Abstract
As diversified reaction paths exist over practical catalysts towards CO2 hydrogenation, it is highly desiderated to precisely control the reaction path for developing efficient catalysts. Herein, we report that the ensemble of Pt single atoms coordinated with oxygen atoms in MIL-101 (Pt1@MIL) induces distinct reaction path to improve selective hydrogenation of CO2 into methanol. Pt1@MIL achieves the turnover frequency number of 117 h−1 in DMF under 32 bar at 150 °C, which is 5.6 times that of Ptn@MIL. Moreover, the selectivity for methanol is 90.3% over Pt1@MIL, much higher than that (13.3%) over Ptn@MIL with CO as the major product. According to mechanistic studies, CO2 is hydrogenated into HCOO* as the intermediate for Pt1@MIL, whereas COOH* serves as the intermediate for Ptn@MIL. The unique reaction path over Pt1@MIL not only lowers the activation energy for the enhanced catalytic activity, but also contributes to the high selectivity for methanol.
Similar content being viewed by others
Introduction
Hydrogenation of CO2 into fuels and useful chemicals serves as an important process which helps to alleviate the dearth of fossil fuels1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17. According to both experimental and theoretical studies, CO2 hydrogenation involves various reaction paths4,5,6. Even for the first step to activate CO2, CO2 can be decomposed into CO*, transformed into carboxyl intermediate (COOH*), or hydrogenated into formate intermediate (HCOO*)6,7,8,9. Over practical catalysts, diversified reaction paths inevitably coexist, which induces the formation of different products such as CO, methane, formic acid, methanol, higher alcohols, and even gasoline, generally limiting the selectivity for the target product10,11,12,13,14. For instance, Cu/ZnO/Al2O3 catalysts have already been applied to realize gas-phase CO2 hydrogenation into methanol in industry, but suffer from the limited selectivity (<70%) for methanol and stringent reaction conditions (50–100 bar, 200–300 °C)15,16. Catalyzed by Cu/ZnO/Al2O3, CO2 undergoes reverse water-gas shift path (RWGS) to produce CO* via the formation of COOH* species17. Since such catalysts also involve high energy barrier for the transformation of CO*, a large proportion of CO* is directly desorbed from the catalyst surfaces to form gaseous CO, competing over further hydrogenation into methanol17. Recently, constructing highly active interfaces such as Cu/CeOx, Cu/ZnO, and Cu/ZrO2 interfaces has been reported to efficiently lower the energy barrier for the transformation of CO*, which not only increases the selectivity for methanol but also elevates the catalytic activity18,19,20,21,22,23. In addition, another pivotal approach is based on engineering the coordination environment of active metal atoms by alloying24,25. For instance, the fabrication of NiGa catalysts was found to suppress the RWGS path, thereby facilitating the production of methanol26. Co4N nanosheets were reconstructed into Co4NHx during aqueous CO2 hydrogenation into methanol, wherein the amido-hydrogen atoms directly added CO2 to form HCOO*4,. Despite of these achievements, it is still highly desiderated to efficiently control the reaction path for developing highly active and selective catalysts toward CO2 hydrogenation.
Herein, we demonstrate that metal-ligand cooperativity in Pt single atoms encapsulated in MIL-101 (Pt1@MIL) varies the reaction path and improves the selective hydrogenation of CO2 into methanol relative to nanocrystal counterparts (Ptn@MIL). In Pt1@MIL, every Pt single atom and its coordinated O atoms compose an active center. During CO2 hydrogenation, the turnover frequency (TOF) number of Pt1@MIL reaches 117 h−1 in DMF under 32 bar at 150 °C, being 5.6 times as high as that (21 h−1) of Ptn@MIL. Moreover, the selectivity for methanol reaches 90.3% over Pt1@MIL, whereas the major product for Ptn@MIL is CO with the selectivity of 57.5%. The cooperativity between Pt single atoms and their coordinated O atoms in Pt1@MIL enables the dissociation of H2 to form O–H groups. The hydroxy H atoms add into CO2 to produce HCOO* as the intermediates. As for Ptn@MIL, Pt–H is formed, wherein H atoms hydrogenate CO2 into COOH* as the intermediates. The unique reaction path over Pt1@MIL not only lowers the activation energy for the enhanced catalytic activity, but also contributes to the high selectivity for methanol.
Results
Synthesis and structural characterizations of Pt1@MIL
MIL-101 is a typical MOF which consists of μ3-oxo bridged Cr(III)-trimers cross linked by terephthalic acid27,28. The high specific surface area of SLangmuir = 5900 m2 g−1 and large pore volume of ca. 2.0 cm3 g−1 render MIL-101 high adsorption capacities, making it an attractive candidate for gas adsorption. As such, MIL-101 with an average particle size of 500 nm was synthesized based on previous literatures27,28 (Supplementary Fig. 1), serving as the support in this work. In a typical synthesis of Pt1@MIL, K2PtCl4 and NaBH4 aqueous solutions were added into the flask containing MIL-101 aqueous solution via a syringe pump under magnetic stirring. In Pt1@MIL, the Pt mass loading was determined as 0.2% by inductively coupled plasma–atomic emission spectroscopy (ICP–AES). Figure 1a shows a high-angle annular dark-field scanning transmission electron microscopy (HAADF–STEM) image of Pt1@MIL. As manifested by brightness, Pt atoms were atomically dispersed in MIL-101 in the absence of nanoparticles. To highlight the dopant atoms, the magnified HAADF–STEM image and its corresponding color-coded intensity map were shown in Fig. 1b, c, indicating the isolated distribution of Pt atoms. After Pt1@MIL was further washed for different rounds, the ratio of Pt to Cr remained almost unchanged, confirming that Pt single atoms were indeed anchored in MIL-101, rather than serving as a residual (Supplementary Fig. 2). By simply increasing the concentrations of K2PtCl4 and NaBH4 aqueous solutions, Ptn@MIL with the Pt mass loading of 1.0% was facilely synthesized. As shown in the HAADF–STEM image of Ptn@MIL, Pt nanocrystals with an average size of 1.8 nm were observed (Supplementary Fig. 3). The ratio of surface Pt atoms in Ptn@MIL was determined as 41.5% by CO pulse chemisorptions (Supplementary Fig. 4). To characterize the electronic properties of the obtained samples, we conducted X-ray photoelectron spectroscopy (XPS) measurements of Pt1@MIL and Ptn@MIL. As shown in Fig. 1d, the Pt species in Pt1@MIL was determined to be at Pt2+ state. By comparison, most of Pt species in Ptn@MIL was at the metallic state, whereas a small portion of Pt was oxidized.
