere2,3,4, for which no full-atmosphere model can self-consistently predict via exclusively gas-phase chemistry5. This has led to the conclusion that there must be some currently unknown chemical pathway responsible for the depletion of SO2 in the middle atmosphere. One abiotic solution has been proposed which hypothesises that the delivery of hydroxide salts to the cloud layer in the form of mineral dust can enable reduction of SO2 within the cloud droplets to reproduce the observed trend6. It is not yet known whether this in-droplet mineral chemistry is occurring in the clouds of Venus, or whether the delivery of mineral dust to the cloud droplets is sufficient for this mechanism to produce the observed SO2 depletion. This raises the possibility of an alternative hypothesis: the observed depletion of SO2 in the clouds of Venus is due to the bio-chemistry of sulfur-metabolising life in an extant aerial biosphere harboured within the cloud layer.
While the present day surface of Venus is uninhabitable, the lower-cloud layer lies in the region of the atmosphere where pressure-temperature conditions are suitable for life (47–57 km altitude). This has led to suggestions of microbial life being a possible explanation for the existence of large and irregularly shaped (‘mode 3’) aerosol particles in the lower clouds and being the identity of the unknown UV-absorber7,8,9. It has recently been suggested that the metabolic activity of microbial life in the clouds could also be the origin of the reduced chemical species PH3 and NH3, which are biotically produced on Earth and have been tentatively observed in the Venusian atmosphere at abundances many orders of magnitude greater than those predicted given their short photochemical lifetimes10,11,12,13. A plausible life cycle for a strictly aerial biosphere in the clouds has been investigated11, and a suite of mission concepts for a future Venus Life Finder Mission have been recently presented to the community14. It it therefore timely to provide novel and quantitative tests of the potential metabolisms of a hypothetical aerial biosphere to better understand the requirements and limits of life in the clouds, if it exists. Given that the terrestrial biosphere has had an inexorable effect on the Earth’s atmospheric chemistry throughout geological time, investigating Venusian biochemistries self-consistently with the atmospheric chemistry is crucially important to assessing the viability of the aerial life-hypotheses.
It has historically been proposed that hypothetical Venusian life could have originated at the planet’s surface or been seeded from Earth, if Venus was ever habitable in its past. This surface-dwelling life would have subsequently migrated to the temperate region of the cloud layer as Venus underwent runaway greenhouse warming, residing in the clouds today as an aerial biosphere7,8,15. Runaway greenhouse warming is instigated by an increase in the surface temperature of a planet which causes an increase in the evaporation of surface water to the atmosphere. Since H2O is a strong greenhouse gas, the increase in atmospheric H2O further exacerbates the warming effect, creating a positive climate feedback. Atmospheric H2O is eventually lost via photo-dissociation followed by hydrogen loss to space, and the record of the past water content is left imprinted in the atmospheric deuterium to hydrogen ratio (D/H). The enhanced D/H ratio measured in the atmosphere of Venus provides evidence of past water loss16, however it is unclear whether this water was ever present as liquid at the surface or always as gas in a hot steam atmosphere. If liquid water was present at the surface of Venus in its past then the planet’s slow rotation rate would have enabled a stabilising cloud-climate feedback mechanism to be established17, maintaining habitable surface conditions despite the increasing Solar luminosity over time. This phase of habitability could have lasted for up to ~900 Myrs if Venus has always been in a stagnant lid tectonic regime18, or if early Venus had Earth-like plate tectonics then this habitable period could have lasted for up to 4 Gyrs, until runaway greenhouse was instigated by voluminous magmatism17. In contrast, other modelling has suggested that, due to its proximity to the Sun, Venus could never have cooled sufficiently for liquid water to condense at the surface following its magma ocean phase, and thus a stabilising cloud feedback could never have been established19. In this case, life in the clouds would likely require an aerial origin of life scenario within cloud droplets, utilising very different bio-chemistry to terran life.
Regardless of the proposed origins or adaptations of hypothetical life on Venus, the requirement of life to obtain energy and materials from its environment is universal. A stable biosphere must consume metabolic feedstock molecules and release metabolic products to liberate energy regardless of the particulars of the biochemical machinery. A Venusian aerial biosphere has been suggested to be dependent on a sulfur-based energy-metabolism20,21 for three reasons: (1) the chemical energy liberated via currently proposed sulfur-based metabolisms20,21 is sufficient to meet the energy requirements of a cell22; (2) the relatively high abundance of sulfur species (e.g. SO2, OCS, H2S) in the Venusian atmosphere means these are the abundant feedstock molecules that could support a global biosphere; and (3) terrestrial acidophillic organisms provide a precedent for life in an acid environment which uses sulfur-metabolism as a primary source of energy (e.g. acidithiobacillus ferooxidans). Further support for sulfur-metabolising life in Venus’s clouds comes from an extensive analysis into the spectral absorptivities of proteins and cofactors used by terrestrial sulfur-metabolising microbes. Significant overlap was found between the spectral appearance of these molecules and the UV features of Venus’s spectrum, currently attributed to the unknown absorber9.
Three sulfur-based energy-metabolisms for putative Venusian microbes have been proposed by Schulze-Makuch et al. and Schulze-Makuch and Irwin20,21, each of which utilise the relatively abundant sulfur species in the atmosphere and can, in principle, induce the depletion of SO2 as a result of their respective biochemical effects on the atmospheric chemistry. One proposed metabolism, ‘A’, is a primitive anoxygenic photosystem used by microbes on Earth, oxidising hydrogen sulfide (H2S) to elemental sulfur (S) meanwhile fixing carbon from atmospheric CO2 into organics20:
where (CH2O) represents carbon locked in organic molecules (e.g. glucose). We couple this photosystem to reaction (2), where the organics produced by the anabolism of reaction (1) are consumed in catabolism to liberate chemical energy:
We have constructed reaction (2) as a hypothetical variant of terrestrial oxygenic respiration with O2 replaced by SO2. The widespread use of O2 for respiration by life on Earth is believed to be an adaptation of anoxygenic life following oxygenation of the Earth’s atmosphere to contain abundant O223. In contrast, O2 is not observed in the upper atmosphere of Venus leading to an inferred upper limit on its abundance of <2.8 ppm4. One measurement of O2 within the cloud layer suggests an abundance of ~70 ppm24 however this detection remains controversial, and is discrepant with the results of modelling6 and the subsequent non-detection of O225. Taking the widely accepted view that there is not substantial O2 in the atmosphere of Venus, we assume that hypothetical Venusian life would have evolved to utilise the abundant SO2 in the atmosphere in an analogous fashion to terrestrial oxygenic life’s respiration of atmospheric O2. The net metabolic reaction for metabolism A is therefore:
Reaction (3) is expressed in it’s simplest stoichiometric ratio. In reality life is more likely to be performing higher multiples of reaction (3), constructing and burning longer chain organic molecules, and releasing longer sulfur allotropes, rather than S and S2. We use this representation of the metabolism for modelling simplicity when coupling bio-chemistry to the atmospheric chemistry, and this does not impact any of the results that we will present.
