Abstract
Ethylene plays essential roles during adaptive responses to water-saturating environments in rice, but knowledge of its signaling mechanism remains limited. Here, through an analysis of a rice ethylene-response mutant mhz1, we show that MHZ1 positively modulates root ethylene responses. MHZ1 encodes the rice histidine kinase OsHK1. MHZ1/OsHK1 is autophosphorylated at a conserved histidine residue and can transfer the phosphoryl signal to the response regulator OsRR21 via the phosphotransfer proteins OsAHP1/2. This phosphorelay pathway is required for root ethylene responses. Ethylene receptor OsERS2, via its GAF domain, physically interacts with MHZ1/OsHK1 and inhibits its kinase activity. Genetic analyses suggest that MHZ1/OsHK1 acts at the level of ethylene perception and works together with the OsEIN2-mediated pathway to regulate root growth. Our results suggest that MHZ1/OsHK1 mediates the ethylene response partially independently of OsEIN2, and is directly inhibited by ethylene receptors, thus revealing mechanistic details of ethylene signaling for root growth regulation.
Similar content being viewed by others
Introduction
Ethylene plays essential roles in plant growth and development. In the model plant Arabidopsis, a linear ethylene signaling pathway has been established based on the characterization of a series of triple response mutants1,2,3,4. The pathway contains a family of endoplasmic reticulum (ER) membrane-bound ethylene receptors, a Ser/Thr kinase CTR1 (constitutive triple response 1), a central ER membrane protein EIN2 and transcription factors EIN3/EIL1 and ERF15,6,7,8,9,10,11,12,13.
In the absence of ethylene, the ethylene receptors are in active state and CTR1 is likely activated to phosphorylate the C-terminal domain of EIN2 to repress ethylene response. In the presence of ethylene, the C-terminus of EIN2 is cleaved and translocated to nucleus for activation of downstream EIN3/EIL1 transcriptional cascade and then ethylene response14,15,16. EIN2 and EIN3/EIL1 are regulated by proteasomal degradation17,18,19,20. Recently, the EIN2 C-terminal domain is also found to be targeted to the cytoplasmic processing-body (P-body) for the translational regulation of EBF1/221,22. EIN2-mediated histone acetylation and deacetylation are also involved in transcriptional regulation in ethylene signaling23,24,25.
Rice (Oryza sativa) is an important monocotyledonous crop and usually lives in water-saturated soil in most of the life cycle. Despite the essential role ethylene plays in the adaptive responses of rice to hypoxia stress, the ethylene signaling pathway in rice has not been systematically studied. Genes homologous to the Arabidopsis ethylene signaling components have been identified and some are characterized in rice, including ethylene receptor gene OsETR2, RTE1-like gene, CTR1-like gene, EIN2-like gene and EIN3-like gene26,27,28,29,30. Adopting an effective screening system, we have isolated a set of rice ethylene-response mutants31. The analyses of these mutants suggest that ethylene signaling in rice and Arabidopsis has both conserved and divergent aspects32,33,34,35.
Histidine kinases (HK) play crucial roles in the regulation of plant development in response to hormones, as well as environmental stimuli36,37. HK-mediated multistep phosphorelay involves hybrid-type HK with both histidine kinase and receiver domains, His-containing phosphotransfer protein (HPt), and response regulator (RR)37,38. Ethylene receptors are structurally similar to bacterial HKs and some receptors such as ETR1 and ERS1 do have canonical HK activity39,40. However, the HK activity of ethylene receptor is not required for ethylene signaling but only plays a modulating role in the pathway41,42. So far, how the ethylene receptor transmits signals remains largely unclear. It has been reported that a non-ethylene receptor HK, Arabidopsis authentic HK5 (AHK5), acts as a negative regulator in the ETR1 dependent signaling pathway in which ethylene and ABA inhibit the root elongation43. In contrast, the maize homolog ZmHK9 acts as a positive regulator in the root growth response to ethylene and ABA in transgenic Arabidopsis44. OsHK1, a rice histidine kinase38, is reported to play roles in root growth and circumnutations through a cytokinin-related pathway45. In these studies, however, little is known about the molecular mechanism by which the HKs regulate the signaling cascade.
In this study, we characterized the rice root-specific ethylene-insensitive mutant mhz1 and found that MHZ1 encodes the rice histidine kinase OsHK1. MHZ1 positively modulates ethylene response in rice roots. Biochemical analysis showed that MHZ1 is a functional hybrid-type HK, which autophosphorylates in a conserved histidine and transfers the phosphoryl group via its receiver domain to OsAHP1/2 and then further to response regulator OsRR21. Genetic evidence demonstrates that the HK activity of MHZ1 and it-mediated phosphorelay are required for regulation of root ethylene response in rice. More interestingly, we discover that the ethylene receptors, via GAF domain, can directly bind to MHZ1 protein and inhibit its kinase activity based on both in vitro and in vivo analyses. These findings reveal a previously unidentified mechanism for the ethylene receptor signal transduction.
Results
Characterization of mhz1 and gene identification
We have isolated a set of rice ethylene-response mutants and the mhz1 exhibited root-specific ethylene-insensitive phenotype31. In air, etiolated seedlings of two allelic mutants mhz1-1 and mhz1-2 were very similar in coleoptile/shoot and root growth to WT. In ethylene, WT root length was drastically reduced whereas mhz1-1 and mhz1-2 root growth was not inhibited, indicating a complete ethylene-insensitive phenotype in primary roots of the two mutants (Fig. 1a). Coleoptile growth of mhz1-1 and mhz1-2 responded normally to ethylene, except that the mutants have slightly longer coleoptiles than WT (Fig. 1a). Light-grown mhz1-1 seedlings had longer roots than WT (Supplementary Fig. 1a). Two additional allelic mutants (mhz1-3 and mhz1-4) were further identified and they resembled mhz1-1 and mhz1-2 in ethylene responses (Supplementary Fig. 1b). These results indicate that mhz1 is insensitive to ethylene in root growth.
a Ethylene response phenotype of mhz1 alleles. Etiolated seedlings were treated with various concentrations of ethylene in darkness. Representative seedlings grown in the air and in 10 ppm ethylene are shown (Left). Coleoptile (Center) and root lengths (Right) are means ± SD, n > 30. Bars indicate 10 mm. b MHZ1 genomic structure and mutation sites of different mhz1 alleles. Colored boxes indicate exons and horizontal lines indicate introns. c Schematic structure of MHZ1 and mutation sites of different mhz1 alleles. d MHZ1 gene expression in WT and mhz1-1. Actin1 was amplified as internal control. e MHZ1 overexpression lines (MHZ1-OE) have constitutive ethylene response. MHZ1 native promoter was used to drive the MHZ1 cDNA for overexpression. Etiolated seedlings were treated with various concentrations of ethylene and 10 ppm 1-MCP under darkness. Bars indicate 10 mm. Root lengths (Right) are means ± SD, n > 30. f Expression of ethylene-inducible genes OsRRA5, OsERF002 OsRAP2.8, OsERF063 and OsERF073 in mhz1-1 and MHZ1-OE lines compared with WT as revealed by qPCR. Data are means ± SD, n = 4. Source data are provided as a Source Data file.
The MHZ1 gene was identified to be LOC_Os06g44410 through TAIL-PCR analysis and the T-DNA was inserted in the fifth intron between 2032 bp and 2033 bp from the start codon of the gene in mhz1-1 (Fig. 1b, c). No MHZ1 expression was detected in mhz1-1 (Fig. 1d). Other alleles were further analyzed (Fig. 1b, c and Supplementary Fig. 1b). Genetic transformation with the WT genomic DNA fragment rescued the ethylene-insensitive phenotype of mhz1-1 (Supplementary Fig. 1c). All these results indicate that MHZ1 corresponds to the locus LOC_Os06g44410, which encodes the histidine kinase OsHK138. The mhz1-1, -2, -3, -4, -5 mutants may be renamed as Oshk1-4, -5, -6, -7, -8 following the Oshk1 mutants identified by Lehner et al.45. For simplicity, the original mutant names were used since these mutants have been named as mao huzi (mhz, Chinese name with an English meaning of cat whiskers)31.
MHZ1/OsHK1 encodes a histidine kinase of 936 amino acids, with a histidine kinase domain (amino acids 365 to 655) and a receiver domain (amino acids 817 to 961) (Fig. 1c and Supplementary Fig. 2a). Phylogenetic analysis indicates that MHZ1 does not belong to ethylene receptor family or cytokinin receptor homologs (Supplementary Fig. 3). MHZ1 is homologous to AHK5 and ZmHK9. Both of the genes were reported to play roles in modulating ethylene responses43,44. MHZ1 is clustered with homologous proteins from monocotyledonous plants (Supplementary Fig. 2b). Coiled-coil and PAS domains were noted in the N-terminal end of these proteins (Supplementary Fig. 2c).
MHZ1 overexpression enhances ethylene response in roots
To study the gene function of MHZ1, we overexpressed MHZ1 in WT rice (Fig. 1e, f and Supplementary Fig. 4d). Compared with the WT, the high-expression MHZ1-OE lines had shorter roots both in air and in ethylene, indicating a constitutive ethylene-response phenotype (Fig. 1e). The short root phenotype of MHZ1-OE lines was not due to elevation of ethylene emission since the ethylene production was not enhanced in these lines (Supplementary Fig. 5). Treatment with 1-methylcyclopropene (1-MCP)46 largely inhibited the short root phenotype of these lines, indicating that ethylene receptor signaling is required for MHZ1 function (Fig. 1e).