Structural characterizations of Pt@MIL. a HAADF-STEM image of Pt1@MIL. b, c Magnified HAADF-STEM image and its corresponding color-coded intensity map of Pt1@MIL. Pt single atoms marked in yellow circles were uniformly anchored in MIL-101. d XPS spectra of Pt 4 f for Pt1@MIL and Ptn@MIL. e Pt L3-edge XANES profiles of Pt foil, Pt1@MIL, and Ptn@MIL. f Pt L3-edge EXAFS spectra in R space of Pt foil, Pt1@MIL, and Ptn@MIL. Pt foil was used as the reference. g Structural model of Pt1@MIL simulated by DFT calculations. Blue, violet, red, gray, and white spheres represent for Pt, Cr, O, C, and H atoms, respectively. The yellow spheres represent the oxygen atoms which are fixed at their positions in crystal
The X-ray absorption near-edge spectroscopy (XANES) and extended X-ray absorption fine structure (EXAFS) were measured to determine the electronic and coordination structures of Pt atoms in Pt@MIL catalysts. The Pt L3-edge XANES profiles in Fig. 1e indicate that the Pt species in Pt1@MIL were in a higher oxidation state than those in Ptn@MIL, according to the stronger intensity for white line. It is worth noting that the spectrum of Ptn@MIL was similar to that of Pt foil, indicating the metallic state of Pt species in Ptn@MIL, consistent with the XPS results (Fig. 1d). As shown in EXAFS in R space (Fig. 1f), Pt1@MIL exhibited a prominent peak at 2.01 Å from the Pt–O shell with a coordination number (CN) of 3.7 (Supplementary Table 1). No other typical peaks for Pt–Pt contribution at longer distances (>2.5 Å) were observed, revealing the isolated dispersion of Pt atoms throughout the whole Pt1@MIL. As for Ptn@MIL, a new peak at 2.76 Å was observed, corresponding to the Pt–Pt metallic bond with a CN of 7.4.
To investigate the location and coordination of Pt single atoms in MIL-101, density functional theory (DFT) calculations were performed to establish the atomic model of Pt1@MIL. The building unit of MIL-101 comprises terephthalic linkers and an inorganic trimer. The trimer consists of three Cr atoms in an octahedral coordination. The vertices of octahedral coordination around each Cr atom are occupied by four O atoms from carboxylate groups in terephthalic linkers, one μ3O atom in the middle of Cr trimers, and one O atom from the terminal water or fluorine group. Both XPS and XANES results indicate an oxidation state assignment of Pt2+ for Pt1@MIL (Fig. 1d, e). In Pt1@MIL, the individual Pt atom was stabilized by O atoms in a planar four-coordinate geometry with one O atom in carboxyl, one dangling O connected with a Cr atom, and one O2 moiety (Fig. 1g and Supplementary Fig. 5). This optimized model is consistent with the oxidation state of Pt2+ indicated by XPS and XANES results, because Pt2+ generally adopts the dsp2 hybridization which results in the planar four-coordinate geometry. To further confirm the Pt oxidation states in this model, we have compared the charge of the Pt atom in Pt1@MIL with that in isolated Pt–O and O–Pt–O clusters as representatives of Pt2+ and Pt4+ (Supplementary Fig. 6). Based on Mullinken analysis, the charges of Pt atoms in Pt–O and O–Pt–O clusters are +0.72 and +1.52 e, respectively. The Pt single atom in Pt1@MIL is + 0.59 e charged, approximating to that in Pt–O cluster. Thus, the oxidation state of Pt in Pt1@MIL was assigned to +2. Moreover, the model of the Pt single atom coordinated with four O atoms in Pt1@MIL matches our EXAFS result in terms of the CN of Pt–O.
