Abstract
Metagenomic sequencing provides a window into microbial community structure and metabolic potential; however, linking these data to exogenous metabolites that microorganisms process and produce (the exometabolome) remains challenging. Previously, we observed strong exometabolite niche partitioning among bacterial isolates from biological soil crust (biocrust). Here we examine native biocrust to determine if these patterns are reproduced in the environment. Overall, most soil metabolites display the expected relationship (positive or negative correlation) with four dominant bacteria following a wetting event and across biocrust developmental stages. For metabolites that were previously found to be consumed by an isolate, 70% are negatively correlated with the abundance of the isolate’s closest matching environmental relative in situ, whereas for released metabolites, 67% were positively correlated. Our results demonstrate that metabolite profiling, shotgun sequencing and exometabolomics may be successfully integrated to functionally link microbial community structure with environmental chemistry in biocrust.
Similar content being viewed by others
Introduction
In soils, which harbor the largest terrestrial pool of organic carbon1, organic matter is largely processed by complex microbial communities. The impact of climate change on these communities and their activities is uncertain2. Given the importance of these systems, vast amounts of sequencing data have been and continue to be collected. While metagenomic sequencing provides important insights into community structure and metabolic potential, if unconstrained, such data are often open to multiple interpretations. New approaches are needed to help link the now readily available sequencing data to in situ metabolism in order to better understand the dynamic reciprocity between carbon cycling and microbial community structure.
Soil organic matter (SOM) content and moisture have long been recognized as important factors controlling soil microbial community structure and carbon cycling3,4. For example, microbial community diversity and richness are positively correlated with soil organics across diverse ecosystems including polar soils5, agricultural soils6 and arid soils7. Similarly, soil wetting events are well-known to dramatically alter community structure8,] including establishing cascades of microbial abundances9. Arid lands account for over 40% of Earth’s terrestrial surface10 and are especially sensitive to SOM and moisture content. It is predicted that the aridity of drylands will increase, reducing SOM and microbial community diversity, and that this will impact ecosystem productivity7,11. This strong coupling between soil moisture, SOM and community structure is especially important in the arid land topsoil microbial communities known as biological soil crusts (biocrusts), which cover a large fraction of arid regions and are critical in nutrient cycling12. Biocrusts exist in a dormant desiccated state and only become metabolically active during infrequent rainfall events13 and like in other soils, organic matter plays a vital role in retaining moisture and increasing microbial diversity14.
The mechanisms linking SOM composition and microbial community structure are poorly understood. It is now thought that the organic matter that is cycled by soil microbes is a complex mixture of microbial metabolites15,16 that can be characterized in detail using soil metabolomics17,18. The composition of these exometabolites has a strong impact on community structure, and in turn, these microbes impact the metabolite pool. For example, in some cases, resource competition can reduce microbial diversity through competitive exclusion, whereas cross-feeding can increase microbial diversity. On the other hand, rich sources of SOM may promote microbial diversity through niche divergence19 and exometabolite niche partitioning20.
Exometabolomics enables direct examination of how microbes transform the small molecule metabolites within their environment, providing new insights into resource competition and cross-feeding21. For this approach, microbes (typically isolates) are cultured in an environmentally-relevant mixture of metabolites and then spent media is profiled to determine the uptake and release of metabolites. Recently, exometabolomics was used to study resource partitioning among sympatric biocrust isolates using complex biocrust-relevant media20. This revealed a high degree of substrate specialization, where 13–26% of the detected metabolites were consumed by individual isolates. As microorganisms from diverse taxa continue to be cultivated and examined, this approach holds substantial potential to provide valuable phenotypic information that can link community structure to SOM composition.
Here we exploited the dynamic and tractable characteristics of biocrust to explore the relationships between soil microbes and metabolites for this particular ecosystem after a laboratory-based wetting event. Liquid chromatography-mass spectrometry (LC/MS) soil metabolomics was used to characterize the dynamic metabolite composition of the biocrust soil water and shotgun metagenomic sequencing was used to measure single copy gene markers of the dominant taxa. We then determined the extent to which isolate exometabolite patterns are conserved in situ (within the intact biocrust soil community). While the comparison of a microbe in isolation and in an environmental system is challenging, isolate exometabolomics were performed with media supplemented with lysed cell metabolite extracts to simulate the biocrust environment20. Our findings reveal significant agreement between isolate exometabolomics and in situ microbe–metabolite relationships for dominant taxa in biocrust. To the best of our knowledge, this is the first study using isolate exometabolomics to link microbial community structure to soil chemistry.
Results
Experimental setup
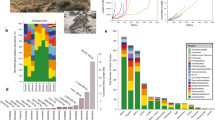
Correlation analyses between microbes and metabolites were facilitated by monitoring biocrust samples across two variables: laboratory-based wetting and ecological succession (Fig. 1a). Four successional stages of biocrust were used, ranging from early/ young (labeled as ‘level A’) to late/ mature (labeled as ‘level D’) (Supplementary Fig. 1). We then compared our current results with previous laboratory-derived knowledge of substrate preferences for four dominant microorganisms by relating the abundance of these bacteria to soil metabolites measured in the intact biocrust system (Fig. 1b). In situ, the general assumption is that as a particular microbe grows and increases in abundance in a community, consumed metabolites will decrease and display a negative-correlation relationship. Conversely, metabolites that are known to be released by a microbe are predicted to concurrently increase and display a positive-correlation relationship with growth (Fig. 1b).
