Abstract
Oxide-ion conductors are important in various applications such as solid-oxide fuel cells. Although zirconia-based materials are widely utilized, there remains a strong motivation to discover electrolyte materials with higher conductivity that lowers the working temperature of fuel cells, reducing cost. Oxide-ion conductors with hexagonal perovskite related structures are rare. Herein, we report oxide-ion conductors based on a hexagonal perovskite-related oxide Ba7Nb4MoO20. Ba7Nb3.9Mo1.1O20.05 shows a wide stability range and predominantly oxide-ion conduction in an oxygen partial pressure range from 2 × 10−26 to 1 atm at 600 °C. Surprisingly, bulk conductivity of Ba7Nb3.9Mo1.1O20.05, 5.8 × 10−4 S cm−1, is remarkably high at 310 °C, and higher than Bi2O3- and zirconia-based materials. The high conductivity of Ba7Nb3.9Mo1.1O20.05 is attributable to the interstitial-O5 oxygen site, providing two-dimensional oxide-ion O1−O5 interstitialcy diffusion through lattice-O1 and interstitial-O5 sites in the oxygen-deficient layer, and low activation energy for oxide-ion conductivity. Present findings demonstrate the ability of hexagonal perovskite related oxides as superior oxide-ion conductors.
Similar content being viewed by others
Introduction
Oxide-ion conducting ceramic materials have attracted much attention due to their applications in solid-oxide fuel cells (SOFCs), oxygen separation membranes, gas sensors, and catalysts1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24. Yttria stabilized zirconia (YSZ) ceramics have widely been used, but the working temperature is high (700–1000 °C). Thus, there remains a strong motivation to explore oxide-ion conductors with higher conductivities at temperatures below 600 °C. High oxide-ion conductivities have been observed in specific structure families such as the fluorite-type, perovskite-type, melilite-type, and apatite-type structures1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24.
The perovskite and its related materials exhibit interesting physical and chemical properties25 and can be classified into four structural groups of (i) AMX3 perovskite-type, (ii) AMX3-related, (iii) hexagonal perovskite-related and (iv) modular structures26 where A and M are larger and smaller cations, respectively, and X is an anion. A number of perovskite-type materials and perovskite related phases belonging to the groups of (i), (ii) and (iv) have been reported to be oxide-ion conductors. The hexagonal perovskite-related structures (iii) have hexagonal close packing of AX3 layers or sequences of hexagonal (h) and cubic (c) AX3 (and/or anion-deficient AX3–x (c′) where x is the anion vacancy content) close-packed layers. The hexagonal perovskite related oxides exhibit a variety of crystal structures26,27,28,29,30,31. However, oxide-ion conductors with hexagonal perovskite-related structures are quite rare. Several mixed ion (oxide-ion and/or proton) and electronic conductors with hexagonal perovskite related structures were reported in the literature32,33,34,35. The hexagonal perovskite related oxides Ba3MNbO8.5–δ (M = Mo and W; δ is the oxygen deficiency) and their solid solutions exhibit significant oxide-ion conductivities23,30,31,36,37,38,39, however, the conductivities are not high at temperatures below 600 °C. The relatively low conductivities are ascribed to relatively high activation energy for conductivity (e.g., 1.2 eV for Ba3MoNbO8.5–δ23). Therefore, we have explored oxide-ion conductors with the hexagonal perovskite related structures. Ba7Nb4MoO20 is a trigonal \(P\bar 3m1\) hexagonal perovskite polytype 7H29,40. Fop et al. found high oxide-ion and proton conductivities of Ba7Nb4MoO2040. Herein, we report higher oxide-ion conductivities, crystal structure and oxide-ion diffusion pathways of the solid solution composition Ba7Nb3.9Mo1.1O20.05. Ba7Nb3.9Mo1.1O20.05 is found to exhibit a bulk conductivity of 5.8 × 10−4 S cm−1 at 310 °C, which is higher than those of the “best” oxide-ion conductors. The present work also demonstrates the two-dimensional (2D) oxide-ion O1–O5 diffusion through the interstitial octahedral O5 and lattice tetrahedral O1 sites at a high temperature of 800 °C.
