Abstract
The material class of rare earth nickelates with high Ni3+ oxidation state is generating continued interest due to the occurrence of a metal-insulator transition with charge order and the appearance of non-collinear magnetic phases within this insulating regime. The recent theoretical prediction for superconductivity in LaNiO3 thin films has also triggered intensive research efforts. LaNiO3 seems to be the only rare earth nickelate that stays metallic and paramagnetic down to lowest temperatures. So far, centimeter-sized impurity-free single crystal growth has not been reported for the rare earth nickelates material class since elevated oxygen pressures are required for their synthesis. Here, we report on the successful growth of centimeter-sized LaNiO3 single crystals by the floating zone technique at oxygen pressures of up to 150 bar. Our crystals are essentially free from Ni2+ impurities and exhibit metallic properties together with an unexpected but clear antiferromagnetic transition.
Similar content being viewed by others
Introduction
The rare earth nickelates RNiO3 (R = rare earth, Y) with the high Ni3+ oxidation state have continued to attract enormous interest due to the famous bandwidth controlled metal-insulator (MI) transition and associated unusual charge and spin-order phenomena occurring in this system1,2 with even the possibility for multiferroicity3. The description of the underlying physics of these phenomena turned out to be a true intellectual challenge and it is not a surprise that important theoretical concepts have been (and still need to be) developed along the way4,5,6,7,8,9,10,11,12. More recently, the prediction of high-T c superconductivity in LaNiO3-based heterostructures13 has triggered a flurry of new activities on LaNiO3–LaMO3 superlattices (M = other metal ion)14,15.
Apart from LaNiO3, all RNiO3 (R = Pr-Lu, Y) compounds exhibit a MI transition at lower temperatures1,2 with an insulating antiferromagnetic ground state. With decreasing R-ionic radius, the octahedral tilts become larger. Thus, the Ni–O–Ni bond angles become smaller, which alters the electronic bandwidth and the magnetic exchange interactions. Whereas an enhancement of the insulating properties and an increase of the MI transition temperature T MI can be observed for decreasing R-ionic radius, the antiferromagnetic transition temperature and the strength of the exchange interactions decrease with decreasing R-ionic radius and Ni–O–Ni bond angles. With La having the largest ionic radius of the series, one might expect the strongest antiferromagnetic properties for LaNiO3. However, so far it was reported that LaNiO3 does not show any magnetic order, and thus, violates the trend. It remains a paramagnetic metal down to lowest temperatures with an enhanced effective mass1,2,16,17.
Here, we report on the successful growth of large LaNiO3 single crystals by the floating zone technique. Electrical resistivity and Hall effect measurements on our single crystals show that LaNiO3 is intrinsically not a bad metal as recently discussed for the RNiO3 (R = Pr-Lu, Y) compounds in their paramagnetic phase10. On the contrary, we found that LaNiO3 has a high conductivity. Moreover, we were able to observe bulk antiferromagnetism in magnetization, specific heat, and neutron scattering experiments. Thus, LaNiO3 appears to be a highly metallic and antiferromagnetic transition metal oxide—a rather rare combination in oxides. Special about LaNiO3 is that it is also very close to an insulating state, making LaNiO3 an intriguing quantum material, probably close to a quantum critical point, where strong local electronic correlations at the Ni sites are likely to interfere in an intricate manner with Fermi surface effects.
Results
Crystal growth and characterization
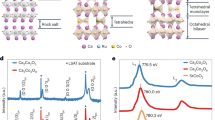
Using high oxygen pressures of 130–150 bar, we were able to grow LaNiO3 single crystals with the floating zone technique, see inset of Fig. 1a. Further details can be found in the Methods section and in Supplementary Notes 1 and 2, in Supplementary Figures 1 and 2, and in Supplementary Movie 1. Powdered crystals exhibit no impurity phases within the accuracy of powder X-ray diffraction measurements, see Fig. 1a. The single crystallinity of our LaNiO3 crystal is confirmed by a series of Laue diffraction patterns from different directions and across the length of this crystal as well as by single crystal X-ray diffraction, see inset of Fig. 1a and Supplementary Figure 1, as well as Supplementary Note 3, Supplementary Figure 3, and Supplementary Table 1. Due to the occurrence of a cubic to rhombohedral phase transition (Pm \(\overline 3 \) m \( \to \) R \(\overline 3 \) c) somewhat below 1100 K2, our floating zone grown single crystals are twinned.
