Abstract
Antimony selenide (Sb2Se3) has a one-dimensional (1D) crystal structure comprising of covalently bonded (Sb4Se6)n ribbons stacking together through van der Waals force. This special structure results in anisotropic optical and electrical properties. Currently, the photovoltaic device performance is dominated by the grain orientation in the Sb2Se3 thin film absorbers. Effective approaches to enhance the carrier collection and overall power-conversion efficiency are urgently required. Here, we report the construction of Sb2Se3 solar cells with high-quality Sb2Se3 nanorod arrays absorber along the [001] direction, which is beneficial for sun-light absorption and charge carrier extraction. An efficiency of 9.2%, which is the highest value reported so far for this type of solar cells, is achieved by junction interface engineering. Our cell design provides an approach to further improve the efficiency of Sb2Se3-based solar cells.
Similar content being viewed by others

Introduction
Among inorganic semiconductor thin film photovoltaics, cadmium telluride (CdTe) and copper indium gallium selenide (Cu(In,Ga)Se2) solar cells have reached power-conversion efficiencies of over 22%1,2. The high device performance is possible due to the enough photon absorption, high bulk lifetime, superior carrier collection efficiency, and excellent junction interface. The chalcogenide antimony selenide (Sb2Se3) recently emerged as a promising alternative light-absorber material for high-efficiency photovoltaic devices due to its attractive properties, such as a single phase structure, proper optical bandgap (1.1–1.3 eV), high light absorption coefficient (105 cm−1 at around 600 nm), low toxicity, and high element abundance3,4,5,6,7,8. The use of the chalcogenide Sb2Se3 avoids the issue of low In and Ga availability. The application of Sb2Se3 in photovoltaic devices as light-absorber was explored by Nair et al. in 2000s, yielding a rather low conversion efficiency of 0.66%9,10. Since the notable efficiency values of 3.21% and 2.26% obtained in 2014 by Choi et al. and Zhou et al., respectively, Sb2Se3-based solar cells have experienced rapid development3,11. A power-conversion efficiencies of 6.0% was reported for a zinc oxide (ZnO)/Sb2Se3 heterojunction and 6.5% for a cadmium sulfide (CdS)/Sb2Se3 heterojunction with PbS quantum dot film as hole-transporting layer, respectively12,13. Moreover, a 7.6% efficiency was reported this year, due to an improved crystallinity of Sb2Se3 thin film absorbers14. However, for Sb2Se3 to become a low cost, high abundancy compound to replace Cu(In,Ga)Se2, this value is still too much behind that of state-of-the-art Cu(In,Ga)Se2 solar cells. We here present a concept based on growing Sb2Se3 nanorod arrays that can lead to fundamentally improved solar cells. This method thus far had led to cells with a certified efficiency of 9.2%.
One attractive feature of Sb2Se3 is that it has a one-dimensional (1D) crystal structure and highly anisotropic properties. The Sb2Se3 crystal consists of ribbon-like (Sb4Se6)n units linked through van der Waals forces in the [010] and [100] direction, while strong covalent Sb–Se bonds make the units holding together in the [001] direction3,15. This apparently direction-dependent bonding nature will result in significant anisotropy. Theoretical calculation revealed that the surfaces parallel to the [001] direction, such as (110), (120) surfaces, have lower formation energies than the other surfaces and were terminated with surfaces free of dangling bonds15. Moreover, theoretical calculations and experimental results exhibited that carrier transport in the [001] direction is much easier than that in other directions15,16. Thus, the devices are expected to offer appealing photoresponse and device performance if the Sb2Se3 absorber consists of (Sb4Se6)n ribbons stacked vertically on the substrate. However, up to date, only quality [221]-oriented absorbers have been fabricated, in which the (Sb4Se6)n ribbons were tilted and have a certain degree with the substrate. On the other hand, the optimal Sb2Se3 absorber thickness for these devices were limited to the range of 0.3–0.6 μm due to the electron diffusion length (Le) of only 0.3 μm in the [221] direction16. Due to this effect, the higher electron diffusion length Le along the [001] direction, which approaches 1.7 μm (five times that along the [221] direction16), could thus far not be fully exploited.
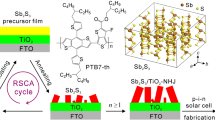
In this work, we address this limitation and grew Sb2Se3 nanorod arrays and solar cells with [001]-orientation on Mo-coated glass substrates using the close spaced sublimation (CSS) technique. A growth model is presented to investigate the mechanism covering the stages from atom absorption at the Mo surface to growth of the thin film structure towards the formation of aligned 1D Sb2Se3 nanorod arrays. We investigated the junction structure of the CdS/Sb2Se3 nanorod interface. We here reveal the migration of element antimony (Sb) into the whole CdS buffer layer if no specific precautions are taken. Subsequently, we introduce a very thin titanium oxide (TiO2) layer deposited by atomic layer deposition (ALD) technique at the CdS/Sb2Se3 junction interface. The interface engineering with TiO2 leads to an independently verified record power-conversion efficiency of 9.2% for the Sb2Se3 solar cells (ZnO:Al/ZnO/CdS/TiO2/Sb2Se3 nanorod arrays/MoSe2/Mo) with an absorber thickness over 1000 nm while maintaining a high fill factor of 70.3%. The values of external quantum efficiency (EQE) are higher than 85% in a wide spectral range from 550 to 900 nm, approximating the values of well-developed CdS/Cu(In,Ga)Se2 thin film solar cells. This work can facilitate the preparation and application of patterned 1D Sb2Se3-based nanostructures for applications in sensor arrays, piezoelectric antenna arrays, and other electronic and optoelectronic devices.