Experimental workflow and biocrust microbe–metabolite relationship predictions. a Biocrust wetup metabolomics and metagenomics experimental setup and analysis. To study microbe–metabolite relationships in situ, biocrusts from four successional stages were wetup and sampled at five time points (total n = 100). Biocrust soil water was removed and analyzed by liquid chromatography/ mass spectrometry (n = 5 for each group) and biocrust DNA was extracted for shotgun sequencing (n = 1 for each group). Metagenome-estimated genome and metabolite abundances were analyzed through Spearman rank correlations to determine microbe–metabolite relationships and compared to the expected relationships based on isolate exometabolomic studies. b Exometabolomics-based in situ microbe–metabolite relationship prediction. The hypothesis is that isolate exometabolomics can be used to predict microbe–metabolite patterns in situ based on microbial abundance: Across wetting and successional stages, microbes change in abundance and negatively correlate with metabolites that they consume and positively correlate with metabolites that they release (metabolites are indicated by dotted lines)
Metabolite and microbe dynamics
The metabolic activity caused by wetting was monitored at various time points ranging from immediate (3 min) to longer-term (49.5 h) across four biocrust successional stages. Biocrust soil water was analyzed by LC/MS, resulting in the identification of 85 metabolites for the experimental data set using authentic chemical standards (Fig. 2 and Supplementary Data 1). All 85 metabolites identified in the active biocrust samples changed at least two-fold (between minimum and maximum peak areas) across both wetting and successional stages (Fig. 2). Wetting duration had a stronger impact on metabolite dynamics vs. successional stage (Supplementary Fig. 2). Hierarchical clustering of metabolite patterns revealed three distinct clusters (Fig. 2). The first cluster (cluster 1, Fig. 2) consisted of most (5 out of 7) of the detected fatty acids (palmitate, myristate, stearate, laurate, decanoate), which were most abundant at the first time point (3 min) for all successional stages, and gradually decreased with time. The largest cluster (cluster 2, Fig. 2) was enriched with the majority of amino acids and nucleobases, which peaked in abundance at the early to early-mid time points. Within this cluster, the earliest metabolites included polar amino acids (glutamine, glutamate, asparagine, 4-oxoproline, aspartate and lysine) and the nucleobases uridine, guanosine and cytidine. The final cluster (cluster 3 in Fig. 2) contained metabolites most abundant at late time points and in more mature biocrust (e.g., salicylate, panthothenate, nicotinate, xanthine, creatinine). These dynamics, especially metabolite consumption, were largely of biological origin as demonstrated by comparison with the killed controls. Thirteen metabolites that were detected in the active samples were not detected in the killed controls presumably due to the lack of biologically activity. Of the 72 metabolites that were detected in the killed controls, all but 19 were significantly different (p < 0.05, by two-way ANOVA and Tukey’s post hoc test) from the active samples and qualitatively all show different dynamics (Supplementary Data 2 and 3; Supplementary Fig. 3).
Metabolite patterns detected in biocrust soil water. Metabolite dynamics (85 metabolites displayed as the average peak area, normalized across each row) were observed in biocrust soil water across wetting and successional stages. Unique patterns are indicated by cluster 1 (early metabolites including fatty acids), cluster 2 (early-to-mid time point metabolites) and cluster 3 (late metabolites). Putative metabolites are indicated by parentheses. n = 2–5 for each group
Biocrust microbial community structure was inferred by shotgun metagenomics using a genome-centric pipeline22. In several recent reports23,24,25,26, ribosomal protein genes were used as phylogenetic markers from shotgun sequencing data (rather than the more classical 16s ribosomal RNA gene) because they exist as single copies in almost all genomes, assemble well from the metagenome data sets (typically better than 16s), are well-conserved and have produced higher resolution phylogenetic trees25. We identified a set of 17 previously-benchmarked single copy universal ribosomal protein genes27 in our biocrust data set and for community analysis, we selected rplO (ribosomal protein L15), which had the most extensive community coverage based on the total number of assembled genes across our data set (Supplementary Table 1).
On the basis of rplO genes, 466 distinct biocrust genomes were identified across all conditions (Supplementary Data 4). As observed for biocrust metabolites, community structure was primarily driven by wetting duration. At the phylum level, the most drastic change was a shift from a Cyanobacteria-dominated community at early time points (17–28% at 3 min to 1–3% by 49.5 h) to a Firmicutes-dominated community by 49.5 h (4–5% at 3 min to 19–39% by 49.5 h) (Supplementary Fig. 4). Other dominant phyla included Proteobacteria and Actinobacteria, which appeared to be indifferent to wetting (i.e., their relative abundance was more evenly-distributed across wetting) (Supplementary Fig. 4).
In order to use previous culture-based isolate exometabolomic analyses to examine the link between microbe and metabolite dynamics in intact biocrust, we conducted an unbiased rplO gene sequence comparison between rplO genes assembled from the native biocrust and previously profiled biocrust bacterial isolates20 (obtained from the same field site). Fortunately, four isolates matched relatively abundant native biocrust bacteria at the species or genus level (Table 1). Further comparative analyses were conducted to calculate genome average nucleotide identity (ANI) between isolate genomes and their related metagenome-assembled genomes (MAGs) to which the rplO gene was co-binned. This validated that each isolate and their closest matching environmental relative were of the same genus or species, following the conventions described in a previous report28 (Table 1). Additional validation was obtained by comparing gene average amino acid identity (AAI) (Supplementary Table 2) and other ribosomal protein genes (Supplementary Data 5) from within each MAG to the corresponding isolate. On the basis of these results, these four isolate-related environmental bacteria were selected for exometabolomics comparisons: Microcoleus sp. (a filamentous Cyanobacterium and primary producer), two Firmicutes (referred to here as Bacillus sp. 1 and Bacillus sp. 2) and Blastococcus sp. (an Actinobacterium) (Table 1, Supplementary Fig. 5).
Remarkably, together, these four bacteria accounted for ~30% of the entire microbial community in many of the biocrust samples (Supplementary Fig. 6). The overall most abundant microorganism, Microcoleus sp. is known to be a pioneer species responsible for initial soil stabilization and biocrust formation29 and in our study was the most dominant in early wetup biocrust, accounting for 10–25% of the entire microbial community at 3 min across all successional stages (Supplementary Fig. 6). The two Firmicutes, Bacillus sp. 1 and Bacillus sp. 2, are likely to be physically-associated with Microcoleus filaments20 and their relative abundance increased during wetting. The most abundant of these, Bacillus sp. 1, was a mid-wetup responder and peaked at 9 h for successional levels A, B and D (16–24% of the community) and at 18 h for successional level C (24% of the community) (Supplementary Fig. 6). Bacillus sp. 2 reached its peak abundance at later time points, accounting for up to 3% of the community by 42 h in successional level C, noting that by the later time points the community was less dominated by any one particular microorganism (Supplementary Fig. 6). Finally, Blastococcus sp. abundance was found to be relatively resistant to wetting and was somewhat evenly distributed across all conditions (0.1–2% of the community) (Supplementary Fig. 6).
Microbe–metabolite relationships in situ vs. in culture
To determine how isolate substrate preferences impact in situ exometabolite composition, we evaluated microbe–metabolite relationships, focusing on metabolites known to be released or consumed by biocrust isolates and their closest matching environmental relatives, Microcoleus sp., Bacillus sp. 1, Bacillus sp. 2 and Blastococcus sp. The general expectation was that released metabolites would be positively correlated with the relative bacterial abundance while consumed metabolites would be negatively correlated (Fig. 1b) across environmental variables, including wetting and successional stage. To link previous isolate exometabolomics data with the current biocrust exometabolome data set (Fig. 2), we determined the direction and degree of correlation between the metabolites that were previously found to be consumed and released by biocrust isolates20 and closest matching environmental relatives (Microcoleus sp., Bacillus sp. 1, Bacillus sp. 2 and Blastococcus sp).