Characterization of LaNiO3. a Powder X-ray diffraction pattern of a powdered LaNiO3 single crystal. The inset shows a photo of the LaNiO3 single crystal and its Laue diffraction pattern. A length scale of 1 cm is indicated by the yellow bar within the photo of the crystal and the distance between two lines in the Laue pattern amounts to 3.75 cm. b The Ni-L2,3 XAS spectra of the LaNiO3 single crystal measured at 90 and 300 K together with the spectrum of a NiO reference compound. c The O-K XAS spectra of our LaNiO3 single crystal measured at 300 K (blue line) together with the spectra of the NiO reference compound (red line)
Figure 1b shows the Ni-L2,3 X-ray absorption spectroscopy (XAS) data of the LaNiO3 single crystal taken at 300 and 90 K together with that of a NiO single crystal serving as a Ni2+ reference compound. We have removed the La-M4 white lines located at 850.6 eV from the LaNiO3 spectra using the La-M4 spectrum of LaCoO3. It is well known that XAS spectra at the L2,3 edge of transition metal oxides are highly sensitive to the valence state, in particular, an increase of the valence state of the transition metal ion by one causes a shift of the XAS L22,3 spectra by one or more eV toward higher energies18. The more than one eV higher energy shift between the spectra of NiO and LaNiO3 indicates the formal Ni3+ valence state in LaNiO3 18,19, with the note that the spectrum cannot be interpreted in terms of a Ni 3d7 configuration, but, rather by a coherent mixture of 3d8 and 3d8 \(\underline L \) \(\underline L \) configurations4,5,9,12, where each \(\underline L \) denotes a hole in the oxygen ligand. Here we can exclude Ni2+ impurities in our LaNiO3 single crystal—otherwise the sharp main peak of the Ni2+ impurity spectrum would have been visible as a sharp shoulder at the leading edge18. At the O-K edge, the pre-edge peak is shifted by about 1 eV to higher energies when going from Ni3+ to Ni2+ 18,19. Figure 1c shows the O-K XAS spectra of our LaNiO3 single crystal (blue) and the NiO reference compound (red). The O-K XAS spectra demonstrate even more clearly that there is no spectral feature from Ni2+ impurities and thus that our as-grown LaNiO3 single crystals are highly stoichiometric. This is further confirmed by thermogravimetric and inductively coupled plasma optical emission spectroscopy (ICP-OES) measurements, see Methods and Supplementary Note 2.
Temperature dependence of physical properties
In Fig. 2a, we show the temperature dependence of the lattice parameters of our LaNiO3 single crystal that has been powdered and measured by means of powder X-ray diffraction. There is no clear indication for the presence of a structural anomaly which otherwise occurs readily in the other nickelates RNiO3 with smaller rare earths (R = Pr-Lu, Y) when cooling through the MI transition1,2.
Temperature dependence of physical properties of LaNiO3. a Lattice parameters of our LaNiO3 as a function of temperature (space group R \(\bar 3\) c). The error bars are the standard deviations obtained from Rietveld refinement using the Fullprof software. b Electrical resistivity of LaNiO3 (gray). The red line indicates a fit to \(\rho _0 + AT^n\) (for T <35 K) and the dashed green line is a fit over the entire temperature range. c Magnetic susceptibility of LaNiO3 under field cooled (FC) and zero-field cooled (ZFC) conditions for applied fields of μ0 H = 0.005 T and 5 T. The high- and low-field measurements both together indicate an antiferromagentic transition at T N that is an intrinsic property of LaNiO3. d Specific heat measurements of LaNiO3 in fields of 0 T, 1 T, and 9 T. These measurements further corroborate that the antiferromagnetic transition is a bulk property of LaNiO3
The electrical resistivity ρ of our LaNiO3 single crystal that is shown in Fig. 2b is in the μΩ cm range, reaching ~6 μΩ cm at low temperatures, i.e., distinctly lower than that of LaNiO3 powder samples1,2,16,17 or LaNiO3 thin films14 or of crystals grown under 50 bar oxygen pressure20. Thus, our LaNiO3 single crystals are more metallic, see also Supplementary Note 4 and Supplementary Figure 4(a). Also the measurement of the Hall effect on the single crystal LaNiO3 (for temperatures between 2 and 300 K) yields high carrier densities of about 4.8 × 1028 m−3, corresponding to ~2.7 (close to 3) electrons per formula unit, consistent with the 3+ valence of the Ni ions in LaNiO3. Up to ~35 K, the low-temperature behavior of the resistivity is Fermi-liquid-like, ρ(T) = \(\rho _0 + AT^n\), with \(\rho _0\) ~6.45 μΩ cm, A = 1.62.10−3 μΩ cm K−2, and n ~2.0, see Fig. 2b. The value of A is of the same order as reported in literature21. A fit over the entire temperature range gives an exponent n ~1.50(1) with the residual resistivity \(\rho _0\) ~4.83 μΩ cm, similar to the value of the fit at low temperatures. Most probably this behavior is a signature that LaNiO3 is close to a quantum critical point (cf. the results for PrNiO3 under pressure21).