Metabolism B is a chemoautotrophic pathway proposed in the literature for hypothetical Venusian life, that exploits the redox disequilibrium between CO, H2 and SO2 in the atmosphere21:
Metabolism C is also a chemoautotrophic pathway proposed in the literature for hypothetical Venusian life, and exploits the redox disequilibrium between CO and SO2 in the atmosphere21:
Both of the proposed chemotrophic metabolisms, B and C, could yield ~240 kJ mol−1 of energy to a putative Venusian microbe, which would be a sufficient source of chemical energy for life to thrive22, provided the reactant species are in high enough abundance.
We present a framework for the coupling of each proposed energy-metabolism to a photochemical-kinetic network for modelling planetary atmospheres in 1D6,26, and self-consistently solve for the atmospheric composition of Venus required to reproduce the observed SO2-profile biochemically. We incorporate each sulfur-based metabolism into our reaction network in turn, impose that the reaction rate can only be non-zero between 47 and 57 km altitude (where ambient atmospheric temperatures are between 0 − 100∘C and aerosols are large enough to accommodate microbial colonies11), and investigate the parameter space of metabolic activity versus availability of metabolic inputs. While plausible metabolic pathways and survival strategies for an extant Venusian biosphere have been suggested in previous studies9,11,15,20,21 none have been investigated self-consistently with the wider atmospheric chemistry. By coupling proposed metabolic requirements with self-consistent models of atmospheric chemistry, we here produce a rigorous test of whether the proposed metabolisms could support a Venusian aerial biosphere. The results of our investigation show that, while proposed metabolic incorporation of gas-phase species can reproduce the sulfur depletion, in every case the resultant atmospheric chemistry violates separate observational constraints on the lower atmosphere of Venus.
Results
Predicting SO2-depletion biochemically
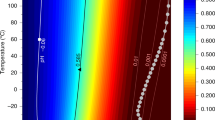
Each of the three metabolisms are capable of reproducing the observed SO2-depletion biochemically in the middle atmosphere of Venus when coupled to our photochemical-kinetics network. For each metabolism, Fig. 1 shows the depletion of the metabolic reagent species, above 47 km altitude, as a percentage of the metabolism-free reference abundance. The figure describes where each metabolic pathway is rate-limited (within the white space) or reagent limited (within the darkest coloured contour for a given reagent species), with transitional regions between each limiting regime. The resultant SO2-depletion is shown with red contours in every panel. We take a surface boundary condition for the initial chemical composition that is consistent with past lower-atmosphere27,28 and full-atmosphere5,6 models as our fiducial initial atmospheric composition. This surface condition corresponds to species that have either been observed or are thermochemically predicted to be present at the base of the Venus atmosphere6. For the fiducial atmospheric composition, each metabolism is either rate-limited or reagent limited: metabolism A is limited by the H2S abundance; metabolism B by the H2 abundance; and metabolism C by the CO abundance. Therefore, none of the three metabolisms initially provide a significant destruction pathway for SO2.
Contour plot showing depletion of metabolic reagents within the cloud layer for each metabolic pathway, (a), (b), and (c). Depletion is shown as a function of net metabolic rate (y-axis) and initial surface mixing ratio of the limiting metabolic reagent species (x-axis). The depletion of each species is calculated with respect to the species' column abundance in a reference model run without the metabolism present. The predicted or observed surface mixing ratios of metabolic reagent species and their 1σ errors are indicated with grey errorbars6, and the metabolic models reproducing SO2-depletion are indicated with a white dotted line.
To fit the SO2 observations, each metabolism must be in the SO2-limiting regime, and thus the abundances of the other limiting metabolic inputs must be increased from the fiducial (and observationally constrained) surface boundary conditions. Figure 1 demonstrates how, for each metabolic pathway, the net metabolic rate and atmospheric composition can be tuned to successfully reproduce the observed SO2 profile (white dotted line inside the SO2-limiting region).
Comparing the resultant atmospheric chemistry with observational constraints
In order to reproduce the observed SO2-depletion biochemically, the predicted abundance of metabolic input species for each of the three metabolisms violates a separate observational constraint in the lower atmosphere of Venus. In Fig. 2 we plot the mixing ratios of SO2, H2S and CO as functions of altitude, for the model atmosphere that best fits the SO2 observations for each metabolic pathway. Alongside, we show the fiducial atmosphere, predicted from thermochemistry, photochemistry and chemical kinetics, which does not correctly predict the SO2 observations, and the abiotic model atmosphere of Rimmer et al. which successfully predicts the SO2 observations by hypothesising aqueous droplet chemistry6. All three proposed metabolisms fit the SO2-depletion with a similar accuracy to the droplet-chemistry hypothesis6, with metabolism A providing the closest fit of any hypothesis to the middle-atmosphere observations (Fig. 2). However, this comes at the cost of the other metabolic reagent species not then fitting their below-cloud observational constraints well.
Atmospheric profiles of (a) SO2, (b) H2S and (c) CO mixing ratios as a function of altitude for the Rimmer et al. models (with and without droplet chemistry)6, compared with the three proposed metabolic pathways20,21 under conditions that reproduce the SO2 observations. Observational data and their 1σ errors are shown in black except for the observation of H2S below 20 km altitude which is shown in grey, as this is possibly a spurious detection35.
Metabolisms B and C each violate well-constrained lower-atmosphere observations of CO, requiring 300 ppm and 400 ppm of CO at the surface respectively, approximately ~15× and ~20× greater than observations indicate. Below-cloud CO has been measured in situ by gas chromatography instruments on-board the Venera 12 and Pioneer Venus descent probes29,30, by remote detection via IR spectra in a 2.3 μm atmospheric transparency window by the Venus Express orbiter31,32, and via remote Earth-based detection33,34. All of the data generally agree on deep atmosphere CO mixing ratios between 17 and 45 ppm and therefore a CO mixing ratio as high as that required by metabolisms B and C would be unexpected.