Catalytic properties of Pt1@MIL for CO2 hydrogenation
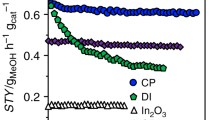
The catalytic properties of the as-obtained Pt@MIL in CO2 hydrogenation were evaluated in a slurry reactor with 10 mL of DMF under 32 bar of CO2/H2 mixed gas (CO2:H2 = 1:3) at 150 °C. As a benchmark, commercial Cu/ZnO/Al2O3 (63 wt% Cu) was directly purchased from Alfa Aesar, with the ratio of surface Cu atoms to total atoms determined as 32.5% (Supplementary Fig. 7). For each catalytic test, the amounts of Pt1@MIL and Ptn@MIL were controlled at 500 and 240 mg, respectively, to keep the same amount (1.0 mg) of exposed Pt atoms, whereas 20 mg of Cu/ZnO/Al2O3 was used. When the reaction was catalyzed by MIL-101, the product was below detection limit (Fig. 2a). As for Pt1@MIL, 0.6 mmol of products were generated after 1 h, whereas the product yield was 0.12 mmol for Ptn@MIL. More importantly, the selectivity for methanol reached 90.3% over Pt1@MIL with the production of formic acid as the by-product (Fig. 2a). With regard to Ptn@MIL, the major product was CO with the selectivity of 57.5%, while the selectivities for formic acid, methanol, and methane were 11.1%, 13.3%, and 18.1%, respectively (Fig. 2a). In general, Pt-based heterogeneous catalysts have been reported to be selective to CO or methane during CO2 hydrogenation29,30. In this case, Pt1@MIL did not give rise to the formation of gaseous products, behaving much differently from conventional Pt-based catalysts. As for Cu/ZnO/Al2O3, 0.16 mmol of methanol, 0.026 mmol of formic acid, and 0.024 mmol of CO were formed after 1 h (Fig. 2a and Supplementary Fig. 8). To compare the catalytic activity more accurately, we calculated the TOF numbers of these catalysts by only taking Pt atoms into account based on the reaction profile at the initial stage. The TOF number of Pt1@MIL reached 117 h−1 in DMF under 32 bar at 150 °C, being around 5.6 times that (21 h−1) of Ptn@MIL and 39 times that (3 h−1) of Cu/ZnO/Al2O3 (Fig. 2b). As a benchmark, the activity of Cu/ZnO/Al2O3 in DMF solvent was comparable with the values obtained in a fixed-bed reactor26,31,32,33. We have further applied deuterated DMF (C3D7NO) as the solvent to replace DMF in CO2 hydrogenation over Pt1@MIL under 32 bar at 150 °C. The TOF number of Pt1@MIL reached 107 h−1 in C3D7NO, almost equal to the corresponding values in DMF (Supplementary Fig. 9). Accordingly, the effect of proton transfer in DMF on CO2 hydrogenation over Pt1@MIL could be neglected.
Catalytic performance of Pt@MIL in CO2 hydrogenation. a Comparison of products obtained by using MIL-101, Pt1@MIL, Ptn@MIL, and commercial Cu/ZnO/Al2O3 in DMF under CO2/H2 mixed gas (CO2:H2 = 1:3, 32 bar) at 150 °C after 1 h. b Comparison of TOF numbers of Pt1@MIL and Ptn@MIL in DMF under CO2/H2 mixed gas (CO2:H2 = 1:3, 32 bar) at different temperatures. c The Arrhenius plots of Pt1@MIL and Ptn@MIL. d Products obtained by conducting in-situ cycles over Pt1@MIL. For each cycle, the catalytic reaction proceeded under 32 bar of CO2/H2 mixed gas (CO2/H2 = 1:3) at 150 °C for 6 h. For each catalytic test, the amounts of Pt1@MIL and Ptn@MIL were controlled at 500 and 240 mg, respectively, to keep the same amount (1.0 mg) of exposed Pt atoms. Error bars represent standard deviation from three independent measurements
To explore the differences in catalytic properties between Pt1@MIL and Ptn@MIL, we conducted a series of catalytic tests under 32 bar of CO2/H2 mixed gas (CO2:H2 = 1:3) at different temperatures. As shown in Fig. 2b, Pt1@MIL exhibited much higher catalytic activity than Ptn@MIL (Supplementary Figs. 10 and 11). Arrhenius plots were obtained based on the linear fitting of lnTOF vs. 1000/T (Fig. 2c). The activation energy for Pt1@MIL was 76.4 kJ mol−1, much lower than that (119.7 kJ mol−1) for Ptn@MIL. To investigate the size effect, Pt nanoparticles with an average size of 1.2 nm on MIL-101 were prepared and evaluated under 32 bar at 150 °C (Supplementary Fig. 12). The TOF number was calculated to be 22 h−1 which was lower than that (117 h−1) of Pt1@MIL and basically the same as that (21 h−1) of Ptn@MIL with 1.8-nm Pt nanoparticles (Supplementary Fig. 13). As such, Pt1@MIL exhibited higher catalytic activity and selectivity for methanol than Ptn@MIL even with tiny Pt clusters.
To explore stability of Pt1@MIL, we performed successive rounds of reaction. For each round, the catalytic reaction proceeded at 150 °C for 1 h. After 10 rounds, the selectivity of methanol kept almost unchanged (Supplementary Fig. 14). Moreover, the HAADF-STEM image of the used Pt1@MIL (“used” refers to the catalyst after 10 rounds) showed that Pt atoms were still atomically dispersed in MIL-101 without the formation of nanoparticles (Supplementary Fig. 15). In addition, only 2.6% of Pt species in Pt1@MIL was leached after 10 rounds as revealed by ICP-AES. To further investigate the potential industrial application, we tested Pt1@MIL for successive in-situ cycles of reaction. For each cycle, the catalytic reaction proceeded under 32 bar of CO2/H2 mixed gas (CO2/H2 = 1:3) at 150 °C for 6 h without the removal of catalysts from the reactor during the whole test. After 16 in-situ cycles (96 h in total), Pt1@MIL led to the generation of about 14.6 mmol of methanol and 1.4 mmol of formic acid in total, exhibiting high long-term stability (Fig. 2d). Accordingly, Pt1@MIL was highly stable during CO2 hydrogenation, thereby exhibiting potential industrial application.
Formation of hydroxyl groups in Pt1@MIL under H2
We conducted DFT calculations to investigate the hydrogen dissociation on Pt1@MIL. Based on DFT calculations, the whole reaction path is divided into three elementary steps that include the dissociation of H2 on Pt (step i), the formation of the first hydroxyl (step ii), and the formation of the second hydroxyl (step iii) (Supplementary Table 2 and Supplementary Fig. 16). Specially, H2 is firstly dissociated on Pt to form Pt–H bonds. Afterwards, the two dissociated H atoms on Pt atoms stepwise migrates to the dangling O2 moiety, resulting in the formation of two hydroxyl groups. Among these steps, step ii exhibits the highest energy barrier with the value of 0.79 eV, indicating that the formation of hydroxyl groups are able to proceed at 150 °C. To support this point, we carried out in-situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) measurements. Figure 3 shows the in-situ DRIFTS spectra of Pt1@MIL and Ptn@MIL after the treatment with H2 at 150 °C for 0.5 h. As for Pt1@MIL, a peak at 3197 cm−1 appeared, corresponding to the stretching vibration of O–H. The observation of O–H and absence of Pt–H are consistent with the DFT results. With regard to Ptn@MIL, the peak at 1949 cm−1 was assigned to the stretching vibration of Pt–H. Therefore, Pt single atom activates its coordinated O atoms to adsorb the dissociated H atoms with the formation of two hydroxyl groups. As for Ptn@MIL, Pt nanoparticles directly dissociate H2 to form Pt–H species.