Of the 85 metabolites identified in the biocrust soil water, 32 matched the isolate exometabolome data set. Nine of these displayed temporal patterns that were not significantly different than the killed control and were excluded from this analysis since abiotic controls on these metabolites could not be ruled out (Supplementary Data 6). Spearman’s rank correlation was used to assess the directionality (positive vs. negative correlations) of biocrust microbe–metabolite relationships. Strikingly, of the 48 microbe–metabolite relationships evaluated (Supplementary Fig. 7), 69% had the expected directionality that would be predicted based on isolate exometabolomics data. This overall observation of correct directionalities is significantly higher than what would be expected by chance (p-value = 0.01 by two-tailed binomial test; Supplementary Data 7).
We next combined our microbe-metabolite correlation data with the isolate exometabolomics data in order to visualize a wetting-induced dynamic exometabolomic foodweb that may result from the release of metabolites by a primary producer (e.g., Microcoleus sp.) and consumption of metabolites by heterotrophs (e.g., Bacillus sp. 1 and 2) (Fig. 3). For simplicity, Blastococcus sp. is not shown in Fig. 3 since this microorganism did not display sequential responses to wetting. It should be noted that the bacteria specified here likely represent metabolically similar groups of organisms that release or consume the same metabolites. Of the set of metabolites that were most highly-released by M. vaginatus PCC 980220, 14 of these were detected in the biocrust soil water and half were positively correlated with Microcoleus sp. across wetting and successional stages (Fig. 3 and Supplementary Fig. 7). While Microcoleus sp. was most abundant immediately following wetting, most of these metabolites (71%) reached their highest level during the first three time points (3 min, 9 h or 18 h; Supplementary Fig. 7) just after the Microcoleus sp. spike, suggesting release by Microcoleus sp. followed by consumption by heterotrophs as they become metabolically active.
Simplified biocrust foodweb for three dominant biocrust bacteria based on combining isolate exometabolomics data with in situ microbe–metabolite relationships. This network displays the relationships between metabolites and three dominant bacteria (or metabolically similar organisms) as they increase and decrease in relative abundance across wetting and successional stages in biocrust. The lower line plot corresponds to real relative abundance measurements for the three bacteria in level C successional stage biocrust. As Microcoleus sp. increases in relative abundance immediately after wetting, many metabolites released by the closest-related isolate are positively correlated with Microcoleus sp. in biocrust (solid red arrows) and as the two Bacilli increase in relative abundance (first Bacillus sp. 1 then Bacillus sp. 2), most metabolites consumed by the closest-related isolate decrease and are negatively correlated with these bacteria in biocrust (solid blue arrows) and most released metabolites are positively correlated (solid red arrows). Dotted arrows indicate metabolites that are released (red) or consumed (blue) by isolates, but did not display the expected relationship with that microorganism in situ. The thickness of the line corresponds to the absolute value of the Spearman’s rho correlation coefficient. The overall expected directionality (solid lines vs. dotted lines) was significant as determined by the exact binomial test (two-tailed p-value = 0.01). *FDR-adjusted p < 0.05 for individual microbe-metabolite Spearman correlations
Consistent with a heterotrophic lifestyle, most metabolites evaluated displayed a negative relationship with the relative abundances of Bacillus sp. 1, Bacillus sp. 2 and Blastococcus sp. Of the metabolites that were consumed by the Bacillus sp. 1. related isolate, D1B51 (Table 1)20, six were detected in the current biocrust soil water samples and reached their highest level early-on (at either 3 min, 9 or 18 h), decreasing just after the Bacillus sp. 1 peak (Supplementary Fig. 7). Four of these metabolites were negatively correlated with Bacillus sp. 1, consistent with metabolite consumption, and all three D1B51-released metabolites were positively correlated with Bacillus sp. 1 (Fig. 3 and Supplementary Fig. 7). As for the less dominant microorganisms, the late-wetup responder, Bacillus sp. 2, was negatively correlated with all seven metabolites that were consumed by the related isolate (L2B47) and positively correlated with all four isolate-released metabolites (Fig. 3 and Supplementary Fig. 7). Furthermore, Blastoccocus sp., was negatively correlated with eight out of the ten metabolites that were consumed by the related isolate (L1B44). Finally, the closest matching environmental relatives to the three remaining exometabolomic-profiled isolates (L1B56, D1B2, and D1B45) accounted for 0.1% or less of the microbial community in our metagenomes and thus, not surprisingly, did not display exometabolite-based microbe–metabolite relationships (data not shown).
Transcriptomics support soil microbe–metabolite relationships
Transcriptomics has the potential to test if gene expression is consistent with predicted substrate utilization and release patterns. As an initial proof of concept, we further analyzed the data obtained from a previous study that evaluated M. vaginatus gene expression following wetup and drydown in biocrusts collected from the same field site30. We found that pathways involved in the biosynthesis of amino acids (KEGG pathways ‘biosynthesis of amino acids’, ‘phenylalanine, tyrosine and tryptophan biosynthesis’ and ‘valine, leucine and isoleucine biosynthesis’) all increased dramatically during early wet-up (Supplementary Fig. 8, Supplementary Data 8). In contrast, pathways involved in the degradation of these same metabolites were relatively constant (‘phenylalanine metabolism’) or only slightly increased (‘tryptophan metabolism’ and ‘valine, leucine and isoleucine biosynthesis’) following wet-up (Supplementary Fig. 8). These observations are consistent with the release of these metabolites by M. vaginatus PCC 980220 and the observed immediate increase of most amino acids in biocrust soil water in the present study.
Discussion
Sequencing has the potential to link exometabolite composition to specific microbes based on genome annotations. However, with these data alone, relating metabolic potential to activity is challenging. Despite this challenge, sequencing and other approaches have started to shed light on the impact individual microorganisms31, microbial genes32 and enzymatic activities33 have on the chemistry within their environment. Here we evaluated exometabolite profiles of individual bacteria in order to link soil exometabolites to bacteria in biocrust, a critical ecosystem that lends itself to studies of community responses to soil wetting.
We found that a biocrust wetting event set in motion an immediate cascade of microbial activities marked by a drastic shift in community structure. The dominance of Cyanobacteria during early time points is consistent with previous reports34 as is the subsequent Firmicutes-bloom9,35. The Firmicutes phylum consists mostly of gram-positive, spore-forming bacteria with rapid generation times, enabling them to ‘bloom’ upon soil wetting36,37. The observed switch from a Cyanobacteria-dominated community to a Firmicutes-dominated community (mostly Bacillus sp. 1 in this study) agrees with our observations of metabolite release by the dominant photoautotroph (Microcoleus sp.)20 followed by consumption and growth of diverse heterotrophs (e.g., Firmicutes), possibly including symbiotic nitrogen-fixers38. While we did not observe evidence of fixed nitrogen transfer into Cyanobacteria, this process may occur during dry-down, when nitrogen-rich nutrients may be released upon the mother cell lysis stage of sporulation39.