Results and discussion
Preparation and characterization of Ba7Nb4MoO20-based oxides
In this work, we studied the electrical and structural properties of Ba7Nb4MoO20-based materials, because (1) the chemical species in Ba7Nb4MoO20 are the same as those in the oxide-ion conductor Ba3MoNbO8.5–δ, (2) Ba7Nb4MoO20 has the hexagonal perovskite related structure similar to Ba3MoNbO8.5–δ28,29,38, (3) the crystal structure of Ba7Nb4MoO20 contains possible oxide-ion conducting Ba–oxygen (c′) layers as does the structure of Ba3MoNbO8.5-δ, and (4) the bond-valence-based energy barrier for oxide-ion migration of Ba7Nb4MoO20 (0.21 eV) is lower than that of Ba3MoNbO8.5–δ (0.51–0.35 eV, See the details in Supplementary Note 1). Ba7Nb3.95Mo1.05O20.025 and Ba7Nb3.9Mo1.1O20.05 were successfully prepared by solid-state reactions. X-ray powder diffraction (XRD) measurements indicated that Ba7Nb3.95Mo1.05O20.025 and Ba7Nb3.9Mo1.1O20.05 have the hexagonal perovskite related structure with trigonal \(P\bar 3m1\) space group (Supplementary Fig. 1). Arrhenius plots of bulk conductivities (σb) of Ba7Nb4MoO2040, Ba7Nb3.95Mo1.05O20.025, and Ba7Nb3.9Mo1.1O20.05 in dry air are shown in Supplementary Fig. 2. The σb of Ba7Nb3.9Mo1.1O20.05 is the highest among the three compositions. Thus, we focus on the Ba7Nb3.9Mo1.1O20.05 composition for further detailed studies.
The cation atomic ratio of Ba: Nb: Mo = 7.11(14): 3.81(3): 1.126(14) for Ba7Nb3.9Mo1.1O20.05 determined through X-ray fluorescence (XRF) analyses agreed with that of the nominal composition where the number in parentheses is the standard deviation in the last digit. X-ray photoelectron spectroscopy (XPS) data for the Ba7Nb3.9Mo1.1O20.05 composition indicated that the valences of Ba, Nb and Mo at room temperature (RT) were +2, +5 and +6, respectively (Ba2+7Nb5+3.9Mo6+1.1O2–20.05; Supplementary Fig. 3). Thermogravimetric measurements of Ba7Nb3.9Mo1.1O20.05 in dry air between 400 and 900 °C indicate very little weight loss and oxygen deficiency δ in Ba7Nb3.9Mo1.1O20.05−δ at high temperatures (Supplementary Fig. 4).
Oxide-ion conduction in Ba7Nb3.9Mo1.1O20.05
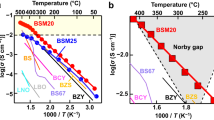
Figure 1a, b shows the typical impedance spectra of Ba7Nb3.9Mo1.1O20.05, which indicates the bulk, grain boundary and electrode responses. Bulk conductivity (σb), grain-boundary conductivity (σgb), and grain-boundary capacitance were obtained by the equivalent circuit fitting (Red lines in Fig. 1a, b, Supplementary Figures 5–9). The σb and σgb were measured in dry O2, dry air and in dry N2 at 295 and 598 °C. They were independent of oxygen partial pressure at these temperatures, which indicates ionic conduction (Supplementary Figure 10). Figure 1c shows the temperature dependencies of the σb and σgb of Ba7Nb3.9Mo1.1O20.05 on cooling in dry air. The activation energy for σb was found to be lower than those for σgb and the DC total electrical conductivity, σtot. The σb was higher than σgb at temperatures below 550 °C and higher than σtot at temperatures below 850 °C. The oxide-ion transport number (tion) was investigated using oxygen concentration cell measurements. The tion values were 1.00 between 700 and 900 °C and 0.99 at 600 °C in air/O2, 0.99 at 800 °C and 1.00 at 900 °C in air/N2, and 0.98 at 600 °C in air/5% H2 in N2 (Fig. 1d). Oxide-ion diffusion in dense Ba7Nb3.9Mo1.1O20.05 was clearly confirmed by the isotope exchange depth profile method41, which calculated a high oxygen tracer diffusion coefficient D* value of 5.35 × 10–9 cm2 s–1 at 700 °C and 7.25 × 10–9 cm2 s–1 at 800 °C (Supplementary Figure 11). The diffusion lengths were about 150 μm and the grain sizes of the Ba7Nb3.9Mo1.1O20.05 samples were 1–5 μm (Supplementary Figure 12), thus, the 18O tracer species encountered a number of grains and grain boundaries. The total DC electrical conductivity (σtot) was independent of the oxygen partial pressure P(O2) between P(O2) = 7 × 10−25 ~ 1 atm at 300 °C, P(O2) = 2 × 10−26 ~ 1 atm at 600 °C, and P(O2) = 5 × 10−18 ~ 1 atm at 900 °C (Fig. 1e). Electronic conductivity was observed in the P(O2) range of 6 × 10−24 ~ 4 × 10−26 atm at 900 °C with the slope of −0.129(19). Therefore, Ba7Nb3.9Mo1.1O20.05 exhibits a wider electrolyte domain compared with Ba7Nb4MoO20 as reported by Fop et al.40. To