We further examined expression of ethylene-responsive genes identified in our previous studies31,32,34,35. qPCR analysis showed that ethylene-induction of OsRRA5, OsERF002 and OsRAP2.8 expression was largely blocked in mhz1, whereas the expression of these genes was substantially enhanced in roots of MHZ1-overexpressing plants in the presence or absence of ethylene (Fig. 1f). In shoots of MHZ1-overexpressing plants, OsRRA5 and OsRAP2.8 expression was not enhanced in air or ethylene; whereas OsERF002 expression was promoted compared to the corresponding WT levels especially in the presence of ethylene (Fig. 1f). The ethylene induction of OsERF063 and OsERF073 was not affected in both roots and shoots of mhz1 or MHZ1-OE plants, suggesting that expression of some genes is independent of MHZ1 (Fig. 1f). These results further confirmed the different ethylene responsiveness of mhz1 mutant and MHZ1-OE plants at molecular levels with some organ and gene specificity.
Ethylene-induced MHZ1 expression requires OsEIN2 and OsEIL1
MHZ1 transcripts are abundant in roots but less in coleoptiles and other organs, and are induced by ethylene in roots (Fig. 2a and Supplementary Fig. 4a). The ethylene induction of MHZ1 requires OsEIN2 and OsEIL1, and OsEIL1 can bind to the ATGTA elements in the MHZ1 promoter and activate the promoter activity in a tobacco transient assay system (Fig. 2a, b, c). Promoter-GUS analysis further reveals that MHZ1 promoter activity is mainly localized in root initiation sites at node, root vascular cylinder and root cortex. The activity is also observed in stem, leaf, grain hull and coleoptile (Supplementary Fig. 4b). Ethylene treatment mildly enhanced the MHZ1 promoter activity especially in the region immediately above the meristem tissue of root tip (Supplementary Fig. 4c).
a MHZ1 expression induced by ethylene is dependent on OsEIN2 and OsEIL1. Two-day-old etiolated seedlings of WT or Osein2 and Oseil1 were treated with 10 ppm ethylene or air. Data are means ± SD, n = 4. b Binding of OsEIL1 protein to the OsEIL1 binding site (EBS) containing region of MHZ1 promoter in vitro. GST-tagged OsEIL1 N-terminus protein was isolated and incubated with biotin-labeled probes carrying EBSs of MHZ1 promoter. An excess of unlabeled probe was added as the competitor. c Transient expression of OsEIL1 in tobacco leaves activated MHZ1 promoter activity. Relative luminescence intensity was calculated, data are means ± SD from three biological replicates. Source data are provided as a Source Data file.
MHZ1 transfers phosphoryl groups to OsRR21 via OsAHPs
Next, we examined whether MHZ1 has HK activity. Different mutant proteins or truncated versions were produced and tested for atuophosphorylation ability (Fig. 3a). The GST-MHZ1 protein (amino acids 365 to 968) containing the kinase domain and receiver domain displays strong kinase activity in the presence of Ca2+ in our phosphorylation assay, and this activity was abolished when the conserved His at 375 position was mutated to Gln (Fig. 3b). A smaller radioactive band was noted below the normal GST-MHZ1, likely representing a degradation product (Fig. 3b). At the physiological level of ATP concentration, GST-MHZ1 had kinase activity in the presence of Ca2+ or Mg2+ (Supplementary Fig. 6a). These results indicate that MHZ1 is a functional HK.
a MHZ1 and its truncated/mutant versions used for phosphorylation analysis. H, G1, G2, and D indicate conserved residues or boxes. MBP indicates maltose-binding protein. b MHZ1 has histidine kinase activity. The radioactive band below the GST-MHZ1 indicates a degradation product. c Trans-phosphorylation between MHZ1 molecules. MBP-G1 has G588A and G590A mutations at G1 box. MBP-G2 has G618A and G620A mutations at G2 box. d MHZ1 can transfer its phosphoryl groups to OsAHP1 and OsAHP2 rather than OsPHP1 or OsPHP2. e Phosphorelay from MHZ1 to OsAHP1 and OsAHP2 was abolished when the conserved histidine was mutated in OsAHP1/2. f MHZ1 can transfer its phosphoryl groups to OsAHP1/2 and further to OsRR21. D68E mutation in OsRR21 disrupted its phosphoryl-accepting ability. g D824A mutation in MHZ1 receiver domain blocked its phosphorelay to OsAHP1/2, and OsRR26 cannot accept phosphoryl groups transferred from MHZ1 and OsAHP1/2. h Time course of the phosphorelay from MHZ1 to OsAHP1 (left panel) and further to OsRR21 (right panel). After the reaction in the left panel was finished, the OsRR21 was added and incubated for various times in the right panel. i MHZ1 but not its mutant versions rescued the ethylene-insensitive phenotype of mhz1 (mhz1-1) mutant. cDNAs of MHZ1 and its mutant versions MHZ1(G1), MHZ1(H375Q) and MHZ1(D824A), fusioned with a 3 × FLAG sequence, driven by MHZ1 native promoter, were transformed into the mhz1-1 to observe the root ethylene response. Total proteins of each line were immunoblotted for MHZ1-FLAG with anti-FLAG antibody. A non-specific band was used as a loading control. MHZ1 gene expression was examined by RT-PCR and Actin1 was amplified as control. Bar indicates 10 mm. j Ethylene response of Osahp1 Osahp2 double-mutant. Osahp1 Osahp2 double-mutant was segregated from the self-bred progenies of an Osahp1 (heterozygous)/Osahp2 (homozygous) plant. “ + ” indicates wild-type OsAHP1. k Ethylene response of OsRR21 mutants and overexpression lines (OE). For j and k, etiolated seedlings were treated with 10 ppm ethylene or air for 2.5 days. Bars indicate 10 mm. Source data are provided as a Source Data file.
When the receiver domain was removed, GST-KD or MBP-KD containing kinase domain (amino acids 365 to 655) still had autophosphorylation activity (Fig. 3c). When the conserved G1 or G2 box for ATP binding in the kinase domain was mutated in MBP-G1(G588A, G590A) or MBP-G2(G618A, G620A), the kinase activity was disrupted (Fig. 3c). However, the two mutated proteins can be phosphorylated by the normal kinase domain GST-KD (Fig. 3c), suggesting that MHZ1 phosphorylation can occur in trans manner.
Next, we investigated whether MHZ1 can transfer phosphoryl groups to putative downstream HPt and B-type RR components in rice. Rice has five HPts (OsAHP1/2, OsPHP1/2/3) and more B-type RRs47,48. Results showed that GST-MHZ1 can transfer phosphoryl groups to OsAHP1 and OsAHP2 instead of OsPHP1 or OsPHP2 (Fig. 3d), possibly implying substrate specificity. The OsPHP3 was not tested because OsPHP3 is a more diverged pseudogene47. H79Q and H80Q mutations in OsAHP1 and OsAHP2, respectively, disrupted their phosphorylation by GST-MHZ1 (Fig. 3e), suggesting that these residues are likely to be the phosphoryl-accepting sites.
Among the rice B-type response regulators, we selected the originally identified six (OsRR21 to OsRR26)47 to express in E. coli. Only OsRR21 and OsRR26 were successfully expressed, and the OsRR21 but not OsRR26 can accept the phosphoryl group transferred from the phosphorylated OsAHP1 or OsAHP2 (Fig. 3f, g), suggesting substrate specificity. D68E mutation of OsRR21 abolished its phosphorylation (Fig. 3f, right panel), suggesting that D68 may be the phosphoryl-accepting site. D824A mutation in the receiver domain of GST-MHZ1 disabled the phosphorelay from MHZ1 to OsAHP1 or OsAHP2 (Fig. 3g, the left two lanes), indicating that the D824 residue of MHZ1 is indispensable for phosphotransfer from GST-MHZ1 to OsAHP1 or OsAHP2. All these results support that MHZ1 can autophosphorylate and transfer the phosphoryl group via its receiver domain to OsAHP1/OsAHP2 and then further to OsRR21 through phosphorelay.
Time-course analysis of the phosphorelay between GST-MHZ1, OsAHP1/2 and OsRR21 was performed to verify the MHZ1-mediated phosphorelay system. GST-MHZ1 was autophosphorylated first in the presence of [γ-32P]ATP before being incubated with OsAHP1. Over the time course, GST-MHZ1 autophosphorylation level was gradually decreased whereas the OsAHP1 phosphorylation was steadily enhanced (Fig. 3h, left panel), suggesting an active phosphotransfer from GST-MHZ1 to OsAHP1. After phosphotransfer from GST-MHZ1 to OsAHP1, the OsRR21 was further added to the assay system and its phosphorylation level was also increased (Fig. 3h, right panel). By contrast, as negative controls, GST-MHZ1 plus OsRR21, or OsAHP1 plus OsRR21 did not result in OsRR21 phosphorylation (Fig. 3h, right panel). Similar phosphotransfer also happened from GST-MHZ1 to OsAHP2 and further to OsRR21 (Supplementary Fig. 6b). Altogether, all these data indicate that the activated GST-MHZ1 could transfer its phosphoryl group to OsAHP1/OsAHP2 and further to the downstream RRs, e.g., OsRR21.
Ethylene signaling requires MHZ1-mediated phosphorelay
We further analyzed whether MHZ1 kinase activity and it-mediated phosphorelay is essential for ethylene response in rice roots. The coding region of MHZ1 gene tagged with FLAG and driven by MHZ1 native promoter, was mutated in the G1 box (G588A, G590A), the H375 (H375Q) or the D824 (D824A) sites and transformed into mhz1-1. No ethylene response was observed in the roots of homozygous transgenic lines harboring the mutated MHZ1 genes, although the MHZ1-FLAG protein was detected in each homozygous line (Fig. 3i). As a positive control, the roots of homozygous ProMHZ1:MHZ1-FLAG transgenic line in mhz1-1 background displayed normal ethylene response (Fig. 3i). These results indicate that MHZ1 kinase activity, the conserved H375 phosphorylation site and D824 phosphoryl-accepting site are all required for its function in the regulation of root ethylene response in rice.