It has been suggested that copiotrophic microorganisms (e.g., many Firmicutes) are superior competitors for a limited number of compounds, whereas oligotrophs (e.g., many Actinobacteria) support a more stable population by using a wider range of substrates40. Our previous isolate exometabolomics work is consistent with this view by showing that the two Firmicutes isolates depleted the narrowest range of substrates (10%), whereas the two Actinobacteria used almost twice as many20. During biocrust wetting, we found that, unlike the boom-bust cycle of Firmicutes, the Actinobacteria phylum (such as Blastococcus sp.) appeared more resistant to wetting duration. This provides limited evidence that utilization of diverse substrates, which is consistent with oligotrophy, may enable slow but continuous growth under conditions with highly dynamic exometabolite pools.
The wetting-induced initiation of microbial community dynamics coincided with an immediate release of metabolites into biocrust soil water. Killed controls were used to differentiate biotic processes of interest from abiotic dynamics driven by metabolite leakage, slow diffusion, mineral sorption, thermolysis and photolysis. As expected, the active biocrusts exhibited extremely different dynamics, with dramatic, near complete depletion of many metabolites, whereas the killed controls (with the exception of the antioxidant ergothionine) show a gradual increase in most metabolites presumably as they are released from the dead biomass (Supplementary Fig. 3). Some likely biological mechanisms involved in the active metabolite “pulse” include osmotic stress induced release of solutes (e.g., proline, betaine and disaccharides) to maintain cell integrity41,42 as well as photosynthate release from the primary producer. These metabolite dynamics that followed wetting resulted in strong microbe–metabolite relationships that were tracked across time and interestingly, most of these strong relationships (correlations) were conserved from one successional stage to another for the four bacteria that were examined (Microcoleus sp., Bacillus sp. 1 and 2 and Blastococcus sp.) (Supplementary Fig. 9). This supports the notion that the content of water-soluble SOM in these biocrusts, to a large degree, originates from and is controlled by microbes15 and the composition of this pool may be predictable if a change in microbial community structure is anticipated. This finding has particular significance for biocrusts, since changes in temperature and rainfall are expected to shift microbial community structure43,44. As a result, these alterations are expected to impact SOM cycling, especially if there is loss of taxa responsible for utilization or production of specific SOM components.
Next, we explored the connection between the observed microbe–metabolite relationships in biocrust and culture-based exometabolite profiles, focusing on metabolites found to be controlled by biological mechanisms. Comparison of the two data sets was facilitated by the fact that the isolates were obtained from the same field site. Therefore, we used the phylogenetic marker, rplO (which had the most coverage in our data set) to relate exometabolite-profiled isolates to native biocrust bacteria. Isolate genomes and their closest matching environmental MAGs had between 73–94% genome ANI values and were 86–92% identical in their rplO sequences (Table 1). These relationships were further confirmed by comparing other ribosomal protein genes, which all ranged between 72–100% identical between each isolate and its environmental MAG (Supplementary Data 5). While there is a general lack of consensus of valid isolate-to-native population comparisons, our values indicate matching at approximately the genus or species level28. Much of the difficulty in these comparisons is due to genomic heterogeneity within environmental samples45. However, despite these complexities with determining exact phylogenetic distances, we observed functional similarity between the isolate and environmental exometabolomics data sets, which is consistent with the view that many metabolic traits are conserved at the genus level46. An exciting alternative explanation for our observations is that the clades that these isolates are members of exhibit the same cohesive dynamics as the closest-matching bacteria in the biocrust47.
Overall, we found that isolate exometabolomic patterns were conserved in the intact biocrust soil microbial community. The expected directionality (positive or negative microbe–metabolite relationships) (solid arrows in Fig. 3) was significantly higher than predicted by chance, indicating a linkage between laboratory observations and in situ soil activities. While most metabolites displayed the expected patterns, some biocrust soil water metabolites (e.g., uracil, N6-acetyl-lysine, hypoxanthine and xanthine) behaved inconsistently with M. vaginatus PCC 9802 exometabolite profiles. However, these were also released by and positively correlated with Bacillus sp. 1 and 2. Deconvoluting this may be possible using dynamic utilization models48,49 to account for the relative contributions of the two microorganisms. Ultimately, this same approach could be used to account for rare community members that may also have an impact on the exometabolite pool or may alter the metabolism of other microbes50,51. Although outside of the scope of the current work, we anticipate that these substrate-genome linkages could be further tested and refined by using other approaches. Stable isotope probing coupled with labeled DNA sequencing38,52 and integrated NanoSIMS and FISH imaging53,54 may be used to examine the spatial localization of microbes and their activities.
We next used the biocrust microbe–metabolite relationships to display a simplified dynamic exometabolomics web describing the wetting response of three dominant bacteria in biocrust (Fig. 3). While this network portrays the dynamic substrate preferences of three specific native biocrust bacteria, there are many functionally-similar microorganisms that could fall into the categories of ‘early responders’, ‘mid-responders’ and ‘late-responders’ (grouping with Microcoleus sp. and the two Bacilli sp., respectively) for a wetting event. However, based on our available data and analyses, this network displays the role of Microcoleus sp. as the biocrust primary producer, releasing many metabolites, which stimulates the growth and metabolite consumption by heterotrophs (in this case, the two heterotrophic Firmicutes). Our findings suggest unique microorganismal roles in the biocrust foodweb, including the release of nucleobases (uracil, hypoxanthine and xanthine) by the two Firmicutes, which is consistent with our earlier reports of heterotrophs releasing these compounds55. This may reflect a nitrogen-scavenging mechanism by consuming N-containing substrates (cytosine, adenine, guanine, and histidine), producing uracil, hypoxanthine, and xanthine as byproducts, which may then be consumed by late-responding microorganisms. Even though these observed functional linkages are for a fraction of the biocrust microbial community, these small yet significant pieces of the puzzle have the potential to help understand and predict nutrient cycling in terrestrial microbial ecosystems56 analogously to the many microorganisms that have been linked to specific transformations within marine ecosystems. For example, Cyanobacteria release and reuptake organic carbon57, a variety of uncultured taxa utilize dissolved proteins52 and SAR11 bacteria assimilate amino acids and dimethylsulfoniopropionate58.