Figure 2c displays the temperature dependence of the magnetic susceptibility χ of our LaNiO3 single crystal, see also Supplementary Note 4 and Supplementary Figure 4(b). First of all, we notice that it shows a significantly smaller low-temperature upturn than reported previously for powder and ceramic samples16,17, which confirms unprecedented high quality of our single crystals. Surprisingly, χ exhibits an anomalous kink at ~157 K, which we take as an indication for a hitherto unknown antiferromagnetic transition in LaNiO3. That this anomaly is not simply caused by a signal from a tiny fraction of a magnetic impurity phase (which is so small that it is not visible in our powder X-ray diffraction measurements) can be excluded by our specific heat (C p) measurements. As can be seen in Fig. 2d, there is a small but clearly visible anomalous peak at ~157 K in \(C_{\mathrm{p}}/T\). Also the resistivity data excludes that the anomaly in the susceptibility is caused by the presence of an oxygen-deficient LaNiO3 minority phase that becomes antiferromagnetic and insulating at low temperatures22,23: we do not observe an upturn or a slowing down of the decrease in the resistivity on cooling. Moreover, our samples have resistivities in the μΩ cm range and have conductivities higher than reported so far. All these support the notion that the transition is an intrinsic bulk property and not due to an impurity phase. The low-temperature behavior of the specific heat supports a Fermi-liquid type of the ground state of LaNiO3, C p(T) = \(\gamma T + \beta T^3\), with \(\gamma \) ~17 mJ mol−1 K−2, consistent with the value 18 mJ mol−1 K−2 reported before17, and with β = 1.87(5) × 10−4 J mol−1 K−4, thus yielding a Debye temperature \({\theta _{\mathrm{D} }= (12\pi ^4NR/5\beta )^{1/3}}\) = 373 K, where N is the number of atoms in the chemical formula and R is the ideal gas constant.
Neutron scattering experiments
The availability of sizeable LaNiO3 single crystals also enabled us to study this intriguing system by means of neutron diffraction and inelastic neutron scattering. These experiments were performed at the Thales, IN12 and IN8 spectrometers at the ILL in Grenoble, France. Within elastic scans, we were able to observe quarter-integer peaks at low temperatures, see Fig. 3a. These quarter-integer peaks (in pseudocubic notation) resemble those found in powder neutron-diffraction experiments within the insulating regime of RNiO3 24. The propagation vector observed for the insulating antiferromagnetic regime of RNiO3 amounts to (1/4 1/4 1/4) in pseudocubic notation (or (1/2 0 1/2) in orthorhombic notation)2. Moreover, the study of the temperature dependence of these quarter-integer peaks indicates an onset temperature that coincides with the magnetic ordering temperature T N that we observed in susceptibility and specific heat measurements of LaNiO3, see Fig. 3b.
Single crystal neutron-diffraction measurements. a Neutron scattering intensities within the (H H L) plane of reciprocal space (in pseudocubic notation) of the LaNiO3 single crystal. These intensites were obtained at 1.6 K. b The temperature dependence of a quarter-integer peak as measured in (3/4 3/4 L) scans for different temperatures. The dashed line indicates the transition temperature (T N) observed in the specific heat
The magnetic origin of these quarter-integer peaks in LaNiO3 could be also confirmed by polarized neutrons at the IN12 spectrometer. In Fig. 4a & b L-scans across two quarter-integer peaks are shown for the three spin-flip (SF) channels and for a non-spin-flip channel. Only in the SF channels, neutron scattering intensities \(\sigma _{x_ix_i}\) can be detected. This unambiguously shows the magnetic nature of these quarter-integer reflections in LaNiO3. Although we cannot detect a symmetry lowering from our high-resolution powder X-ray diffraction measurements—see Fig. 2a—the pseudocubic propagation vector for rhombohedral LaNiO3 is the same as the pseudocubic propagation vector for the orthorhombic insulating nickelates RNiO3 (R = Pr-Lu, Y). Based on the magnetic symmetry analysis for the high symmetry cubic structure with space group Pm \(\overline 3 \) m and for the propagation vector (1/4 1/4 1/4), a helical magnetic structure with moments spiraling perpendicular to the propagation vector is consistent with our single crystal neutron data, see Supplementary Note 2, Supplementary Tables 2 and 3, and Supplementary Figure 6. The size of the ordered moment amounts to ~0.3B, which indicates that magnetism in LaNiO3 is a bulk property and does not originate from a tiny (insulating) impurity phase. This small magnetic moment might explain why paramagnetic properties have been reported for LaNiO3 in the past1,2,16,17.