To investigate the role played by H atoms in hydroxyl groups, we have explored the kinetic isotope effect (KIE) with the use of D2 in catalytic tests. When the reaction proceeded under CO2/D2 mixed gas (CO2:D2 = 1:3) at 150 °C, the TOF numbers of Pt1@MIL and Ptn@MIL were 21 h−1 and 13 h−1, respectively (Supplementary Fig. 17a–b). The KIE value refers to the ratio of the TOF using H2 as the reactant gas to that using D2 (kH/kD). As shown in Supplementary Fig. 17c, the KIE value of Pt1@MIL was 5.6 much higher than that (1.6) of Ptn@MIL. As such, the bond cleavage of O-D rather than Pt-D was involved in the reaction over Pt1@MIL.
CO2 hydrogenation paths
The different adsorption properties of H atoms on Pt1@MIL and Ptn@MIL induced the variation of reaction paths in CO2 hydrogenation. To investigate the reaction path, we calculated the addition of the first H atom into CO2. For Pt1@MIL, the energy of HCOO* is 0.26 eV lower than that of COOH* (Supplementary Fig. 18a–b and Tables 3–4). In this case, the first H atom adds to the C atom in CO2 to form HCOO* as the stable intermediate on Pt1@MIL. In comparison, Ptn@MIL favors the generation of COOH* whose energy is 0.34 eV lower than that of HCOO* (Supplementary Fig. 18c–d and Tables 3–4). As such, the transformation of CO2 into COOH* serves as the dominating reaction channel on Ptn@MIL. To support this point, we conducted in-situ DRIFTS measurements of Pt1@MIL and Ptn@MIL after the treatment with H2 and CO2 in sequence at 150 °C. For Pt1@MIL, the peak for O–H at 3197 cm−1 was still observed, but its intensity was weakened relative to the case when the sample was only treated with H2 (Fig. 3). The peak at 1364 cm−1 was observed, indicating the formation of CO2δ- species4,18. Moreover, the peaks at 2919, 1631, and 1228 cm−1 also emerged, which were assigned to the stretching vibration of C–H, the asymmetrical and symmetrical stretching vibrations of the bidentate O–C–O in HCOO* species, respectively (Fig. 3). As for Ptn@MIL treated with H2 and CO2 in sequence relative to the one solely exposed to H2, the peak for Pt–H disappeared. Moreover, besides the peak for CO2δ-, the peaks at 3203, 1705, and 1530 cm−1 appeared, corresponding to the stretching vibration of O–H, the stretching vibration of C=O, and the bending vibration of C–O in COOH* species, respectively (Fig. 3). The in-situ DRIFTS results indicate that HCOO* and COOH* served as the intermediates for Pt1@MIL and Ptn@MIL, respectively, consistent with the theoretical results.
The alteration of intermediates in CO2 hydrogenation over Pt1@MIL and Ptn@MIL was further supported by quasi-situ XPS. Quasi-situ XPS measurements were conducted after the treatment of the samples with H2 and CO2 in sequence at 150 °C in a reaction cell attached to the XPS end-station. As shown in Fig. 4a, the C 1 s spectra exhibited four typical peaks at 284.6, 288.6, 289.5, and 291.0 eV, corresponding to benzene ring in MIL-101 (Ph), carboxyl group in MIL-101 (Ph-COO), HCOO*, and COOH*, respectively18,34,35,36. In addition, different species could also be distinguished in O 1s spectra. Specifically, the peaks at 531.5 and 532.4 eV derived from the oxygen atoms in Ph–COO and HCOO*, respectively (Fig. 4b). Two peaks at 530.0 and 531.1 eV were assigned to the carbonyl and hydroxyl species in COOH*, respectively (Fig. 4b)34,35,36. As indicated by quasi-situ XPS spectra, HCOO* or COOH* was not observed for MIL-101. Therefore, the intermediates for Pt1@MIL and Ptn@MIL were determined as HCOO* and COOH*, respectively, consistent with in-situ DRIFTS results.
Mechanistic insight into remarkable activity of Pt1@MIL. a, b Quasi-situ XPS spectra of C 1 s and O 1 s for MIL-101, Pt1@MIL, and Ptn@MIL after the treatment with H2 and CO2 in sequence at 150 °C, respectively. c, d C and O K-edge XANES spectra for MIL-101, Pt1@MIL, and Ptn@MIL before/after the treatment with H2 and CO2 in sequence at 150 °C, respectively
The intrinsic difference between HCOO* and COOH* lies in the exclusive existence of C–H bond in HCOO* and O–H bond in COOH*, which could be verified by C and O K-edge XANES measurements. Figure 4c shows the C K-edge XANES profiles of Pt1@MIL and Ptn@MIL before/after the treatment with H2 and CO2 in sequence at 150 °C. Before gas treatment, both Pt1@MIL and Ptn@MIL revealed the same XANES profiles which exhibited seven characteristic features. In detail, three features at 285.0, 285.9, and 290.3 eV were assigned to the excitations of the C=C π antibonding orbital for benzene ring in MIL-101 (π*(Ph))37. Besides, the peaks at 287.6, 288.5, 289.4, and 293.1 eV corresponded to the excitations of the C–H antibonding orbital (σ*(C–H)), the C = O π antibonding orbital (π*(C=O)), the C–O antibonding orbital (σ*(C–O)), and the σ antibonding orbital for benzene ring in MIL-101 (σ*(Ph)), respectively37. After the treatment of MIL-101 with H2 and CO2 in sequence at 150 °C, the features were almost the same as the corresponding ones before the treatment. After the sequential gas treatment, the profiles of Pt1@MIL and Ptn@MIL both showed stronger feature of π*(C=O) relative to that without gas treatment, indicating the transformation of CO2. Moreover, the feature of σ*(C–H) became stronger for Pt1@MIL, but remained almost unchanged for Ptn@MIL. As such, only the species generated over Pt1@MIL contained C–H bond. As for O K-edge XANES, the features at 531.1 and 533.7 eV corresponded to the excitations of the π*(C=O), while the features at 536.1 and 539.1 eV were assigned to the O–H antibonding orbital (σ*(O–H)) and the C–O antibonding orbital (σ*(C–O)), respectively38 (Fig. 4d). After the exposure to H2 and CO2 in sequence at 150 °C, the feature of σ*(O–H) became significantly stronger for Ptn@MIL, but stayed almost the same for Pt1@MIL. Accordingly, only Ptn@MIL induced the generation of species containing O–H groups.