We attribute much of the success of this study to the suitability of the relatively simplified biocrust soil ecosystem. One such advantage is that the biocrust used in this study is primarily quartz sand, facilitating metabolite analysis compared to many other soils which are typically rich in clays and other strongly-sorptive mineral surfaces59. Accurately representing the competition between microbes and mineral surfaces would require additional studies examining mineral-metabolite sorption dynamics60,61. Another simplifying factor is that the biocrust community, unlike many other soils, is dominated by a few bacteria, greatly enabling accurate correlations between taxa and metabolites. We anticipate that in order to more accurately predict microbe–metabolite relationships for more diverse communities and complex environments, a large number of relevant taxa would need to be subjected to exometabolite profiling. Additionally, accounting for switching between metabolic states will require profiling under diverse environmental conditions. For example, the discrepancy between metabolites that were released by M. vaginatus PCC 980220, but were not correlated with Microcoleus sp. abundance in biocrust may be due to different metabolic processes occurring during the day (photosynthesis) vs. night (respiration) (i.e., Diel cycle)30,62. Thus, modeling approaches will be required to account for metabolic state switching among other processes. One exciting possibility of expanded exometabolomic data sets, is that knowledge of uptake and release of metabolites can be used as boundary constraints for flux-balance analysis in metabolic models63 and to inform trait-based models64,] providing a genome-scale approach for linking soil metabolites with metagenomic data. For example, OptCom65, a multi-level and multi-objective flux balance analysis framework to understand metabolism within microbial communities, which currently primarily relies on genomic information, could be used in conjunction with exometabolomic data.
In conclusion, this study suggests that, for biocrust, many genotype-to-phenotype relationships are conserved from test tube to soil. These conserved isolate exometabolite patterns begin to provide a functional link between in situ community structure and metabolite composition. We expect that exometabolomic characterization of additional taxa and determination of mineral-metabolite sorption dynamics, under a range of environmentally relevant conditions (e.g., day/night cycles), integrated with modeling approaches will further enhance the predictive power of these relationships. These studies may help pave the way for interpretation and use of metagenomic and metatranscriptomic approaches for linking soil chemistry to soil microbiomes to define exometabolite webs of microbes in complex ecosystems.
Methods
Materials
LC/MS-grade water and LC/MS-grade methanol (CAS 67-56-1) were from Honeywell Burdick & Jackson (Morristown, NJ). LC/MS-grade acetonitrile (CAS 75-05-8) and ammonium acetate (CAS 631-61-8) were from Sigma-Aldrich (St. Louis, MO). LC/MS internal standards included MOPS (CAS 1132-61-2), HEPES (CAS 7365-45-9), 3,6-dihydroxy-4-methylpyridazine (CAS 5754-18-7), 4-(3,3-dimethyl-ureido)benzoic acid (CAS 91880-51-2), d5-benzoic acid (CAS 1079-02-3) and 9-anthracenecarboxylic acid (CAS 723-62-6) from Sigma-Aldrich.
Sample collection
Petri dishes (6 cm2 × 1 cm depth) were used to core biocrust samples from the Green Butte Site near Canyonlands National Park (38°42'54.1'' N, 109°41'27.0'' W, Moab, UT, USA). Samples were collected along an apparent maturity gradient of Cyanobacteria-dominated biocrusts ranging from light, young (level A) to darker, more mature (level D) (Supplementary Fig. 1). Dry samples collected in the field were maintained in a dark desiccation chamber in the laboratory for approximately 11 months until lab-based experiments were performed consistent with previous reports66. Biocrusts are adapted to prolonged desiccation though it is important to note that the duration of desiccation likely affects metabolite composition and biological responses.
Biocrust wetting
Biocrust (0.5 g) was transferred to each well within 12-well plates. Sterile LC/MS-grade water (1 mL) was added to each sample and placed under a 12 h light (~300 μmol photons/m2s)/ 12 h dark cycle. Microcosms were completely enclosed by aluminum foil to prevent infiltration by outside light sources. At each time point (3 min, 9 h, 18 h, 42 h and 49.5 h), biocrust and soil water were removed and placed in 2 mL Eppendorf tubes and 500 μL of additional water was used to rinse out the wells and added to the sample. Tubes were centrifuged at 5000 × g for 5 min and supernatant (biocrust soil water) was pipetted and placed in new 2 mL tubes. Remaining biocrust was stored at −80 °C until nucleic acid extraction was performed. There were five biological replicates, five time points and four successional stages of biocrust resulting in 100 total samples. An extraction control (three replicates of water, no soil) was included and sampled at 49.5 h to evaluate background contamination. To control for abiotic metabolite processes (such as sorption, photodegradation, etc), a separate experiment was performed where soil water was sampled along the same time points (5 sampling time points in triplicate) from killed (autoclaved four times) biocrust from a late successional stage.
Metabolite extraction and LC/MS analysis
Biocrust soil water samples (1.5 mL) were lyophilized and resuspended in methanol (200 μL) containing internal standards (2–10 μg/mL) and filtered through 96-well Millipore filter plates (0.2 μm PVDF) by centrifuging at 1500 × g for 2 min. Samples were analyzed using normal-phase LC/MS with a ZIC-pHILIC column (150 × 2.1 mm, 3.5 μm 200 Å, Merck Sequant, Darmstadt, Germany) using an Agilent 1290 series UHPLC (Agilent Technologies, Santa Clara, California, USA). Chromatographic separation was achieved using two mobile phases, 5 mM ammonium acetate in water (A) and 90% acetonitrile with 5 mM ammonium acetate (B) at a flow rate of 0.25 mL/min with the following gradient: 100% B for 1.5 min, decreased to 50% B by 25 min, held until 29.9 min then returned to initial conditions by 30 min with a total runtime of 40 min. Column temperature was maintained at 40 °C. For MS, negative mode data were acquired on an Agilent 6550 quadrupole time-of-flight mass spectrometer and positive mode data were acquired on a Thermo QExactive (Thermo Fisher Scientific, Waltham, MA). Fragmentation spectra (MS/MS) were acquired for some metabolites using collision energies of 10–40 eV. For the killed biocrust controls, samples were analyzed using an Agilent 1290 series UHPLC with a SeQuant ZIC-HILIC column (150 × 2.1 mm, 5 μm, 200 Å, MilliporeSigma, Billerica, MA). The two mobile phases used were 5 mM ammonium acetate in water (A) and 95% acetonitrile with 5 mM ammonium acetate (B) at a flow rate of 0.45 mL/min with the following gradient: 100% B for 1.5 min, decreased to 65% B by 15 min then 0% B by 18 min, held until 23 min then returned to initial conditions by 25 min with a total runtime of 30 min. The negative and positive MS data for the killed controls were obtained using a Thermo QExactive.
