Abstract
The social intelligence hypothesis (SIH) posits that within-group interactions drive cognitive evolution, but it has received equivocal support. We argue the SIH overlooks a major component of social life: interactions with conspecific outsiders. Competition for vital resources means conspecific outsiders present myriad threats and opportunities in all animal taxa across the social spectrum (from individuals to groups). We detail cognitive challenges generated by conspecific outsiders, arguing these select for ‘Napoleonic’ intelligence; explain potential influences on the SIH; and highlight important considerations when empirically testing these ideas. Including interactions with conspecific outsiders may substantially improve our understanding of cognitive evolution.
Similar content being viewed by others
Introduction
Cognitive evolution is one of the most hotly debated topics in biology, with considerable uncertainty remaining about the likely drivers. Some of the earliest hypotheses for the evolution of intelligence in non-human animals focused on ecological factors, such as the cognitive demands associated with locating spatially and temporally unpredictable food sources1. Several comparative studies have supported the predictions of the so-called ecological intelligence hypothesis; for instance, environmental variation and diet have been identified as key selection pressures for brain-size evolution in birds and primates, respectively2,3,4. However, a major body of conceptual and empirical work indicates that the social environment can also play a crucial role in cognitive evolution5,6,7.
To date, predictions about the social drivers of cognitive evolution have largely focused on within-group interactions8. Byrne and Whiten5 hypothesised that competitive interactions among groupmates (e.g., for food and/or mates) select for cognitively demanding behaviours, such as tactical deception, social manipulation and political manoeuvring. Conversely, Dunbar6 argued that the cooperative aspects of group living, including the need to create functional, cohesive, bonded groups to solve ecological problems, select for greater cognition. Positive actions between group members, such as the trading of commodities (e.g., grooming for coalitionary support) and the contingent rewarding of cooperative acts, may also generate cognitive demands9,10,11. Collectively, these within-group aspects of social life are argued to drive cognitive evolution12,13 and form the basis of the social intelligence hypothesis (SIH) (see Dunbar and Shultz14 for a review of the SIH and its variants). The SIH was initially conceived with primates in mind and there is strong evidence to suggest that the social environment explains some of the cognitive variation observed in this taxonomic group (e.g., a positive relationship between group size and brain size)15. However, some primate studies have not found this relationship2,4 and it is also not so apparent in other taxa; pair-bondedness (indicative of strong social relationships) is associated with large brains in some avian16 and non-primate mammalian17 species, but there are also a number of studies reporting no relationship between sociality and cognition in these taxa3. The SIH, as currently framed, therefore receives somewhat equivocal support and leaves a significant amount of cognitive variation unexplained.
We argue that considerable cognitive variation may have remained unexplained because the SIH, and empirical tests of it, overlook a second major axis of social variation in animals: interactions with conspecific outsiders. From solitary species to those living in complex groups, in all animal taxa from invertebrates to primates, interactions with conspecific outsiders are commonplace18,19,20 and their profound effects on sociality have become increasingly recognised21,22,23,24. Rival individuals, pairs or groups may compete for valuable resources such as mating opportunities, breeding positions, food or territories25,26,27. Such competition and associated threats and opportunities likely create selection pressures for cognitive traits that aid success. The idea that conspecific outsiders could influence cognitive evolution has been proposed before with respect to human evolution28,29. However, discussion of the cognitive consequences of outsider threats in non-human animals has largely focused on the challenges posed by predators15; the possible influence of conspecific outsiders on cognitive evolution has been largely ignored and untested (for notable exceptions, see refs. 30,31). Conspecific outsiders differ in several important ways from predators. For example, although predators represent only a threat, conspecific outsiders can present threats and opportunities. In addition, some threats posed by outsiders, such as those to mating or breeding positions, are unique to conspecifics. Finally, the roles of ‘predator’ and ‘prey’ are split between different species, whereas the roles of both parties (i.e., threatener and threatened) are played by members of the same species in interactions with conspecific outsiders. Although interactions with heterospecifics—both antagonistic ones (e.g., with predators) and those of a cooperative nature (e.g., between cleanerfish and clients)—undoubtedly pose cognitive challenges and are worthy of study, we focus here on conspecific outsiders. We term the ability to exploit interactions with conspecific outsiders to an actor’s advantage as ‘Napoleonic’ intelligence, given the strategic intelligence synonymous with Napoleon Bonaparte.
Here we offer a case for the full inclusion of conspecific outsider interactions and resulting Napoleonic intelligence in the SIH. We begin by detailing the cognitive challenges presented by conspecific outsiders (hereafter referred to as ‘outsiders’), considering both those relevant across the whole social spectrum and additional challenges pertinent to group-living species. We then explain the potential influences of outsider interactions on the SIH: we describe the different selection pressures exerted by groupmates and outsiders, indicate how these social axes can interact additively or synergistically to influence cognition and assess intraspecific variation (e.g., between sexes) in selection pressures. Finally, we outline how the extension of the SIH to include outsider interactions can be tested empirically. Our argument is that both within-group and outsider interactions are crucial drivers of social intelligence (Fig. 1).
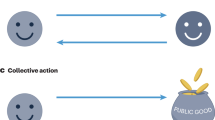
Within-group social intelligence is the capacity to succeed in within-group interactions (a); it represents the well-established basis of the SIH as currently framed5, 6. We argue that the capacity to succeed in interactions with conspecific outsiders should be included in the SIH for a more complete representation of the social environment. Significant cognitive challenges with respect to outsider threats and opportunities include those arising from b conflict with rivals over resources and territory space, c conflict with rivals over breeding or mating opportunities, d contest dynamics when there are adversarial interactions between rivals and e the evaluation of secondary cues (e.g., faecal deposits) containing information about conspecifics. NB: here we use the dwarf mongoose (Helogale parvula), a cooperatively breeding species, to illustrate some of the cognitive challenges posed by outsiders. However, such challenges and resulting Napoleonic intelligence are relevant across the social spectrum (including solitary and pair-bonded species, and those living in a variety of group structures). Dwarf mongoose illustrations: Martin Aveling. Landscape: David Clode.