In Pt1@MIL, every Pt single atom and its coordinated O atoms composed an active center. The metal-ligand cooperativity leads to the dissociation of H2 to form hydroxyl groups, wherein hydroxy H atoms added into CO2 to produce HCOO* as the intermediates. With regards to Ptn@MIL, Pt hydrides formed, offering H atoms to hydrogenate CO2 into COOH* as the intermediates. In general, HCOO* is reported to be hydrogenated into HCOOH* and finally converted into methanol6,9. By comparison, besides forming HCOOH*, COOH* is also apt to lose the hydroxyl species to generate CO* species6,18. CO* can be either directly desorbed to form gaseous CO, or further hydrogenated into methane6. Thus, experiencing the reaction path via COOH* intermediate over Ptn@MIL leads to the formation of gaseous products.
Discussion
In conclusion, the metal-ligand cooperativity in Pt1@MIL was found to induce distinct reaction path and improve selective hydrogenation of CO2 into methanol relative to Ptn@MIL. According to mechanistic studies, CO2 was hydrogenated into HCOO* as the intermediate for Pt1@MIL, whereas COOH* served as the intermediate for Ptn@MIL. The unique reaction path over Pt1@MIL both lowered the activation energy for the enhanced activity and led to the high selectivity for methanol. Upgrading the catalysts from nanocrystals to single atoms not only enhances the atomic utilization efficiency, but also alters the catalytic mechanisms such as the adsorption of reactants or intermediates on catalysts and the reaction path. This strategy offers a powerful means to improve the catalytic performance for CO2 hydrogenation, and extends our understanding of single-atom catalysis.
Methods
Synthesis of MIL-101
The synthesis of MIL-101 followed a reported method28. Specifically, 2 g of Cr(NO3)3·9H2O, 830 mg of terephthalic acid, and 0.225 mL of HF aqueous solution (40.0% in mass fraction) were added into 25 mL of water in a 50-mL beaker, followed by stirring at room temperature for 15 min. Then, the solution was transferred into a 50-mL Teflon-lined autoclave and heated at 220 °C for 8 h. After the solution was cooled to room temperature, the product was collected by centrifugation, washed three times with water. For further purification, the solid product was sequentially stirred in 200 mL of ethanol solution (95% ethanol with 5% H2O) at 80 °C for 24 h, 300 mL of NH4F aqueous solution (30 mM) at 70 °C for 24 h, and 200 mL of water at 90 °C for 3 h. The product was collected by centrifugation, and then dried at 60 °C under vacuum.
Synthesis of Pt@MIL
In a typical synthesis of Pt1@MIL, 100 mg of MIL-101 were dispersed in 60 mL of water in a 250-mL flask under magnetic stirring for 10 min. 10.1 mL of K2PtCl4 aqueous solution (0.1 mM) and 10.1 mL of NaBH4 aqueous solution (0.1 mM) were added into the flask through a two-channel syringe pump at a rate of 2 mL h−1 under magnetic stirring at room temperature. The sample was washed three times with water, and dried at 60 °C under vacuum. Further ICP result determined that the mass loading of Pt was 0.2%. The synthetic procedure for Ptn@MIL with the Pt loadings of 1.0% was similar to that for Pt1@MIL, except for increasing the amounts of K2PtCl4 and NaBH4. For Ptn@MIL, 10.4 mL of K2PtCl4 aqueous solution (0.5 mM) and 10.4 mL of NaBH4 aqueous solution (0.5 mM) were added into the flask through a two-channel syringe pump at a rate of 10 mL h−1 under magnetic stirring at room temperature. The synthesis of Pt nanoparticles with an average size of 1.2 nm was similar to that of Ptn@MIL except for the concentration of precursors. Specially, 100 mg of MIL-101 were dispersed in 60 mL of water in a 250-mL flask under magnetic stirring for 10 min. 10.4 mL of K2PtCl4 aqueous solution (0.3 mM) and 10.1 mL of NaBH4 aqueous solution (0.5 mM) were added into the flask through a two-channel syringe pump at a rate of 10 mL h−1 under magnetic stirring at room temperature.
XAFS measurements
The XAFS measurements were carried out at Pt L3-edge (11564 eV) on the BL14W1 beamline39 of Shanghai Synchrotron Radiation Facility. The XAFS data of Pt@MIL were recorded in fluorescence mode by using a Ge solid-state detector. The calibration of the energy was based on the absorption edge of pure Pt foil. The data extraction and fitting were carried out by using Athena and Artemis codes. As for XANES, the normalized absorption refers to the experimental absorption coefficients vs. energies μ(E) after background subtraction and normalization procedures. As for EXAFS, the first-shell approximation was adopted to analyze the Fourier transformed data in R space of the Pt–O shell, while metallic Pt model was used to analyze that of the Pt–Pt shell. To determine the passive electron factors (S02), the experimental Pt foil data were fitted while the CN of Pt–Pt was fixed as 12. Afterwards, S02 was fixed for further analysis. The parameters such as bond distance (R), CN, and Debye Waller (D.W.) factor around the absorbed atoms were variable during the fitting process.
