Abstract
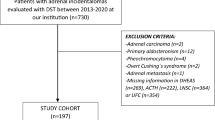
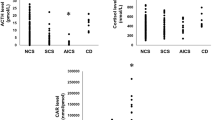
Patients with adrenal Cushing’s syndrome have a poor prognosis due to the autonomous hypersecretion of endogenous cortisol from the adrenal gland. Although several clinical examinations have been developed to manifest the autonomous hypersecretion of endogenous cortisol in this syndrome, it is unknown which parameter of cortisol overproduction is best associated with the adrenal cortisol content and bone and vascular complications of this syndrome. Therefore, the present study was undertaken to evaluate the parameter(s) associated with the adrenal cortisol content in patients with adrenal Cushing’s syndrome, as well as to assess the associations between these parameters and the vascular and bone complications of this syndrome. Cardiovascular factors such as blood pressure and pulse rate, glucose metabolism, lipid metabolism, renal function, and indices of arteriosclerosis, except for diastolic blood pressure, failed to show an association with the adrenal cortisol content. Twenty-four-hour urinary free cortisol excretion (ρ = 0.893, P = 0.007) and plasma cortisol levels after the 1-mg (ρ = 0.857, P = 0.014) or 8-mg (ρ = 0.900, P = 0.037) overnight dexamethasone suppression test, but not plasma cortisol levels in the morning or late at night or indices of 131I-adosterol single-photon emission computed tomography-computed tomography, were significantly and positively associated with the adrenal cortisol content. Twenty-four-hour urinary free cortisol excretion, and not plasma cortisol levels after the 1- or 8-mg overnight dexamethasone suppression test, was significantly and negatively associated with lumbar spine bone density (lumbar spine bone mineral density, ρ = −0.786, P = 0.036; lumbar spine T score, ρ = −0.883, P = 0.009; and lumbar spine Z score, ρ = −0.883, P = 0.009). These results indicate that 24-h urinary free cortisol excretion is best associated with a decrease in bone density via increased adrenal cortisol overproduction and suggest that it could be used as a marker for osteoporosis in these patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
12 August 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41440-022-00991-4
References
Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, et al. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93:1526–40.
Tanaka M. Improving obesity and blood pressure. Hypertens Res. 2020;43:79–89.
Pivonello R, De Martino MC, De Leo M, Tauchmanova L, Faggiano A, Lombardi G, et al. Cushing’s syndrome: aftermath of the cure. Arq Bras Endocrinol Metab. 2007;51:1381–91.
Masuda T, Nagata D. Recent advances in the management of secondary hypertension: chronic kidney disease. Hypertens Res. 2020;43:869–75.
Schoina M, Loutradis C, Memmos E, Dimitroulas T, Pagkopoulou E, Doumas M, et al. Microcirculatory function deteriorates with advancing stages of chronic kidney disease independently of arterial stiffness and atherosclerosis. Hypertens Res. 2021;44:179–87.
Chiu NKH, Ehgoetz Martens KA, Mok VCT, Lewis SJG, Matar E. Prevalence and predictors of mood disturbances in idiopathic REM sleep behaviour disorder. J Sleep Res. 2021;30:e13040.
Ghatage T, Goyal SG, Dhar A, Bhat A. Novel therapeutics for the treatment of hypertension and its associated complications: peptide- and nonpeptide-based strategies. Hypertens Res. 2021;44:740–55.
Hisamatsu T, Segawa H, Kadota A, Ohkubo T, Arima H, Miura K. Epidemiology of hypertension in Japan: beyond the new 2019 Japanese guidelines. Hypertens Res. 2020;43:1344–51.
Arnaldi G, Angeli A, Atkinson AB, Bertagna X, Cavagnini F, Chrousos GP, et al. Diagnosis and complications of Cushing’s syndrome: a consensus statement. J Clin Endocrinol Metab. 2003;88:5593–602.
Stratakis CA. Cushing syndrome caused by adrenocortical tumors and hyperplasias (corticotropin- independent Cushing syndrome). Endocr Dev. 2008;13:117–32.
Bolland MJ, Holdaway IM, Berkeley JE, Lim S, Dransfield WJ, Conaglen JV, et al. Mortality and morbidity in Cushing’s syndrome in New Zealand. Clin Endocrinol (Oxf). 2011;75:436–42.
Ahn CH, Kim JH, Park MY, Kim SW. Epidemiology and comorbidity of adrenal Cushing syndrome: a nationwide cohort study. J Clin Endocrinol Metab. 2021;106:e1362–e1372.
Javanmard P, Duan D, Geer EB. Mortality in patients with endogenous Cushing’s syndrome. Endocrinol Metab Clin North Am. 2018;47:313–33.
Di Dalmazi G, Vicennati V, Garelli S, Casadio E, Rinaldi E, Giampalma E, et al. Cardiovascular events and mortality in patients with adrenal incidentalomas that are either non-secreting or associated with intermediate phenotype or subclinical Cushing’s syndrome: a 15-year retrospective study. Lancet Diabetes Endocrinol. 2014;2:396–405.
Debono M, Bradburn M, Bull M, Harrison B, Ross RJ, Newell-Price J. Cortisol as a marker for increased mortality in patients with incidental adrenocortical adenomas. J Clin Endocrinol Metab. 2014;99:4462–70.
Patrova J, Kjellman M, Wahrenberg H, Falhammar H. Increased mortality in patients with adrenal incidentalomas and autonomous cortisol secretion: a 13-year retrospective study from one center. Endocrine. 2017;58:267–75.
Payne RB, Little AJ, Williams RB, Milner JR. Interpretation of serum calcium in patients with abnormal serum proteins. Br Med J. 1973;4:643–6.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Shirai K, Utino J, Otsuka K, Takata M. A novel blood pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI). J Atheroscler Thromb. 2006;13:101–7.
Binkley N, Kiebzak GM, Lewiecki EM, Krueger D, Gangnon RE, Miller PD, et al. Recalculation of the NHANES database SD improves T-score agreement and reduces osteoporosis prevalence. J Bone Min Res. 2005;20:195–201.
Tamura Y, Okinaga H, Takami H. Glucocorticoid-induced osteoporosis. Biomed Pharmacother. 2004;58:500–4.
Suzuki Y, Ichikawa Y, Saito E, Homma M. Importance of increased urinary calcium excretion in the development of secondary hyperparathyroidism of patients under glucocorticoid therapy. Metabolism. 1983;32:151–6.
Acknowledgements
We thank Noriko Morishima for her technical support with the measurement of cortisol concentrations in the adrenal tumors and Asanuma Masayasu and Youko Kizuka for their technical support with the 131I-adosterol SPECT/CT analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The original version of this article Contained errors in Table 2. In it, several numbers were incorrectly given in columns corresponding to the Endocrinological examination.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kimura Takiguchi, M., Morimoto, S., Watanabe, D. et al. Urinary free cortisol excretion is associated with lumbar bone density in patients with adrenal Cushing’s syndrome. Hypertens Res 45, 1632–1642 (2022). https://doi.org/10.1038/s41440-022-00939-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-022-00939-8