examine the possible proton conduction of this phase, the DC electrical conductivities, σtot, of Ba7Nb3.9Mo1.1O20.05 were measured in wet air (H2O partial pressure, P(H2O) = 2.3 × 10−2 atm) and in dry air (P(H2O) < 1.8 × 10−4 atm). The contribution of protons to σtot of Ba7Nb3.9Mo1.1O20.05 was small, even in wet air where the proton transport number was 0.03 − 0.12 (Supplementary Fig. 13). Water incorporation behavior was also investigated and the results are shown in Supplementary Fig. 14 and Supplementary Note 2. A significant but small amount of water uptake was observed for Ba7Nb3.9Mo1.1O20.05 compared with Ba7Nb4MoO20, which is responsible for the lower transport number of protons in Ba7Nb3.9Mo1.1O20.05. These results indicate that the oxide ion is the dominant charge carrier and that Ba7Nb3.9Mo1.1O20.05 is an oxide-ion conductor. No change was observed in the X-ray powder diffraction patterns before and after the oxygen concentration cell measurements (Supplementary Fig. 15), which demonstrates the high phase stability of Ba7Nb3.9Mo1.1O20.05 at high temperatures and in the wide P(O2) region between P(O2) = 1.2 × 10−27 and 1 atm. The σb of Ba7Nb3.9Mo1.1O20.05 was found to be higher than those of Ba7Nb4MoO2040 (Fig. 1c) and YSZ, and comparable to those of the best oxide-ion conductors (Fig. 1f). It should be noted that the σb of Ba7Nb3.9Mo1.1O20.05 was higher than the best oxide-ion conductors at temperatures of around 300 °C, due to the low activation energy of Ba7Nb3.9Mo1.1O20.05 (0.185-0.454 eV as shown in Fig. 1c). These results indicate the high potential of the hexagonal perovskite related oxide Ba7Nb3.9Mo1.1O20.05 as a superior oxide-ion conductor.
a, b Complex impedance plots of Ba7Nb3.9Mo1.1O20.05 recorded in dry air at (a) 309 °C and (b) 598 °C. c Arrhenius plots of bulk conductivity σb, grain-boundary conductivity σgb and DC σtot of Ba7Nb3.9Mo1.1O20.05 in dry air. Activation energy for σb of Ba7Nb3.9Mo1.1O20.05 decreases with temperature from 0.454 to 0.185 eV as shown by the red numbers in panel c. Green line represents σb of Ba7Nb4MoO20 reported by Fop et al.40. d Oxygen transport number of Ba7Nb3.9Mo1.1O20.05. e Oxygen partial pressure P(O2) dependence of σtot of Ba7Nb3.9Mo1.1O20.05. f Comparison of bulk conductivities of Ba7Nb3.9Mo1.1O20.05 and other oxide-ion conductors.
Structural origin of the high oxide-ion conductivity in Ba7Nb3.9Mo1.1O20.05
Next, we discuss the structural origin of the high oxide-ion conductivity of Ba7Nb3.9Mo1.1O20.05, based on its refined crystal structure and neutron scattering length density (NSLD) at 800 °C (Fig. 2). In the Rietveld refinements of the neutron-diffraction data, the Mo6+ and Nb5+ cations were assumed to be disordered, since they have quite similar neutron scattering lengths. By the trigonal \(P\bar 3m1\) hexagonal perovskite polytype 7H (c′hhcchh; Fig. 2a), the crystal structure of Ba7Nb3.9Mo1.1O20.05 was successfully refined by Rietveld analyses of the neutron-diffraction data measured in situ at a temperature of 800 °C in vacuum on the super-high-resolution diffractometer, SuperHRPD42,43 at J-PARC, Japan (Fig. 3 and Supplementary Table 1). In order to examine the oxide-ion diffusion pathway and to validate the crystal structure of Ba7Nb3.9Mo1.1O20.05, the NSLD was analysed using the maximum-entropy method (MEM) and structure factors obtained through the Rietveld analysis. It is well known that the MEM is a powerful tool to study the structural disorder and ion-diffusion pathways in various ionic conductors16,19,31. Oxygen atoms were found to partially occupy the octahedral interstitial O5 site in the Ba1(O1)2−x(O5)0.05+x layer (Fig. 2a), because (i) the Rietveld fit for the structural model with the O5 atom (weighted profile reliability factor Rwp = 2.39%) was lower than those without the O5 atom (Rwp = 2.47%) and (ii) the MEM NSLD distribution clearly shows the O5 site (Fig. 2b, d). Here the x in Ba1(O1)2−x(O5)0.05+x is the vacancy content at the O1 site in the unit cell. We applied the split-atom model for the tetrahedral O1 site, because the atomic displacement parameter was quite high for the non-split-atom model and the Rietveld fit for the split-atom model (Rwp = 2.39%) was better than that for the non-split atom model (Rwp = 2.44%).