DFT calculations
Density functional theory (DFT) calculations were performed using Gaussian program40 to explain the interaction mechanism between CO2 and Pt1@MIL, a cluster with 30 O atoms, 48 C atoms, 3 Cr atoms, 32 H atoms, and 1 Pt atom was adopted to simulate Pt1@MIL. The six carboxylate terminals on the top of benzene ring were saturated by hydrogen atoms (COO− → COOH) and the twelve oxygen atoms were fixed at their crystal positions during geometric optimization. We made calculations with B3LYP-D3 functional41 implemented in Gaussian with the combination of the Grimme’s third generation dispersion correction (D3)42. For structural optimization, the standard Gaussian-type basis sets 6–31 G(d)43 was used for C, O, and H atoms and LANL2DZ44 sets for metal atoms. As for the calculation of reaction paths, we used the optimized structure for a higher-precision single-point energy calculation. In the level, the basis sets for describing C, O, and H atoms was replaced by 6–311++G(d, p)45, whereas the basis sets for metal atoms still adopted LANL2DZ. In order to simulate the periodic Pt (111), we used Vienna ab initio simulation package (VASP)46,47. The projector augmented wave (PAW) method was used to describe the interaction between ions and electrons48. The nonlocal exchange correlation energy was evaluated using the Perdew–Burke–Ernzerhof functional49. A plane wave basis set with a cutoff energy of 400 eV and a 1 × 1 × 1 k-point grid generated by the Monkhorst–Pack method were used to describe the Brillouin zone for geometric optimization. The atomic structures were relaxed using either the conjugate gradient algorithm or the quasi-Newton scheme as implemented in the VASP code until the forces on all unconstrained atoms were ≤0.02 eV Å−1.
Catalytic tests of Pt@MIL catalysts in CO2 hydrogenation
The hydrogenation of CO2 was conducted in a 100-mL slurry reactor (Parr Instrument Company). In a typical catalytic test, the reactor was charged and discharged with 32 bar of mixed gas (CO2:H2 = 1:3) at room temperature for three times, after the addition of 10 mL of DMF and certain amounts of catalysts into the Teflon inlet. For a catalytic test, the weights of Pt1@MIL and Ptn@MIL were controlled at 500 and 240 mg, respectively, so as to keep the same amount (1.0 mg) of exposed Pt in each catalyst. The reaction proceeded under stirring with a rate of 300 rpm and 32 bar of mixed gas (CO2:H2 = 1:3) at 150 °C. After the completion of the reaction, the gaseous products were determined by GC-FID and GC-TCD. The reaction mixture in liquid phase was collected by centrifugation at 11,180 × g for 2 min. The test solution contained 50 μL of chloroform, as an internal standard, and 1 mL of the reaction mixture. 100 μL of the test solution was dissolved in 0.4 mL of DMSO-d6 for the measurements of 1H NMR. For each data point, the catalytic tests were repeated thrice. For the stability of Pt1@MIL, we applied two methods including successive reaction rounds and in-situ cycles. The addition of catalysts, solution, and reaction gases followed the same procedure as a typical catalytic test. Successive reaction rounds were carried out with the collection of catalysts for each round. Specially, after the proceeding of reaction at 150 °C for 1 h, both gaseous and liquid products were detected. Meanwhile, the catalysts were collected via centrifugation and washed thrice with DMF, followed by being re-added to the slurry reactor for the next round. In-situ cycles were carried out without opening the slurry reactor to collect the catalysts. For each cycle, after the proceeding of reaction at 150 °C for 6 h, both gaseous and liquid products were taken through tubes for detection. The TOF was calculated based on the equations.
In this equation, nCO2 represents the mole of converted CO2 molecules. t is the reaction time. nsurface Pt atoms represents the mole of surface Pt atoms. μPt is the weight of one mole of Pt atoms. msurface is the weight of surface Pt species in the catalysts. mcat is the weight of catalysts. w represents the mass loading of Pt species. rsurface represents the ratio of surface Pt atoms to total Pt atoms. For Pt1@MIL, we assumed that all the Pt atoms were exposed on the surface, so that rsurface is 100.0%. For Ptn@MIL, rsurface was determined as 41.5% via CO pulse chemisorptions
In-situ DRIFTS tests
The instrument for in-situ DRIFTS experiments was composed of an elevated-pressure cell (DiffusIR Accessory PN 041-10XX) and a Fourier transform infrared spectrometer (TENSOR II Sample Compartment RT-DLaTGS). The resolution of wavenumber was 4 cm−1 at 150 °C. The background spectrum was obtained after a N2 flow (1 bar) for 0.5 h at 150 °C. Then, 1 bar of H2 was allowed to flow into the cell at the rate of 30 sccm at 150 °C for 0.5 h, followed by flowing with 1 bar of N2 at the rate of 30 sccm at 150 °C for 0.5 h. To detect the species generated after the treatment of the samples with H2 and CO2 in sequence, 1 bar of H2 was allowed to flow into the cell at the rate of 30 sccm at 150 °C for 0.5 h, followed by flowing with 1 bar of N2 at the rate of 30 sccm at 150 °C for 0.5 h. Then, 1 bar of CO2 was allowed to flow into the cell at 25 °C for 30 min, followed by purging with 1 bar of N2 at 25 °C for 30 min.