Polarized neutron-diffraction measurements. a, b Polarized neutron scattering experiments for the LaNiO3 single crystal at 2 K. Spin-flip (SF) and non-spin-flip (NSF) channels were measured for neutron spins parallel to the scattering vector (x-direction), perpendicular to the scattering plane (z-direction) or perpendicular to x and z (y-direction). c A structural scan at 2 K showing no indications for any charge ordering peak at the (0.5 0.5 0.5) position in pseudocubic notation (indicated by the green arrow). The intensity error bars are statistical error bars calculated by the square root of intensity
We also have been able to study and observe the magnetic excitations in LaNiO3 by means of inelastic neutron scattering, thereby also providing further support that the antiferromagnetism is a bulk property. As can be seen in Fig. 5, magnetic excitations are clearly visible up to at least 12 meV. With increasing energy transfer, the magnetic peaks become somewhat broader and damped, which is indicative for the appearance of fluctuations in this itinerant antiferromagnetic system. These fluctuations could be also responsible for a reduced ordered moment of ~0.3B in LaNiO3.
Magnetic excitations in LaNiO3. Inelastic neutron scattering intensities measured at 75 K for 3, 6, 9, and 12 meV energy transfer. The horizontal bars indicate the shift of the data in vertical direction. Up to 12 meV, the magnetic peaks become broader and damped, which is indicative for the appearance of fluctuations in this itinerant antiferromagnetic system. The intensity error bars are statistical error bars calculated by the square root of intensity
Discussion
LaNiO3 appears to be a rare case of an antiferromagnetic and metallic transition metal oxide with a fully three-dimensional (3D) crystal and electronic structure. Other systems like (La,Sr)3Mn2O7 and Ca3Ru2O7 have a lower electronic and structural dimensionality where the antiferromagnetic order is resulting from a stacking of ferromagnetic layers25,26. So far, the chromate system ACrO3 (A = Ca, Sr) and very recently, RuO2 are known to be the only other intrinsically antiferromagnetic and metallic transition metal oxides with such a fully 3D crystal and electronic structure27,28. The systems CaCrO3 and LaNiO3 have in common that the oxidation state of the transition metal ion is very high. Thus, the oxygen 2p to transition metal 3d charge-transfer energy here is apparently negative5,12,29 resulting in extreme 2p-3d covalency where the presence of holes in the oxygen band can effectively prevent the opening of the conductivity gap and at the same time mediate strongly the magnetic exchange interactions. However, in contrast to CaCrO3 27, the rare earth nickelate LaNiO3 in single crystalline form is much more metallic and shows conductivities in the μΩ cm range. This is probably also true for RuO2 28. Unique for LaNiO3 is that it is close to the insulating phase of RNiO3 (R = Pr-Lu, Y) indicating the importance of strong correlation effects, making it rather exceptional among all transition metal oxides.
Although charge order, which plays a very important role in nickelates RNiO3 with small rare earth ions R, cannot be observed in our present X-ray diffraction (XRD) and neutron-diffraction measurements—see Fig. 4c—the presumably associated symmetry lowering of the structure that goes along with the antiferromagnetic order may produce only very weak new peaks in diffraction experiments. Some hints for this may in fact be found in a recent pair distribution function study using neutron diffraction30. Nevertheless, with the charge order effects being so weak, one could infer that LaNiO3 is perhaps better described using Fermi surface arguments7,28, while the other insulating nickelates RNiO3 with the smaller R-ionic sizes and with higher charge ordering temperatures can be more intuitively understood in terms of local charge or bond disproportionations5,6,8,9,11,12. Note, that recent ab initio calculations31 reproduced metallic and antiferromagnetic state of LaNiO3, and also predicted charge (or rather bond) disproportionation, whose magnitude is however below our experimental detection limit. One needs special dedicated experiments to probe for the eventual symmetry lowering with the appearance of inequivalent Ni ions. However, our main conclusion—the existence of antiferromagnetic ordering in highly metallic single crystals of LaNiO3, is confirmed by these calculations. According to our findings, we now also present a tentative RNiO3 phase diagram in Fig. 6.