Metabolism A requires 300 ppm of H2S at the surface in order for life to be responsible for the observed SO2-depletion. H2S has been measured at an abundance of 3 ppm below the cloud layer via a mass spectrometer instrument on-board the Pioneer Venus descent probe35, however this reported value is one to two orders of magnitude larger than the currently expected surface mixing ratio of ~10–100 ppb from thermochemical modelling6,27. The H2S abundance otherwise only has an inferred upper limit at the cloud top derived from non-detection by Earth-based platforms36. From Fig. 2 we find that, even when the H2S mixing ratio is on the order of 100′s ppm in the cloud layer, H2S is so efficiently photolysed by the irradiating Solar flux that the profile always remains consistent with the inferred upper limit at the cloud top (with the H atoms liberated forming H2, and the sulfur atoms liberated mainly forming sulfur allotropes, Sx for x = 1, . . . , 8). Given the relative lack of observational constraints on the deep atmosphere H2S abundance, it remains a remote possibility that H2S exists with the required surface mixing ratio for SO2-depletion via metabolism A. However, assuming a volcanic source of SO2 and H2S in the Venus atmosphere37, this would require that the molar ratio of volcanically degassed H2S/SO2 is ~2. This is in disagreement with the oxidising nature of the Venus atmosphere, the observation of oxidised sulfur-containing minerals at the Venera 13, Venera 14, and Vega 2 landing sites37,38,39, and the inference that the volcanic H2S abundance should be lower than that of SO2 due to the very low water content in the atmosphere37.
Discussion
Our results demonstrate that the three sulfur-based metabolic pathways proposed for Venusian aerial life are capable of reproducing the observed SO2-depletion in the cloud layer of Venus, but in each case they require a source of chemical reducing power roughly equal in abundance to below-cloud SO2: the atmosphere otherwise does not possess enough reducing power for life to exploit to generate the observed SO2-depletion.
It is possible that our understanding of the below-cloud and in-cloud atmosphere is not correct. There is a consensus picture of Venus’s atmosphere, however this picture still has several mysteries that models are not able to solve, and certain observational data are discrepant with this picture. Discrepant measurements include in situ and ground-based detections of H2, H2O, H2S, and O2, at higher abundances than expected24,30,35,40, although these data are not consistent with each other without introducing spatial and temporal variability or significant systematic error. If the discrepant measurements were not made in error, then they must somehow be reconciled with the known atmospheric chemistry and thus reveal potential uncertainties in the consensus picture. However, even if these outliers represented the global atmosphere of Venus, they would still be inconsistent with these metabolic pathways explaining the sulfur depletion in Venus’s atmosphere. If, as seems likely, the standard description of the lower-atmosphere composition of Venus is broadly correct6, then we must conclude that either the SO2-depletion is due to a different metabolic pathway, or SO2-depletion on Venus is not related to life.
If any diagnostic feature of a biosphere utilising the proposed sulfur energy-metabolisms is being imprinted in the atmospheric chemistry of Venus, then the enigmatic cloud-layer SO2-depletion is likely to be it. This is because the observed SO2-depletion does not yet have a proven abiotic solution, even though the rest of the atmospheric sulfur chemistry can be explained by abiotic models to within at least an order of magnitude accuracy6. It may be that the products of these metabolisms could in principle provide good biosignatures, but only in the case that their presence or abundances are not explained by abiotic sources and sinks. This criteria for metabolic products to be diagnostic biosignatures is not met by the three suggested metabolisms that we investigate here: the presence of CO2, H2O, H2S, OCS, and sulfur allotropes in the atmosphere of Venus is already consistent with an abiotic source from volcanic degassing37; the enhanced abundance of OCS and H2S produced via metabolisms B and C remain undetectable above the cloud layer due to efficient photochemical destruction by the Solar flux; and any H2O produced via metabolism A would very likely be retained by the hypothetical microorganism as part of its survival strategy rather than being released back to the atmosphere, given the environmental stress posed by the relative desiccation of Venus’s atmosphere compared to the Earth’s atmosphere41. Since there are limited ways to utilise SO2 as a metabolic input, and since all other reactants would seem to exist in even more limited abundances than those investigated in the present study (CO, OCS, H2S, H2), this strongly suggests there is no unknown sulfur-metabolism responsible for the observed SO2-depletion.
Alternatively, a Venusian biosphere may exist and not be influencing the atmospheric chemistry in a diagnostic and observable way, in which case we can now place upper limits on the potential biomass density in the cloud layer before observational constraints are explicitly violated for the three proposed metabolisms. In order to calculate this limiting biomass density, an alternative removal mechanism for cloud-layer SO2 must be incorporated into the model to self-consistently predict the SO2-depletion. One alternative biologically-driven mechanism for removing SO2 has been suggested by Bains et al.42 which hypothesises that life in the clouds, fixing nitrogen from N2 and using atmospheric H2O, can produce NH3 that reacts with SO2 to form ammonium sulfites and sulfates, however this has not yet been demonstrated with fully self-consistent chemical modelling. An abiotic route to removing SO2 in the cloud layer has been suggested by Rimmer et al. by incorporating aqueous droplet chemistry. The droplet chemistry hypothesis posits that an additional source of hydrogen could be delivered to the clouds as hydroxide salts in mineral dust. This would enable aqueous reactions within the cloud droplets to provide a destruction pathway for aqueous SO2 dissolved in the droplets from the gas-phase. A dust flux to the clouds of ~16 Gt/yr containing 5 wt.% salt would be sufficient to explain the observed SO2-depletion. For more details see Rimmer et al.6. We here repeat our metabolic analysis, now coupling each metabolism in turn to the abiotic model that incorporates droplet chemistry in order to reproduce the observed SO2-depletion self-consistently6, and investigate how far we can increase the metabolic activity and abundance of metabolic input species within observational limits.
The first observational constraints to be exceeded as the biomass density is increased are the constraints on the abundances of metabolic inputs below the clouds. Figure 3 shows the three metabolic models with their metabolic input abundances increased as far they can be while remaining just within an observed upper limit, or within observational error. For metabolism B this corresponds to a ~10 ppm upper limit on H2 below the cloud layer30,43, and for metabolism C the upper end of the estimated error on the largest measured CO mixing ratio below the clouds (~60 ppm). For metabolism A, assuming that the in situ observation of 3 ppm at the surface is correct, the maximum below-cloud H2S within observational uncertainty corresponds to ~5 ppm H2S35.
Atmospheric profiles of (a) H2, (b) H2S and (c) CO mixing ratios as a function of altitude for the fiducial model and droplet-chemistry (abiotic) model6, compared with the three proposed metabolic models at the limit of violating an observational constraint in the atmosphere. Observational data and their 1σ errors are shown in black except for the observation of H2S below 20 km altitude35, and the observation of H2 at 50 km altitude40, which are shown in grey, as these are possibly spurious detections.