Since OsAHP1/2 and OsRR21 accept phosphoryl groups from MHZ1, we tested whether they are involved in ethylene signaling. Mutants of OsAHP1, OsAHP2 and OsRR21 were generated through CRISPR/Cas9 and their ethylene responses were examined (Fig. 3j, k and Supplementary Fig. 7a, b, c, d). Results showed that while Osahp1 and Osahp2 single mutants had normal ethylene responses (Supplementary Fig. 7a), Osahp1 Osahp2 double-mutant, which was segregated from the self-bred progenies of the Osahp1 (heterozygous)/Osahp2 (homozygous) plant, exhibited ethylene-insensitive root growth (Fig. 3j and Supplementary Fig. 7c), suggesting that OsAHP1 and OsAHP2 may play redundant roles in ethylene signal transduction. Transgenic plants overexpressing OsRR21 exhibited shorter roots compared with WT both in air and in ethylene (Fig. 3k and Supplementary Fig. 7b), and expression of ethylene-responsive genes are enhanced in these overexpression lines compared with WT (Supplementary Fig. 7e), suggesting that OsRR21 positively modulates ethylene response. Meanwhile, two mutant lines of OsRR21 exhibited normal ethylene responses (Fig. 3k and Supplementary Fig. 7b, d), indicating that response regulators may play redundant roles in regulating ethylene response. In WT protoplast, OsRR21 enhanced OsRAP2.8 promoter activity and this enhancement is abolished in the protoplast of mhz1, suggesting that OsRR21 function requires signal from MHZ1 (Supplementary Fig. 7f). Ethylene induction of several other response regulator genes suggests that these additional genes may also contribute to regulation of ethylene response (Supplementary Fig. 7g). Altogether, these results indicate that the MHZ1-AHP1/2-OsRR21 phosphorelay pathway is required for ethylene response in rice roots.
MHZ1 genetically acts at OsERS2
Genetic analyses were performed to study how MHZ1 interacts with the canonical ethylene signaling pathway. Double-mutant analysis showed that the ethylene hypersensitivity in the roots of Osers2 and Osetr2 ethylene receptor loss-of-function mutants was abolished by mhz1 mutation, suggesting that MHZ1 is required for the ethylene-response phenotype of the receptor mutants.
Next, we used a dominant OsERS2 gain-of-function mutant Osers2d to test its genetic interaction with MHZ1. Osers2d was identified as mhz12 from our ethyl methanesulphonate (EMS)-mutagenized population and harbors a dominant mutation (A32V) at the transmembrane domain of OsERS2, which is equivalent to Arabidopsis etr1-349 (Supplementary Fig. 8). The mutant showed ethylene-insensitive phenotype (Fig. 4b and Supplementary Fig. 8). The Osers2d mutation was introduced into MHZ1 overexpression background by crossing and by transgenic approach, and this introduction resulted in complete masking of the constitutive and ethylene-induced short root phenotype of MHZ1-OE plants, without altering the abundance of MHZ1 protein (Fig. 4b, c). These results indicate that gain-of-function mutation of Osers2d suppressed MHZ1 function in root ethylene response. Combining with the fact that mhz1 suppressed the short root phenotype of Osers2, we propose that MHZ1 may genetically function at the OsERS2 level or they may form a complex.
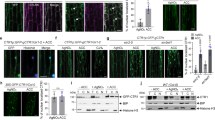
a mhz1 suppressed the ethylene hypersensitivity of Osetr2 (Dongjin) and Osers2 (Dongjin) loss-of-function mutants. (Left) Seedlings were treated with air or 1 ppm ethylene. (Right) Data are means ± SD, n > 30. **P < 0.01; Student’s t-test. DJ, Dongjin; Nip, Nipponbare. Bars indicate 10 mm. b Ethylene response of Osers2d/MHZ1-OE lines. Etiolated seedlings were treated with air or 10 ppm ethylene. Bars indicate 10 mm. c Root length quantification of each line in b. Total proteins of each line were immunoblotted for MHZ1-FLAG with anti-FLAG antibody. Data are means ± SD, n > 30. **P < 0.01; Student’s t-test. d Schematic structures of MHZ1 and OsERS2 and their truncated versions. e Split-ubiquitin Y2H assay for interaction of MHZ1 and OsERS2. Combination of pTSU2-APP and pNubG-Fe65 (provided in the kit) was used as a positive control. f Pull-down of MBP-MHZ1 with GST-OsERS2ΔTM in E. coli. g Co-IP assays for interaction of MHZ1 and OsERS2. The constructs were cotransformed into rice protoplasts. Total proteins were immunoprecipitated with GFP-Trap and immunoblotted with anti-GFP, anti-FLAG, and anti-UGPase antibodies. h OsERS2 facilitates the ER membrane localization of MHZ1. MHZ1-GFP and OsERS2-mCherry proteins were transiently expressed in tobacco leaf epidermal cells. Bars indicate 20 μm. i Co-IP assays for interaction domain mapping of MHZ1 and OsERS2. The constructs were cotransformed into rice protoplasts. Total proteins were immunoprecipitated with GFP-Trap and immunoblotted with anti-FLAG, anti-GFP antibodies. j OsERS2 inhibits MHZ1 kinase activity. GST was added as a control. Values at the bottom indicate relative phosphorylation levels of GST-MHZ1. k OsERS2 inhibited phosphate groups relayed to OsAHP1. Values at the bottom indicate relative phosphorylation levels of GST-AHP1. l GAF domain of OsERS2 inhibits MHZ1 kinase activity. GST was added as a control. Values at the bottom indicate relative phosphorylation levels of GST-MHZ1. m Histidine phosphorylation of MHZ1 is suppressed by OsERS2 and Osers2d. Vectors carrying OsERS2-myc and Osers2d-myc were transformed into protoplasts of MHZ1OE 4-4. Total protein was immunoprecipitated with anti-FLAG affinity gel and immunoblotted with anti-FLAG or anti-1-p-His (Millipore, MABS1330) antibodies. Values at the bottom indicate relative phosphorylation levels of MHZ1. Protoplasts of ProMHZ1:MHZ1(H375Q) transgenic line transformed with the control vector were used as negative control. Source data are provided as a Source Data file.
OsERS2 interacts with MHZ1 to inhibit its kinase activity
Genetically, we demonstrated that MHZ1 and OsERS2 function at the same level. Given that they are all HKs in structure (Fig. 4b, c, d), we tested the possibility of protein–protein interaction between MHZ1 and OsERS2. Membrane-based yeast two-hybrid assay showed that yeast cells co-expressing OsERS2-Cub and NubG-MHZ1 were able to grow well on the selective media, suggesting that MHZ1 interacts with OsERS2 in yeast cells (Fig. 4e). GST pull-down assay and coimmunoprecipitation (Co-IP) assay in rice protoplasts further reveals that OsERS2 can interact with MHZ1 both in vitro and in rice cells (Fig. 4f, g). Since MHZ1 lacked the transmembrane domain and is predicted to be a cytoplasmic protein, we examined whether its interaction with OsERS2 could help it localize to the ER membrane through the membrane recruitment assay (MeRA)50. MHZ1-GFP and OsERS2-mCherry proteins were transiently expressed in tobacco leaf epidermal cells and fluorescence was examined. When expressed separately, OsERS2-mCherry was mainly detected in a reticular network-like structure, while MHZ1-GFP was mainly detected in the cytoplasm. When expressed together, MHZ1-GFP was found to co-localize with OsERS2-mCherry, suggesting that OsERS2 facilitated the ER membrane localization of MHZ1 (Fig. 4h). In addition, protein fractionation assay shows that quite amounts of MHZ1 protein were detected in the membrane fractions especially in the presence of gain-of-function Osers2d, further supporting the association of MHZ1 with membrane-bound OsERS2 (Supplementary Fig. 9a). Consistently, 1-MCP treatment caused abundance of MHZ1 in membrane fraction, whereas ethylene treatment led to decrease of MHZ1 in membrane fraction (Supplementary Fig. 9b). Furthermore, Osers2d appeared to have stronger interaction with MHZ1 than wild-type OsERS2 in yeast two-hybrid assay (Supplementary Fig. 9c). To examine which domain of OsERS2 mediates the interaction with MHZ1, we generated truncated versions of OsERS2 (Fig. 4d). Co-IP assay shows that the GAF domain, but not the kinase domain (KD) of OsERS2, is actually responsible for the interaction of OsERS2 with MHZ1 (Fig. 4i).
Next, we examined the effects of this interaction on MHZ1 activity. In the phosphorylation assay, addition of increasing amount of OsERS2 (GST-ERS2ΔTM) drastically reduced the MHZ1 histidine kinase activity, whereas inclusion of GST itself did not significantly affect this activity (Fig. 4j), indicating that OsERS2 can inhibit the autophosphorylation of MHZ1 in vitro. Since autophosphorylated MHZ1 can transfer its phosphoryl group to OsAHPs (Fig. 3), we investigated whether OsERS2 could affect this process. Compared with GST, addition of the OsERS2 apparently reduced the OsAHP1 phosphorylation by MHZ1-mediated phosphorelay (Fig. 4k). The inhibitory effect of OsERS2 on MHZ1 kinase activity and the phosphorelay may not be due to an competitive binding of [γ-32P]ATP, as suggested by our results that OsERS2 only had very limited kinase activity in the presence of Ca2+ (Fig. 4k, right-most panel, Supplementary Fig. 10a). Given that the GAF domain mediates the interaction of OsERS2 with MHZ1, we examined the effect of GAF domain on MHZ1 autophosphorylation. Phosphorylation assay showed that the GAF domain exerted an inhibitory effect on MHZ1 autophosphorylation while the kinase domain of OsERS2 (GST-ERS2KD) did not show a significant effect (Fig. 4l). All these results indicate that OsERS2 can inhibit both MHZ1 autophosphorylation and MHZ1-mediated phosphorelay likely via its GAF domain.