Tentative RNiO3 phase diagram. The blue triangles indicate the metal-insulator transition temperatures T MI associated with the onset of charge ordering and the appearance of monoclinic distortions. The red symbols indicate the Néel temperatures T N associated with the onset of antiferromagnetic ordering. Data points for other R than La were taken from literature24,32. The gray dashed curve subdivides orthorhombic (monclinic) and rhombohedral regimes
Methods
Chemical synthesis
The LaNiO3 single crystal was grown under high oxygen pressures of 130–150 bar with a growth speed of 6–7 mm per h in a mirror furnace from Scidre that was equipped with a 5000 W Xe lamp and with counter-rotation of feeding and seeding rods—see Supplementary Movie 1. The seeding and feeding rods were synthesized by pressing appropriate mixtures of La2O3 and NiO into rods with roughly 10 cm length and 6 mm diameter. These rods were sintered at 800–1000 °C for several days. The temperature of the melting zone was measured in situ by means of a pyrometer and is close to 1800 °C at ~140 bar \({\rm {p}_O{_{_2}}}\).
X-ray diffraction
Laue diffraction measurements were performed on a multiwire real-time back-reflection Laue camera from Multiwire Laboratories; see also Supplementary Note 1 and Supplementary Figure 1.
Powder X-ray diffraction measurements were performed on a Bruker D8 Discover A25 diffractometer, which is equipped with a Johansson monochromator for Cu Kα1 radiation. A closed cycle helium cryostat (Phenix of Oxford Cryosystems) was used for temperature-dependent measurements.
Single crystal X-ray diffraction measurements have been performed on a twined single crystal of LaNiO3 using a Bruker D8 VENTURE single crystal X-ray diffractometer equipped with a bent graphite monochromator for Mo K α radiation (about 3× intensity enhancement) and a Photon CMOS large area detector. A crystal with roughly 20 μm diameter has been measured and a multi-scan absorption correction has been applied to the data (minimum and maximum transmission: 0.6184 and 0.7519, respectively). A total of 12,328 (observed) reflections (H: −12→13, K: −13→9 and L: −32→31) have been collected with an internal R-value of 6.70%, a redundancy of 36.8, and with 98.85% coverage up to \(2\Theta _{{\mathrm{max}}}\) = 123.6°. For the refinement, the Jana2006 program package was used. The goodness of fit of our crystal structure refinement amounts to 1.93, and the R- and weighted R-values amount to 2.29% and 5.79%, respectively. The refinement of the Ni and La occupancies yields an almost stoichiometric composition: La0.995(12)Ni1.000(14)O3. The structural parameters are listed in Supplementary Table 1 and the crystal structure is visualized in Supplementary Figure 3.
Composition determined by ICP and TG measurements
Inductively coupled plasma optical emission spectroscopy measurements yields the following composition of LaNiO3: La1.001(11)Ni0.999(4)O3+δ . Moreover, thermogravimetric measurements confirm an almost perfectly stoichiometric oxygen content with δ = −0.002, see Supplementary Note 2.
X-ray absorption spectroscopy
X-ray absorption spectroscopy measurements have been performed at the 08B beamline of the National Synchrotron Radiation Research Center (NSRRC), Taiwan.
Magnetization measurements
The magnetic properties were studied using a Quantum Design Inc. MPMS-5XL SQUID magnetometer.
Electrical conductivity
The measurements of electrical resistivity and specific heat were carried out using a four-probe and a standard thermal relaxation calorimetric method in a Quantum Design Inc. Physical Property Measurement System.