We estimate the corresponding biomass density in the cloud layer for the three limiting metabolic models using a simple energetic argument. The reactions of metabolisms B and C are chemotrophic and therefore the ΔGr liberated by each metabolic reaction is already known. If we make an assumption about the wavelength of light absorbed by the photosystem of metabolism A then an effective ΔGr liberated by the net metabolic reaction can be assumed, give or take some efficiency factor and some numerical factor for the ratio of photons required per metabolic input molecule consumed in the synthesis of long-chain organic molecules (these numerical factors would act to reduce the Gibbs free energy per mole for metabolism A, thus our estimate here is a relevant upper limit). The average volumetric rate of the net metabolic reaction thus provides us with an upper limit on the net energy released via the metabolic activity. We can therefore make an estimate of the maximum biomass density as a function of cell power, under the assumption that cells are ~0.5 μm in radius9 and the density of cellular material is of the order ~1 g cm−3. The results are shown in Fig. 4, and further details of this calculation can be found in the ‘Methods’ section.
Biomass density in the cloud layer for metabolisms A, B, and C, as a function of cell power. The red dashed line indicates the estimated minimum limit on cell power for terrestrial microorganisms44 and the red hatch region is the excluded region of the parameter space. The blue shaded region indicates the range of usual cell maintenance power requirements, measured in a laboratory setting, for terrestrial microorganisms22.
The possible biomass range in our model is critically dependent on the power requirements of a single cell. We can first obtain an absolute upper bound on the possible biomass density in the Venusian cloud layer for each proposed energy-metabolism, by adopting that \({P}_{{{{{{{{\rm{cell}}}}}}}}}\,\approx\, {P}_{{{{{{{{\rm{cell,min}}}}}}}}}\). The red dashed line in Fig. 4 shows the estimated minimum cell-power, \({P}_{{{{{{{{\rm{cell,min}}}}}}}}} \,\sim\, 1{0}^{-21}\,{{{{{{{\rm{W}}}}}}}}\) for terrestrial microbial life44. The biomass density estimates corresponding to this \({P}_{{{{{{{{\rm{cell,min}}}}}}}}}\) are 2 × 105 mg m−3 for metabolism A, 3 × 106 mg m−3 for metabolism B, and 5 × 106 mg m−3 for metabolism C. These estimates are inconsistent with the mass loading estimate of ~0.1–100 mg m−3 for the Venusian lower-cloud droplets themselves9, which is problematic as these droplets would host the microbial life in the hypothesised life cycle11. This treatment must therefore be a large overestimate of the maximum possible biomass because of the minimum power assumption we have made: Life in Venus’s clouds would inevitably have far greater power requirements than the minimum estimated power requirements of terrestrial life.
More realistic values for the power requirements of terrestrial microorganisms are those of cell maintenance power—the power per cell required to prevent population decay—estimated in a laboratory setting to be between 0.019 and 4700 × 10−15 J s−1 cell−1 (indicated with a blue shaded region in Fig. 4)22. Using these values, we calculate a range of biomass estimates between ~3 × 10−5 and 8 mg m−3 for metabolism A, ~6 × 10−4–160 mg m−3 for metabolism B, or ~1 × 10−3–270 mg m−3 for metabolism C. The majority of values in these ranges of maximum biomass density would be consistent with the mass loading in the Venusian cloud layer, and the values at the upper end of the ranges are comparable to the past estimate of 14 mg m−39, and to the estimated biomass density of the Earth’s own aerial biosphere, 44 mg m−39.
We can instead express this result in terms of the maximum cell concentration within the aerosol substrate by considering the number density of cells in the cloud layer and the aerosol mass loading of the Venusian lower-cloud droplets (see the Methods section for details). For our maximum biomass density ranging between order 10−5–102 mg m−3, and aerosol mass loading estimates ranging from 0.1 to 100 mg m−39, we obtain an estimated range of values for the maximum possible cell concentration between 105 and 1015 cells mL−1. Comparing to the measured values of cell concentrations of 103–105 cells mL−1 from hyperarid environments on Earth such as the Atacama desert45, we find that the lower end of the maximum cell concentration range for a Venusian aerial biosphere intersects the uppermost value measured in hyperarid terran environments, and otherwise all other values of maximum cell concentration in the range exceed cell concentrations observed in hyperarid terran environments. A Venusian biosphere more abundant than a terrestrial biosphere, even in a hyperarid environment, would be unexpected given extreme stresses in the Venusian environment: to loss of biomass out of the cloud layer; and, to desiccation in the sulfuric acid of the cloud droplets. We note, however, that hyperarid terran environments consist of dry soils whereas the hyperarid environment of the Venusian cloud layer consists of liquid sulfuric acid and thus there is no true analog environment on Earth that can be used as a close comparator to that of Venus.
The vast chemical challenges that the acidic environment of Venus would pose to an aerial biosphere would likely result in maintenance power requirements that are larger than any encountered on Earth, which would favour biomass density and cell concentration values below even the lower limit of our estimated biomass ranges (i.e. ≲3 × 10−5, 6 × 10−4, and 1 × 10−3 mg m−3 for metabolism A, B, and C, respectively). We note also that these estimates have not factored in limitations on biomass from metabolic processes that are not related to energy-capture but are still necessary for life: fixation of nutrients from the atmosphere for growth, for example the energetically costly process of nitrogen fixation from atmospheric N2, and the incorporation and availability of metals for use in enzymes to catalyze reactions. This may pose a problem for the life hypothesis proposed by Bains et al. which relies on life fixing nitrogen from atmospheric N2 in a non-energy-capture metabolism to explain the SO2-depletion biochemically42. Given these additional challenges life faces, we emphasise again that the analysis we present here is the most optimistic scenario possible for the hypothetical life that we have considered, in which the only limit imposed on a hypothetical biosphere is the availability of metabolic inputs required to capture chemical energy. These arguments suggest a very high cell-power requirement should be favoured for hypothetical Venusian aerial life, with the consequence that any such sulfur-metabolising biosphere would then have to be extremely low in biomass density, at least four orders of magnitude lower than the terrestrial aerial biosphere, to remain consistent with atmospheric observations.
We can compare the above estimated biomass densities to the abundance of matter required to produce the unexplained spectral properties observed at UV wavelengths in the cloud layer, which are attributed to an ‘unknown UV-absorber’. It has been suggested that the unknown UV-absorber may be explained by the presence of sulfur-metabolising microorganisms harboured in the clouds, with significant overlap found between the spectral appearance of various proteins and photosynthetic pigments used by some sulfur-metabolising microorganisms on Earth9. Alternatively, one abiotic contender to explain the UV-absorber is suggested to be a 1 % solution of FeCl3 dissolved within 75 % H2SO4 cloud droplets46,47. Performing a comparison of our biomass density estimates to this abiotic suggestion, 1 % of the mass-loading of the lower clouds is equal to 10−3–1 mg m−39. If the maximum biomass density we have calculated above is indeed close to or less than the lower end of our estimated range of values (i.e. large cell-power requirements with respect to known terrestrial values), then, assuming the biotic matter can produce similar strength spectral properties as the suggested FeCl3 molecules, only metabolism C could possibly intersect the lower limit of the absorber’s required mass-loading range (10−3 mg m−3) and metabolisms A and B fall short by at least one order of magnitude. However, if the biological pigments can produce much stronger absorption than simple salts then the potential biomass density we have constrained in the Venusian cloud layer may possibly still be a viable explanation to the unknown UV-absorber. Future studies would be needed to compare the standard absorbance of such proposed biological pigments to the observed spectral properties of the cloud aerosol.