Results
Characterization of Sb2Se3 nanorod arrays
It is worth noting that, to our knowledge, the fabrication of high quality ribboned Sb2Se3 nanorod arrays on Mo-coated glass substrate by the CSS technique has not been previously reported. The surface and cross-sectional morphologies of the as-deposited Sb2Se3 nanorod arrays were characterized by scanning electron microscope (SEM) in Fig. 1a, b, respectively. A high density array of Sb2Se3 nanorods grown vertically on the substrate with diameters ranging from 100 to 300 nm and lengths of about 1200 nm was observed. The crystal structure and phase purity of the Sb2Se3 nanorod arrays were measured by X-ray diffraction (XRD) as depicted in Fig. 1c. The arrays exhibit the orthorhombic crystal geometry belonging to the space group of Pbnm (JCPDS 15-0861) with no detectable impurities of other phases. It is important to note that only strong (hk1) and (hk2) diffraction peaks are observed in the XRD pattern, suggesting that the Sb2Se3 nanorod arrays have a preferred orientation along the c-axis direction. The intensity ratios of I101/I221 and I002/I221 for the nanorod arrays reached 0.42 and 0.73, respectively. These ratios are much higher than those of thin films with the (221)-preferred orientation in previous reports12,17. Since the (221)-oriented grain consists of (Sb4Se6)n ribbons grown vertically to the substrate with a tilt angle, the increased I101/I221 and I002/I221 values hint that the Sb2Se3 nanorod arrays are grown with enhanced preference along the c-axis [001] direction and at a higher tilt angle between (Sb4Se6)n ribbons and the substrate, compared to the (221)-oriented thin films4,15. We further relied on high-resolution transmission electron microscopy (HRTEM) to reveal the crystal orientation of the individual Sb2Se3 nanorods. Samples were cross-sectioned by focused ion beam and a TEM image of the nanorod array is shown in Fig. 1d. The interplanar d-spacings of 0.389 nm and 0.521 nm correspond to the (001) and (210) planes of orthorhombic Sb2Se3, respectively, as shown in Fig. 1e, which is consistent with the 1D single-crystalline Sb2Se3 nanostructures synthesized by chemical synthesis methods18,19. The corresponding selected-area electron diffraction (SAED) pattern (Fig. 1f) exhibited the vertical relationship of the (001) and (210) planes, indicating the \(\left[ {1\bar 20} \right]\) crystallographic axis of the Pbnm space group and the Sb2Se3 nanorod, suggesting that the Sb2Se3 nanorod arrays in this work grow along the [001] direction. Analysis on additional Sb2Se3 nanorods further supported that the Sb2Se3 nanorod arrays were grown along the [001] direction (Supplementary Figure 1). The SAED characterization provides a direct observation of the atomic arrangement of the Sb2Se3 nanorod and echoes previous XRD and SEM results.
Microscopy and spectroscopy of Sb2Se3 nanorod array. a–c Top-view (a), cross-sectional (b), SEM images and X-ray diffraction pattern (c) of the Sb2Se3 nanorod arrays grown on Mo-coated glass substrate. d–f TEM image (d), high resolution TEM (HRTEM) image (e), and the corresponding selected-area electron diffraction (SAED) pattern (f) of the Sb2Se3 nanorod array
Growth model of Sb2Se3 nanorod arrays on Mo substrate
As shown in Fig. 2, a series of plan-view and cross-sectional SEM images of Sb2Se3 grown with different durations on Mo substrate exhibit the morphological evolution of Sb2Se3. It was found that with increasing growth durations from 60 to 180 s the morphologies of Sb2Se3 samples vary from a compact thin film structure to an aligned nanorod array structure. As seen from the corresponding cross-sectional images, the thickness of the Sb2Se3 layer was 200, 600, 1000 and 2000 nm for the samples grown for 60, 120, 160 and 180 s, respectively. It indicates that both thickness and growth rate are increased as the deposition proceeds. The CSS-processed Sb2Se3 is a smooth and compact film composed of grains with uniform grain size of about 100 nm in the first 60 s (Fig. 2a, e). When the growth time increases to 120 s, the grain size increases to 200–300 nm and the sample still displays film structure morphology, though the surface becomes porous and some craters can be observed (Fig. 2b, f). For the sample grown for 160 s (Fig. 2c, g), it is observed that the Sb2Se3 consists of a compact bottom layer and a nanorod-array top layer vertical to the substrate. The vertical nanorod array appears to grow on top of the compact bottom layer. As the growth times increases further, to 180 s, the thickness of the top nanorod-array layer increases while the compact bottom layer thickness shrinks (Fig. 2d, h).