Cognitive challenges arising from interactions with conspecific outsiders
Outside threats and opportunities present a variety of cognitively demanding challenges that likely generate consequences both plastically within the lifetime of individuals and on an evolutionary scale. Animal contests in general have been proposed to favour cognitive traits underpinning three broad skillsets—opponent evaluation, own status evaluation and assessment strategies31— all of which are applicable to interactions with outsiders. Individuals benefit from a capacity to assess rival characteristics and intent in relation to their own characteristics and to be able to appraise and respond to developments during encounters. More broadly, as individuals navigate the social context of competition with outsiders, there may be value in possessing abilities to change behaviour based on previous encounters, to remember rivals and spatial locations, to process varied, infrequent and unreliable information, and to make informed decisions. Cognitive traits relating to perception, learning and memory are therefore as applicable to interactions with outsiders as other conflict scenarios32. We consider in turn those cognitive challenges presented by outsiders that are relevant across the full social spectrum (from solitary and pair-bonded animals to those living in groups) and those cognitive challenges that are of particular relevance to group-living species.
Cognitive challenges across the social spectrum
To maximise opportunities and to reduce the threats and costs associated with outsiders, decisions are needed about whether and how to engage with rivals. Decision-making regarding interactions with outsiders is often influenced by their identity. For example, opportunities to obtain breeding positions or extra-pair/extra-group matings vary between neighbouring territories25,33. Similarly, from a threat perspective, it may be adaptive to avoid rivals of greater resource-holding potential (RHP); relative RHP, and therefore the odds of winning a contest, can depend on the size of individuals, the strength of a pair bond or the size and composition of groups27,34,35. Furthermore, the decision to engage in territorial behaviour may require an ability to infer relatedness to specific groups36 (and therefore indirect fitness consequences of outsider interactions). In general, opponent recognition can arise from such processes as habituation learning37, categorisation of different classes and even individual recognition38. Deciding whether to engage in contests with conspecific outsiders is made all the more important given the inherent risks: although the potential benefits (e.g., in terms of resource acquisition) are high, the costs can be substantial as there may be escalation to violence and possible injury or death25,39. Even when the costs are not this extreme, engagement takes time and energy, and can have consequences for the harmony of pairs or groups21,40. During outsider interactions, strategic behaviour may be modified in relation to different third-party audiences41 (both insiders and outsiders). Acquisition, retention and assessment of relevant information helps individuals make complex decisions about, for instance, whether to invest in resource defence, attack a territorial neighbour or attempt dispersal or sneaky matings, and how to react during contests.
Enhanced spatial memory is also likely to be important in the context of outsider conflict and opportunities (e.g., remembering the position of territory boundaries and the locations of previous interactions). For example, the use of cognitive maps can allow avoidance of areas where costly contests with rivals tend to occur42 or behavioural preparation for potential encounters when in zones of conflict43. Conversely, this information may also be used to identify opportunities for territory expansion. In many species, neighbours and non-neighbours (‘strangers’) present different threat levels; in some cases, strangers are more threatening (contexts in which neighbours are known as ‘dear enemies’), whereas sometimes neighbours present the greater threat (contexts in which neighbours are known as ‘nasty neighbours’)44,45. Knowledge about the location of specific neighbouring territories is important to ensure a suitable response to neighbours vs. strangers, but also because, in dear-enemy situations, stronger responses are favoured towards neighbours on the ‘wrong’ territorial boundary compared to where they are expected spatially46. Selection should favour a capacity to obtain, update and retain this spatial information.
To add to the cognitive challenge of monitoring and assessing outside threats and opportunities, relationships are dynamic and information about outsiders may only be available infrequently and often just from secondary cues. In territorial species, mortality, take-overs and dispersal mean that the identity of neighbours will change across time. The relationship with a given neighbour can also alter with repeated interactions44. For instance, a recent incomer might initially be viewed as a nasty neighbour but could become a dear enemy as knowledge and familiarity increase; responses can also change dynamically if outsiders intrude more frequently or become more aggressive47,48. More generally, and equally relevant to non-territorial species, threats and opportunities can fluctuate with context44; mating opportunities, e.g., will only be available when there are fertile females in the population26. On a spatial scale, territorial boundaries can change, as owners expand or contract the area over which they control access19. Compounding the cognitive challenge of tracking and reassessing such situations, information regarding outsiders might be received relatively infrequently; encounters with rivals or indicators of their presence may occur only every few days or weeks49. Relevant information can be obtained from deliberate exchanges between rivals34,50 or from secondary cues (e.g., faecal deposits51,52). The cognitive demands of signal detection, deception, interpretation and response, as well as integrating information of differing reliability from direct and indirect sources, is likely considerable53.
Additional cognitive challenges arising in group-living species
Group-living species encompass a range of social structures, from territorial, cooperatively breeding animals where a ‘group’ is easily defined to fission–fusion societies (where the ‘group’ is more ambiguous and could refer to the ‘parent group’ or ‘sub-groups’) and societies where ‘group’ identity varies with context. However, all can experience additional, and qualitatively different, cognitive challenges to those arising in species with simpler social structures. For instance, in the context of dynamic outsider characteristics, there can be changes in the identity of a whole neighbouring group, a subset of the group or just a single member. This alters both the collective threat and the potential threats and opportunities presented to individuals (e.g., from individual roving males seeking breeding opportunities54). Furthermore, information about multiple group members, available from individually distinct scent marks and calls55,56, requires more elaborate cognitive processing than that from single outsiders. Additional inferential challenges arise when others shift between ‘insider’ and ‘outsider’ status depending on social context (e.g., foraging groups vs. breeding groups).