a Refined crystal structure and b corresponding yellow isosurface of maximum-entropy method neutron scattering length densities (MEM NSLDs) at 0.36 fm Å−3 of Ba7Nb3.9Mo1.1O20.05 at 800 °C. Refined crystal structure (c) and corresponding MEM NSLD distribution (d) on the ab plane at z = 0 of Ba7Nb3.9Mo1.1O20.05 at 800 °C. In d, the contour lines from 0 to 2 fm Å−3 by the step of 0.2 fm Å−3. Thermal ellipsoids in panels (a) and (c) are drawn at the 90% probability level. Arrows in c denote the directions of oxide-ion O1-to-O5 migration.
Rietveld patterns of neutron-diffraction data taken with the (a) backscattering bank (d = 1.1−3.4 Å) and (b) 90o bank (d = 1.6−5.1 Å) of the SuperHRPD diffractometer in vacuum at 800 °C. The observed and calculated intensities and difference plots are shown by red marks, blue and black solid lines, respectively. Black tick marks stand for calculated Bragg peak positions.
The crystal structure of Ba7Nb3.9Mo1.1O20.05 consists of an oxide-ion conducting Ba1(O1)2−x(O5)0.05+x layer (c′), two Ba2(O2)3 layers (h), two Ba4(O4)3 layers (h), two Ba3(O3)3 layers (c), and Nb and Mo cations at the Nb/Mo1, Nb/Mo2 and Nb/Mo3 sites (Fig. 2a). A striking feature of the MEM NSLD distribution of Ba7Nb3.9Mo1.1O20.05 at 800 °C is the connected oxide-ion diffusional pathway between the tetrahedral O1 and interstitial octahedral O5 sites on the oxide-ion conducting Ba1(O1)2−x(O5)0.05+x layer (c′) (Fig. 2b, d). The oxide ions two-dimensionally migrate through both lattice O1 and interstitial O5 sites, which indicates the interstitialcy mechanism of oxide-ion diffusion. The bond-valence-based energy barriers for oxide-ion migration, Eb, for the refined crystal structure of Ba7Nb3.9Mo1.1O20.05 at 800 °C also supported this 2D feature, because the Eb along the ab plane (0.19 eV) is much lower than Eb along the c axis (1.54 eV). Ba7Nb3.9Mo1.1O20.05 has an excess oxygen of x = 0.05 (O20+x or O0.05 in Ba7Nb3.9Mo1.1O20.05) compared with the mother material Ba7Nb4MoO20, which leads to a larger amount of interstitial oxygen and the higher oxide-ion conductivity of Ba7Nb3.9Mo1.1O20.05 (Fig. 1c).
In conclusion, we have discovered a structure family of rare-earth-free oxide-ion conductors based on the hexagonal perovskite related oxide Ba7Nb4MoO20. Ba7Nb3.9Mo1.1O20.05 shows a wide stability range and predominantly oxide-ion conduction in the oxygen partial pressure range from 2 × 10–26 to 1 atm at 600 °C. The bulk conductivity of Ba7Nb3.9Mo1.1O20.05 is as high as 5.8 × 10−4 S cm−1 at 310 °C. This high conductivity is ascribed to the interstitial O5 oxygen, 2D oxide-ion O1 − O5 diffusion through the lattice tetrahedral O1 and interstitial O5 octahedral oxygen sites on the ab plane at z = 0 and to the low activation energy for oxide-ion conductivity. The (tetrahedral O1)–(octahedral O5) diffusion pathways in Ba7Nb3.9Mo1.1O20.05 are along the [\(1\bar 10\)], [120] and [\(\bar 2\bar 10\)] directions (Arrows in Fig. 2c), which are the same as those for the (tetrahedral O3)–(octahedral O2) migration paths in the hexagonal perovskite related oxide Ba3MoNbO8.5–δ31. This strongly suggests that the (tetrahedral)–(octahedral) oxide-ion migration pathway along the [\(1\bar 10\)], [120] and [\(\bar 2\bar 10\)] directions on the oxygen deficient c′ layer is a common feature of the oxide-ion conductors with hexagonal perovskite related structures. This feature would be a guide for design of oxide-ion conductors with the hexagonal perovskite-related structures. The present finding of high oxide-ion conductivities in rare-earth-free Ba7Nb3.9Mo1.1O20.05 suggests the ability of various hexagonal perovskite related oxides as superior oxide-ion conductors.