To examine whether OsERS2 inhibits MHZ1 phosphorylation in vivo, we transfected vectors harboring the OsERS2-myc and Osers2d-myc into rice protoplasts isolated from MHZ1OE 4-4 line. With similar MHZ1-FLAG protein levels in each material group (Fig. 4m, middle panel), the histidine phosphorylation level of the MHZ1-FLAG protein is significantly and differentially reduced by expressing OsERS2 or Osers2d compared to vector control as revealed by immunoblot analysis using the anti-1-p-His antibody (Fig. 4m, top panel). While OsERS2 and Osers2d had similar protein levels (Fig. 4m, bottom panel), Osers2d had an apparently stronger inhibitory effect on MHZ1 phosphorylation than wild-type OsERS2 does, probably due to a stronger interaction of Osers2d with MHZ1 compared with wild-type OsERS2 (Supplementary Fig. 9a, b, c). In mhz1:MHZ1(H375Q) plant cells, no signal of MHZ1 histidine phosphorylation was detected. Transfection of MHZ1-FLAG into WT and Osers2d protoplasts further revealed that Osers2d had stronger inhibitory effect on MHZ1 histidine phosphorylation than the control (Supplementary Fig. 9d). All these results clearly demonstrate that OsERS2 and Osers2d inhibit MHZ1 histidine phosphorylation in rice cells.
In addition to OsERS2, other ethylene receptors such as OsERS1 and OsETR2 also displayed mild interaction with MHZ1 and moderate inhibitory effect on MHZ1 autophosphorylation (Supplementary Fig. 10b, c, d), suggesting similar roles of ethylene receptors in modulating MHZ1 kinase activity.
Genetic interaction of MHZ1 with OsEIN2
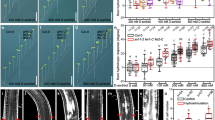
We further examined the genetic relation of MHZ1 with OsEIN2. Rice Osein2 mutant is insensitive to ethylene in both root and coleoptile growth, and overexpression of OsEIN2 in WT seedlings resulted in strong constitutive and enhanced ethylene responses31. We generate the mhz1/OsEIN2-OE plants by crossing. OsEIN2-OE partially suppressed the ethylene-insensitive root growth of mhz1 in mhz1/OsEIN2-OE seedlings in ethylene (Fig. 5a, b), while the mhz1/OsEIN2-OE seedlings still have longer adventitious roots at the node above the mesocotyl compared to the OsEIN2-OE seedlings, indicating that activated OsEIN2-mediated signaling pathway can partially restore the ethylene response of mhz1 mutant (Fig. 5c).
a Ethylene response of mhz1/OsEIN2-OE in comparison with mhz1 and OsEIN2-OE. Bars indicate 10 mm. b Quantification of root length of the mutants in a. Root lengths are means ± SD, n > 30. **P < 0.01; Student’s t-test. c Enlargement of root features of the mutants treated with 10 ppm ethylene in a. d Ethylene response of Osein2/MHZ1-OE in comparison with Osein2 and MHZ1-OE. Rice seedlings were grown in dark for 2 days with air or 10 ppm ethylene. Bars indicate 10 mm. e Quantification of root length (above) and relative root length (below) of the mutants in d. Data are means ± SD, n > 30. **P < 0.01; Student’s t-test. f Comparison of MHZ1-, OsEIN2-, and OsEIL1-regulated ethylene-response genes. Etiolated seedlings of WT, mhz1, Osein2 and Oseil1 were grown for 2 days before treated with air or 10 ppm ethylene for 3 h. Roots were subjected to RNA-seq analysis with three biological replicates. Ethylene-response genes (ERGs) were identified in WT according to the gene expression levels with at least relative twofold changes (q-value < 0.05) in ethylene treatment compared to those in the air. A total of 1789 ERGs were identified. g A proposed working model for MHZ1-mediated ethylene signaling in rice. Rice has the conserved components of ethylene signaling as those in Arabidopsis, including ethylene receptors, OsCTR2, OsEIN2, and OsEIL1. Besides the conserved receptor-CTR2-OsEIN2-OsEIL1 signaling pathway, our results suggests a novel MHZ1-AHP1/2-OsRR21 phosphorelay pathway, through which ethylene receptors could regulate the root growth of rice by suppressing the kinase activity of MHZ1. In the absence of ethylene, ethylene receptors are in active conformations, which may facilitate their interaction with MHZ1 and suppress MHZ1 activity. With ethylene, ethylene receptors possibly released the repression effect on MHZ1 and the phosphorelay pathway is activated. The MHZ1-mediated phosphorelay pathway and the OsEIN2-mediated pathway may work together to regulate a subset of downstream gene to modulate root growth. MHZ1 and OsRRs can also be transcriptionally induced by ethylene through the OsEIN2-mediated pathway, which may facilitate the maintenance of MHZ1-mediated signaling after activation. The orange arrow indicates transcriptional activation. In addition, H2O2 may also function through MHZ1 to participate in the ethylene-regulated root growth. Source data are provided as a Source Data file.
We further crossed MHZ1-OE with Osein2 to generate Osein2/MHZ1-OE plants (Fig. 5d). In ethylene, the roots of Osein2/MHZ1-OE seedlings are longer than that of MHZ1-OE seedlings, but are still significantly shorter compared with its air control (Fig. 5d, e). The results indicate that Osein2 mutation cannot completely block the enhanced ethylene response conferred by MHZ1 overexpression, implying that MHZ1 may have the ability to accept signal from upstream components, e.g., ethylene receptors, independent of OsEIN2 function.
To further elucidate the relationship between MHZ1- and OsEIN2-mediated pathways, we compared the downstream ethylene-response genes (ERGs) regulated by MHZ1, OsEIN2, or OsEIL1 in rice roots through transcriptome analysis. RNA-seq data suggested that 85% (719) of MHZ1-dependent ERGs were also regulated by OsEIN2 and OsEIL1 (Fig. 5f and Supplementary Data 1). This is consistent with the fact that MHZ1 is transcriptionally downstream of OsEIN2 and OsEIL1 (Fig. 2). As MHZ1-dependent ERGs only account for half of OsEIN2-dependent ERGs (Fig. 5f and Supplementary Data 1), we further compared the ethylene responsiveness of mhz1 and Osein2 by checking the expression of several ERGs, including OsRRA5, OsRAP2.8, OsERF002, OsERF063, and OsERF073. Whereas the ethylene induction of all five genes were abolished or hampered in Osein2, only OsRRA5, OsRAP2.8, and OsERF002 expression was affected by mhz1 (Supplementary Fig. 11a, b), suggesting that the ethylene responsiveness of mhz1 and Osein2 is differential in terms of gene expression. These results suggest that MHZ1-mediated pathway shares a subset of ERGs with the OsEIN2 signaling pathway for regulating root growth in rice, although genetically MHZ1 is partially independent of OsEIN2. A GO analysis for the subset of MHZ1-dependent ERGs in comparison to total ERGs was performed using BiNGO51. Results showed that compared with total ERGs, MHZ1-dependent ERGs are mainly enriched in auxin signaling pathway and responses to different stimuli (Supplementary Fig. 11c). This is in line with former findings that ethylene functions upstream of auxin signaling to regulate root growth52,53,54, indicating that MHZ1 may be involved in the crosstalk between ethylene, auxin and different stimuli to regulate root growth.
During an effort to screen for the suppressor of OsEIN2-OE plants, three suppressors lines (SOE-7407, -410, -9744) were found to be very similar to the mhz1/OsEIN2-OE seedlings after ethylene treatment (Supplementary Fig. 12). MHZ1 was identified to harbor the mutation sites in these suppressors by sequencing (Supplementary Fig. 12). These findings further support the genetic relationship between MHZ1 and OsEIN2.
Discussion
Through mutant analysis, we identified MHZ1 as a positive modulator of root ethylene response in rice. MHZ1 has autophosphorylation ability and can transfer its phosphoryl group to OsRR21 via OsAHP1 and OsAHP2. Ethylene receptor OsERS2 physically interacts with MHZ1 to inhibit MHZ1 autophosphorylation and signaling. We propose that in the absence of ethylene, the ethylene receptors are in active conformations, which facilitates their interaction with MHZ1 and MHZ1 kinase activity is suppressed. Upon ethylene perception, the receptors may release their inhibition on MHZ1, triggering MHZ1-mediated phosphorelay for regulation of root growth. This pathway may be a branch in parallel with the OsEIN2-mediated one, and both act downstream of ethylene receptor signaling to regulate root growth in rice (Fig. 5g).
Our study reveals a previously unidentified mechanism, by which ethylene receptor OsERS2 binds to the histidine kinase MHZ1 and suppressed its autophosphorylation and phosphorelay, adding knowledge toward how the ethylene receptors transmit signals. This conclusion may be in line with those from bacteria studies. In Pseudomonas aeruginosa, virulence is partially controlled by the response regulator protein GacA, which receives phosphoryl group from upstream histidine kinase GacS. The kinase activity of GacS is proved to be inhibited by another histidine kinase RetS through direct binding, and the binding did not require the conserved phosphorylation site in RetS55,56,57. The interaction of the two histidine kinases facilitates the bacteria to integrate environmental signals to control complex adaptive processes. Plant ethylene receptors are structurally related histidine kinase-like proteins. Although OsERS2 (Supplementary Fig. 10a) and OsETR2 showed kinase activity29, it is not clear if these kinase activities would have any roles in inhibition of MHZ1 signaling. Actually, the GAF domain in OsERS2 is sufficient for mediating the interaction with MHZ1 and plays a major role in inhibition of MHZ1 kinase activity (Fig. 4). GAF domain has been found in all ethylene receptors and the function has not been identified so far. Our study discloses its function in connecting downstream histidine kinase MHZ1 during ethylene signaling in rice roots. Previously, the N-terminal part (residues 1–349) of ETR1 containing the GAF domain has been found to largely rescue the ctr1-1 and ctr1-2 mutant phenotypes, possibly suggesting an alternative pathway bypassing CTR1 in Arabidopsis58. A recent research also suggested that ETR1 is involved in the multistep phosphorelay pathway by interacting with AHP proteins through its RD domain59. Whether the Arabidopsis AHK543, a homolog of MHZ1, is involved in the above pathway needs to be further investigated. Ethylene transcriptionally induces the expression of ethylene receptor genes ERS1, ERS2, and ETR2 in Arabidopsis11 and OsETR2 in rice (Supplementary Data 1). It is possible that ethylene-induced receptor gene expression may function as a desensitizing approach at later stage for ethylene receptor to re-lock MHZ1 after the initial biochemical triggering of the signaling and completeness of ethylene response.