Direct contests between groups may generate cognitively demanding dynamics. When groups fight, two main strategies are possible: members of a larger group could concentrate attacks on single-rival individuals or individuals from opposing groups could compete in one-on-one battles. Lanchester57 proposed that the former is governed by a ‘square law’, which predicts that numerical superiority leads to victory, whereas the latter is governed by a ‘linear law’ such that the fighting ability of individuals is the more important factor. Although initially conceived with humans in mind, Lanchester’s laws have been found to operate across a wide range of taxa, from primates58 to insects59. Thus, assessment of the size and ability of rival forces is likely crucial when deciding how to engage with them in contests, both initially and if a change in strategy is required during an interaction. As relative group size is often a strong determiner of between-group contest outcomes60, selection may favour a capacity to assess numerical differences (‘numerosity’) between own and rival groups50. In general, learning will be important in developing contest skills, including signalling components, fighting tactics or the ability to adjust behaviour in response to ongoing dynamics31.
Outsider threats can also affect within-group interactions. The motivations of individual actors with respect to between-group interactions have been repeatedly shown to differ substantially depending on, e.g., their age, sex, reproductive strategy, body condition and dominance status60,61,62,63. For instance, females often engage in between-group contests more frequently than males when the dispute is over resources such as territory or food62, whereas males of many species engage in between-group contests more readily when disputes are over access to females62,64. Similarly, dominant and subordinate group members may have more or less to gain from defence against different outsiders60,65. Resolving such conflicts of interest can foster the emergence of within-group punishment and deterrence either during or after between-group interactions21,61. Conversely, when the interests of individual group members are aligned, individuals participating more in contests with rivals may receive rewards, such as increased affiliative behaviour21,66,67. The tracking of individual contributions and the decision-making processes leading to resultant within-group agonistic and affiliative behaviours likely requires considerable investment in memory and accounting about the status of different relationships. Furthermore, monitoring whether these behaviours have any effect on subsequent contributions to interactions with outsiders66, especially interactions that occur relatively infrequently, introduces inferential challenges associated with between-group conflict.
Integration of outsider interactions in the SIH
A clear relationship between sociality and cognition, the main prediction of the SIH (Fig. 2a), has not consistently emerged2,3,8,12 and studies often report much unexplained variation. For instance, species with similar group sizes often show large variation in brain size that is not explicable when focusing solely on social intelligence associated with interactions within groups2,4. Existing proposed explanations for this brain-size variation include (1) differences in non-social ecological factors2, (2) physical constraints on brain size12 and (3) resource constraints on energetically expensive brains12. However, as the extent and nature of outsider interactions varies considerably between species, we argue that there are several reasons why including this social axis (and resulting Napoleonic intelligence) may improve the predictive power of the SIH.
a The classic social intelligence hypothesis focuses only on one axis of sociality (within-group interactions, often using group size as a proxy) in driving cognitive evolution; variation in conspecific outsider threats is ignored. Combining within-group and outsider interactions could produce b an additive or c a synergistic effect on cognitive evolution.
Outsider interactions can select for higher cognitive abilities in both similar and different ways to within-group interactions. On the one hand, both require optimal responses to the behaviour (or predicted behaviour) of others. Here, the two axes may combine to promote higher cognition through selection on the same specific domain. On the other hand, outsider interactions often differ qualitatively from within-group interactions (Table 1). For instance, conflict within groups may favour a capacity for transitive reasoning in dominance hierarchies (based on individual recognition)31,68, which is likely often to be of limited use with respect to outsiders (but see ref. 69 for transitive inference between territory-holders). By contrast, conflict with outsiders may favour long-term spatial memory, which is likely to be of limited use in terms of within-group conflict. In such cases, the two social axes may promote different domain-specific cognitive abilities and the combined pressure on multiple domains may contribute to enhanced selection on domain-general intelligence.
The selective pressures imposed by the two axes of sociality might interact in different ways. An additive scenario would involve a step along each axis boosting cognition by a fixed amount regardless of the value on the other axis (Fig. 2b). Alternatively, a synergistic scenario would involve both axes combining to drive disproportionately high cognitive abilities in contexts where high levels of within-group conflict are matched by high levels of outsider conflict (Fig. 2c). Two forms of synergistic effects can be envisaged. First, synergy may be a result of the way domain-general intelligence arises within the brain. Exposure to both within-group and outsider interactions may increase the diversity of social tasks. Consequent selection on several cognitive domains in parallel may boost domain-general intelligence more than selection on only a limited number of cognitive tasks. A second form of synergy may arise when an increase along one axis of sociality pushes the organism further along the other axis. Indeed, outsider threats may amplify the value of within-group social intelligence: within-group conflict can arise, for instance, through access to outgroup mates54 or disagreements about the optimum investment in between-group aggression70. Such non-additive effects may reduce the chances of confirming the predictions of the SIH as currently framed: e.g., in a species with large groups and low outsider threat, the effects of group size on general intelligence (and, by implication, brain size) may be disproportionately lower than in an equivalent species with high outsider threat.
It is likely that the selection pressures exerted by the social environment on cognitive evolution will vary between individuals. For instance, threats from outsiders may generate sex-specific challenges that result in sexually dimorphic cognitive traits. Indeed, there is evidence that sexual selection drives sexual dimorphism in the brain71 and one element of outsider threat, extra-pair or extra-group paternity, has already been linked to sexually dimorphic brains in birds72. High levels of breeding threat might select for behaviours such as territorial defence, elaborate courtship and mate guarding; as these behaviours entail several cognitive challenges, larger brains could evolve in the threatened sex. Particular components of outsider threats may generate cognitive selection pressures on specific types or classes of individuals.