Metabolite identification and statistical analysis
Metabolomics data were analyzed using Metabolite Atlas67 (https://github.com/biorack/metatlas) in conjunction with the Python programming language to construct extracted ion chromatograms corresponding to metabolites previously detected in biocrust or contained within our in-house standards library. Note that this same analysis could be done with other open-source software packages. Authentic standards were then used to validate assignments based on two orthogonal data (accurate mass less than 15 p.p.m., retention time within 1 min and/or MS/MS matching major fragments) relative to standards and/or the Metlin database68,69 and are provided in Supplementary Data 1. Metabolites that did not match orthogonal measures were classified as putative and are indicated by parentheses in figures. Internal standards were assessed from each sample to ensure peak area and retention times were consistent from sample-to-sample. Quality control mixtures were included at the beginning, end and throughout the runs to ensure proper instrument performance (m/z accuracy and retention time and peak area stability). Sample QC failed (internal standards were not present within the specified retention time window) for some replicates including all 9 h wetup level D samples and were not included for further analyses.
To explore the degree of variation in biocrust metabolite profiles across wetting and successional stages, biocrust samples were PCA-ordinated based on their metabolite profiles. Significance between temporal patterns from active biocrust and killed control samples was analyzed using the anovan and multcompare functions in Matlab R2016A with an alpha of 0.05 corresponding to 95% confidence level and Tukey’s honestly significant difference test where time is considered a continuous variable and succession is considered a categorical variable (Supplementary Data 3).
DNA extraction, sequencing and microbial annotation
DNA was extracted from biocrust (0.25 g) using the MoBio Powersoil DNA isolation kit (MoBio Laboratories, Inc, Carlsbad, CA) resulting in 100 µL of eluted DNA. Library preparation and sequencing were done at the QB3 facility at the University of California, Berkeley using Illumina HiSeq4000 (see supplementary methods for details on metagenome analysis). In recent studies24,25,70, ribosomal protein genes have been used as phylogenetic markers as an alternative to the more classical 16s ribosomal RNA gene. Ribosomal protein genes exist as single copies in almost all genomes, assemble well from metagenome datasets, are well-conserved and have produced higher resolution phylogenetic trees25. Given these advantages, the 50S ribosomal protein L15 (rplO) gene had the most extensive community coverage for our data set and was therefore used as a phylogenetic marker to examine the relative abundance of individual microorganisms within the microbial community across wetting and successional stages. The rplO genes from the genomes of the seven exometabolite profiled biocrust bacterial isolates20 were compared to all the rplO genes recovered from biocrust MAGs. Those with the highest percent similarity were considered the “closest matching environmental relatives” to the isolates and are reported in Table 1. Average nucleotide and amino acid identity metrics were calculated using the ‘enveomics’ tool71.
Microbe-metabolite correlations
Correlations were used to identify microbe–metabolite relationships across both wetting and successional stages. Spearman’s rank (rho) correlation coefficients for every pairwise (microbe–metabolite) relationship and p values (unadjusted and FDR-adjusted) were calculated using the cor() stats function in R. A Spearman’s rho value greater than or equal to 0.5 was considered “highly correlated” and less than or equal to −0.5 was considered “highly negatively correlated”. To test if the overall observed directionality (positive vs. negative correlations) was due to chance rather than as would be predicted based on exometabolomics (release vs. consumption), the exact binomial test was conducted using R (binom.test) with a total of 48 “trials” or observed microbe-metabolite interactions (Supplementary Data 7).
Exometabolomics comparison and analysis
Of the 85 metabolites detected in biocrust soil water, 32 were previously analyzed for consumption and release by biocrust isolates20. Of those metabolites, 23 were selected for further analyses since they were considered to be biologically-controlled (they displayed temporal patterns that were significantly different from the killed controls). For continued analyses of the isolate exometabolomics data here, fold-change was calculated by dividing the average peak area of each metabolite in (isolate) inoculated spent media by the non-inoculated control spent media (raw data can be found in the Supplemental Table in Baran et al.20). A metabolite was considered consumed if the fold-change was 0.5 or less and released metabolites had a fold-change of 2 or greater.
Microcoleus gene expression analysis
Microcoleus genes from Supplementary Table 6 in Rajeev et al.30 were categorized into KEGG pathways (for genes containing KEGG ID numbers), which are summarized in Supplementary Data 8. Analyses focused on pathways that are primarily anabolic or catabolic for metabolites that were released by Microcoleus PCC 9802. The average fold-change (relative to dry biocrusts) and standard errors were calculated for all genes belonging to pathways of interest.
Data availability
Sequencing data that support the findings of this study are available at the NCBI Sequence Read Archive (project accession number PRJNA395099; sample accession numbers SRX3024638 to SRX3024657, https://www.ncbi.nlm.nih.gov/bioproject/395099) and metabolomics data have been deposited to the EMBL-EBI MetaboLights database72 with the identifier MTBLS492 (http://www.ebi.ac.uk/metabolights/MTBLS492). Other relevant data supporting the findings of the study are available in this article and its Supplementary Information files, or from the corresponding author upon request.
References
Paustian, K. et al. Climate-smart soils. Nature 532, 49–57 (2016).
Friedlingstein, P. et al. Climate–carbon cycle feedback analysis: results from the C4MIP model intercomparison. J. Clim. 19, 3337–3353 (2006).
Judd, K. E., Crump, B. C. & Kling, G. W. Variation in dissolved organic matter controls bacterial production and community composition. Ecology 87, 2068–2079 (2006).
Collins, S. L. et al. A multiscale, hierarchical model of pulse dynamics in arid-land ecosystems. Annu. Rev. Ecol. Evol. Syst. 45, 397–419 (2014).
Siciliano, S. D. et al. Soil fertility is associated with fungal and bacterial richness, whereas pH is associated with community composition in polar soil microbial communities. Soil Biol. Biochem. 78, 10–20 (2014).
Berthrong, S. T., Buckley, D. H. & Drinkwater, L. E. Agricultural management and labile carbon additions affect soil microbial community structure and interact with carbon and nitrogen cycling. Microb. Ecol. 66, 158–170 (2013).
Maestre, F. T. et al. Increasing aridity reduces soil microbial diversity and abundance in global drylands. Proc. Natl. Acad. Sci. USA 112, 15684–15689 (2015).
Barnard, R. L., Osborne, C. A. & Firestone, M. K. Responses of soil bacterial and fungal communities to extreme desiccation and rewetting. ISME J. 7, 2229–2241 (2013).