Methods
Synthesis and characterization
Ba7Nb3.95Mo1.05O20.025 and Ba7Nb3.9Mo1.1O20.05 were prepared by the solid-state reactions. High-purity (> 99.9%) BaCO3, Nb2O5, and MoO3 were mixed and ground using an agate mortar and pestle as ethanol slurries and dry powders repeatedly for 0.5–2 h. The obtained mixtures were calcined at 900 °C for 10–12 h in static air. The calcined samples were crushed and ground using an agate mortar and pestle as ethanol slurries and dry powders repeatedly for 0.5–2 h. The powders thus obtained were uniaxially pressed into pellets at 62–150 MPa and subsequently sintered in static air at 1100 °C for 24 h. Parts of the sintered pellets were crushed and ground into white powders to measure X-ray powder diffraction, atomic absorption spectroscopy (AAS, Hitachi Z-2300), inductively coupled plasma optical emission spectroscopy (ICP-OES, Hitachi PS3520UVDD), and thermogravimetric (TG) data. To identify the existing phases, X-ray powder diffraction patterns of Ba7Nb3.95Mo1.05O20.025 and Ba7Nb3.9Mo1.1O20.05 were measured at RT with an X-ray powder diffractometer (BRUKER D8 Advance, Cu Kα radiation, 2θ range: 5−90°). The chemical composition of Ba7Nb3.9Mo1.1O20.05 was examined by energy dispersive XRF analyses (Rigaku, NEX DE). XPS spectra of Ba7Nb3.9Mo1.1O20.05 were measured using an X-ray photoelectron spectrometer (ULVAC PHI 5000 Versa Probe III). TG analysis was carried out in dry air using a Bruker-AXS 2020SA instrument at the heating and cooling rates of 10 °C min−1. The heating and cooling cycle was repeated three times to negate the influence of absorbed species, such as water and to confirm the reproducibility of the measurement.
Measurements of electrical conductivity, oxygen diffusion coefficient and transport properties
The electrical conductivities of Ba7Nb3.9Mo1.1O20.05 were measured as a function of temperature by AC impedance spectroscopy in flowing dry air, N2, and O2 gases (100 mL min−1) using a sintered pellet (20 mm in diameter, 2.7 mm in thickness, relative density of 100−98%) with Pt electrodes. Impedance spectra were recorded with a Solartron 1260 impedance analyser in the frequency range of 10 MHz−1 Hz at an applied alternating voltage of 100 mV. The activation energies, Ea, for the conductivities were estimated using the Arrhenius equation:
where A0, k, and T are the pre-exponential factor, Boltzmann constant, and absolute temperature, respectively. Oxygen concentration cell measurements were conducted to investigate the oxygen transport number tion using a sintered pellet (20 mm in diameter, 4.5 mm in height, and relative density of 100−98%) attached to an alumina tube with a glass seal. One side of the pellet was exposed to flowing dry air and the other side to flowing dry O2 (Air/O2), N2 (Air/N2), or 5% H2 in N2 (Air/5% H2 in N2) gases at high temperatures. The electromotive forces of the concentration cell were recorded with a Keithley model 617 electrometer. The following Nernst equation was utilized to estimate the tion:
where F is the Faraday constant, R is the gas constant, T is the absolute temperature, p(O2) is the oxygen partial pressure of the gas of O2, N2, 5% H2 in N2, and p0(O2) (= 0.21 atm) is the oxygen partial pressure of dry air. After the oxygen concentration cell measurements, the surface of the pellet was ground with sandpaper carefully to remove the Pt paste and then crushed and ground into powder. X-ray diffraction patterns of the resulting powders were measured to investigate the phase stability at high temperatures and different atmospheres.
The total electrical conductivity σtot of the Ba7Nb3.9Mo1.1O20.05 pellet (relative density: 95%) was measured by a DC-4-probe method with Pt electrodes at various oxygen partial pressure p(O2). The p(O2) was controlled using a mixture of O2, N2, and 5% H2 in N2 and p(O2) was monitored by an oxygen sensor.
18O tracer diffusion measurements of dense Ba7Nb3.9Mo1.1O20.05 pellets (relative density of 98-100%) were carried out using the line scan method by secondary ion mass spectrometry (SIMS)41. Each sample prepared was polished with diamond spray media down to a finish of 0.25 µm. Samples were pre-annealed in dry research grade oxygen (BOC 99.996%) of natural isotopic abundance for a duration of 10 times that of the isotopic exchange. The samples were subsequently annealed for 2 h in 18O-enriched gas at a pressure of ≃200 mbar. After the exchange anneal, the samples were cut perpendicular to the original surface and the exposed cross-sections polished to 0.25 μm finish, as above. The oxygen diffusion profiles were measured by Time-of-Flight Secondary Ion Mass Spectrometry (ToF-SIMS) using a ToF-SIMS.5 instrument (IONTOF GmbH) using Bi+ ions at 25 keV energy. Values of oxygen self-diffusion, D*, and surface exchange, k, coefficients were obtained by fitting the experimental data to Crank’s solution of Fick’s 2nd law of diffusion41,44 using the TraceX software45. The microstructure of the Ba7Nb3.9Mo1.1O20.05 pellet used for the 18O tracer diffusion measurements was observed by a scanning electron microscope (KEYENCE VE-8800).