Downstream of ethylene receptors, two branch pathways are proposed (Fig. 5g). Apparently, the conserved OsCTRs-OsEIN2-OsEIL1 pathway should play major roles in both roots and aerial parts, whereas the MHZ1-OsAHP1/2-OsRR21 pathway may specifically play roles in rice roots. Ethylene-induced OsEIN2 accumulation is not affected in mhz1 or mhz1/OsEIN2-OE plants, further supporting a separate role of the MHZ1 in root ethylene response (Supplementary Fig. 13). Transcriptionally, MHZ1 and OsRR21 can be induced by the conserved OsEIN2-OsEIL1 pathway and this feature may facilitate maintenance of MHZ1-mediated signaling after activation (Fig. 2 and Supplementary Fig. 7g). In our RNA-seq analysis, 719 ERGs were identified to be shared by MHZ1, OsEIN2 and OsEIL1 (Fig. 5f and Supplementary Data 1). It is proposed that the two pathways may work together to modulate the expression of these ERGs, thus regulating root growth. Interruption of either pathway would abolish the ethylene induction of these ERGs, causing an ethylene-insensitive phenotype of rice roots. On the other hand, when either pathway is interrupted, constitutive activation of the other pathway could partially restore the induction of downstream ERGs.
Two-component systems also participate in cytokinin signaling in Arabidopsis and similar mechanism may exist in rice38,47,60,61,62,63. Considering that rice may have only two functional HPts (OsAHP1 and OsAHP2) for signaling47,48, it is hence possible that these two genes play roles in both ethylene signaling and cytokinin signaling in specific manners depending on distinct treatments, times, cell types, tissues and/or organs. Actually, only limited number of homozygous Osahp1 Osahp2 seedlings were identified from 168 self-bred progenies of the Osahp1 (heterozygous)/Osahp2 (homozygous) plant, implying that the Osahp1 Osahp2 double-mutant may have some degree of defect in embryo development, which may be caused by a defect in cytokinin signaling. Other possibilities cannot be excluded. Similar sharing of the histidine-containing phosphotransmitter has been reported in filamentous fungi Aspergillus nidulans64.
Arabidopsis has a MHZ1 homolog AHK5. Mutation of the gene caused mildly enhanced ethylene response in Arabidopsis ahk5 roots43. This response is in contrast with the complete ethylene-insensitive response in the present rice mhz1 root, suggesting that rice may have evolved to adopt the MHZ1 pathway in a different mechanism to strongly control ethylene-regulated root growth for adaptation in water environment. ahk5 is also sensitive to ABA, and our present mhz1 is slightly insensitive to ABA (Supplementary Fig. 14), suggesting that MHZ1 may also participate in ABA- or other stress-related processes in rice. A maize homolog ZmHK9 has been characterized and the gene is highly expressed in roots and induced by drought and ABA treatment44. Overexpression of the gene in transgenic Arabidopsis plants caused hypersensitivity to ABA and ethephone, and led to drought tolerance through regulation of stomatal density and stomatal closure44. These studies suggest that MHZ1 may also have other functions in addition to its roles in ethylene response.
In the N-terminal end of MHZ1, a PAS domain is noted (Supplementary Fig. 2c). This domain is usually involved in ligand binding and/or protein interaction, suggesting other sensing possibilities in addition to be regulated by ethylene receptors. The possibility that MHZ1 serves as a cytoplasmic co-receptor of ethylene signaling cannot be excluded since both MHZ1 and ethylene receptors are histidine kinases or structurally similar proteins. It should be noted that, Arabidopsis AHK5, several AHPs and response regulators can form signaling network to modulate stomatal closure in response to H2O2 and/or ethylene65,66,67. Root growth of mhz1 is slightly insensitive to H2O2 (Supplementary Fig. 15), suggesting that H2O2 may partially function through MHZ1 to inhibit root growth of rice. Given that ethylene induces H2O2 production in Arabidopsis68, it is possible that H2O2 may also be involved in the proposed pathway (Fig. 5g). This result is consistent with the GO analysis that MHZ1-dependent ERGs are enriched in auxin signaling pathway and also responses to different stimuli. OsHK1/MHZ1 is previously reported to play roles in rice large radius root tip circumnutations through a cytokinin-related pathway45. As ethylene is also demonstrated to stimulate nutations in Arabidopsis in an auxin transport-dependent manner69, it is possible that MHZ1 is also involved in the crosstalk between ethylene and auxin, cytokinin or H2O2 to regulate root growth.
Collectively, we identified the histidine kinase MHZ1 as a regulator of ethylene signaling in rice. Ethylene receptor OsERS2 interacts with MHZ1 through GAF domain and inhibits MHZ1-mediated signaling. The MHZ1-mediated pathway may function as a branch in parallel with the conserved pathway to regulate root growth especially under semi-aquatic environment (Fig. 5g). Our data provide valuable insights into the mechanism of ethylene signaling in rice and should facilitate improvement of stress adaptation and relevant agronomic traits in crops.
Methods
Materials, ethylene treatment, and gene identification
The rice (Oryza sativa L.) mutants mhz1, Osein2/mhz7-1, Oseil1/mhz6, and Osers2d/mhz12 mutants were previously identified by Ma31. OsEIN2-OE lines were generated by Ma31. All the mhz1 alleles are in Nip background, and the Osers2 and Osetr2 mutants are in DJ background. Material propagation and crosses were carried out in the Experimental Station of the Institute of Genetics and Developmental Biology in Beijing from May to October of each year. For ethylene-response assay, seeds were soaked at 37 °C for 2 days and the germinated seeds were placed on stainless net for ethylene treatment at 28 °C in dark for 3 days if not specified, with a water level below the seeds31. Lengths of roots and/or coleoptiles were measured. For ABA treatment, stock solution of ABA was prepared in ethanol and diluted into solutions of different concentrations with water. Equivalent volumes of ethanol were added to the control. The MHZ1 gene was identified by TAIL-PCR method. To generate MHZ1-OE lines, MHZ1 CDS driven by the native promoter (3 kb sequence upstream of ATG) was transformed into WT rice and homozygous transgenic lines with higher MHZ1 expression levels were analyzed. The ethylene receptor loss-of-function mutants Osers2 and Osetr2 were purchased and identified through PCR with primers suggested (http://cbi.khu.ac.kr/RISD_DB.html) (Supplementary Table 1). The mhz1-1 was used as male parent and crossed with Osers2, Osetr2, Osein2, and OsEIN2-OE transgenic lines to generate double mutants for genetic interaction analysis. The Osers2d/MHZ1OE 4-4 line was derived from crossing Osers2d with MHZ1OE 4-4 and the Osers2d/MHZ1OE 3-5 line was generated by overexpressing MHZ1 in Osers2d mutant through transgenic approach. To generate the MHZ1-OE/Osein2 double-mutant, MHZ1-OE transgenic line was used as male parent and crossed with Osein2. F2 populations were used for genotyping and F3 or F4 populations were phenotypically and/or genotypically analyzed. The mhz1-5 mutant with Tos17 insertion was requested from the rice mutant database in Japan (https://pc7080.abr.affrc.go.jp/~miyao/pub/tos17/index.html.en). The Osahp1, Osahp2 and Osrr21 single mutants were generated through an CRISPR/Cas9 approach. SG sequences are as follows: OsAHP1, GTTGAGCTGGCTGGTCAGCG; OsAHP2, GATCTCGTTGATGATCCTGT; OsRR21, GGGACAGATATCGTTATGAA. To generate the Osahp1 Osahp2 double-mutant, Nip rice was transformed with vectors carrying two SG sequences targeting OsAHP1 and OsAHP2. After sequencing the genomic sequences of the two genes in 24 transgenic T0 lines, no Osahp1 Osahp2 double-mutant was identified. An Osahp1 (heterozygous)/Osahp2 (homozygous) plant was self-crossed and the self-bred progenies (168 seeds) were germinated and grown under 10 ppm ethylene. After phenotype observation, seedlings were numbered and OsAHP1 and OsAHP2 genomic sequences were sequenced. Homozygous Osahp1 Osahp2 plants were identified.
Gene expression analysis by real-time PCR
Two or three-day-old etiolated rice seedlings were treated with air or ethylene before roots and shoots were harvested for RNA extraction. Total RNA was extracted using TRIZOL reagent (Invitrogen). The complementary DNAs (cDNAs) were synthesized using cDNA Synthesis Kit (M-MLV Version) (TaKaRa) and then subjected to real-time PCR. Real-time PCR was conducted according to the instructions of TransStart Green qPCR SuperMix (TransGen Biotech, China). OsActin2 was used for internal control. The primers are listed in Supplementary Table 1. The experiments were repeated independently for at least three times and the results were consistent. One set of results were shown.
GUS Staining
Tissues and organs were fixed in 90% acetone on ice for 15 min. After washing with staining buffer (100 mM Na3PO4 buffer pH 7.0, 10 mM EDTA, 5 mM potassium ferricyanide, 5 mM potassium ferrocyanide, 0.1% Triton X-100), the samples were soaked in staining solution (staining buffer containing 0.5 mg/mL X-Gluc (Sigma, B8049) for 10 min in a vacuum system. The samples were incubated at 37 °C in the dark. Green tissues were decolorized with 70% ethanol. The samples were observed using stereo microscopy (Leica, M165 FC).