One final consideration that could be made is that of the ecological stoichiometry of a combined ecosystem, featuring a combination of metabolisms in a steady state where the fluxes of metabolic products and inputs consumed are equally balanced. For any significant biosphere an ecosystem is needed and nutrient recycling is critical. Given that the terrestrial biosphere would collapse within a short time period in the absence of nutrient recycling between different species in a diverse ecosystem, it is also unlikely that one species of organism could exist by itself in the cloud layer of Venus, with the speculative exception perhaps of an autotrophic species using photosynthesis and only maintaining a very low biomass, such as that described by metabolism A. We have demonstrated that each of the metabolic hypotheses investigated here can be individually ruled out on the basis of violating lower-atmosphere constraints on the required metabolic input abundances, and we will now demonstrate how our results also show that a multi-metabolism ecosystem recycling the required chemical species exclusively within the cloud layer cannot explain the sulfur depletion in Venus’s clouds.
If we were to take, for example, the net effect of metabolism A and B together then the flux of H2S consumed by life with metabolism A, could be produced at the same rate by life utilising metabolism B. This results in a net reaction:
Even if arguments could be posed for the efficient production of CO from the abundant CO2 in the atmosphere via photolysis or perhaps another metabolism in a hypothetical ecosystem, the reaction inevitably still requires input of H2 to the atmosphere, inconsistent with observations. It has been suggested that H2 could be replenished in the atmosphere via hydration reactions of ferrous iron oxide minerals at the surface48. Even if there were sufficient H2O in the atmosphere to enable this, the resulting below-cloud H2 mixing ratio would violate the observational upper limit of <10 ppm30. Production of H2 via hydration of ferrous iron oxide mineral dust within the cloud layer exclusively would not be possible because this mineral dust would be dissolved in the sulfuric acid cloud droplets and thus any hydrogen in the reaction would exist as aqueous H+ ions. Mineral dust therefore cannot provide a source of H2 localised within the cloud layer as a potential metabolic input and H2 could only be replenished by surface-atmosphere processes. The observational upper limit on H2 abundance below the clouds therefore poses a strict upper limit on the productivity of any biosphere recycling nutrients in ecosystem interactions. In the absence of ecosystem interactions, the requirement for metabolic inputs to be replenished by surface-atmosphere processes extends to all chemical species and therefore below-cloud observations of any limiting input species poses a strict upper limit on the productivity of the biosphere.
Ultimately there simply is not enough reducing power in the lower atmosphere of Venus for the suggested forms of life to exploit in order to deplete SO2 and thus be of sufficient biomass to influence the global atmospheric chemistry. We have demonstrated this result for life using each of the energy-capture metabolisms proposed in the literature so far, however our results extend generally to: (a) chemotrophs exploiting energy gradients from gaseous atmospheric species; or (b) phototrophs utilising gaseous atmospheric species as electron donors. The exception to this conclusion is phototrophic life that captures light energy not for CO2 reduction but to support general cellular demands, in which case the potential biomass density in the clouds cannot be constrained via the availability of metabolic inputs in the atmosphere, and future studies are needed to constrain this potential biomass via the available light flux. There may also still plausibly be a low mass biosphere present, the only observable effect of which is to release relatively small amounts of trace metabolic gases. The recent detections of phosphine gas in the atmosphere of Venus may be consistent with this, in the absence of any abiotic phosphine production pathway10,12,49,50,51.
Upcoming missions to Venus may be able to determine whether or not the cloud layer hosts a high mass biosphere using a currently unidentified metabolism, a low mass biosphere producing only trace metabolic products, or no biosphere at all. However, the prospect remains that other, possibly wetter, Venus-like exoplanets could host a habitable niche in the temperate region of their atmospheres. Aerial biospheres in general therefore have significant implications on the number and observability of potentially habitable planets beyond the Solar System. The hypothetical ‘habitable zone’ around a star has resulted in the search for Earth-sized exoplanets within the range of orbital distances for which an Earth-like atmosphere could permit condensation of liquid water at the surface52. Interior to the habitable zone is the region where surface temperatures are too high for water to condense at the surface, and this region is more likely to be populated by Venus-like exoplanets53. For Venus-like exoplanets with permanent cloud cover in the temperate region of the atmosphere, the possibility of an aerial biosphere extends the inner edge of the habitable zone much closer to the host star, where orbital periods are shorter and transiting planetary signals have enhanced signal to noise ratios, compared to planets within the surface-water habitable zone. The presence of a biosphere at high altitude also means that biologically induced features in the atmospheric chemistry will be imprinted in the observationally accessible region of the atmosphere, compared to surface life obscured by thick cloud cover. It has been demonstrated that the metabolic inputs H2S (metabolism A) and SO2 (metabolisms A, B, and C), and metabolic products OCS (metabolism C) and H2S (metabolism B), will not be depleted photochemically in the observable region of a Venus-like planet’s atmosphere around cooler host stars, in the same way they are in Venus’s atmosphere54. This means that with high precision retrievals of the upper atmosphere, the aerial biosphere hypothesis may be possible to test on Venus-like exoplanets. The framework that we present in this paper, self-consistently coupling metabolic reaction networks with a photochemical-kinetics network for the atmosphere of Venus, is equally applicable to simulating the effect of an aerial biosphere on the atmospheric chemistry of potentially habitable Venus-like exoplanets. If the new and exotic biome of a strictly aerial biosphere is possible in principle, then given the sheer number and diversity of exoplanets, we might expect that the first detected sign of life beyond the Solar System will originate from an aerial biosphere harboured on a world interior to the surface-water habitable zone.