Neutron-diffraction measurements of Ba7Nb3.9Mo1.1O20.05 at 800 °C, Rietveld and MEM analyses
High-temperature neutron-diffraction measurements of Ba7Nb3.9Mo1.1O20.05 pellets (8.7 mm in diameter, 43 mm in height) in a Ti-Zr alloy holder were carried out in vacuum using a super-high-resolution time-of-flight (TOF) neutron diffractometer (SuperHRPD) installed at the Materials and Life Science Experimental Facility of J-PARC, Japan42,43. The absorption correction was performed using the method given by Rouse and Cooper46. The diffraction data were analysed by the Rietveld method using the Z-Rietveld program47. The neutron scattering length density distribution was investigated using the MEM. The MEM analysis was carried out with computer program, Dysnomia48, using the structure factors obtained in the Rietveld refinement of the neutron diffraction data at 800 °C. The MEM calculations were performed with the unit cell divided into 60 × 60 × 168 pixels.
Computation of the bond-valence-based energy barrier for oxide-ion migration
The bond-valence-based energy landscapes for a test oxide ion were calculated using the SoftBV49 software with a spatial resolution of 0.1 Å.
The refined crystal structure, MEM neutron scattering length density distributions, and bond-valence-based energy landscape were depicted using VESTA50.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abraham, F., Boivin, J. C., Mairesse, G. & Nowogrocki, G. The BIMEVOX series: a new family of high performances oxide ion conductors. Solid State Ion. 40–41, 934–937 (1990).
Goodenough, J. B., Ruiz-Diaz, J. E. & Zhen, Y. S. Oxide-ion conduction in Ba2In2O5 and Ba3In2MO8 (M = Ce, Hf, or Zr). Solid State Ion. 44, 21–31 (1990).
Ishihara, T., Matsuda, H. & Takita, Y. Doped LaGaO3 perovskite type oxide as a new oxide ionic conductor. J. Am. Chem. Soc. 116, 3801–3803 (1994).
Nakayama, S., Kageyama, T., Aono, H. & Sadaoka, Y. Ionic conductivity of lanthanoid silicates, Ln10(SiO4)6O3 (Ln= La, Nd, Sm, Gd, Dy, Y, Ho, Er and Yb). J. Mater. Chem. 5, 1801–1805 (1995).
Nakayama, S. & Sakamoto, M. Electrical properties of new type high oxide ionic conductor RE10Si6O27 (RE = La, Pr, Nd, Sm, Gd, Dy). J. Eur. Ceram. Soc. 18, 1413–1418 (1998).
Huang, K., Feng, M. & Goodenough, J. B. Synthesis and electrical properties of dense Ce0.9Gd0.1O1.95 ceramics. J. Am. Ceram. Soc. 81, 357–362 (1998).
Lacorre, P., Goutenoire, F., Bohnke, O., Retoux, R. & Laligant, Y. Designing fast oxide-ion conductors based on La2Mo2O9. Nature 404, 856–858 (2000).
Skinner, S. J. & Kilner, J. A. Oxygen diffusion and surface exchange in La2−xSrxNiO4+δ. Solid State Ion. 135, 709–712 (2000).
Kilner, J. A. Fast oxygen transport in acceptor doped oxides. Solid State Ion. 129, 13–23 (2000).
Esaka, T. Ionic conduction in substituted scheelite-type oxides. Solid State Ion. 136–137, 1–9 (2000).
Imanaka, N., Kamikawa, M. & Adachi, G.-Y. A carbon dioxide gas sensor by combination of multivalent cation and anion conductors with a water-insoluble oxycarbonate-based auxiliary electrode. Anal. Chem. 74, 4800–4804 (2002).
Huang, K., Tichy, R. S. & Goodenough, J. B. Superior perovskite oxide-ion conductor; strontium- and magnesium-doped LaGaO3: I, phase relationships and electrical properties. J. Am. Ceram. Soc. 81, 2565–2575 (2005).
León-Reina, L. et al. High oxide ion conductivity in Al-doped germanium oxyapatite. Chem. Mater. 17, 596–600 (2005).
Kwon, O. H. & Choi, G. M. Electrical conductivity of thick film YSZ. Solid State Ion. 177, 3057–3062 (2006).
Kuang, X. et al. Interstitial oxide ion conductivity in the layered tetrahedral network melilite structure. Nat. Mater. 7, 498–504 (2008).
Yashima, M. Diffusion pathway of mobile ions and crystal structure of ionic and mixed conductors: a brief review. J. Ceram. Soc. Jpn. 117, 1055–1059 (2009).