Proteins expression and phosphorylation assay
The cDNA fragments encoding various protein versions were fused with GST in pGEX-6p-1 vector or maltose-binding protein (MBP) gene in pMAL-2C vector and proteins were expressed in BL21(DE3) pLysS. GST-MHZ1(365–968 aa) containing kinase domain and receiver domain, and its mutant versions, GST-KD containing only the kinase domain (365–655 aa), and MBP-KD or its mutant versions were all expressed. Full-length MHZ1(1–968 aa) was not successfully expressed. Four OsHPts were expressed except OsPHP3 because the OsPHP3 may be a pseudogene. The full-length OsPHP1 (Os01g54050), OsAHP1 (Os08g44350), OsAHP2 (Os09g39400) and OsPHP2 (Os05g09410) gene were fused with GST and similarly expressed. The fragments encoding the receiver domain of OsRR21 (1–129 aa, Os03g12350) or OsRR26 (1–123 aa, Os01g67770) were fused with MBP and expressed. Different truncated versions of ethylene receptors (ERS2ΔTM, ERS2GAF, ERS2HK, ERS1ΔTM, ETR2GAF + KD) were fused with GST and expressed. All genes encoding the mutant protein versions were generated by site-directed mutagenesis through overlapping extension PCR. The transfected E. coli cells were cultured at 25 °C and proteins were induced by addition of 0.8 mM IPTG. The proteins were purified with Glutathione sepharose for GST fusions or with Amylose Resin for MBP fusions.
Purified proteins were used for protein kinase assay according to our previous protocol29. Around 1–5 μg purified MHZ1 proteins, 5 μg purified OsHPts and/or OsRRs were used for autophosphorylation assay and/or phosphorelay analysis. Reactions were started by adding [γ-32P]ATP, incubated at 25 °C for 45 min and then terminated by the addition of 5 × Loading buffer. Cation dependence of GST-MHZ1 kinase activity was tested at a physiological level of ATP (0.5 mM)40. To test the inhibition effect of ethylene receptors on MHZ1 kinase activity, 0.5–4 μg truncated OsERS2 or other ethylene receptor proteins were added to the reaction systems before adding [γ-32P]ATP. Samples were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) using 10% polyacrylamide gels. After electrophoresis, the gel was dried for 4 h at 80 °C by the gel dryer and then the dry gel was exposed to X-ray films for autoradiography.
Rice transformation and phenotype analysis
The vector pCambia2300 was used for construction of complementation vector and overexpression vector. For complementation, the vector contained the MHZ1 genomic sequence (5.623 kb), the 4.414 kb sequence upstream of MHZ1 ATG (promoter) and the 1 kb sequence downstream of MHZ1 TGA. This full-length sequence (11.037 kb) was cloned by adding three separated regions: 1–2249 bp region with Sse8387 I and BamH I sites, the middle region (2249-8668 bp) with BamH I and EcoR V, and the last region with EcoR V and Sal I. For overexpression, the MHZ1-coding sequence fused with sequence encoding FLAG tag was inserted into the pCambia2300 and the gene was driven by the MHZ1 native promoter (3 kb). The 3 kb MHZ1 native promoter was also fused with GUS gene in pCambia2300 for rice transformation and examination of promoter activity. Mutations of the G1 box (G588A, G590A) in the kinase domain of MHZ1, conserved His-containing site (H375Q) and conserved Asp (D824A) in receiver domain were generated in MHZ1 cDNA by site-directed mutagenesis using overlapping extension PCR. To construct the OsRR21 overexpression vector, open reading frame of the gene with restriction sites was amplified by PCR and cloned into cloning vector pEASY-Blunt (TRANSGENE BIOTECH), and subsequently cloned into binary vector pCambia2300-35S-OCS at the sites of BamH I/Sal I. Plasmids were transfected into agrobacterium EHA105 and further subjected to rice transformation following previous protocol29.
OsEIL1 binding and activation of the MHZ1 promoter
GST-N-terminal OsEIL1 (amino acids 1–350) recombinant protein was expressed and purified according to our previous description34. Single-stranded complementary oligonucleotide fragments harboring the EBS elements (222 bp and 452 bp upstream of MHZ1 start codon) were synthesized (Invitrogen) and labeled by biotin using the Biotin 3′-end DNA-labeling Kit (Thermo Fisher Scientific). Following assay was carried out according to manufacturer’s protocol (LightShift Chemiluminescent EMSA Kit; Thermo Fisher Scientific).
For testing of the transactivation of the MHZ1 promoter activity in tobacco leaves, the open reading frame of OsEIL1 was cloned into pCambia2300-35S-OCS to generate Pro35S:OsEIL1 vector. MHZ1 promoter (2.16 kb DNA sequence upstream of ATG) was cloned to drive luciferase (LUC) gene expression. A combination of vectors containing Pro35S:OsEIL1 and ProMHZ1:LUC were cotransformed into A. tumefaciens stain EHA105 and subsequently infiltrated into young leaves of Nicotiana tabacum. Plants were grown for 2.5 days with 16 h of light/8 h of dark at 24 °C before charge-coupled device (CCD) imaging. LUC activity was observed with a low-light cooled CCD imaging apparatus (iXon; Andor Technology). A combination of vector control and vector containing ProMHZ1:LUC was used as negative control.
Membrane-based Y2H assay
Coding sequence of OsERS2 was cloned into the bait vector pBT3-STE (OsERS2-Cub) and MHZ1 into the prey vector pPR3-N (NubG-MHZ1) from the DUAL membrane starter kit SUC (Dualsystem Biotech). The bait and prey constructs were then cotransformed into the yeast strain NMY51. Positive transformants were selected on SD-Trp-Leu medium, and protein–protein interactions were detected on SD-Trp-Leu-His-Ade medium. Combination of pTSU2-APP and pNubG-Fe65 (provided in the kit) was used as a positive control. Combinations of NubG-MHZ1 and pTSU2-APP, OsERS2-Cub and pNubG-Fe65 were used as negative controls.
Pull-down of MBP-MHZ1 with GST-ERS2ΔTM
To carry out the GST pull-down assay, the coding sequences of MHZ1 and OsERS2ΔTM were cloned into the pMAL-c2 and pGEX-6P-1 plasmids, respectively, to make MBP-MHZ1 and GST-ERS2ΔTM constructs. The constructs were then transformed into BL21(DE3). The transfected E. coli cells were cultured at 25 °C and proteins were induced by addition of 0.2 mM IPTG. After sonication and centrifugation, lysate supernatants containing GST-ERS2ΔTM recombinant protein were incubated with Glutathione sepharoses at 4 °C for 2 h. The sepharoses were washed with phosphate-buffered saline (PBS; pH 7.3) for three times before being incubated with supernatants containing MBP-MHZ1 recombinant protein at 4 °C for 2 h. The sepharoses were washed with PBS (pH 7.3) for three times. Harvested beads were boiled with 2 × SDS loading buffer before running the SDS-PAGE and immunoblotted with anti-GST, anti-MBP antibodies.
Co-IP assays
For coimmunoprecipitation of MHZ1 with ERS2ΔTM, constructs containing MHZ1-FLAG and ERS2ΔTM-GFP were cotransformed into rice protoplasts. Total proteins were extracted by homogenizing the protoplasts in 0.5 mL IP buffer (50 mM HEPES pH 7.5, 150 mM NaCl, 0.5 mM EDTA, 0.5% NP-40, 50 μM MG132, 2% (v/v) protease inhibitor cocktail) and incubating the samples on ice for 15 min. The samples were centrifuged at 12,000 rpm for 10 min at 4 °C before the supernatants were incubated with 25 μL of GFP-Trap beads for 2 h at 4 °C. After being washed for three times with wash buffer (10 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.5 mM EDTA), the beads were collected and resuspended with 50 μL 2 × SDS-PAGE loading buffer and heated at 95 °C for 5 min. The eluted immunoprecipitates were immunoblotted with anti-GFP, anti-FLAG, and anti-UGPase antibodies. For interactions of MHZ1 with OsERS1 and OsETR2, constructs containing MHZ1-FLAG and OsERS1ΔTM/OsETR2ΔTM-GFP were cotransformed into rice protoplasts. Total proteins were immunoprecipitated with GFP-Trap and immunoblotted with anti-GFP, anti-FLAG, and anti-Actin antibodies.
For interaction domain mapping of MHZ1 and OsERS2, constructs containing MHZ1-FLAG, ERS2KD/GAF-GFP were cotransformed into rice protoplasts. Total proteins were immunopreciptated with anti-GFP affinity gel and immunoblotted with anti-FLAG, anti-GFP antibodies.
Membrane recruitment assay
To perform the membrane recruitment assay, MHZ1-GFP and OsERS2-mCherry proteins were expressed separately or together in tobacco leaf epidermal cells and fluorescence was examined. The images were taken using a confocal microscopy (Zeiss LSM 710). Excitation/emission wavelengths were set at 488 nm/500-530 nm for GFP and 561 nm/582-639 nm for mCherry.
Histidine phosphorylation state detection
To detect the histidine phosphorylation state of MHZ1 in Osers2d mutant, vector carrying MHZ1-FLAG was transformed into protoplasts of WT and Osers2d. Total proteins were extracted and MHZ-FLAG protein was immunopreciptated with anti-FLAG affinity gel and immunoblotted with anti-FLAG or anti-1-p-His antibodies (Millipore, MABS1330).
Total and membrane protein isolation
To analyze the localization of MHZ1 protein, vector carrying MHZ1-FLAG was transformed into protoplasts of WT and Osers2d mutant. Total and membrane proteins were isolated as described by Ma33. Equal amounts of total protein (T), soluble protein (S), and microsomal membranes (M) were immunoblotted for MHZ1, BIP (ER membrane marker), and UGPase (cytoplasm marker).