Methods
The model
We model the atmosphere of Venus using a photochemical-diffusion model6. The model is composed of a 1D Lagrangian solver, ARGO, and a network of reactions, STAND2020, that treats H/C/N/O/S/Cl chemistry accurately within a temperature range of 100–30, 000 K26,55. The model has been shown to reproduce the known atmosphere of Venus within approximately an order of magnitude of observations given the inclusion of a scheme of aqueous chemistry that aids the gas-phase depletion of SO2 and H2O6. The chemical network, STAND2020, is a list of reactants, products, and rate constants, for every chemical reaction considered in the atmosphere. For thermochemical reactions the reaction rates are temperature dependent, and for photochemical reactions the reaction rates are dependent on the photon flux as a function of wavelength incident at a given layer of the atmosphere. These chemical reactions determine the chemical production and loss rates of each species. At each altitude step in the atmosphere the reactions are solved by ARGO as a set of time-dependent, coupled, non-linear differential equations:
where, at a given height z (cm) and time t (s), nX (cm−3) is the number density of species X, PX (cm−3 s−1) is the rate of production of species X, LX (s−1) is the rate constant for loss of species X, and ∂ΦX/∂z (cm−3 s−1) describes the divergence of the vertical diffusion flux, encapsulating both eddy- and molecular-diffusion.
ARGO follows a parcel of gas as it rises from the surface to the top of the atmosphere and back down again. An initial condition for the chemical composition is input at the base of the atmosphere, listed in Table 1. At every altitude step on the journey upwards ARGO solves Eq. (7) for all species in the atmosphere, based on the pressure, temperature, and species’ abundances at that altitude in the atmosphere. The time interval over which ARGO solves for is prescribed by the eddy-diffusion profile which parametrises vertical transport through the atmosphere. The pressure-temperature profile and eddy-diffusion profile for modern Venus are each taken from past photochemical-kinetics models of the lower27 and middle56 atmosphere. At the top of the atmosphere, ARGO takes the incident stellar spectrum and, at each step on the journey downwards, includes photochemical reactions driven by the stellar flux irradiating that layer of the atmosphere. ARGO iterates this procedure until a convergence criteria is met and thus a global solution is found.
We test the effect of each of the net metabolic reactions, (3), (4) and (5) in turn, by coupling them to our atmospheric network and solving for the atmospheric composition of Venus. For each proposed metabolism the individual net metabolic reaction is added to the STAND2020 reaction network with an effective rate constant that is unconstrained. This rate is not synonymous with the biochemical rate of the metabolic reaction itself, rather it is an ‘effective’ net volumetric rate of all microbial activity in our one dimensional and diurnally averaged model. In this sense, the effective rate of the reaction relates to the biomass density in the model. The effective reaction is constrained to occur within the temperate region of the cloud layer by prescribing a condition that depends on atmospheric temperature. For a given effective rate, k, the reaction constant in the network depends on temperature (and thus altitude / pressure) as:
With each metabolism coupled to the STAND2020 network in turn, we iterate ARGO over a grid of models to explore the parameter space of net metabolic rate versus reagent abundance. The SO2 abundance remains fixed at it’s fiducial value. For metabolism A we gradually increase the H2S abundance from 10 ppb up to 500 ppm. For metabolism B we gradually increase the H2 abundance from 3 ppb up to 10 ppm, and then increase both the H2 and CO abundances in tandem in the ratio 2:1 respectively, from 10 ppm up to 300 ppm for H2 and from 20 ppm up to 600 ppm for CO. For metabolism C we gradually increase the CO abundance from 20 ppm up to 650 ppm.
Biomass density calculation
For the model atmospheres that incorporate the metabolic activity of a putative Venusian biosphere at the maximum possible limit before transgressing observational constraints, we can estimate the maximum biomass density averaged over the volume of the cloud layer, under the assumption that the availability of metabolic inputs is the limiting factor on the productivity of the biosphere (in practise other factors, such as availability of metals for catalytic enzymes, will likely limit the productivity of the biosphere). For volumetric reaction rate R (s−1 m−3) of the metabolic reaction in the model, we convert to the rate of energy liberated metabolically per unit volume in the cloud layer, ϵ (W m−3):
where ΔGr (J mol−1) is the Gibbs free energy release from the metabolic reaction and NA is Avogadro’s constant. The average cell number density in the cloud layer, ncell (cells m−3), is then given by:
and the average biomass density:
where Pcell (W cell−1) represents the power requirement of a single microbe of mass Mcell (mg). This allows for a comparison to be made to the measured mass loading of aerosol in the atmosphere, and to the estimated mass loading of microbes in the Earth’s (transiently) aerial biosphere. We can further make comparison to measured cell concentrations in analogue terrestrial environments that are most similar to the hyperarid environment of the Venusian cloud layer. To do so, we estimate the volume of aerosol per unit volume of the atmosphere, vaero (mL m−3), within the cloud layer, from the reported aerosol mass loading maero (mg m−3) and an assumed density of aerosol material, ρaero (g mL−1):
The cell concentration within the aerosol substrate, ncell, aero (cells mL−1), is then given by:
Combining Eqs. (9)–(13) and evaluating when the volumetric metabolic rate and metabolic input abundances are at the limit of transgressing observational data, gives us our final set of equations used to evaluate the range of maximum possible biomass densities and maximum possible cell concentrations referenced in the main text:
The analysis here refers primarily to the chemotrophic metabolisms where ΔGr ~ 240 kJ mol−1 is known a priori. Photosynthetic life would obtain energy from sunlight rather than a redox disequilibrium in the atmosphere, however the phototrophic metabolism can be treated with the same analysis if an assumption is made about the wavelength of light absorbed in reaction (1). For example, the energy delivered in sunlight at a wavelength of 500 nm is approximately ~240 kJ mol−1 (i.e. per NA of photons absorbed where NA is Avogadro’s constant) and so the above analysis can be repeated exactly for the phototroph (metabolism A) give or take some efficiency factor, and some numerical factor for the different possible wavelengths of light being utilised (the exception to this energetic analysis would be phototrophic life that captures light energy for cell maintenance without any redox chemistry occuring, in which case the potential biomass cannot be constrained by the rate of consumption of metabolic input species). Recent radiative transfer modelling has shown that, below ~ 59 km altitude, while biologically-damaging UV radiation is screened by the cloud layer, photosynthetically active light at longer wavelengths (>400 nm) continues to penetrate57. We therefore assume ΔGr ~ 240 kJ mol−1 for each of the three net metabolic reactions. While our choice of 500 nm for the wavelength utilised in the photosynthetic metabolism is for simplicity in calculating the biomass, this value sits well within the range of common terrestrial photosynthetic wavelengths (~400–1000 nm) including the wavelengths utilised by anoxygenic phototrophs in deep water columns58. We further assume a typical value for cell density of a microbe to be of the order ρcell ~ 1 g cm−3 and typical size of rcell ~ 0.5 μm in order to evaluate Mcell. We can then investigate how the choice of cell-power requirement, Pcell, affects our estimates for the biomass density, in Fig. 4.