Quasi-situ XPS measurements
Quasi-situ XPS measurements were performed at the photoemission end-station on the BL10B beamline of National Synchrotron Radiation Laboratory (NSRL, China). The samples were exposed to 1 bar of H2 and CO2 in sequence at 150 °C for 0.5 h. Afterwards, the samples were moved to the analysis chamber for further XPS analysis.
Instrumentations
TEM, HAADF–STEM, and STEM–EDX images were collected on a JEOL ARM-200F field-emission transmission electron microscope operating at 200 kV accelerating voltage. ICP-AES (Atomscan Advantage, Thermo Jarrell Ash, USA) was used to determine the concentration of metal species. NMR spectra were recorded on a Brucker-400 MHz spectrometer. The C and O K-edge X-ray absorption spectra were measured at beamline B12b of National Synchrotron Radiation Laboratory (NSRL, China) in the total electron yield (TEY) mode by collecting the sample drain current under a vacuum better than 1 × 10–7 Pa.
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Gao, P. et al. Direct conversion of CO2 into liquid fuels with high selectivity over a bifunctional catalyst. Nat. Chem. 9, 1019–1024 (2017).
Vogt, C. et al. Unravelling structure sensitivity in CO2 hydrogenation over nickel. Nat. Catal. 1, 127–134 (2018).
Liu, Q. et al. Direct catalytic hydrogenation of CO2 to formate over a Schiff-base-mediated gold nanocatalyst. Nat. Commun. 8, 1407 (2017).
Wang, L. et al. Incorporating nitrogen atoms into cobalt nanosheets as a strategy to boost catalytic activity toward CO2 hydrogenation. Nat. Energy 2, 869–876 (2017).
Kar, S., Sen, R., Goeppert, A. & Prakash, G. K. S. Integrative CO2 capture and hydrogenation to methanol with reusable catalyst and amine: toward a carbon neutral methanol economy. J. Am. Chem. Soc. 140, 1580–1583 (2018).
Li, Y., Chan, S. H. & Sun, Q. Heterogeneous catalytic conversion of CO2: A comprehensive theoretical review. Nanoscale 7, 8663–8683 (2015).
Peng, Y. et al. Molecular-level insight into how hydroxyl groups boost catalytic activity in CO2 hydrogenation into methanol. Chem 4, 613–625 (2018).
Gao, G., Jiao, Y., Waclawik, E. & Du, A. Single atom (Pd/Pt) supported on graphitic carbon nitride as an efficient photocatalyst for visible-light reduction of carbon dioxide. J. Am. Chem. Soc. 138, 6292–6297 (2016).
Kattel, S. et al. Active sites for CO2 hydrogenation to methanol on Cu/ZnO catalysts. Science 355, 1296–1299 (2017).
Li, H. et al. Synergetic interaction between neighbouring platinum monomers in CO2 hydrogenation. Nat. Nanotechnol. 13, 411–417 (2018).
Nielsen, D. U., Hu, X.-M., Daasbjerg, K. & Skrydstrup, T. Chemically and electrochemically catalysed conversion of CO2 to CO with follow-up utilization to value-added chemicals. Nat. Catal. 1, 244–254 (2018).
Zhang, X. et al. Product selectivity in plasmonic photocatalysis for carbon dioxide hydrogenation. Nat. Commun. 8, 14542 (2017).
Nie, L. et al. Activation of surface lattice oxygen in single-atom Pt/CeO2 for low-temperature CO oxidation. Science 358, 1419–1423 (2017).
Wei, J. et al. Directly converting CO2 into a gasoline fuel. Nat. Commun. 8, 15174 (2017).
Spencer, M. S. The role of zinc oxide in Cu/ZnO catalysts for methanol synthesis and the water-gas shift reaction. Top. Catal. 8, 259–266 (1999).
Kuld, S. et al. Quantifying the promotion of Cu catalysts by ZnO for methanol synthesis. Science 352, 969–974 (2016).
Behrens, M. et al. The active site of methanol synthesis over Cu/ZnO/Al2O3 industrial catalysts. Science 336, 893–897 (2012).
Graciani, J. et al. Highly active copper-ceria and copper-ceria-titania catalysts for methanol synthesis from CO2. Science 345, 546–550 (2014).
Liu, C. et al. Carbon dioxide conversion to methanol over size-selected Cu4 clusters at low pressures. J. Am. Chem. Soc. 137, 8676–8679 (2015).
Liao, F. et al. Electronic modulation of a copper/zinc oxide catalyst by a heterojunction for selective hydrogenation of carbon dioxide to methanol. Angew. Chem. Int. Ed. 51, 5832–5836 (2012).
Wang, J. et al. A highly selective and stable ZnO-ZrO2 solid solution catalyst for CO2 hydrogenation to methanol. Sci. Adv. 3, e1701290 (2017).
Li, C. S. et al. High-performance hybrid oxide catalyst of manganese and cobalt for low-pressure methanol synthesis. Nat. Commun. 6, 6538 (2015).
Matsubu, J. C. et al. Adsorbate-mediated strong metal-support interactions in oxide-supported Rh catalysts. Nat. Chem. 9, 120–127 (2017).
Khan, M. U. et al. Pt3Co octapods as superior catalysts of CO2 hydrogenation. Angew. Chem. Int. Ed. 55, 9548–9552 (2016).
Van den Berg, R. et al. Structure sensitivity of Cu and CuZn catalysts relevant to industrial methanol synthesis. Nat. Commun. 7, 13057 (2016).
Studt, F. et al. Discovery of a Ni-Ga catalyst for carbon dioxide reduction to methanol. Nat. Chem. 6, 320–324 (2014).
Liu, X. H., Ma, J. G., Niu, Z., Yang, G. M. & Cheng, P. An efficient nanoscale heterogeneous catalyst for the capture and conversion of carbon dioxide at ambient pressure. Angew. Chem. Int. Ed. 54, 988–991 (2015).