Measurement of ethylene production
To test the ethylene production of different mutants, seedlings were grown in 40-mL uncapped vials for 7 days in dark at 28 °C. The vials were then sealed with rubber syringe caps for 17 h. One milliliter of headspace of each vial was measured by using gas chromatography (GC-2014; Shimadzu)35.
Screening for supperssors of OsEIN2 (SOE)
For generation of SOE lines, seeds of OsEIN2 overexpression line OsEIN2OE-44 were soaked in water for 16 h at room temperature. The seeds were then treated with 0.6% EMS (Sigma, M0880) for 8 h at room temperature. The EMS-treated seeds were germinated at 37 °C and grown in the field. M2 generation seeds of the EMS-mutagenized lines were used for mutant screening31.
Statistical analysis
The relative root or coleoptile length of each mutant is analyzed relative to the length in untreated conditions. All of the data were analyzed using a one-way ANOVA (LSD t-test) for the test groups.
RNA-seq analysis
To carry out the RNA-seq analysis, etiolated seedlings of WT, mhz1, Osein2/mhz7 and Oseil1/mhz6 were grown in the dark at 28 °C for 2 days before treated with air or 10 ppm ethylene for 3 h. Roots of WT and different mutants were subjected to RNA-seq analysis with three biological replicates. The clean data was mapped to rice genome by TopHat and analyzed with Cufflinks software. In WT and different mutants, genes with at least twofold changes in ethylene compared with those in air are marked as ethylene-inducible genes [log2(fold change) ≥ 1, q-value < 0.05] or ethylene-repressible genes [log2(fold change) ≤ –1, q-value < 0.05] genes. In WT, ethylene inducible and repressible genes are defined as ethylene-response genes (ERGs). In mhz1, Osein2 and Oseil1 mutants, ERGs that no longer respond to ethylene [q-value ≥ 0.05 or |log2(fold change)| < 1, q-value < 0.05], or exhibit an opposite ethylene response pattern compared with WT (induced by ethylene in WT, repressed by ethylene in mutants or repressed by ethylene in WT, induced by ethylene in WT) were identified as MHZ1-, OsEIN2-, or OsEIL1-dependent ERGs, respectively. The test status of each gene indicates whether it is calculated. Genes with a test status of “NOTEST” indicates that there are not enough alignments for testing.
Locus identifiers
The locus identifiers of genes referred to in this article is as follows: Os06g44410 (MHZ1), Os01g54050 (OsPHP1), Os08g44350 (OsAHP1), Os09g39400 (OsAHP2), Os05g09410 (OsPHP2), Os03g12350 (OsRR21), Os06g08400 (OsRR22), Os02g55320 (OsRR23), Os02g08500 (OsRR24), Os01g67770 (OsRR26), Os06g08340 (OsERF002), Os09g11480 (OsERF063), Os09g11460 (OsERF073), Os11g05740 (OsRAP2.8), Os07g26720 (OsRRA5), Os05g06320 (OsERS2), Os04g08740 (OsETR2), Os07g06130 (OsEIN2), Os03g20790 (OsEIL1).
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The authors declare that all data supporting the findings of this study are available within the manuscript and the Supplementary Files or are available from the corresponding authors upon request. The source data underlying Figs. 1a, 1e, 1f, 2a-c, 3i, 4a-c, 4g-m, 5b, 5e and Supplementary Figs. 4,a, 4,d, 5, 7,a, 7,b, 7,e-g, 8,b, 9,a, 9,b, 10,c, 10,d, 11,a, 11,b, 14 and 15 are provided as a Source Data file. RNA sequence data reported in this paper have been deposited in the NCBI Sequence Read Archive (https://www.ncbi.nlm.nih.gov/sra) under BioProject accession PRJNA597369 and BioSample accessions (SAMN13673841–13673864). Data has also been deposited in the Genome Sequence Archive in BIG Data Center, Beijing Institute of Genomics (BIG), Chinese Academy of Sciences, under accession numbers CRA002197.
References
Bleecker, A. B. & Kende, H. Ethylene: a gaseous signal molecule in plants. Annu. Rev. Cell Dev. Biol. 16, 1–18 (2000).
Ju, C. L. & Chang, C. Advances in ethylene signalling: protein complexes at the endoplasmic reticulum membrane. AoB Plants 2012, pls031 (2012).
Merchante, C., Alonso, J. M. & Stepanova, A. N. Ethylene signaling: simple ligand, complex regulation. Curr. Opin. Plant Biol. 16, 554–560 (2013).
Shakeel, S. N., Wang, X. M., Binder, B. M. & Schaller, G. E. Mechanisms of signal transduction by ethylene: overlapping and non-overlapping signalling roles in a receptor family. AoB Plants 5, plt010 (2013).
Alonso, J. M., Hirayama, T., Roman, G., Nourizadeh, S. & Ecker, J. R. EIN2, a bifunctional transducer of ethylene and stress responses in Arabidopsis. Science 284, 2148–2152 (1999).
Chang, C., Clark, K., Wang, X. & Stewart, R. ‘Two-component’ ethylene signaling in Arabidopsis. Symp. Soc. Exp. Biol. 51, 59–64 (1998).
Chang, C., Kwok, S. F., Bleecker, A. B. & Meyerowitz, E. M. Arabidopsis ethylene-response gene ETR1: similarity of product to two-component regulators. Science 262, 539–544 (1993).
Chang, C. & Stadler, R. Ethylene hormone receptor action in Arabidopsis. Bioessays 23, 619–627 (2001).
Chao, Q. M. et al. Activation of the ethylene gas response pathway in Arabidopsis by the nuclear protein ETHYLENE-INSENSITIVE3 and related proteins. Cell 89, 1133–1144 (1997).
Hua, J. & Meyerowitz, E. M. Ethylene responses are negatively regulated by a receptor gene family in Arabidopsis thaliana. Cell 94, 261–271 (1998).
Hua, J. et al. EIN4 and ERS2 are members of the putative ethylene receptor gene family in Arabidopsis. Plant Cell 10, 1321–1332 (1998).
Kieber, J. J., Rothenberg, M., Roman, G., Feldmann, K. A. & Ecker, J. R. CTR1, a negative regulator of the ethylene response pathway in Arabidopsis, encodes a member of the Raf family of protein kinases. Cell 72, 427–441 (1993).
Sakai, H. et al. ETR2 is an ETR1-like gene involved in ethylene signaling in Arabidopsis. Proc. Natl Acad. Sci. USA 95, 5812–5817 (1998).
Ju, C. L. et al. CTR1 phosphorylates the central regulator EIN2 to control ethylene hormone signaling from the ER membrane to the nucleus in Arabidopsis. Proc. Natl Acad. Sci. USA 109, 19486–19491 (2012).
Qiao, H. et al. Processing and subcellular trafficking of ER-tethered EIN2 control response to ethylene gas. Science 338, 390–393 (2012).
Wen, X. et al. Activation of ethylene signaling is mediated by nuclear translocation of the cleaved EIN2 carboxyl terminus. Cell Res. 22, 1613–1616 (2012).
An, F. Y. et al. Ethylene-induced stabilization of ETHYLENE INSENSITIVE3 and EIN3-LIKE1 is mediated by proteasomal degradation of EIN3 binding F-Box 1 and 2 that requires EIN2 in Arabidopsis. Plant Cell 22, 2384–2401 (2010).
Guo, H. W. & Ecker, J. R. Plant responses to ethylene gas are mediated by SCF (EBF1/EBF2)-dependent proteolysis of EIN3 transcription factor. Cell 115, 667–677 (2003).
Potuschak, T. et al. EIN3-dependent regulation of plant ethylene hormone signaling by two Arabidopsis F box proteins: EBF1 and EBF2. Cell 115, 679–689 (2003).
Qiao, H., Chang, K. N., Yazaki, J. & Ecker, J. R. Interplay between ethylene, ETP1/ETP2 F-box proteins, and degradation of EIN2 triggers ethylene responses in Arabidopsis. Gene Dev. 23, 512–521 (2009).
Li, W. Y. et al. EIN2-directed translational regulation of ethylene signaling in Arabidopsis. Cell 163, 670–683 (2015).
Merchante, C. et al. Gene-specific translation regulation mediated by the hormone-signaling molecule EIN2. Cell 163, 684–697 (2015).
Zhang, F. et al. EIN2-dependent regulation of acetylation of histone H3K14 and non-canonical histone H3K23 in ethylene signalling. Nat. Commun. 7, 13018 (2016).
Zhang, F., Wang, L. K., Ko, E. E., Shao, K. & Qiao, H. Histone deacetylases SRT1 and SRT2 interact with ENAP1 to mediate ethylene-induced transcriptional repression. Plant Cell 30, 153–166 (2018).
Zhang, F. et al. EIN2 mediates direct regulation of histone acetylation in the ethylene response. Proc. Natl Acad. Sci. USA 114, 10274–10279 (2017).
Jun, S. H. et al. OsEIN2 is a positive component in ethylene signaling in rice. Plant Cell Physiol. 45, 281–289 (2004).
Mao, C. Z., Wang, S. M., Jia, Q. J. & Wu, P. OsEIL1, a rice homolog of the Arabidopsis EIN3 regulates the ethylene response as a positive component. Plant Mol. Biol. 61, 141–152 (2006).
Wang, Q., Zhang, W., Yin, Z. M. & Wen, C. K. Rice CONSTITUTIVE TRIPLE-RESPONSE2 is involved in the ethylene-receptor signalling and regulation of various aspects of rice growth and development. J. Exp. Bot. 64, 4863–4875 (2013).
Wuriyanghan, H. et al. The ethylene receptor ETR2 delays floral transition and affects starch accumulation in rice. Plant Cell 21, 1473–1494 (2009).
Zhang, W., Zhou, X. & Wen, C. K. Modulation of ethylene responses by OsRTH1 overexpression reveals the biological significance of ethylene in rice seedling growth and development. J. Exp. Bot. 63, 4151–4164 (2012).