Morphology evolution of Sb2Se3: from thin film to nanorod array. a–d Top-view SEM images of Sb2Se3 with different deposition times, a 60 s, b 120 s, c 160 s, and d 180 s. e–h The corresponding cross-sectional images of Sb2Se3 with different deposition time, e 60 s, f 120 s, g 160 s, and h 180 s. The scale bar for a–h is 1 μm
Based on the above observation, we propose a model to understand the mechanism governing the transition in the growth process from Sb2Se3 thin film to nanorod array. The growth process of Sb2Se3 can be divided into four stages: surface absorption, film growth, splitting, and nanorod array growth stage. For the first (surface absorption) stage, we have generated an atomistic model shown in Fig. 3a based on the following considerations: first, Sb2Se3 possesses a 1D crystal structure and is comprised of (Sb4Se6)n ribbons. Considering the combination between the (Sb4Se6)n ribbon and the substrate surface, we calculated the atom displacement distributions by the Vienna ab initio Simulation Package (VASP)20. The calculated results show that the Sb and Se atoms are dispersed from Sb4Se6 and scattered on the Mo surface and that the ribboned structure of Sb4Se6 collapses if the Sb4Se6 unit runs parallel to the Mo (110) surface (Supplementary Figure 2 and Supplementary Figure 3a). On the contrary, when the Sb4Se6 unit is standing vertically on the Mo (110) plane, the simulated results display that the unit is stable with lower distortion (Supplementary Figure 3b). Second, despite the decomposition of Sb2Se3 during the thermal process, the absorption of Sb or Se atoms at the Sb4Se6/Mo interface is also taken into account. The degree of lattice deformation for the Sb4Se6/Mo, Sb4Se6/Sb/Mo and Sb4Se6/Se/Mo absorption models, respectively, is 0.755, 0.642 and 0.534. This indicates that on the Mo surface the absorption of one Se atom layer prior to (Sb4Se6)n ribbons is favored rather than the vertical growth of (Sb4Se6)n ribbons (Supplementary Figure 3c, 3d and Supplementary Table 1).
Growth model of the Sb2Se3 nanorod arrays on Mo substrate. a Atomistic model of Sb4Se6 unit on the (110) plane of Mo. b–d Schematics of the Sb2Se3 at different growth stages, b thin film growth, c split, and d nanorod array growth (top part exhibits obvious nanorod array morphology and bottom is compact layer)
During film growth, splitting, and nanorod growth stages, the Sb2Se3 grains grow bigger as Sb2Se3 vapor continuously evaporates from the Sb2Se3 source, and then the transition from thin film to nanorod growth occurs when the generated lateral stress beyond the tolerance of the van der Waals forces between the (Sb4Se6)n ribbons in the deposited Sb2Se3 films. The nanorods get longer and more in number and the splitting goes deeper into the film as the growth time proceeds (Fig. 3d), which could be attributed to the higher growth rate in the ribbon direction due to the stronger covalent Sb–Se bonds internally in the ribbon.
Device performance and characterization
To investigate the effect of different absorber morphologies on the performance of the Sb2Se3 solar cells, the devices were finished by successively depositing the CdS buffer, high-resistance (HR) and low-resistance (LR) ZnO layer, and front Ag contact. The devices were divided into three groups according to the thicknesses and morphologies of the CSS-processed Sb2Se3 absorbers. For description clarity, we denoted the Sb2Se3 thin film absorbers with thickness between 200 and 600 nm as TF-Sb2Se3, the Sb2Se3 thickness in the range of 650 to 1100 nm, comprising a double layer (vertical nanorod-array top layer and compact-film bottom layer) as M-Sb2Se3, and Sb2Se3 absorbers thicker than 1100 nm with nearly an entire nanorod-array structure as NA-Sb2Se3.
Figure 4a displays representative current density-voltage (J-V) curves of the solar cells employing the TF-Sb2Se3, M-Sb2Se3, and NA-Sb2Se3 absorbers, respectively. Typical J-V characterizations performed under standard test conditions (STC) yielded an optimal conversion efficiency of 4.78% for the M-Sb2Se3 solar cell with an open circuit voltage (VOC) of 0.370 V, short circuit current density (JSC) of 27.43 mA cm−2, and fill factor (FF) of 47.46% (see Table 1). The NA-Sb2Se3 samples show substantially reduced FF, which may be attributed to strong CdS/Sb2Se3 interface recombination.
The presence of the nanorod structure in the absorbers increases the JSC of the solar cells, which is mainly due to the enhanced long wavelength response (Fig. 4b). The rough surface of the thicker nanorod absorber enhances the light harvesting and thus reduces the optical reflection (Supplementary Figure 4)21. At the same time, the [001] preferential orientation of the nanorods facilitates long-range carrier transport along the (Sb4Se6)n ribbons and thus guarantees carrier extraction and high JSC16. This is also supported by the EQE and biased EQE results. As shown in Fig. 4b, the EQE spectrum of the TF-Sb2Se3 device reaches a maximum value of 80% at about 550 nm, then declines both at shorter and longer wavelength due to the strong absorption of the CdS buffer and the insufficient generation and/or collection of carriers at the back side, respectively. This observation is consistent with previous reports of Sb2Se3-based thin film solar cells22,23. For the M-Sb2Se3 device, the maximum value of EQE reaches 88% at approximately 550 nm, higher than that of the TF-Sb2Se3 device, partly due to its lower reflectance. The EQE spectra of the NA-Sb2Se3 device demonstrates a relatively wide EQE plateau with values approaching 87% between 550 and 900 nm, and a gradual decrease towards longer wavelengths. EQE spectra were also measured under bias-voltage conditions (−0.5 V), and the curves describing the ratio of EQE (−0.5 V) over EQE (0 V) are shown in Fig. 4c. For the NA-Sb2Se3 device, the EQE ratio is approximately unity over the whole spectral range, while that of the TF-Sb2Se3 and M-Sb2Se3 devices is strongly bias dependent, especially at long wavelength. This indicates that the photogenerated carriers in the latter devices are not collected completely and the collection requires an internal electric field. The high and wide plateau and its weak bias-voltage dependence of the EQE spectrum of NA-Sb2Se3 device reveals that the carrier collection is highly efficient for the Sb2Se3 nanorod array structure along the [001] direction, explaining the higher JSC value of the NA-Sb2Se3 device compared to that of the M-Sb2Se3 device.