Finally, we suggest a form of intrigue that may arise with respect to outsider interactions in group-living species: cognitive free-riding, where individuals might exploit the cognitive investment of others. For instance, a need to invest substantial cognitive effort into spatial memory to recall and monitor territorial boundaries may generate a collective-action problem73. This is because cognition is individually costly74—investment in cognition may come at the expense of lifespan75 or investments in other fitness-boosting functions, including digestion76 and immunity77—and at least some of the benefits are realised by the group as a whole. Group members who ‘parasitise’ the cognitive investments of other group members may therefore enjoy a within-group advantage, freeing cognitive resources for self-interest. Parasitically outsourcing cognitive abilities is rational when they do not confer a significant within-group advantage to individuals (e.g., spatial memory; Table 1). Thus, investment in aspects of cognition may itself become a source of within-group conflict, driving the evolution of divergent cognitive strategies78 within species.
Testing predictions
To test our proposed expansion of the SIH, including both major axes of sociality, we advocate careful consideration of the metrics used to assess outsider interactions and when they are applicable, and the use of a complementary suite of intraspecific and interspecific approaches.
Outsider metrics
Clear metrics of outsider threats and opportunities are needed. We suggest three main categories to capture the level of competition with outsiders, all of which are relevant to individuals, pairs and groups (Fig. 3); as with predation79, these range from measures of the overall risk level to those relating to specific events. First, quantification of the broad context—the landscape of outsider pressure (e.g., number of neighbours and territorial turnover rate). A high threat (or ‘shadow’) of conflict may actually cause neighbours to avoid escalation in their interactions but still play an important role in shaping social dynamics80 and, in turn, cognition. Second, quantification of the frequency of interactions within this contextual landscape, both in terms of the encounter rate between rivals (e.g., between-group interaction frequency) and the rate at which specific outcomes arise (e.g., extra-pair or extra-group paternity). Third, quantification of parameters relating to actual contests with outsiders (e.g., duration and level of escalation).
a ‘Context’ refers to the social landscape in which conspecific outsider interactions occur. In solitary western fence lizards (Sceloporus occidentalis), e.g., long-term observations reveal considerable home-range and territorial overlap98. Lizard photograph: D. A. Hofmann. b ‘Frequency’ refers to the rate of relevant interactions with conspecific outsiders. Many socially monogamous birds have high levels of extra-pair mating; for instance, in pair-bonded yellow-breasted chats (Icteria virens), telemetry reveals nocturnal visits to other territories for likely mating opportunities99. Yellow-breasted chat photograph: E. Willoughby. c ‘Contests’ refers to the characteristics of actual interactions between conspecific rivals. In chimpanzees (Pan troglodytes), e.g., violent between-group contests can have dramatic consequences for social evolution100. Chimpanzee photograph: A. Sandel.
No single factor measures competition with outsiders in its entirety. One reason for this is that different types of threat, which are not necessarily strongly correlated, may be captured by different metrics. For instance, challenges to reproduction may best be indicated by extra-pair or extra-group paternity rates72, whereas threats to territory space may be better captured by the occurrence of agonistic encounters between rival individuals, pairs or groups81,82. Moreover, a single metric may not provide a clear-cut reflection of competition level across species. For example, one primate study of brain size has used greater home-range overlap to indicate greater between-group competition for resources30, but overlap could equally be indicative of higher tolerance in some species. Similarly, although pressure exerted by neighbours is often measured as the rate of between-group encounters81,82, low rates might either represent low outsider pressure or indicate avoidance by rivals. Accordingly, using a plurality of metrics will be necessary to capture variation in outsider conflict within and between species; for any given comparison, the same metrics will be needed for all species being considered. Those metrics could be used as independent factors in analyses, potentially comparing between different threat types, or could be combined to form an index of overall threat. Recent work on between-group interactions in chimpanzees, for instance, has developed a single ‘neighbour pressure index’, integrating estimates of between-group interaction frequency, territorial position (with intrusions to the territory core being viewed as more threatening than those on the periphery) and disputed resource value (as indicated by usage by the resident group)18. This index was then coupled with a group’s competitive ability and within-group competition to consider influences on reproductive output18. It is this integrated approach, considering both interactions within groups and with outsiders, that we are advocating with respect to cognition.
Careful thought is also needed about the categorisation of outsiders and when these outsider metrics should be measured. In the majority of cases, most obviously in those species that defend all-purpose territories, the classification of outsiders is clear: it is those individuals not part of the territorial unit (be that the individual, pair or group). In some species, individuals breed in pairs, defending a nest site or breeding territory together, but feed with others in temporary groups; here, outsiders in the breeding context may be groupmates while foraging, so we would advocate measurement of threat metrics with respect to just the former. Likewise, there are examples of group-living species that require further consideration as to what constitutes an outsider (e.g., fission–fusion societies, where group composition shifts dynamically as groups divide into sub-groups and later reassemble). In these cases, it might be valuable to quantify outsider threats at multiple levels of social organisation, as is already often the case with neighbours vs. strangers in territorial species.