Ma, B. et al. Identification of rice ethylene-response mutants and characterization of MHZ7/OsEIN2 in distinct ethylene response and yield trait regulation. Mol. Plant 6, 1830–1848 (2013).
Ma, B. et al. Ethylene-induced inhibition of root growth requires abscisic acid function in rice (Oryza sativa L.) seedlings. PLoS Genet. 10, e1004701 (2014).
Ma, B. et al. Membrane protein MHZ3 stabilizes OsEIN2 in rice by interacting with its Nramp-dlike domain. Proc. Natl Acad. Sci. USA 115, 2520–2525 (2018).
Yang, C. et al. MAOHUZI6/ETHYLENE INSENSITIVE3-LIKE1 and ETHYLENE INSENSITIVE3-LIKE2 regulate ethylene response of roots and coleoptiles and negatively affect salt tolerance in rice. Plant Physiol. 169, 148–165 (2015).
Yin, C. C. et al. Ethylene responses in rice roots and coleoptiles are differentially regulated by a carotenoid isomerase-mediated abscisic acid pathway. Plant Cell 27, 1061–1081 (2015).
Mizuno, T. Two-component phosphorelay signal transduction systems in plants: from hormone responses to circadian rhythms. Biosci. Biotech. Bioch. 69, 2263–2276 (2005).
Nongpiur, R., Soni, P., Karan, R., Singla-Pareek, S. L. & Pareek, A. Histidine kinases in plants: cross talk between hormone and stress responses. Plant Signal. Behav. 7, 1230–1237 (2012).
Tsai, Y. C. et al. Characterization of genes involved in cytokinin signaling and metabolism from rice. Plant Physiol. 158, 1666–1684 (2012).
Gamble, R. L., Coonfield, M. L. & Schaller, G. E. Histidine kinase activity of the ETR1 ethylene receptor from Arabidopsis. Proc. Natl Acad. Sci. USA 95, 7825–7829 (1998).
Moussatche, P. & Klee, H. J. Autophosphorylation activity of the Arabidopsis ethylene receptor multigene family. J. Biol. Chem. 279, 48734–48741 (2004).
Hall, B. P. et al. Histidine kinase activity of the ethylene receptor ETR1 facilitates the ethylene response in Arabidopsis. Plant Physiol. 159, 682–695 (2012).
Wang, W. Y., Hall, A. E., O’Malley, R. & Bleecker, A. B. Canonical histidine kinase activity of the transmitter domain of the ETR1 ethylene receptor from Arabidopsis is not required for signal transmission. Proc. Natl Acad. Sci. USA 100, 352–357 (2003).
Iwama, A. et al. AHK5 histidine kinase regulates root elongation through an ETR1-dependent abscisic acid and ethylene signaling pathway in Arabidopsis thaliana. Plant Cell Physiol. 48, 375–380 (2007).
Wang, B. et al. A novel histidine kinase gene, ZmHK9, mediate drought tolerance through the regulation of stomatal development in Arabidopsis. Gene 501, 171–179 (2012).
Lehner, K. R. et al. A histidine kinase gene is required for large radius root tip circumnutation and surface exploration in rice. bioRxiv. https://doi.org/10.1101/437012 (2018).
Serek, M., Tamari, G., Sisler, E. C. & Borochov, A. Inhibition of ethylene-induced cellular senescence symptoms by 1-Methylcyclopropene, a new inhibitor of ethylene action. Physiol. Plant. 94, 229–232 (1995).
Pareek, A. et al. Whole-genome analysis of Oryza sativa reveals similar architecture of two-component signaling machinery with Arabidopsis. Plant Physiol. 142, 380–397 (2006).
Schaller, G. E. et al. Nomenclature for two-component signaling elements of rice. Plant Physiol. 143, 555–557 (2007).
Hall, A. E., Chen, Q. H. G., Findell, J. L., Schaller, G. E. & Bleecker, A. B. The relationship between ethylene binding and dominant insensitivity conferred by mutant forms of the ETR1 ethylene receptor. Plant Physiol. 121, 291–299 (1999).
Grefen, C. et al. Subcellular localization and in vivo interactions of the Arabidopsis thaliana ethylene receptor family members. Mol. Plant 1, 308–320 (2008).
Maere, S., Heymans, K. & Kuiper, M. BiNGO: a cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics 21, 3448–3449 (2005).
Ruzicka, K. et al. Ethylene regulates root growth through effects on auxin biosynthesis and transport-dependent auxin distribution. Plant Cell 19, 2197–2212 (2007).
Stepanova, A. N., Yun, J., Likhacheva, A. V. & Alonso, J. M. Multilevel interactions between ethylene and auxin in Arabidopsis roots. Plant Cell 19, 2169–2185 (2007).
Swarup, R. et al. Ethylene upregulates auxin biosynthesis in Arabidopsis seedlings to enhance inhibition of root cell elongation. Plant Cell 19, 2186–2196 (2007).
Chambonnier, G. et al. The hybrid histidine kinase LadS forms a multicomponent signal transduction system with the GacS/GacA two-component system in Pseudomonas aeruginosa. Plos Genet. 12, e1006032 (2016).
Goodman, A. L. et al. Direct interaction between sensor kinase proteins mediates acute and chronic disease phenotypes in a bacterial pathogen. Gene Dev. 23, 249–259 (2009).
Lapouge, K., Schubert, M., Allain, F. H. T. & Haas, D. Gac/Rsm signal transduction pathway of gamma-proteobacteria: from RNA recognition to regulation of social behaviour. Mol. Microbiol. 67, 241–253 (2008).
Qiu, L. P., Xie, F., Yu, J. & Wen, C. K. Arabidopsis RTE1 is essential to ethylene receptor ETR1 amino-terminal signaling independent of CTR1. Plant Physiol. 159, 1263–1276 (2012).
Zdarska, M. et al. ETR1 integrates response to ethylene and cytokinins into a single multistep phosphorelay pathway to control root growth. Mol. Plant. https://doi.org/10.1016/j.molp.2019.1005.1012 (2019).
Hwang, I., Sheen, J. & Muller, B. Cytokinin signaling networks. Annu. Rev. Plant Biol. 63, 353–380 (2012).
Sharan, A., Soni, P., Nongpiur, R. C., Singla-Pareek, S. L. & Pareek, A. Mapping the ‘Two-component system’ network in rice. Sci. Rep. 7, 9287 (2017).
Yamburenko, M. V., Kieber, J. J. & Schaller, G. E. Dynamic patterns of expression for genes regulating cytokinin metabolism and signaling during rice inflorescence development. PLoS ONE 12, e0176060 (2017).
Sun, L. et al. Two rice authentic histidine phosphotransfer proteins, OsAHP1 and OsAHP2, mediate cytokinin signaling and stress responses in rice. Plant Physiol. 165, 335–345 (2014).
Azuma, N. et al. In vitro analysis of His-Asp phosphorelays in Aspergillus nidulans: the first direct biochemical evidence for the existence of His-Asp phosphotransfer systems in filamentous fungi. Biosci. Biotechnol. Biochem. 71, 2493–2502 (2007).
Desikan, R. et al. The histidine kinase AHK5 integrates endogenous and environmental signals in Arabidopsis guard cells. PLoS ONE 3, e2491 (2008).
Mira-Rodado, V. et al. Identification of two-component system elements downstream of AHK5 in the stomatal closure response of Arabidopsis thaliana. Plant Signal. Behav. 7, 1467–1476 (2012).
Pham, J., Liu, J., Bennett, M. H., Mansfield, J. W. & Desikan, R. Arabidopsis histidine kinase 5 regulates salt sensitivity and resistance against bacterial and fungal infection. N. Phytol. 194, 168–180 (2012).
Desikan, R. et al. Ethylene-induced stomatal closure in Arabidopsis occurs via AtrbohF-mediated hydrogen peroxide synthesis. Plant J. 47, 907–916 (2006).
Binder, B. M., O’Malley, R. C., Wang, W. Y., Zutz, T. C. & Bleecker, A. B. Ethylene stimulates nutations that are dependent on the ETR1 receptor. Plant Physiol. 142, 1690–1700 (2006).
Acknowledgements
This work is supported by the National Natural Science Foundation of China (31530004, 31670274, 31600980), the National Key Basic Research Project (2015CB755702), and the State Key Lab of Plant Genomics.
Author information
Authors and Affiliations
Contributions
J.S.Z., H.Z., K.X.D., B.M. and S.Y.C. designed the research; H.Z. and K.X.D. performed most of the research; B.M. isolated the mutants and performed some research. C.C.Y., C.Y. and H.C. helped analyze the ethylene response phenotype of the mutants. J.J.T., Y.H.H. and W.Q.C identified the SOE lines. Z.G.Z. and T.G.L. provided the original population for screening; Y.H. performed RNA-seq analysis; S.J.H. conducted some rice transformation; W.K.Z and X.Y.W contributed to material preparation and data analysis; J.S.Z., K.X.D., H.Z. and B.M. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, H., Duan, KX., Ma, B. et al. Histidine kinase MHZ1/OsHK1 interacts with ethylene receptors to regulate root growth in rice. Nat Commun 11, 518 (2020). https://doi.org/10.1038/s41467-020-14313-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-14313-0
This article is cited by
-
OsRR26, a type-B response regulator, modulates salinity tolerance in rice via phytohormone-mediated ROS accumulation in roots and influencing reproductive development
Planta (2024)
-
Multiplex CRISPR-Cas9 knockout of EIL3, EIL4, and EIN2L advances soybean flowering time and pod set
BMC Plant Biology (2023)
-
A translational regulator MHZ9 modulates ethylene signaling in rice
Nature Communications (2023)
-
The role of histidine kinase signalling in response to salt stress
Plant and Soil (2023)
-
Comparison of anther transcriptomes in response to cold stress at the reproductive stage between susceptible and resistant Japonica rice varieties
BMC Plant Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.