We then turned to the issue of the junction properties of the TF-Sb2Se3, M-Sb2Se3 and NA-Sb2Se3 solar cells. In order to understand their AC behavior, an equivalent circuit model was introduced. It consists of serial conductance, junction conductance and the capacitance element, which mainly includes the junction interface and trapping state induced capacitance. (Supplementary Figure 5). The junction capacitance is frequency independent while trapping capacitance is strongly frequency-dependent24,25,26. In comparison with the TF-Sb2Se3, the M-Sb2Se3 and NA-Sb2Se3 device exhibit smaller and less frequency dependent capacitances, indicating that the growth of Sb2Se3 nanorods reduces the defect density in the Sb2Se3 absorber or at its surface.
We further performed the capacitance–voltage (C-V) profiling and deep-level capacitance profiling (DLCP) measurements on these devices for characterizing the defects. In general, the C-V measurement is relation to free carriers, junction interface defects and bulk defects, while DLCP measurement is less sensitive to the junction interface defects27. As shown in Fig. 4d, the NDLCP values for these three devices are in the range of 4 × 1014 to 2 × 1015cm−3, which are lower than the values obtained for reference samples of Sb2Se3 grown on ZnO or TiO2 layer as well as for Sb2Se3 deposited by thermal evaporation on Mo substrate (4.6 × 1015 to 1.1 × 1017cm−3)12,17,28. This suggests that the CSS-processed Sb2Se3 absorbers on Mo substrate have a lower bulk defect density. On the other hand, NDLCP for TF-Sb2Se3 device was a little higher than that for M-Sb2Se3 and NA-Sb2Se3 devices, indicating the reduced bulk defect densities due to the evolution of Sb2Se3 from thin films to nanorod array structure. However, the NCV values were much higher than the NDLCP values for these three devices, indicating serious interface defects present at the CdS/Sb2Se3 interface. The depletion width (Wd) is mainly located in the Sb2Se3 region at the CdS/Sb2Se3 junction interface since the doping density of CdS is much higher than that of the Sb2Se3 absorbers16,29,30. Hence, the interfacial defect density could be calculated to be 2.77 × 1012 cm−2, 2.85 × 1012 cm−2and 3.21 × 1012 cm−2 for TF-Sb2Se3, M-Sb2Se3 and NA-Sb2Se3 devices, respectively. These values are higher than those of CdS/Sb2Se3, ZnO/Sb2Se3 or TiO2/Sb2Se3 in superstrate configurations, indicating that much more interface state activity can be expected for CBD-CdS buffer grown on Sb2Se3 absorbers12.
CdS/Sb2Se3 junction interface
To explore the coverage of CBD-CdS layer coated on the Sb2Se3 nanorod surface and the inter-diffusion of elements at the CdS/Sb2Se3 interface, we employed SEM, TEM, and high-angle annular dark-field scanning transmission electron microscope (HAADF-STEM) equipped with energy-dispersive spectroscopy (EDX) to characterize the interface of our CdS-coated Sb2Se3 nanorod array samples. As shown in Fig. 5a, b, the CBD growth procedure yields a uniform, dense, and pin-hole free CdS film, and the CdS layer completely covers the Sb2Se3 nanorod array surface, yielding a CdS/Sb2Se3 core-shell structure. The morphology of the CdS film reveals a fine-grain accumulated structure. The TEM image (Fig. 5c, d) displays that the thickness of the CdS coated at the top of the Sb2Se3 nanorods is about 50 to 60 nm. More details on the CdS film growth on the Sb2Se3 nanorod arrays reveal that the CBD-CdS is not only present on top of the nanorods but also penetrates into the space between the nanorods and conformally coats the sidewalls of the nanorods and in the valleys on the bottom compact layer, making the cell at least partially a radial junction cell. The uniform and complete coverage of CdS layer suggests good adhesion and well defined junction formation between the Sb2Se3 nanorod array and the CdS buffer layer.