Neutron measurements
Unpolarized neutron measurements have been performed at the IN8 and Thales Spectrometers at the ILL in Grenoble, France. For the elastic and inelastic measurements on the Thales spectrometer, a pyrolytic graphite (PG) (002) monochromator and analyzer as well as a velocity selector were used (k f = 1.8 Å−1). On the IN8 spectrometer, very first quick inelastic scans have been made using a Si monochromator and a Flatcone analyzer array. Polarized neutron measurements have been performed at the IN12 Spectrometer at the ILL in Grenoble, France. A velocity selector was used for choosing the incident neutron wavevector k i of 2.25 Å−1. A horizontally and vertically focussing PG (002) monochromator and Heusler (111) analyzer have been used. The incident neutron beam was polarized by a transmission polarizer (cavity) in the neutron guide and the measured flipping ratio amounts to 22.2.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Torrance, J. B., Lacorre, P., Nazzal, A. I., Ansaldo, E. J. & Niedermayer, C Systematic study of insulator-metal transitions in perovskites RNiO3 (R=Pr,Nd,Sm,Eu) due to closing of charge-transfer gap. Phys. Rev. B 45, 8209–8212 (1992).
Medarde, M. L. Structural, magnetic and electronic properties of RNiO3 perovskites (R=rare earth). J. Phys. Condens. Matter 9, 1679–1707 (1997).
Giovannetti, G., Kumar, S., Khomskii, D., Picozzi, S. & van den Brink, J. Multiferroicity in rare-earth nickelates RNiO3. Phys. Rev. Lett. 103, 156401 (2009).
Mizokawa, T. & Fujimori, A. Unrestricted Hartree-Fock study of transition-metal oxides: spin and orbital ordering in perovskite-type lattice. Phys. Rev. B 51, 12880–12883 (1995).
Mizokawa, T., Khomskii, D. I. & Sawatzky, G. A. Spin and charge ordering in self-doped Mott insulators. Phys. Rev. B 61, 11263–11266 (2000).
Mazin, I. I. et al. Charge ordering as alternative to Jahn-Teller distortion. Phys. Rev. Lett. 98, 176406 (2007).
Lee, S. B., Chen, R. & Balents, L. Landau theory of charge and spin ordering in the nickelates. Phys. Rev. Lett. 106, 016405 (2011).
Park, H., Millis, A. J. & Marianetti, C. A. Site-selective Mott transition in rare-earth-element nickelates. Phys. Rev. Lett. 109, 156402 (2012).
Johnston, S., Mukherjee, A., Elfimov, I., Berciu, M. & Sawatzky, G. A. Charge disproportionation without charge transfer in the rare-earth-element nickelates as a possible mechanism for the metal-insulator transition. Phys. Rev. Lett. 112, 106404 (2014).
Jaramillo, R., Ha, S. D., Silevitch, D. M. & Ramanathan, S. Origins of bad-metal conductivity and the insulator metal transition in the rare-earth nickelates. Nat. Phys. 10, 304–307 (2014).
Subedi, A., Peil, O. E. & Georges, A. Low-energy description of the metal-insulator transition in the rare-earth nickelates. Phys. Rev. B 91, 075128 (2015).
Green, R. J., Haverkort, M. W. & Sawatzky, G. A. Bond disproportionation and dynamical charge fluctuations in the perovskite rare-earth nickelates. Phys. Rev. B 94, 195127 (2016).
Chaloupka, J. & Khaliullin, G. Orbital order and possible superconductivity in LaNiO3/LaMO3 superlattices. Phys. Rev. Lett. 100, 016404 (2008).
Scherwitzl, R. et al. Metal-insulator transition in ultrathin LaNiO3 films. Phys. Rev. Lett. 106, 246403 (2011).
Middey, S. et al. Physics of ultrathin films and heterostructures of rare-earth nickelates. Ann. Rev. Mater. Res. 46, 305–334 (2016).
Xu, X. Q., Peng, J. L., Li, Z. Y., Ju, H. L. & Greene, R. L. Resisitivity, thermopower, and susceptibility of RNiO3 (R=La,Pr). Phys. Rev. B 48, 1112–1118 (1992).
Zhou, J. S., Marshall, L. G. & Goodenough, J. B. Mass enhancement versus stoner enhancement in strongly correlated metallic perovskites: LaNiO3 and LaCuO3. Phys. Rev. B 89, 245138 (2014).
Hu, Z. et al. Hole distribution between the Ni3d and O2p orbitals in Nd2−x Sr x NiO4−δ . Phys. Rev. B 61, 3739–3744 (2000).
Hu, Z. et al. On the electronic structure of Cu(III) and Ni(III) in La2Li1/2Cu1/2O4, Nd2Li1/2Ni1/2O4, and Cs2KCuF6. Chem. Phys. 232, 63–74 (1998).