Malavasi, L., Fisher, C. A. J. & Islam, M. S. Oxide-ion and proton conducting electrolyte materials for clean energy applications: structural and mechanistic features. Chem. Soc. Rev. 39, 4370–4387 (2010).
Jung, D. W., Duncan, K. L. & Wachsman, E. D. Effect of total dopant concentration and dopant ratio on conductivity of (DyO1.5)x-(WO3)y-(BiO1.5)1-x-y. Acta Mater. 58, 355–363 (2010).
Yashima, M., Sirikanda, N. & Ishihara, T. Crystal structure, diffusion path and oxygen permeability of a Pr2NiO4-based mixed conductor (Pr0.9La0.1)2(Ni0.74Cu0.21Ga0.05)O4+δ. J. Am. Chem. Soc. 132, 2385–2392 (2010).
Li, M. et al. A family of oxide ion conductors based on the ferroelectric perovskite Na0.5Bi0.5TiO3. Nat. Mater. 13, 31–35 (2014).
López, C. A., Pedregosa, J. C., Lamas, D. G. & Alonso, J. A. The strongly defective double perovskite Sr11M o4O23: Crystal structure in relation to ionic conductivity. J. Appl. Crystallogr. 47, 1395–1401 (2014).
Fujii, K. et al. New perovskite-related structure family of oxide-ion conducting materials NdBaInO4. Chem. Mater. 26, 2488–2491 (2014).
Fop, S. et al. Oxide ion conductivity in the hexagonal perovskite derivative Ba3MoNbO8.5. J. Am. Chem. Soc. 138, 16764–16769 (2016).
Zhang, W. et al. Oxide-ion conduction in the Dion−Jacobson phase CsBi2Ti2NbO10-δ. Nat. Comm. 11, 1224 (2020).
Schaak, R. E. & Mallouk, T. E. Perovskites by design: a toolbox of solid-state reactions. Chem. Mater. 14, 1455–1471 (2002).
Tilley, R. J. D. Perovskites: Structure-property relationships. (Wiley, Chichester, UK, 2016).
Darriet, J. & Subramanian, M. A. Structural relationships between compounds based on the stacking of mixed layers related to hexagonal perovskite-type structures. J. Mater. Chem. 5, 543–552 (1995).
García-González, E., Parras, M. & González-Calbet, J. M. Electron microscopy study of a new cation deficient perovskite-like oxide: Ba3MoNbO8.5. Chem. Mater. 10, 1576–1581 (1998).
García-González, E., Parras, M. & González-Calbet, J. M. Crystal structure of an unusual polytype: 7H-Ba7Nb4MoO20. Chem. Mater. 11, 433–437 (1999).
Fop, S. et al. Investigation of the relationship between the structure and conductivity of the novel oxide ionic conductor Ba3MoNbO8.5. Chem. Mater. 29, 4146–4152 (2017).
Yashima, M. et al. Direct evidence for two-dimensional oxide-ion diffusion in the hexagonal perovskite-related oxide Ba3MoNbO8.5− δ. J. Mater. Chem. A 7, 13910–13916 (2019). 2019.
Jing, X.-P. & West, A. R. AC impedance and gas concentration cell measurements for Ba12Y4.67Ti8O35. Acta Phys. –Chim. Sin. 18, 617–623 (2002).
Kuang, X. et al. Oxygen vacancy ordering phenomena in the mixed-conducting hexagonal perovskite Ba7Y2Mn3Ti2O20. Chem. Mater. 19, 2884–2893 (2007).
Ling, C. D. et al. Structures, phase transitions, hydration, and ionic conductivity of Ba4Nb2O9. Chem. Mater. 21, 3853–3864 (2009).
Ling, C. D. et al. Structures, phase transitions, hydration, and ionic conductivity of Ba4Ta2O9. Chem. Mater. 22, 532–540 (2010).
Bernasconi, A., Tealdi, C., Mühlbauer, M. & Malavasi, L. Synthesis, crystal structure and ionic conductivity of the Ba3Mo1−xWxNbO8.5 solid solution. J. Solid State Chem. 258, 628–633 (2018).
Bernasconi, A., Tealdi, C. & Malavasi, L. High-temperature structural evolution in the Ba3Mo1−xWxNbO8.5 system and correlation with ionic transport properties. Inorg. Chem. 57, 6746–6752 (2018).
Fop, S., McCombie, K., Wildman, E., Skakle, J. & Mclaughlin, A. Hexagonal perovskite derivatives: a new direction in the design of oxide ion conducting materials. Chem. Comm. 55, 2127–2137 (2019).