The required introduction of the cell-power requirement term provides the largest uncertainty to this energetic analysis, varying over at least 10 orders of magnitude for terrestrial microbial life alone. It is for this reason why we must evaluate a range of possible estimates for \({m}_{{{{{{{{\rm{bio}}}}}}}},\max }\) and \({n}_{{{{{{{{\rm{cell,aero,max}}}}}}}}}\). Assuming that the stresses posed by the acidic and hyperarid Venusian environment pose greater survival challenges to life compared to conditions encountered on Earth, we can significantly constrain our estimated range of values of mbio and ncell, aero at the limiting metabolic reaction rate, by imposing that \({P}_{{{{{{{{\rm{cell}}}}}}}}}\,\gtrsim\, {P}_{\max ,\bigoplus }\):
Taking \({P}_{\max ,\bigoplus }\,=\,4700\,\times\, 1{0}^{-15}\,{{{{{{{\rm{J}}}}}}\,{{{{{{\rm{s}}}}}}}^{-1}}}\,{{{{{{{{\rm{cell}}}}}}}}}^{-1}\) we evaluate limits \({m}_{{{{{{{{\rm{bio}}}}}}}},\max }\,\lesssim\, 3\,\times\, 1{0}^{-5}\,{{{{{{{\rm{mg}}}}}}}}\,{{{{{{{{\rm{m}}}}}}}}}^{-3}\) for metabolism A, \({m}_{{{{{{{{\rm{bio}}}}}}}},\max }\,\lesssim\, 6\,\times\, 1{0}^{-4}\,{{{{{{{\rm{mg}}}}}}}}\,{{{{{{{{\rm{m}}}}}}}}}^{-3}\) for metabolism B, and \({m}_{{{{{{{{\rm{bio}}}}}}}},\max }\,\lesssim\, 1\,\times\, 1{0}^{-3}\,{{{{{{{\rm{mg}}}}}}}}\,{{{{{{{{\rm{m}}}}}}}}}^{-3}\) for metabolism C, as quoted in the abstract.
Data availability
The data generated in this study have been deposited in the Harvard online database under accession code (https://doi.org/10.7910/DVN/AKUTME).
References
Vandaele, A. C. et al. Sulfur dioxide in the Venus atmosphere: I. Vertical distribution and variability. Icarus 295, 16–33 (2017).
Demore, W. B. & Yung, Y. L. Catalytic processes in the atmospheres of Earth and Venus. Science 217, 1209–1213 (1982).
Parkinson, C. D. et al. Photochemical control of the distribution of Venusian water. Planet. Space Sci. 113, 226–236 (2015).
Marcq, E., Mills, F. P., Parkinson, C. D. & Vandaele, A. C. Composition and chemistry of the neutral atmosphere of Venus. Space Sci. Rev. 214, 10 (2018).
Bierson, C. J. & Zhang, X. Chemical cycling in the Venusian atmosphere: a full photochemical model from the surface to 110 km. J. Geophys. Res. Planets 125, e06159 (2020).
Rimmer, P. B. et al. Hydroxide salts in the clouds of Venus: their effect on the sulfur cycle and cloud droplet pH. Planet. Sci. J. 2, 133 (2021).
Morrowitz, H. & Sagan, C. Life in the clouds of Venus? Nature 215, 1259–1260 (1967).
Grinspoon, D. H. Venus revealed: a new look below the clouds of our mysterious twin planet. (Addison-Wesley, 1997).
Limaye, S. S. et al. Venus’ spectral signatures and the potential for life in the clouds. Astrobiology 18, 1181–1198 (2018).
Greaves, J. S. et al. Phosphine gas in the cloud decks of Venus. Nat. Astron. 5, 655–664 (2021).
Seager, S. et al. The Venusian lower atmosphere haze as a depot for desiccated microbial life: a proposed life cycle for persistence of the Venusian aerial biosphere. arXiv e-prints, https://doi.org/10.48550/arXiv.2009.06474 (2020).
Bains, W. et al. Phosphine on Venus cannot be explained by conventional processes. Atrobiology 21, 1277–1304 (2021).
Mogul, R., Limaye, S. S., Way, M. J. & Cordova, J. A. Venus’ mass spectra show signs of disequilibria in the middle clouds. Geophys. Res. Lett. 48, e91327 (2021).
Seager, S. et al. Venus life finder mission study. arXiv e-prints, https://doi.org/10.48550/arXiv.2112.05153 (2021).
Cockell, C. S. Life on Venus. Planet. Space Sci. 47, 1487–1501 (1999).
de Bergh, C. et al. Deuterium on Venus: observations from Earth. Science 251, 547–549 (1991).
Way, M. J. & Del Genio, A. D. Venusian habitable climate scenarios: modeling Venus through time and applications to slowly rotating Venus-like exoplanets. J. Geophys. Res. (Planets) 125, e06276 (2020).
Höning, D., Baumeister, P., Grenfell, J. L., Tosi, N. & Way, M. J. Early habitability and crustal decarbonation of a stagnant-lid Venus. J. Geophys. Res. (Planets) 126, e06895 (2021).
Turbet, M. et al. Day-night cloud asymmetry prevents early oceans on Venus but not on Earth. Nature 598, 276–280 (2021).
Schulze-Makuch, D., Grinspoon, D. H., Abbas, O., Irwin, L. N. & Bullock, M. A. A sulfur-based survival strategy for putative phototrophic life in the Venusian atmosphere. Astrobiology 4, 11–18 (2004).
Schulze-Makuch, D. & Irwin, L. N. The prospect of alien life in exotic forms on other worlds. Naturwissenschaften 93, 155–172 (2006).
LaRowe, D. E. & Amend, J. P. Catabolic rates, population sizes and doubling/replacement times of microorganisms in natural settings. Am. J. Sci. 315, 167–203 (2015).
Soo, R. M. et al. On the origins of oxygenic photosynthesis and aerobic respiration in Cyanobacteria. Science 355, 1436–1440 (2017).
Oyama, V. I. et al. Laboratory corroboration of the pioneer Venus gas chromatograph analyses. Science 205, 52–54 (1979).
Mills, F. P. A spectroscopic search for molecular oxygen in the Venus middle atmosphere. J. Geophys. Res. 104, 30757–30764 (1999).
Rimmer, P. B. & Helling, C. A Chemical Kinetics Network for Lightning and Life in Planetary Atmospheres. Astrophysical J. Suppl. Ser. 224, 9 (2016).
Krasnopolsky, V. A. Chemical kinetic model for the lower atmosphere of Venus. Icarus 191, 25–37 (2007).
Krasnopolsky, V. A. S3 and S4 abundances and improved chemical kinetic model for the lower atmosphere of Venus. Icaurs 225, 570–580 (2013).