Characterization of CdS/Sb2Se3 junction interface. a, b Top-view (a) and cross-sectional (b) SEM images of CdS buffer deposited on Sb2Se3 nanorod arrays. c–e TEM (c, d) and HAADF-STEM image and energy-dispersive spectroscopy elemental mapping (e) of the CdS/Sb2Se3 junction interface. Elements detected: Sb L, Se L, Cd L, and S L
A rectangular area in the Z-contrast HAADF cross-sectional image at the CdS/Sb2Se3 nanorod interface was chosen to analyze the Sb, Se, Cd, and S element distribution. As shown in Fig. 5e, element spatial mapping of Se, Cd, and S shows sharp edges, indicating negligible interfacial inter-diffusion of these three elements. On the contrary, the Sb element mapping exhibits an obvious two-zone behavior in the CdS/Sb2Se3 nanorod interface region, suggesting Sb-diffusion into the CBD-CdS layer. This phenomenon is quite different from the superstrate CdS/Sb2Se3 heterojunction case, in which the Cd, S, Sb, and Se elements mix together to form a thin n-type inter-diffusion layer and a buried homojunction at the interface, dictating charge separation and device performance in superstrate CdS/Sb2Se3 thin film solar cells. The presence of Sb in the whole CBD-processed CdS buffer layer can be attributed to the dissolution of Sb2Se3 in the alkaline precursor solution (Supplementary Table 2). During the CBD process, some ammonia was added into the precursor solution to supply a suitable environment for the chemical reactions, and thus it also reacted with the precursor to form surface growth complexes31,32. For reference, the metal chalcogenide was dissolved in hydrazine or ammonia sulfide solution through the formation of highly soluble metal chalcogenide complexes at a molecular level33,34,35. A similar dissolution process is expected to occur in the reaction of Sb2Se3 with NH4+ during the deposition of the CdS layer in an ammonia solution.
Surface modification of Sb2Se3 nanorod arrays by thin ALD-TiO2
In order to address the issue of Sb diffusion and the concomitant high interface defect density, a very thin atomic layer deposited (ALD) TiO2 layer was introduced between the Sb2Se3 nanorod array absorber and the CdS buffer to protect the Sb2Se3 from directly contacting the NH4+ ions during the deposition of the CdS layer by CBD method. The EDX line scan analysis shows that the Sb content in the CdS layer was reduced for the CdS shell grown on ALD-TiO2 modificated Sb2Se3 nanorod (Supplementary Figure 6). The decrease of Sb content in the CdS shell indicated that the thin ALD-TiO2 could efficiently reduce the dissolution of Sb2Se3 during the CBD process. Moreover, the corrosion rate of Sb2Se3 layer in the ammonia solutions is slightly decreased after performing 20 cycles of TiO2 (Supplementary Figure 7 and Supplementary Table 2). Figure 6b, c exhibit the top-view and cross-sectional images of the Sb2Se3 solar cells after applying all steps to a successfully completed fabrication. The device exhibits a stamp-like nanopatterned surface morphology and fewer holes and gaps are observed in the cross-sectional image, suggesting that the CBD-processed CdS and sputtered ZnO/ZnO:Al completely covers the top of the Sb2Se3 nanorods as well as the lower parts within the space between nanorods.
Solar cell structure and mechanistic investigation of ALD-TiO2 on Sb2Se3 nanorod arrays. a Schematic of the Sb2Se3 nanorod arrays on Mo-coated glass and finished Sb2Se3/CdS core/shell nanorod array solar cells. b, c Cross-sectional (b) and top view (c) SEM images of the completed CdS/Sb2Se3 solar cells. d, e J-V curve (d) and EQE spectrum (e) of the champion device (area = 0.2603 cm2). f Histogram of device efficiency over 100 individually fabricated solar cells. g VOC decay curves of the solar cells with and without ALD-TiO2 layer
Figure 6d displays the J-V curve of our best device in this work under simulated AM1.5 G solar illumination. This device was fabricated with 20 cycles of ALD TiO2 on the Sb2Se3 absorber prior to the deposition of the CdS buffer. The cell exhibits a VOC of 0.40 V, a JSC of 32.58 mA cm−2, a FF of 70.3%, resulting in an overall power-conversion efficiency of 9.2%, which has independently been verified by National Institute of Metrology of China (Supplementary Figure 8). A histogram of the device efficiencies obtained from 100 individually fabricated devices is shown in Fig. 6f. The average VOC, JSC, FF, and conversion efficiency were 399 ± 33 mV, 29.80 ± 3.36 mA cm−2, 64.46 ± 12.01% and 7.69 ± 1.56%, respectively. Figure 6e depicts the corresponding EQE spectrum for the champion solar cell. It exhibits a broad plateau of over 85% between 550 nm and 900 nm and the integrated current density reaches a value as high as 31.48 mA cm−2. The photoresponse in the plateau region is higher than that of Sb2Se3 solar cells in a superstrate configuration and is comparable with that of CdS/CIGS thin film solar cells prepared in our laboratory with an efficiency of 15%, as shown in Supplementary Figure 9. Nonetheless, there is large current loss at wavelengths below 550 nm due to strong parasitic absorption of the CdS buffer since the electron-hole pairs generated in the CdS layer are not collected. Therefore, it is desirable to replace the CdS with another wide band gap buffer material for further optimization.