Auckett, J. E., Milton, K. L. & Evans, I. R. Cation distributions and anion disorder in Ba3MNbO8.5 (M = Mo, W) materials: implications for oxide ion conductivity. Chem. Mater. 31, 1715–1719 (2019).
Fop, S. et al. High oxide ion and proton conductivity in a disordered hexagonal perovskite. Nat. Mater. 19, 752–757 (2020).
Kilner, J. A., Skinner, S. J. & Brongersma, H. H. The isotope exchange depth profiling (IEDP) technique using SIMS and LEIS. J. Solid State Electrochem. 15, 861–876 (2011).
Torii, S. et al. Super high resolution powder diffractometer at J-PARC. J. Phys. Soc. Jpn 80, SB020 (2011).
Torii, S. et al. Improvement of instrument devices for super high resolution powder diffractometer at J-PARC. J. Phys. Conf. Ser. 502, 012052 (2014).
Crank, J. The mathematics of diffusion. (Clarendon Press, Oxford, 1975).
Cooper, S. J. Quantifying the Transport Properties of Solid Oxide Fuel Cell Electrodes. (Imperial College London, 2015).
Rouse, K. D., Cooper, M. J., York, E. J. & Chakera, A. Absorption corrections for neutron diffraction. Acta Crystallogr. Sect. A 26, 682–691 (1970).
Oishi, R. et al. Rietveld analysis software for J-PARC. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers, Detect. Assoc. Equip. 600, 94–96 (2009).
Momma, K., Ikeda, T., Belik, A. A. & Izumi, F. Dysnomia, a computer program for maximum-entropy method (MEM) analysis and its performance in the MEM-based pattern fitting. Powder Diffr. 28, 184–193 (2013).
Chen, H., Wong, L. L. & Adams, S. SoftBV – a software tool for screening the materials genome of inorganic fast ion conductors. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 75, 18–33 (2019).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
We thank Dr. M. Hagihala, Dr. S. Lee, and Dr. P. Miao for the assistance in the neutron diffraction experiments. We express special thanks to Dr. T. Murakami, Dr. E. Niwa, Mr. M. Shiraiwa, Mr. K. Hibino, Dr. W. Zhang, Mr. H. Yaguchi, Mr. M. Matsui, Mr. R. Inoue, Mr. H. Tejima, and Mr. Y. Suzuki for useful discussion and assistance in the experiments/analyses. We would like to acknowledge to Kojundo Chemical Laboratory Co. Ltd. for arrangements of the XPS and ICP measurements. We thank the Ookayama Materials Analysis Division of Tokyo Institute of Technology for their assistance in the XRF measurements. The neutron-diffraction measurements were carried out by the project approval (J-PARC MLF Proposal No. 2017A0111 and 2017L1301). This study was partly supported by Grants-in-Aid for Scientific Research (KAKENHI, No. JP15H02291, JP16H00884, JP16H06293, JP16H06440, JP16H06441, JP16H06438, JP16K21724, JP17K17717, JP17H06222, and JP19H00821) from the Ministry of Education, Culture, Sports, Science and Technology of Japan, Adaptable and Seamless Technology transfer Program through Target-driven R&D (A-STEP) No. JPMJTM19AY from Japan Science and Technology Agency (JST) and JSPS Core-to-Core Program, A. Advanced Research Networks (Solid Oxide Interfaces for Faster Ion Transport).
Author information
Authors and Affiliations
Contributions
M.Y. and T.T. designed research. K.F., S.T., T.T. and T.K. measured the neutron data. Y.Y. and Y.Z. carried out the oxygen diffusion measurements based on the supervision by S.J.S. T.T., Y.S. and Y.Y. prepared the samples and measured the X-ray diffraction data, electrical conductivity, TG data, and transport number. T.T., Y.S., Y.Y. and K.F. analyzed the data and made figures and Tables. M.Y. wrote and edited the manuscript and response to the referees’ comments. All the authors read the manuscript. Funding acquisition and supervision: M.Y. and S.J.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks Peter Slater and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yashima, M., Tsujiguchi, T., Sakuda, Y. et al. High oxide-ion conductivity through the interstitial oxygen site in Ba7Nb4MoO20-based hexagonal perovskite related oxides. Nat Commun 12, 556 (2021). https://doi.org/10.1038/s41467-020-20859-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-20859-w
This article is cited by
-
High proton conductivity within the ‘Norby gap’ by stabilizing a perovskite with disordered intrinsic oxygen vacancies
Nature Communications (2023)
-
High proton conduction in Ba2LuAlO5 with highly oxygen-deficient layers
Communications Materials (2023)
-
Hidden chemical order in disordered Ba7Nb4MoO20 revealed by resonant X-ray diffraction and solid-state NMR
Nature Communications (2023)
-
Borates as a new direction in the design of oxide ion conductors
Science China Materials (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.