Complementary approaches
With respect to intraspecific investigations of Napoleonic intelligence, we suggest three priority areas. First, we advocate a focus on the causes of individual variation in cognitive traits in relation to outsider interactions. This can be achieved by testing cognitive performance on psychometric tasks7,83 or using neuroanatomical measures, such as brain size84, size of brain regions85 or neuron density86. In addition, longitudinal studies, quantifying cognition at regular time steps over the course of an individual’s life87, may help to identify an effect of outsider threat on cognitive development. Quantifying individual cognitive performance in both domain-general cognitive tasks (e.g., associative learning87) and socio-cognitive tasks (e.g., social competence88) will be important in determining whether the cognitive consequences of outsider interactions are domain specific or if a ‘general intelligence factor’ exists. Second, investigation into the consequences of individual variation in cognition will help to determine whether outsider threats can have evolutionary implications. This can be achieved by examining the relationship between cognition and measures of fitness (such as reproductive success87). Third, comparisons of sub-populations experiencing different selection pressures offer a window into the factors governing cognitive variation89. As with predation risk85, natural variation in outsider threat levels provides one option and it may also be possible to manipulate experimentally the level of threat. Recent studies examining the short-term consequences of outgroup conflict, in both laboratory and field settings, have demonstrated the feasibility of simulating outsider threats using call playbacks, faecal presentations and controlled intrusions80,90,91.
Interspecific approaches, using phylogenetic comparisons at varying scales, can complement intraspecific studies by revealing long-term evolutionary trends in cognitive evolution2,3. Using phylogenetic regressions, measures of brain size (relative or total brain size, or size of certain brain regions) may be modelled as a function of outsider threat as determined from the metrics outlined in Fig. 3. The use of neuroanatomical measures as cognitive indicators is a controversial issue92 (e.g., links between brain size and cognitive performance have been identified in some cases74,84,93, but other researchers have argued that is unclear how they translate to differences in cognition92). However, these proxies are the most viable option currently available for large-scale comparisons (e.g., across birds3 or primates2). In addition, small-scale phylogenetic comparisons on clades of closely related species could use cognitive performance quantified in all the relevant species using identical psychometric methods and tests. Quantifying cognitive performance within a radiation would allow comparative analyses both to use direct measures of cognition and to mitigate the confounding effects that arise in making interspecific comparisons across disparate taxa. A combination of intra- and interspecific techniques would allow a powerful test of the central predictions about Napoleonic intelligence specifically and the expanded SIH more generally.
Conclusion
For over 50 years, sociality has been hypothesised as an evolutionary driver of animal cognition5,6. However, the focus has been predominantly on interactions occurring within groups. Although this focus has advanced our understanding of cognition in diverse taxa94, the complexity of social interactions with conspecific outsiders (and their often surprising consequences for social evolution) is increasingly being recognised65,70,95,96,97. Conflict with conspecific outsiders is prevalent across the social spectrum (from individuals to pairs, to groups) and represents a powerful selective force; adaptations have arisen that minimise the risks and allow exploitation of the opportunities arising from these outsider interactions. We argue that a complementary focus of the SIH should therefore be those pressures arising from conspecific outsider interactions: considering both major axes of sociality will likely improve our understanding of social intelligence and cognitive evolution.
Data availability
No new data were used in the preparation of this manuscript.
Change history
24 November 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41467-020-20090-7
References
Milton, K. in Machiavellian Intelligence: Social Expertise and the Evolution of Intellect in Monkeys, Apes, and Humans 285–305 (Clarendon, Oxford, 1988).
DeCasien, A. R., Williams, S. A. & Higham, J. P. Primate brain size is predicted by diet but not sociality. Nat. Ecol. Evol. 1, 0112 (2017).
Sayol, F. et al. Environmental variation and the evolution of large brains in birds. Nat. Commun. 7, 13971 (2016).
Powell, L. E., Isler, K. & Barton, R. A. Re-evaluating the link between brain size and behavioural ecology in primates. Proc. R. Soc. B Biol. Sci. 284, 1–8 (2017).
Byrne, R. W. & Whiten, A. in Machiavellian Intelligence: Social Expertise and the Evolution of Intellect in Monkeys, Apes, and Humans. https://doi.org/10.2307/2804121 (Clarendon, Oxford, 1988).
Dunbar, R. I. M. The social brain hypothesis. Evol. Anthropol. Rev. 6, 178–190 (1998).
Ashton, B. J., Thornton, A. & Ridley, A. R. An intraspecific appraisal of the social intelligence hypothesis. Philos. Trans. R. Soc. B Biol. Sci. 373, 20170288 (2018).
Holekamp, K. E. Questioning the social intelligence hypothesis. Trends Cogn. Sci. 11, 65–69 (2007).
Kern, J. M. & Radford, A. N. Experimental evidence for delayed contingent cooperation among wild dwarf mongooses. Proc. Natl Acad. Sci. USA 115, 6255–6260 (2018).
Borgeaud, C. & Bshary, R. Wild vervet monkeys trade tolerance and specific coalitionary support for grooming in experimentally induced conflicts. Curr. Biol. 25, 3011–3016 (2015).
Moll, H. & Tomasello, M. Cooperation and human cognition: the Vygotskian intelligence hypothesis. Philos. Trans. R. Soc. B Biol. Sci. 362, 639–648 (2007).
van Schaik, C. P., Isler, K. & Burkart, J. M. Explaining brain size variation: from social to cultural brain. Trends Cogn. Sci. 16, 277–284 (2012).
Lucas, J. R., Gentry, K. E., Sieving, K. E. & Freeberg, T. M. Communication as a fundamental part of Machiavellian intelligence. J. Comp. Psychol. 132, 442–454 (2018).
Dunbar, R. I. M. & Shultz, S. Why are there so many explanations for primate brain evolution? Philos. Trans. R. Soc. B Biol. Sci. 372, 20160244 (2017).
Dunbar, R. I. M. & Shultz, S. Understanding primate brain evolution. Philos. Trans. R. Soc. B Biol. Sci. 362, 649–658 (2007).
Shultz, S. & Dunbar, R. I. M. Social bonds in birds are associated with brain size and contingent on the correlated evolution of life-history and increased parental investment. Biol. J. Linn. Soc. 100, 111–123 (2010).