Compared with the device without ALD-TiO2 (Fig. 4, Supplementary Figure 10), the enhancement in conversion efficiency mainly results from an increase in VOC and FF, which is tentatively attributed to the reduction of dissolution of Sb2Se3 during the CBD process and/or the reduction of shunt paths by the ALD-TiO2 of the surface defects on the Sb2Se3 nanorods (the dangling bonds at the tips of the (Sb4Se6)n nanoribbons). As shown in Supplementary Figure 11, Kelvin probe force microscope (KPFM) was employed to study the surface properties of the Sb2Se3 nanorod array surfaces before and after the deposition of 20 cycles of TiO2. While the average roughness stays at the same value (100 nm), the average surface potential difference decreases from 28.8 to 10.4 mV after the deposition of the thin ALD-TiO2. This suggests that a thin layer of ALD-TiO2 improves the surface band bending at the side walls and reduces the surface defects at the tips of the Sb2Se3 nanorods15,36. A surface potential difference of 280 mV was observed between the Sb2Se3 layer before and after thin ALD-TiO2 modification. Taking into account of the valence band maximum (VBM) and band gap of Sb2Se3, we obtained the energy level diagram of the CdS/(TiO2) Sb2Se3 interface (Supplementary Figure 12). The conduction band minimum (CBM) of Sb2Se3 layer is shifted by about 0.13 eV towards to the vacuum level after ALD-TiO2 modification. The downshifted of the CBM could decrease the conduction band offset at buffer/absorber interface, and lead to the increased fill factor. Furthermore, the possible shunt paths for the CdS/Sb2Se3 junction with and without ALD-TiO2 were detected by conductive atomic force microscopy (C-AFM). For the sample without ALD-TiO2 some white dots, representing the detected current, are observed (Supplementary Figure 13), indicating the poor coverage of CdS and the presence of shunt leakage due to local discontinuity or pinholes in the CdS buffers. On the contrary, with the insertion of thin ALD-TiO2 between the CdS buffer and the Sb2Se3 nanorod array absorber, the white dotted area decreases or even vanishes, suggesting reduced shunt leakage.
The ALD-TiO2 layer may also passivate the surface defects of the Sb2Se3 layer. This can be confirmed by the VOC decay measurement, which is related to the carrier recombination rate and the carrier lifetimes. Figure 6g displays the VOC decay curves of two representative Sb2Se3 solar cells, with and without ALD-TiO2 thin layer. The cell with 20 cycles of ALD-TiO2 layer exhibits an obvious longer decay time than the cell without ALD-TiO2. Furthermore, as the thin ALD-TiO2 layer is compact and has excellent film conformity due to its layer-by-layer growth, it is expected to reduce or even prevent the chemical reaction of Sb2Se3 with the growth solution during the CBD deposition of the CdS buffer layer, leading to a more pure CdS buffer layer. The influence of doping of Sb in CdS buffer layers has not been exclusively demonstrated thus far and requires more investigation in the near future. We investigate the stability of the Sb2Se3 nanorod array based solar cells. As shown in Fig. 7, the normalized efficiency of the CdS/Sb2Se3 solar cell with TiO2 modification hold a slightly higher value (~97% of its initial value) than that of the device without TiO2 modification (~94% of its initial value) after storage in air for more than 500 h.
Discussion
In summary, we have demonstrated the fabrication of high quality solar cells employing a 1D Sb2Se3 nanorod array absorber with a height of more than 1000 nm in the substrate configuration. TEM analysis indicated that the growth of nanorods is along the [001] direction. We propose a split growth model based on the morphology evolution from the thin film to a nanorod array. The solar cells exhibited excellent EQE spectra in the whole working wavelength range (higher than 85% between 550 and 900 nm), indicating that there is long-range carrier transport along the [001] direction. Furthermore, we found that Sb diffuses into the CdS buffer due to the solubility of Sb2Se3 in the alkaline solution during the CBD process. A very thin TiO2 layer deposited by ALD was introduced prior to the deposition of CdS buffer layer, leading to an improved VOC, FF as well as conversion efficiency. This cell design and these results provide important progress towards the understanding and application of 1D-structured Sb2Se3 crystals.
Methods
Solar cell fabrication
The bilayer Mo back contacts were prepared by a two-step magnetron sputtering process, which consisted of high working pressure (2.0 Pa) and low working pressure (0.3 Pa) process. The total thickness of Mo was about 1000 nm. A Mo selenization process was carried out at 620°C for 20 min to form about 20 nm thick MoSe2 layer prior to the deposition of Sb2Se317. The Sb2Se3 absorber layers were grown on selenized Mo-coated glass by using a homemade CSS system. In CSS system, the thermocouple was inserted into the graphite plate to directly detect the temperatures of substrate and evaporation source, respectively. The temperatures of Sb2Se3 source and substrate holder were controlled by two sets of lamp heaters and thermocouples. The distance between the source and the sample holder was 11 mm. We started the deposition when the pressure was below 10−2 Pa. First, the source and sample holder were warmed up to 480 °C and 270 °C, respectively, in 200 seconds, and maintained at the high temperatures for hundreds of seconds to obtain the desired Sb2Se3 absorber thickness. The thicknesses of the Sb2Se3 layers in the range of 200–2000 nm were controlled by adjusting the duration ranging from 60–180 s at high temperature. The samples were taken out after cooling down to about 150 °C in about 1 h. After that, the Sb2Se3 samples were coated with 60 nm of CdS by chemical bath deposition at a bath temperature of 70 °C. Window layers of HR and LR ZnO films were sputtered from pure ZnO and ZnO:Al targets (Al2O3 2 wt%-doped). Top Ag grids of the solar cells were finally formed by thermal evaporation. The complete Sb2Se3 solar cells have a structure of glass/Mo/MoSe2/Sb2Se3/HR-ZnO/LR-ZnO/Ag. TiO2 was deposited at 150 °C in a homemade ALD reactor system, which using titanium isopropoxide (TTIP) and H2O as Ti and O precursors, respectively. One deposition cycle involves a H2O pulse of 0.5 s, a N2 pulse of 60 s, a TTIP pulse of 0.5 s, and 60 s of N2 purging, and each deposition cycle was started with a H2O pulse and terminated with a TTIP pulse. About 2 nm thickness of TiO2 coating was deposited in 20 cycles.