Zhang, J., Zheng, H., Ren, Y. & Mitchell, J. F. High-pressure floating-zone growth of perovskite nickelate LaNiO3 single crystals. Cryst. Growth Des. 17, 2730–2735 (2017).
Zhou, J.-S., Goodenough, J. B. & Dabrowski, B. Pressure-induced non-fermi-liquid behavior of PrNiO3. Phys. Rev. Lett. 94, 226602 (2005).
Sánchez, R. D. et al. Metal-insulator transition in oxygen-deficient LaNiO3−x perovskites. Phys. Rev. B 54, 16574–16578 (1996).
Gayathri, N., Raychaudhuri, A. K., Xu, X. Q., Peng, J. L. & Greene, R. L. Electronic conduction in LaNiO3−δ: the dependence on the oxygen stoichiometry. J. Phys. Condens. Matter 10, 1323–1338 (1998).
Garca-Muñoz, J. L., Rodrguez-Carvajal, J. & Lacorre, P. Neutron-diffraction study of the magnetic ordering in the insulating regime of the perovskites RNiO3 (R=Pr and Nd). Phys. Rev. B 50, 978–992 (1993).
Argyriou, D. N. et al. Lattice effects and magnetic structure in the layered colossal magnetoresistance manganite La2−2 xSr1+2x Mn2O7 (x=0.3). Phys. Rev. B 59, 8695–8702 (1999).
Yoshida, Y. et al. Crystal and magnetic structure of Ca3Ru2O7. Phys. Rev. B 72, 054412 (2005).
Komarek, A. C. et al. CaCrO3: an anomalous antiferromagnetic metallic oxide. Phys. Rev. Lett. 101, 167204 (2008).
Berlijn, T. et al. Itinerant antiferromagnetism in RuO2. Phys. Rev. Lett. 118, 077201 (2017).
Khomskii, D. I. Transition Metal Compounds (Cambridge University Press, 2014).
Li, B. et al. Insulating pockets in metallic LaNiO3. Adv. Electron. Mater. 2, 1500261 (2016).
Subedi, A. Breathing distortions in the metallic, antiferromagnetic phase of LaNiO3. Preprint at https://arxiv.org/abs/1708.08899 (2017).
Zhou, J. S., Goodenough, J. B. & Dabrowksi, B. Transition from Curie-Weiss to enhanced Pauli paramagnetism in RNiO3 (R=La, Pr,… Gd). Phys. Rev. B 67, 020404 (2003).
Acknowledgements
We thank O. Stockert, G.A. Sawatzky, and A. Fujimori for valuable discussions. We thank M. Schmidt and his team for thermogravimeteric measurements. We thank G. Auffermann and her team for ICP measurements. We acknowledge A. Todorova and G. Ryu for support on preparatory work. We thank the team of H. Borrmann for initial room-temperature powder X-ray diffraction measurements used for phase analysis. The research in Dresden and Cologne is partially supported by the Deutsche Forschungs-Gemeinschaft through FOR 1346.
Author information
Authors and Affiliations
Contributions
Conceiving experiments and project management: A.C.K.; physical experiments: H.G., Z.W.L., L.Z., Z.H., C.F.C., C.-Y.K., W.S., A.P., T.W.P., O.S., and A.C.K.; chemical synthesis: A.C.K. and H.G.; interpretation and manuscript writing: A.C.K., D.I.K., and L.H.T.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, H., Li, Z.W., Zhao, L. et al. Antiferromagnetic correlations in the metallic strongly correlated transition metal oxide LaNiO3 . Nat Commun 9, 43 (2018). https://doi.org/10.1038/s41467-017-02524-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-017-02524-x
This article is cited by
-
Tunable ferroelectricity in oxygen-deficient perovskites with Grenier structure
npj Computational Materials (2023)
-
Unusual double ligand holes as catalytic active sites in LiNiO2
Nature Communications (2023)
-
Ferromagnetic order controlled by the magnetic interface of LaNiO3/La2/3Ca1/3MnO3 superlattices
Scientific Reports (2023)
-
Evidence for charge and spin density waves in single crystals of La3Ni2O7 and La3Ni2O6
Science China Physics, Mechanics & Astronomy (2023)
-
Infinite critical boson non-Fermi liquid on heterostructure interfaces
Quantum Frontiers (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.