Shultz, S. & Dunbar, R. I. M. The evolution of the social brain: anthropoid primates contrast with other vertebrates. Proc. R. Soc. B Biol. Sci. 274, 2429–2436 (2007).
Lemoine, S. et al. Between-group competition impacts reproductive success in wild chimpanzees. Curr. Biol. 30, 312–318.e3 (2020).
Adams, E. S. Approaches to the study of territory size and shape. Annu. Rev. Ecol. Syst. 32, 277–303 (2001).
Hardy, I. C. W. & Briffa, M. Animal Contests (Cambridge Univ. Press, Cambridge, 2013).
Radford, A. N., Majolo, B. & Aureli, F. Within-group behavioural consequences of between-group conflict: a prospective review. Proc. R. Soc. B Biol. Sci. 283, 20161567 (2016).
van Schaik, C. P. in Comparative Socioecology (eds. Standen, V. & Foley, R. A.) 195–218 (Blackwell, 1989).
Wrangham, R. W. An ecological model of female-bonded primate groups. Behaviour 75, 262–300 (1980).
Sterck, E. H. M. The evolution of female social relationships in nonhuman primates. Behav. Ecol. Sociol. 291–309 (1997).
Wilson, M. L. & Wrangham, R. W. Intergroup relations in chimpanzees. Annu. Rev. Anthropol. 32, 363–392 (2003).
Moser-Purdy, C., MacDougall-Shackleton, E. A. & Mennill, D. J. Enemies are not always dear: male song sparrows adjust dear enemy effect expression in response to female fertility. Anim. Behav. 126, 17–22 (2017).
Gherardi, F. Fighting behavior in hermit crabs: the combined effect of resource-holding potential and resource value in Pagurus longicarpus. Behav. Ecol. Sociobiol. 59, 500–510 (2006).
Alexander, R. D. in The Human Revolution (eds. Mellars, P. & Stringer, C.) 455–513 (Edinburgh Univ. Press, Edinburgh, 1984).
Hamilton, W. D. in ASA Studies 4: Biosocial Anthropology (ed. Fox, R.) 133–153 (Malaby, 1975).
Grueter, C. C. Home range overlap as a driver of intelligence in primates. Am. J. Primatol. 77, 418–424 (2015).
Reichert, M. S. & Quinn, J. L. Cognition in contests: mechanisms, ecology, and evolution. Trends Ecol. Evol. 32, 773–785 (2017).
Shettleworth, S. J. Cognition, Evolution, and Behavior (Oxford Univ. Press, 2010).
Young, A. J., Spong, G. & Clutton-Brock, T. Subordinate male meerkats prospect for extra-group paternity: alternative reproductive tactics in a cooperative mammal. Proc. R. Soc. B Biol. Sci. 274, 1603–1609 (2007).
Radford, A. N. & du Plessis, M. A. Territorial vocal rallying in the green woodhoopoe: factors affecting contest length and outcome. Anim. Behav. 68, 803–810 (2004).
Geissmann, T. & Orgeldinger, M. The relationship between duet songs and pair bonds in siamangs, Hylobates syndactylus. Anim. Behav. 60, 805–809 (2000).
Ridley, A. R. in Cooperative Breeding in Vertebrates: Studies of Ecology, Evolution and Behavior. 115–132. https://doi.org/10.1017/CBO9781107338357.008 (Cambridge Univ. Press, 2016).
Bee, M. A. Habituation and sensitization of aggression in bullfrogs (Rana catesbeiana): testing the dual-process theory of habituation. J. Comp. Psychol. 115, 307–316 (2001).
Sheehan, M. J. & Tibbetts, E. A. Selection for individual recognition and the evolution of polymorphic identity signals in Polistes paper wasps. J. Evol. Biol. 23, 570–577 (2010).
Cant, M. A., Otali, E. & Mwanguhya, F. Fighting and mating between groups in a cooperatively breeding mammal, the banded mongoose. Ethology 108, 541–555 (2002).
Braga Goncalves, I. & Radford, A. N. Experimental evidence that intruder and group member attributes affect outgroup defence and associated within-group interactions in a social fish. Proc. R. Soc. B Biol. Sci. 286, 20191261 (2019).
Szipl, G., Ringler, E. & Bugnyar, T. Attacked ravens flexibly adjust signalling behaviour according to audience composition. Proc. R. Soc. B Biol. Sci. 285, 20180375 (2018).
Noser, R. & Byrne, R. W. Mental maps in chacma baboons (Papio ursinus): Using inter-group encounters as a natural experiment. Anim. Cogn. 10, 331–340 (2007).
Radford, A. N. Preparing for battle? Potential intergroup conflict promotes current intragroup affiliation. Biol. Lett. 7, 26–29 (2011).
Christensen, C. & Radford, A. N. Dear enemies or nasty neighbors? Causes and consequences of variation in the responses of group-living species to territorial intrusions. Behav. Ecol. 29, 1004–1013 (2018).
Temeles, E. J. The role of neighbours in territorial systems: when are they ‘dear enemies’? Anim. Behav. 47, 339–350 (1994).
Radford, A. N. Group-specific vocal signatures and neighbour-stranger discrimination in the cooperatively breeding green woodhoopoe. Anim. Behav. 70, 1227–1234 (2005).
Hyman, J. & Hughes, M. Territory owners discriminate between aggressive and nonaggressive neighbours. Anim. Behav. 72, 209–215 (2006).
Monclús, R., Saavedra, I. & de Miguel, J. Context-dependent responses to neighbours and strangers in wild European rabbits (Oryctolagus cuniculus). Behav. Process. 106, 17–21 (2014).