Material and device characterization
SEM observations were performed on a FEI Nova NANOSEM 450 field-emission microscope and the TEM measurements were carried out on a FEI Tecnai G2 transmission electron microscope. The optical properties were recorded using a Perkin-Elmer Lambda 950 spectrophotometer. The XRD data were collected with a Bruker D8 Advance diffractometer. The current density-voltage (J-V) measurement was performed using an AM1.5 solar simulator equipped with a 300 W Xenon lamp (Model No. XES-100S1, SAN-EI, Japan). The EQE was measured by an Enlitech QER3011 system equipped with a 150 W xenon light source. Capacitance-voltage (C-V) measurement was performed on Agilent B1500A Semiconductor device analyzer in the dark at room temperature. Carrier-lifetime measurements were performed using the DN-AE01 Dyenamo toolbox with a white light-emitting diode (Luxeon Star 1W) as the light source37,38.
Simulation methods
All calculations of Sb2Se3 growing on the Mo (110) were calculated by the VASP. The DFT calculations employed the Perdew-Burke-Ernzerhof (PBE) generalized gradient approximation (GGA) exchange-correlation functional and the projector-augmented wave (PAW) method. An energy cut-off of 500 eV was applied for the plane wave expansion of the wave functions. 2 × 4 × 1 Monkhorst-pack mesh for k-point sampling are required to relaxation all models of the Sb2Se3 sheet growing on the Mo (110) with or without Se and Sb layers.
Data availability
The data supporting this study are available from the authors on request.
References
Green, M. A. et al. Solar cell efficiency tables (version 51), Prog. Photovolt. Res. Appl. 26, 3–12 (2018).
Solar Frontier. Solar Frontier achieves world record thin-film solar cell efficiency of 22.9%. Solar Frontier http://www.solar-frontier.com/eng/news/2017/1220_press.html, Accessed June 2018 (2017).
Zhou, Y. et al. Solution-processed antimony selenide heterojunction solar cells. Adv., Energy Mater. 4, 1079–1083. (2014).
Liang, G. X. et al. Thermally induced structural evolution and performance of Sb2Se3 films and nanorods prepared by an easy sputtering method. Sol. Energy Mater. Sol. Cells 174, 263–270 (2018).
Yuan, C., Zhang, L., Liu, W. & Zhu, C. Rapid thermal process to fabricate Sb2Se3 thin film for solar cell application. Sol. Energy 137, 256–260 (2016).
Birkett, M. et al. Band gap temperature-dependence of close-space sublimation grown Sb2Se3 by photo-reflectance. APL Mater. 6, 084901 (2018).
Wang, X., Tang, R., Wu, C., Zhu, C. & Chen, T. Development of antimony sulfide–selenide Sb2(S, Se)3-based solar cells. J. Energy Chem. 27, 713–721 (2018).
Shen, K. et al. Mechanisms and modification of nonlinear shunt leakage in Sb2Se3 thin film solar cells,. Sol. Energy Mater. Sol. Cells 186, 58–65 (2018).
Sarah, M., Nair, M. T. S. & Nair, P. K. Antimony selenide absorber thin films in all-chemically deposited solar cells. J. Electrochem. Soc. 156, H327–H332 (2009).
Bindu, K., Nair, M. T. S. & Nair, P. K. Chemically deposited Se thin films and their use as a planar source of selenium for the formation of metal selenide layers. J. Electrochem. Soc., 153, C526–C534 (2006).
Choi, Y. C. et al. Sb2Se3-sensitized inorganic–organic heterojunction solar cells fabricated using a single-source precursor. Angew. Chem. Int. Ed. 53, 1329–1333 (2014).
Wang, L. et al. Stable 6%-efficient Sb2Se3 solar cells with a ZnO buffer layer. Nat. Energy 2, 17046 (2017).
Chen, C. et al. 6.5% certified efficiency Sb2Se3 solar cells using PbS colloidal quantum dot film as hole-transporting layer. ACS Energy Lett. 2, 2125–2132 (2017).
Wen, X. et al. Vapor transport deposition of antimony selenide thin film solar cells with 7.6% efficiency. Nat. Commun. 9, 2179 (2018).
Zhou, Y. et al. Thin-film Sb2Se3 photovoltaics with oriented one-dimensional ribbons and benign grain boundaries. Nat. Photon 9, 409–415 (2015).
Chen, C. et al. Characterization of basic physical properties of Sb2Se3 and its relevance for photovoltaics, Front. Optoelectron 10, 18–30 (2017).
Li, Z. et al. Sb2Se3 thin film solar cells in substrate configuration and the back contact selenization. Sol. Energy Mater. Sol. Cells 161, 190–196 (2017).
Zhai, T. et al. Single-crystalline Sb2Se3 nanowires for high-performance field emitters and photodetectors. Adv. Mater. 22, 4530–4533 (2010).
Xie, Q. et al. Polymer-controlled growth of Sb2Se3 nanoribbons via a hydrothermal process. J. Cryst. Growth 252, 570–574 (2003).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Zhu, J. et al. Optical absorption enhancement in amorphous silicon nanowire and nanocone arrays. Nano Lett. 9, 279–282 (2009).