Férey, G. et al. A chromium terephthalate-based solid with unusually large pore volumes and surface area. Science 309, 2040–2042 (2005).
Kattel, S., Yan, B. H., Chen, J. G. G. & Liu, P. CO2 hydrogenation on Pt, Pt/SiO2 and Pt/TiO2: importance of synergy between Pt and oxide support. J. Catal. 343, 115–126 (2016).
Beaumont, S. K., Alayoglu, S., Specht, C., Kruse, N. & Somorjai, G. A. A nanoscale demonstration of hydrogen atom spillover and surface diffusion across silica using the kinetics of CO2 methanation catalyzed on spatially separate Pt and Co nanoparticles. Nano Lett. 14, 4792–4796 (2014).
An, B. et al. Confinement of Ultrasmall Cu/ZnOx nanoparticles in metal–Organic frameworks for selective methanol synthesis from catalytic hydrogenation of CO2. J. Am. Chem. Soc. 139, 3834–3840 (2017).
Rungtaweevoranit, B. et al. Copper nanocrystals encapsulated in Zr-based metal–organic frameworks for highly selective CO2 hydrogenation to methanol. Nano Lett. 16, 7645–7649 (2016).
Sun, Q. et al. A novel process for the preparation of Cu/ZnO and Cu/ZnO/Al2O3 ultrafine catalyst: structure, surface properties, and activity for methanol synthesis from CO2 + H2. J. Catal. 167, 92–105 (1997).
Mudiyanselage, K. et al. Importance of the metal-oxide interface in catalysis: In situ studies of the water-gas shift reaction by ambient-pressure X-ray photoelectron spectroscopy. Angew. Chem. Int. Ed. 52, 5101–5105 (2013).
Prosvirin, I. P., Bukhtiyarov, A. V., Bluhm, H. & Bukhtiyarov, V. I. Application of near ambient pressure gas-phase X-ray photoelectron spectroscopy to the investigation of catalytic properties of copper in methanol oxidation. Appl. Surf. Sci. 363, 303–309 (2016).
Deng, X. et al. Surface chemistry of Cu in the presence of CO2 and H2O. Langmuir 24, 9474–9478 (2008).
Stöhr, J. NEXAFS Spectroscopy (Springer, Berlin, 1992).
Gordon, M. L. et al. Inner-shell excitation spectroscopy of the peptide bond: comparison of the C 1s, N 1s, and O 1s spectra of glycine, glycyl-glycine, and glycyl-glycyl-glycine. J. Phys. Chem. A 107, 6144–6159 (2003).
Yu, H. S. et al. The XAFS beamline of SSRF. Nucl. Sci. Tech. 26, 050102 (2015).
Frisch, M. J. et al. Gaussian 09, Revision D.01. (Gaussian, Inc., Wallingford CT, 2013).
Jiao, Y. et al. Electrocatalytically switchable CO2 capture: first principle computational exploration of carbon nanotubes with pyridinic nitrogen. ChemSusChem 7, 435–441 (2014).
Becke, A. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Hehre, W. J., Ditchfield, R. & Pople, J. A. Self-consistent molecular orbital methods. XII. Further extensions of Gaussian-type basis sets for use in molecular orbital studies of organic molecules. J. Chem. Phys. 56, 2257–2261 (1972).
Chiodo, S., Russo, N. & Sicilia, E. LANL2DZ basis sets recontracted in the framework of density functional theory. J. Chem. Phys. 125, 104107 (2006).
Clark, T., Chandrasekhar, J., Spitznagel, G. W. & Schleyer, P. V. R. Efficient diffuse function-augmented basis sets for anion calculations. III. The 3-21 + G basis set for first-row elements. Li–F. J. Comp. Chem. 4, 294–301 (1983).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for Ab Initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Acknowledgements
This work was supported by NSFC (21573206), Key Research Program of Frontier Sciences of the CAS (QYZDB-SSW-SLH017), Anhui Provincial Key Scientific and Technological Project (1704a0902013), Major Program of Development Foundation of Hefei Center for Physical Science and Technology (2017FXZY002), National Key Research and Development Program (2018YFA0208600), and Fundamental Research Funds for the Central Universities. This work was partially carried out at the USTC Center for Micro and Nanoscale Research and Fabrication. The calculations were performed on the supercomputing system in USTC-SCC and Guangzhou-SCC.
Author information
Authors and Affiliations
Contributions
Y.C., H.L., and Wa.Z. equally contributed to this work. Y.C., H.L., and J.Ze. designed the studies and wrote the paper. Y.C., H.L., and Wenb.Z. synthesized catalysts. Y.C., H.L., and J.L. performed catalytic tests. Wa.Z. and Wenh.Z. performed DFT calculations. Y.C., X.Z., and J.Zh. conducted XPS measurements. W.L. and R.S. conducted XAFS measurements. H.L. and W.Y. conducted XANES measurements. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information: Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Y., Li, H., Zhao, W. et al. Optimizing reaction paths for methanol synthesis from CO2 hydrogenation via metal-ligand cooperativity. Nat Commun 10, 1885 (2019). https://doi.org/10.1038/s41467-019-09918-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-019-09918-z
This article is cited by
-
General synthesis and atomic arrangement identification of ordered Bi–Pd intermetallics with tunable electrocatalytic CO2 reduction selectivity
Nature Communications (2024)
-
Recent advances in metal-organic frameworks for catalytic CO2 hydrogenation to diverse products
Nano Research (2022)
-
Enhancing electrical conductivity of single-atom doped Co3O4 nanosheet arrays at grain boundary by phosphor doping strategy for efficient water splitting
Nano Research (2022)
-
Coordination tailoring of Cu single sites on C3N4 realizes selective CO2 hydrogenation at low temperature
Nature Communications (2021)
-
Lattice-strained nanotubes facilitate efficient natural sunlight-driven CO2 photoreduction
Nano Research (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.