Thompson, F. J., Marshall, H. H., Vitikainen, E. I. K. & Cant, M. A. Causes and consequences of intergroup conflict in cooperative banded mongooses. Anim. Behav. 126, 31–40 (2017).
McComb, K., Packer, C. & Pusey, A. Roaring and numerical assessment in contests between groups of female lions, Panthera leo. Anim. Behav. 47, 379–387 (1994).
Descovich, K. A., Lisle, A. T., Johnston, S., Nicolson, V. & Phillips, C. J. C. Differential responses of captive southern hairy-nosed wombats (Lasiorhinus latifrons) to the presence of faeces from different species and male and female conspecifics. Appl. Anim. Behav. Sci. 138, 110–117 (2012).
Christensen, C., Kern, J. M., Bennitt, E. & Radford, A. N. Rival group scent induces changes in dwarf mongoose immediate behavior and subsequent movement. Behav. Ecol. 27, 1627–1634 (2016).
Trimmer, P. C. & Houston, A. I. An evolutionary perspective on information processing. Top. Cogn. Sci. 6, 312–330 (2014).
Mares, R., Young, A. J., Levesque, D. L., Harrison, N. & Clutton-Brock, T. H. Responses to intruder scents in the cooperatively breeding meerkat: sex and social status differences and temporal variation. Behav. Ecol. 22, 594–600 (2011).
Humphries, D. J., Finch, F. M., Bell, M. B. V. & Ridley, A. R. Vocal cues to identity: pied babblers produce individually distinct but not stable loud calls. Ethology 122, 609–619 (2016).
Burgener, N., Dehnhard, M., Hofer, H. & East, M. L. Does anal gland scent signal identity in the spotted hyaena? Anim. Behav. 77, 707–715 (2009).
Lanchester, F. W. W. Aircraft in Warfare: the Dawn of the Fourth Arm. (Constable and Company Limited, 1916).
Wilson, M. L., Britton, N. F. & Franks, N. R. Chimpanzees and the mathematics of battle. Proc. R. Soc. B Biol. Sci. 269, 1107–1112 (2002).
Plowes, N. J. R. & Adams, E. S. An empirical test of Lanchester’s square law: mortality during battles of the fire ant Solenopsis invicta. Proc. R. Soc. B Biol. Sci. 272, 1809–1814 (2005).
Radford, A. N. Territorial vocal rallying in the green woodhoopoe: influence of rival group size and composition. Anim. Behav. 66, 1035–1044 (2003).
van Schaik, C. P. et al. Male monkeys use punishment and coercion to de-escalate costly intergroup fights. Proc. R. Soc. B Biol. Sci. 285, 20172323 (2018).
Boydston, E. E., Morelli, T. L. & Holekamp, K. E. Sex differences in territorial behavior exhibited by the spotted hyena (Hyaenidae, Crocuta crocuta). Ethology 107, 369–385 (2001).
McComb, K., Pusey, A., Packer, C. & Grinnell, J. Female lions can identify potentially infanticidal males from their roars. Proc. R. Soc. B Biol. Sci. 252, 59–64 (1993).
Koch, F., Signer, J., Kappeler, P. M. & Fichtel, C. Intergroup encounters in Verreaux’s sifakas (Propithecus verreauxi): who fights and why? Behav. Ecol. Sociobiol. 70, 797–808 (2016).
Schindler, S., Radford, A. N. & Schindler, S. Factors influencing within-group conflict over defence against conspecific outsiders seeking breeding positions. Proc. R. Soc. B Biol. Sci. 285, 20181669 (2018).
Arseneau-Robar, T. J. M. et al. Female monkeys use both the carrot and the stick to promote male participation in intergroup fights. Proc. R. Soc. B Biol. Sci. 283, 20161817 (2016).
Radford, A. N. Duration and outcome of intergroup conflict influences intragroup affiliative behaviour. Proc. R. Soc. B Biol. Sci. 275, 2787–2791 (2008).
Tibbetts, E. A., Agudelo, J., Pandit, S. & Riojas, J. Transitive inference in Polistes paper wasps. Biol. Lett. 15, 20190015 (2019).
Grosenick, L., Clement, T. S. & Fernald, R. D. Fish can infer social rank by observation alone. Nature 445, 429–432 (2007).
Arseneau-Robar, T. J. M., Taucher, A. L., Schnider, A. B., van Schaik, C. P. & Willems, E. P. Intra- and interindividual differences in the costs and benefits of intergroup aggression in female vervet monkeys. Anim. Behav. 123, 129–137 (2017).
Kotrschal, A., Räsänen, K., Kristjánsson, B. K., Senn, M. & Kolm, N. Extreme sexual brain size dimorphism in sticklebacks: a consequence of the cognitive challenges of sex and parenting? PLoS ONE 7, e30055 (2012).
Garamszegi, L. Z., Eens, M., Erritzøe, J. & Møller, A. P. Sperm competition and sexually size dimorphic brains in birds. Proc. R. Soc. B Biol. Sci. 272, 159–166 (2005).
Willems, E. P. & Van Schaik, C. P. Collective action and the intensity of between-group competition in nonhuman primates. Behav. Ecol. 26, 625–631 (2015).
Kotrschal, A. et al. Artificial selection on relative brain size in the guppy reveals costs and benefits of evolving a larger brain. Curr. Biol. 23, 168–171 (2013).
Kotrschal, A., Corral-Lopez, A. & Kolm, N. Large brains, short life: selection on brain size impacts intrinsic lifespan. Biol. Lett. 15 (2019).
Tsuboi, M. et al. Comparative support for the expensive tissue hypothesis: big brains are correlated with smaller gut and greater parental investment in Lake Tanganyika cichlids. Evolution 69, 190–200 (2015).