Placella, S. A., Brodie, E. L. & Firestone, M. K. Rainfall-induced carbon dioxide pulses result from sequential resuscitation of phylogenetically clustered microbial groups. Proc. Natl. Acad. Sci. USA 109, 10931–10936 (2012).
Belnap, J., Weber, B. & Büdel, B. in Biological Soil Crusts: An Organizing Principle in Drylands (eds. Weber, B., Büdel, B. & Belnap, J.) 3–13 (Springer, Switzerland, 2016).
Maestre, F. T. et al. Structure and functioning of dryland ecosystems in a changing world. Annu. Rev. Ecol. Evol. Syst. 47, 215–237 (2016).
Elbert, W. et al. Contribution of cryptogamic covers to the global cycles of carbon and nitrogen. Nat. Geosci. 5, 459–462 (2012).
Garcia-Pichel, F. & Pringault, O. Cyanobacteria track water in desert soils. Nature 413, 380–381 (2001).
Baldock, J. A. & Nelson, P. N. Soil Organic Matter. In Handbook of Soil Science (ed. Sumner, M. E.) B25–B84 (CRC Press, Boca Raton, FL, 2000).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Kogel-Knabner, I. The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil. Biol. Biochem. 34, 139–162 (2002).
Swenson, T. L., Jenkins, S., Bowen, B. P. & Northen, T. R. Untargeted soil metabolomics methods for analysis of extractable organic matter. Soil. Biol. Biochem. 80, 189–198 (2015).
Warren, C. R. Comparison of methods for extraction of organic N monomers from soil microbial biomass. Soil. Biol. Biochem. 81, 67–76 (2015).
Fierer, N. & Lennon, J. T. The generation and maintenance of diversity in microbial communities. Am. J. Bot. 98, 439–448 (2011).
Baran, R. et al. Exometabolite niche partitioning among sympatric soil bacteria. Nat. Commun. 6, 1–9 (2015).
Silva, L. P. & Northen, T. R. Exometabolomics and MSI: deconstructing how cells interact to transform their small molecule environment. Curr. Opin. Biotechnol. 34, 209–216 (2015).
Prosser, J. I. Dispersing misconceptions and identifying opportunities for the use of ‘omics’ in soil microbial ecology. Nat. Rev. Microbiol. 13, 439–446 (2015).
Yutin, N., Puigbo, P., Koonin, E. V. & Wolf, Y. I. Phylogenomics of prokaryotic ribosomal proteins. PLoS ONE 7, e36972 (2012).
Wu, M. & Eisen, J. A. A simple, fast, and accurate method of phylogenomic inference. Genome Biol. 9, R151 (2008).
Hug, L. A. et al. A new view of the tree of life. Nat. Microbiol. 1, 1–6 (2016).
Anantharaman, K. et al. Thousands of microbial genomes shed light on interconnected biogeochemical processes in an aquifer system. Nat. Commun. 7, 1–11 (2016).
Sorek, R. et al. Genome-wide experimental determination of barriers to horizontal gene transfer. Science 318, 1449–1452 (2007).
Rodriguez-R, L. M. & Konstantinidis, K. T. Bypassing cultivation to identify bacterial species. Microbe 9, 111–118 (2014).
Garcia-Pichel, F. & Wojciechowski, M. F. The evolution of a capacity to build supra-cellular ropes enabled filamentous cyanobacteria to colonize highly erodible substrates. PLoS ONE 4, e7801–e7806 (2009).
Rajeev, L. et al. Dynamic cyanobacterial response to hydration and dehydration in a desert biological soil crust. Isme. J. 7, 2178–2191 (2013).
Walsh, A. M. et al. Microbial succession and flavor production in the fermented dairy beverage kefir. mSystems 1, e00052 (2016).
Ding, J. et al. Integrated metagenomics and network analysis of soil microbial community of the forest timberline. Sci. Rep. 5, 7994–8010 (2015).
Trivedi, P. et al. Microbial regulation of the soil carbon cycle: Evidence from gene-enzyme relationships. ISME J. 10, 2593–2604 (2016).
Garcia-Pichel, F., Lopez-Cortes, A. & Nubel, U. Phylogenetic and morphological diversity of cyanobacteria in soil desert crusts from the Colorado Plateau. Appl. Environ. Microbiol. 67, 1902–1910 (2001).
Angel, R. & Conrad, R. Elucidating the microbial resuscitation cascade in biological soil crusts following a simulated rain event. Environ. Microbiol. 15, 2799–2815 (2013).
Nicholson, W. L., Munakata, N., Horneck, G., Melosh, H. J. & Setlow, P. Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol. Mol. Biol. Rev. 64, 548–572 (2000).
Nicholson, W. L. Roles of Bacillus endospores in the environment. Cell. Mol. Life. Sci. 59, 410–416 (2002).
Pepe-Ranney, C. et al. Non-cyanobacterial diazotrophs mediate dinitrogen fixation in biological soil crusts during early crust formation. Isme. J. 10, 287–298 (2016).
Hosoya, S., Lu, Z., Ozaki, Y., Takeuchi, M. & Sato, T. Cytological analysis of the mother cell death process during sporulation in bacillus subtilis. J. Bacteriol. 189, 2561–2565 (2007).
Upton, A. C. & Nedwell, D. B. Nutritional flexibility of oligotrophic and copiotrophic antarctic bacteria with respect to organic substrates. Fems. Microbiol. Ecol. 62, 1–6 (1989).
Morbach, S. & Krämer, R. Body shaping under water stress: osmosensing and osmoregulation of solute transport in bacteria. Chembiochem 3, 384–397 (2002).
Kell, D. B., Swainston, N., Pir, P. & Oliver, S. G. Membrane transporter engineering in industrial biotechnology and whole cell biocatalysis. Trends Biotechnol. 33, 237–246 (2015).
Garcia-Pichel, F., Loza, V., Marusenko, Y., Mateo, P. & Potrafka, R. M. Temperature drives the continental-scale distribution of key microbes in topsoil communities. Science 340, 1574–1577 (2013).
Ferrenberg, S., Reed, S. C. & Belnap, J. Climate change and physical disturbance cause similar community shifts in biological soil crusts. Proc. Natl. Acad. Sci. USA 112, 12116–12121 (2015).
Allen, E. E. & Banfield, J. F. Community genomics in microbial ecology and evolution. Nat. Rev. Microbiol. 3, 489–498 (2005).
Martiny, J. B. H., Jones, S. E., Lennon, J. T. & Martiny, A. C. Microbiomes in light of traits: A phylogenetic perspective. Science 350, aac9323 (2015).