Gelman, B. G. et al. An analysis of the chemical composition of the atmosphere of Venus on an AMS of the Venera-12 using a gas chromatograph NASASTI/Recon Technical Report N. (1979).
Oyama, V. I. et al. Pioneer Venus gas chromatography of the lower atmosphere of Venus. J. Geophys. Res. 85, 7891–7902 (1980).
Marcq, E. et al. A latitudinal survey of CO, OCS, H2O, and SO2in the lower atmosphere of Venus: Spectroscopic studies using VIRTIS-H. J. Geophys. Res. (Planets) 113, E00B07 (2008).
Tsang, C. C. C. et al. Variability of CO concentrations in the Venus troposphere from Venus Express/VIRTIS using a Band Ratio Technique. Icaurs 201, 432–443 (2009).
Bezard, B., de Bergh, C., Crisp, D. & Maillard, J. P. The deep atmosphere of Venus revealed by high-resolution nightside spectra. Nature 345, 508–511 (1990).
Pollack, J. B. et al. Near-infrared light from Venus’ nightside: a spectroscopic analysis. Icarus 103, 1–42 (1993).
Hoffman, J. H., Hodges, R. R., Donahue, T. M. & McElroy, M. M. Composition of the Venus lower atmosphere from the Pioneer Venus mass spectrometer. J. Geophys. Res. 85, 7882–7890 (1980).
Krasnopolsky, V. A. High-resolution spectroscopy of Venus: detection of OCS, upper limit to H2S, and latitudinal variations of CO and HF in the upper cloud layer. Icarus 197, 377–385 (2008).
Fegley, B. In Treatise on Geochemistry (Second Edition) (eds Holland, H. D. & Turekian, K. K.) 2nd edn, 127–148 (Elsevier, Oxford, 2014). ISBN:978-0-08-098300-4. https://www.sciencedirect.com/science/article/pii/B9780080959757001224.
Surkov, Y. A., Barsukov, V. L., Moskalyeva, L. P., Kharyukova, V. P. & Kemurdzhian, A. L. New data on the composition, structure, and properties of Venus rock obtained by Venera 13 and Venera 14. Lunar Planet. Sci. Conf. Proc. 89, B393–B402 (1984).
Surkov, Y. A. et al. Venus rock composition at the Vega 2 landing site. J. Geophys. Res. 91, E215–E218 (1986).
Mukhin, L. M. et al. VENERA-13 and VENERA-14 gas chromatography analysis of the Venus atmosphere composition. Sov. Astron. Lett. 8, 216–218 (1982).
Hallsworth, J. E. et al. Water activity in Venus’s uninhabitable clouds and other planetary atmospheres. Nat. Astron. 5, 665–675 (2021).
Bains, W. et al. Production of ammonia makes Venusian clouds habitable and explains observed cloud-level chemical anomalies. Proc. Natl Acad. Sci. 118, 52 (2021).
Donahue, T. M., Grinspoon, D. H., Hartle, R. E. & Hodges R. R., J. Ion/neutral Escape of Hydrogen and Deuterium: Evolution of Water in Venus II: Geology, Geophysics, Atmosphere, and Solar Wind Environment, 385 (eds Bougher, S. W., Hunten, D. M. & Phillips, R. J.) (1997).
LaRowe, D. E. & Amend, J. P. Power limits for microbial life. Front. Microbiol. 6, 718 (2015).
Connon, S. A. et al. Bacterial diversity in hyperarid Atacama Desert soils. J. Geophys. Res. (Biogeosciences). 112, G04S17 (2007).
Zasova, L. V., Krasnopolskii, V. A. & Moroz, V. I. Vertical distribution of SO2 in upper cloud layer of Venus and Origin of U.V.-absorption. Adv. Space Res. 1, 13–16 (1981).
Krasnopolsky, V. A. On the iron chloride aerosol in the clouds of Venus. Icarus 286, 134–137 (2017).
Watson, A. J., Donahue, T. M. & Kuhn, W. R. Temperatures in a runaway greenhouse on the evolving Venus: implications for water loss. Earth Planet. Sci. Lett. 68, 1 (1984).
Greaves, J. S. et al. Re-analysis of phosphine in Venus’ clouds. arXiv e-prints, https://doi.org/10.48550/arXiv.2011.08176 (2020).
Greaves, J. S. et al. On the robustness of phosphine signatures in Venus’ clouds. arXiv e-prints, https://doi.org/10.48550/arXiv.2012.05844 (2020).
Greaves, J. S. et al. Recovery of Spectra of Phosphine in Venus’ Clouds. arXiv e-prints, https://doi.org/10.48550/arXiv.2104.09285 (2021).
Kasting, J. F., Whitmire, D. P. & Reynolds, R. T. Habitable Zones around main sequence stars. Icarus 101, 108–128 (1993).
Kane, S. R., Kopparapu, R. K. & Domagal-Goldman, S. D. On the frequency of potential venus analogs from Kepler data. Astrophysical J. Lett. 794, L5 (2014).
Jordan, S. et al. Photochemistry of Venus-like planets orbiting K- and M-dwarf stars. Astrophys. J. 922, 44 (2021).
Hobbs, R. et al. Sulfur chemistry in the atmospheres of warm and hot Jupiters. Monthly Not. R. Astronomical Soc. 506, 3186–3204 (2021).
Krasnopolsky, V. A. A photochemical model for the Venus atmosphere at 47-112 km. Icarus 218, 230–246 (2012).
Patel, M. R., Mason, J. P., Nordheim, T. A. & Dartnell, L. R. Constraints on a potential aerial biosphere on Venus: II. Ultraviolet radiation. Icarus 373, 114796 (2022).
Haas, S. et al. Low-light anoxygenic photosynthesis and Fe-S-biogeochemistry in a microbial mat. Front. Microbiol. 9, 858 (2018).
Rimmer, P. B. & Rugheimer, S. Hydrogen cyanide in nitrogen-rich atmospheres of rocky exoplanets. Icarus 329, 124–131 (2019).
Acknowledgements
S.J. thanks the Science and Technology Facilities Council (STFC) for the PhD studentship (grant reference ST/V50659X/1). P.B.R. thanks the Simons Foundation for funding (SCOL awards 599634).
Author information
Authors and Affiliations
Contributions
S.J., O.S., and P.B.R. conceived of the study and contributed extensively to the paper. S.J. implemented the simulations and analysed the output data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Janusz Oschlisniok and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jordan, S., Shorttle, O. & Rimmer, P.B. Proposed energy-metabolisms cannot explain the atmospheric chemistry of Venus. Nat Commun 13, 3274 (2022). https://doi.org/10.1038/s41467-022-30804-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-30804-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.