Kotrschal, A., Kolm, N. & Penn, D. J. Selection for brain size impairs innate, but not adaptive immune responses. Proc. R. Soc. B Biol. Sci. 283 (2016).
Gervais, M. M., Kline, M., Ludmer, M., George, R. & Manson, J. H. The strategy of psychopathy: primary psychopathic traits predict defection on low-value relationships. Proc. R. Soc. B Biol. Sci. 280, 20122773 (2013).
Creel, S. & Christianson, D. Relationships between direct predation and risk effects. Trends Ecol. Evol. 23, 194–201 (2008).
Bruintjes, R., Lynton-Jenkins, J., Jones, J. W. & Radford, A. N. Out-group threat promotes within-group affiliation in a cooperative fish. Am. Nat. 187, 274–282 (2015).
Mosser, A. & Packer, C. Group territoriality and the benefits of sociality in the African lion, Panthera leo. Anim. Behav. 78, 359–370 (2009).
Crofoot, M. C., Gilby, I. C., Wikelski, M. C. & Kays, R. W. Interaction location outweighs the competitive advantage of numerical superiority in Cebus capucinus intergroup contests. Proc. Natl Acad. Sci. USA 105, 577–581 (2008).
Shaw, R. C., Boogert, N. J., Clayton, N. S. & Burns, K. C. Wild psychometrics: evidence for ‘general’ cognitive performance in wild New Zealand robins, Petroica longipes. Anim. Behav. 109, 101–111 (2015).
Buechel, S. D., Boussard, A., Kotrschal, A., van Der Bijl, W. & Kolm, N. Brain size affects performance in a reversal-learning test. Proc. R. Soc. B Biol. Sci. 285, 20172031 (2018).
Kotrschal, A., Deacon, A. E., Magurran, A. E. & Kolm, N. Predation pressure shapes brain anatomy in the wild. Evol. Ecol. 31, 619–633 (2017).
Herculano-Houzel, S. & Lent, R. Isotropic fractionator: a simple, rapid method for the quantification of total cell and neuron numbers in the brain. J. Neurosci. 25, 2518–2521 (2005).
Ashton, B. J., Ridley, A. R., Edwards, E. K. & Thornton, A. Cognitive performance is linked to group size and affects fitness in Australian magpies. Nature 61, 5985–5991 (2018).
Taborsky, B. & Oliveira, R. F. Social competence: an evolutionary approach. Trends Ecol. Evol. 27, 679–688 (2012).
Gonda, A., Herczeg, G. & Merilä, J. Evolutionary ecology of intraspecific brain size variation: a review. Ecol. Evol. 3, 2751–2764 (2013).
Morris-Drake, A. et al. Experimental field evidence that out-group threats influence within-group behavior. Behav. Ecol. 30, 1425–1435 (2019).
Hellmann, J. K. & Hamilton, I. M. Intragroup social dynamics vary with the presence of neighbors in a cooperatively breeding fish. Curr. Zool. 65, 21–31 (2018).
Healy, S. D. & Rowe, C. A critique of comparative studies of brain size. Proc. R. Soc. B Biol. Sci. 274, 453–464 (2007).
Kotrschal, A. et al. The benefit of evolving a larger brain: big-brained guppies perform better in a cognitive task. Anim. Behav. 86, e4–e6 (2013).
Whiten, A. Social, Machiavellian and cultural cognition: a golden age of discovery in comparative and evolutionary psychology. J. Comp. Psychol. 132, 437–441 (2018).
Radford, A. N. & Bruintjes, R. Expanding the link between out-group threats and in-group behavior (a reply to Kavaliers and Choleris). Am. Nat. 189, 459–462 (2017).
Brown, M. Food and range defence in group-living primates. Anim. Behav. 85, 807–816 (2013).
Mirville, M. O. et al. Factors influencing individual participation during intergroup interactions in mountain gorillas. Anim. Behav. 144, 75–86 (2018).
Sheldahl, L. A. & Martins, E. P. The territorial behavior of the western fence lizard, Sceloporus occidentalis. Herpetologica 56, 469–479 (2000).
Ward, M. P., Alessi, M., Benson, T. J. & Chiavacci, S. J. The active nightlife of diurnal birds: extraterritorial forays and nocturnal activity patterns. Anim. Behav. 88, 175–184 (2014).
Feldblum, J. T., Manfredi, S., Gilby, I. C. & Pusey, A. E. The timing and causes of a unique chimpanzee community fission preceding Gombe’s “Four-Year War”. Am. J. Phys. Anthropol. 166, 730–744 (2018).
Acknowledgements
We are grateful to Ines Braga Goncalves, Amy Morris-Drake and Harry Suter for beneficial discussions about the topic of outsider consequences. This work was supported by a European Research Council Consolidator Grant (project number 682253) awarded to A.N.R.
Author information
Authors and Affiliations
Contributions
B.J.A., P.K. and A.N.R. conceived the concepts of this paper together and contributed equally to the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks Thomas Bugnyar, Michael Cant and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ashton, B.J., Kennedy, P. & Radford, A.N. Interactions with conspecific outsiders as drivers of cognitive evolution. Nat Commun 11, 4937 (2020). https://doi.org/10.1038/s41467-020-18780-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-18780-3
This article is cited by
-
Why are ravens smart? Exploring the social intelligence hypothesis
Journal of Ornithology (2024)
-
When neighbors become family: the dear-enemy effect of swimming crab and the verification of the formation hypothesis
Behavioral Ecology and Sociobiology (2024)
-
Mechanisms for individual, group-based and crowd-based attention to social information
Nature Reviews Psychology (2022)
-
Search performance and octopamine neuronal signaling mediate parasitoid induced changes in Drosophila oviposition behavior
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.