Herren, C. M. & McMahon, K. D. Cohesion: a method for quantifying the connectivity of microbial communities. ISME J. 11, 2426–2438 (2017).
Erbilgin, O. et al. Dynamic substrate preferences and predicted metabolic properties of a simple microbial consortium. BMC Bioinformatics, 18, 1–12 (2017).
Behrends, V., Ebbels, T. M. D., Williams, H. D. & Bundy, J. G. Time-resolved metabolic footprinting for nonlinear modeling of bacterial substrate utilization. Appl. Environ. Microbiol. 75, 2453–2463 (2009).
Seth, E. C. & Taga, M. E. Nutrient cross-feeding in the microbial world. Front. Microbiol. 5, 1–6 (2014).
Ponomarova, O. & Patil, K. R. Metabolic interactions in microbial communities: untangling the Gordian knot. Curr. Opin. Microbiol. 27, 37–44 (2015).
Orsi, W. D. et al. Diverse, uncultivated bacteria and archaea underlying the cycling of dissolved protein in the ocean. ISME J. 10, 2158–2173 (2016).
Fike, D. A., Gammon, C. L., Ziebis, W. & Orphan, V. J. Micron-scale mapping of sulfur cycling across the oxycline of a cyanobacterial mat: a paired nanoSIMS and CARD-FISH approach. ISME J. 2, 749–759 (2008).
Woebken, D. et al. Revisiting N2 fixation in Guerrero Negro intertidal microbial mats with a functional single-cell approach. ISME J. 9, 485–496 (2015).
Baran, R. et al. Metabolic footprinting of mutant libraries to map metabolite utilization to genotype. ACS Chem. Biol. 8, 189–199 (2013).
Gougoulias, C., Clark, J. M. & Shaw, L. J. The role of soil microbes in the global carbon cycle: tracking the below-ground microbial processing of plant-derived carbon for manipulating carbon dynamics in agricultural systems. J. Sci. Food Agric. 94, 2362–2371 (2014).
Stuart, R. K. et al. Cyanobacterial reuse of extracellular organic carbon in microbial mats. ISME J. 10, 1240–1251 (2016).
Malmstrom, R. R., Kiene, R. P., Cottrell, M. T. & Kirchman, D. L. Contribution of SAR11 bacteria to dissolved dimethylsulfoniopropionate and amino acid uptake in the North Atlantic Ocean. Appl. Environ. Microbiol. 70, 4129–4135 (2004).
Kaiser, M. et al. The influence of mineral characteristics on organic matter content, composition, and stability of topsoils under long-term arable and forest land use. J. Geophys. Res. 117, G02018 (2012).
Swenson, T. L., Bowen, B. P., Nico, P. S. & Northen, T. R. Competitive sorption of microbial metabolites on an iron oxide mineral. Soil Biol. Biochem. 90, 34–41 (2015).
Lv, J. et al. Molecular-scale investigation with ESI-FT-ICR-MS on fractionation of dissolved organic matter induced by adsorption on iron oxyhydroxides. Environ. Sci. Technol. 50, 2328–2336 (2016).
Diamond, S., Jun, D., Rubin, B. E. & Golden, S. S. The circadian oscillator in Synechococcus elongatus controls metabolite partitioning during diurnal growth. Proc. Natl. Acad. Sci. USA 112, E1916–E1925 (2015).
Orth, J. D., Thiele, I. & Palsson, B. Ø. What is flux balance analysis? Nat. Biotechnol. 28, 245–248 (2010).
Allison, S. D. A trait-based approach for modelling microbial litter decomposition. Ecol. Lett. 15, 1058–1070 (2012).
Zomorrodi, A. R. & Maranas, C. D. OptCom: A multi-level optimization framework for the metabolic modeling and analysis of microbial communities. PLoS Comput. Biol. 8, e1002363 (2012).
Strauss, S. L., Day, T. A. & Garcia-Pichel, F. Nitrogen cycling in desert biological soil crusts across biogeographic regions in the Southwestern United States. Biogeochemistry 108, 171–182 (2011).
Yao, Y. et al. Analysis of metabolomics datasets with high-performance computing and metabolite atlases. Metabolites 5, 431–442 (2015).
Smith, C. A. et al. METLIN: A metabolite mass spectral database. Ther. Drug Monit. 27, 747–751 (2005).
Sumner, L. W. et al. Proposed minimum reporting standards for chemical analysis. Metabolomics 3, 211–221 (2007).
Sharon, I. et al. Accurate, multi-kb reads resolve complex populations and detect rare microorganisms. Genome Res. 25, 534–543 (2015).
Rodriguez-R, L. M. & Konstantinidis, K. T. The enveomics collection: a toolbox for specialized analyses of microbial genomes and metagenomes. https://doi.org/10.7287/peerj.preprints.1900v1 (2016).
Haug, K. et al. MetaboLights—an open-access general-purpose repository for metabolomics studies and associated meta-data. Nucleic Acids Res. 41, D781–D786 (2012).
Acknowledgements
This work was funded by the Office of Science Early Career Research Program, Office of Biological and Environmental Research, of the U. S. Department of Energy under contract number DE-AC02-05CH11231 to Lawrence Berkeley National Laboratory. DNA was sequenced using the Vincent J. Coates Genomics Sequencing Laboratory at UC Berkeley, supported by NIH S10 OD018174 Instrumentation Grant. We thank Rebecca Lau for technical assistance in biocrust sample collection and experimentation.
Author information
Authors and Affiliations
Contributions
T.L.S. and T.R.N. conceived the study, designed the experiments and wrote the manuscript. T.L.S. performed the experiments. T.L.S. and B.P.B. analyzed the metabolomics data. U.K. analyzed the metagenomics data. T.L.S. and J.M.S. conducted correlation and statistical analyses. All co-authors commented on the design of experiments, data analysis and draft manuscripts.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Swenson, T.L., Karaoz, U., Swenson, J.M. et al. Linking soil biology and chemistry in biological soil crust using isolate exometabolomics. Nat Commun 9, 19 (2018). https://doi.org/10.1038/s41467-017-02356-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-017-02356-9
This article is cited by
-
The interplay between microbial communities and soil properties
Nature Reviews Microbiology (2024)
-
Survival and rapid resuscitation permit limited productivity in desert microbial communities
Nature Communications (2024)
-
Biogeographic patterns and drivers of soil viromes
Nature Ecology & Evolution (2024)
-
Changes in Microbial Composition During the Succession of Biological Soil Crusts in Alpine Hulun Buir Sandy Land, China
Microbial Ecology (2024)
-
Predicting metabolomic profiles from microbial composition through neural ordinary differential equations
Nature Machine Intelligence (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.