Liu, X. et al. Improving the performance of Sb2Se3 thin film solar cells over 4% by controlled addition of oxygen during film deposition. Prog. Photovolt. Res. Appl. 23, 2892 (2015).
Liu, X. et al. Enhanced Sb2Se3 solar cell performance through theory-guided defect control. Prog. Photovolt. Res. Appl. 25, 861–870 (2017).
Proskuryakov, Y. Y. et al. Impedance spectroscopy of thin-film CdTe/CdS solar cells under varied illumination. J. Appl. Phys. 106, 024504 (2009).
Walter, T., Herberholz, R., Müller, C. & Schock, H. W. Determination of defect distributions from admittance measurements and application to Cu(In,Ga)Se2 based heterojunctions. J. Appl. Phys. 80, 4411–4420 (1996).
Kneisel, J., Siemer, K., Luck, I. & Braunig, D. Admittance spectroscopy of efficient CuInS2 thin film solar cells. J. Appl. Phys. 88, 5474–5481 (2000).
Duan, H. S. et al. The role of sulfur in solution-processed Cu2ZnSn(S,Se)4 and its effect on defect properties. Adv. Funct. Mater. 23, 1466–1471 (2013).
Chao, C. et al. Accelerated optimization of TiO2/Sb2Se3 thin film solar cells by high-throughput combinatorial approach. Adv. Energy Mater. 7, 1700866 (2017).
Cwil, M., Igalson, M., Zabierowski, P. & Siebentritt, S. Charge and doping distributions by capacitance profiling in Cu(In,Ga)Se2 solar cells. J. Appl. Phys. 103, 063701 (2008).
Decock, K., Zabierowski, P. & Burgelman, M. Modeling metastabilities in chalcopyrite-based thin film solar cells. J. Appl. Phys. 111, 043703 (2012).
Maticiuc, N., Katerski, A., Danilson, M., Krunks, M. & Hiie, J. XPS study of OH impurity in solution processed CdS thin films. Sol. Energy Mater. Sol. Cells 160, 211–216 (2017).
Ortega Borges, R. & Lincot, D. Mechanism of chemical bath deposition of cadmium sulfide thin films in the ammonia-thiourea system: in situ kinetic study and modelization. J. Electrochem. Soc. 140, 3464–3473 (1993).
Xia, Z. et al. Generalized water-processed metal chalcogenide complexes: synthesis and applications. Chem. Mater. 27, 8048–8057 (2015).
Mitzi, D. B. Solution processing of chalcogenide semiconductors via dimensional reduction. Adv. Mater. 21, 3141–3158 (2009).
Protesescu, L. et al. Atomistic description of thiostannate-capped CdSe nanocrystals: retention of four-coordinate SnS4 motif and preservation of Cd-rich stoichiometry. J. Am. Chem. Soc. 137, 1862–1874 (2015).
Jiang, C.-S. et al. How grain boundaries in Cu(In,Ga)Se2 thin films are charged: Revisit. Appl. Phys. Lett. 101, 033903 (2012).
Freitag, M. et al. Dye-sensitized solar cells for efficient power generation under ambient lighting. Nat. Photon. 11, 372 (2017).
Boschloo, G., Häggman, L. & Hagfeldt, A. Quantification of the effect of 4-tert-butylpyridine addition to I−/I3− redox electrolytes in dye-sensitized nanostructured TiO2 solar cells. J. Phys. Chem. B 110, 13144–13150 (2006).
Acknowledgements
This work was supported by the Advanced Talents Incubation Program of the Hebei University (801260201001), National Natural Science Foundation of China (NSFC No.61804040), Scientific Research Foundation for the Returned Overseas Chinese Scholars (CG2015003004), and Natural Science Foundation of Hebei Province (No.E2016201028).
Author information
Authors and Affiliations
Contributions
Z.L. and Y.M. conceived the idea and designed the experiments. Z.L., X.L. and G.L. performed most of the device fabrication and characterization. H.L., H.Z. and W.Y. conducted the TiO2 deposition. K.S. and X.S. assisted in the TEM and EDX mapping characterization and data analysis. J.G. and J.C. carried out the theoretical simulation and analyzed the results. Z.L., R.E.I.S., and Y.M. analyzed the overall results and wrote the paper. Y.M. supervised the project and all authors discussed the experiments and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information: Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Z., Liang, X., Li, G. et al. 9.2%-efficient core-shell structured antimony selenide nanorod array solar cells. Nat Commun 10, 125 (2019). https://doi.org/10.1038/s41467-018-07903-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-018-07903-6
This article is cited by
-
Tri-chalcogenides (Sb2S3/Bi2S3) solar cells with double electron transport layers: design and simulation
Discover Energy (2024)
-
Improved performances in Sb2Se3 solar cells based on CdS buffered TiO2 electron transport layer
Journal of Sol-Gel Science and Technology (2024)
-
Vapor-Phase Epitaxial Growth of Large-Scale High Crystalline Sb2Se3 Nanowires for Photodetector Application
Journal of Electronic Materials (2023)
-
rGO spatially confined growth of ultrathin In2S3 nanosheets for construction of efficient quasi-one-dimensional Sb2Se3-based heterojunction photocathodes
Science China Materials (2023)
-
Hydrogel protection strategy to stabilize water-splitting photoelectrodes
Nature Energy (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.