Abstract
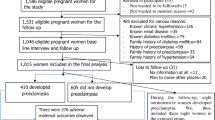
This study aimed to investigate the diagnostic accuracy of the Fetal Medicine Foundation (FMF) Bayes theorem-based model for the prediction of preeclampsia (PE) at 11–13 weeks of gestation in the Japanese population. In this prospective cohort study, we invited 2655 Japanese women with singleton pregnancies at 11–13 weeks of gestation to participate, of whom 1036 women provided written consent. Finally, we included 913 women for whom all measurements and perinatal outcomes were available. Data on maternal characteristics and medical history were recorded. Mean arterial pressure (MAP), uterine artery pulsatility index, and maternal serum placental growth factor (PlGF) were measured. The patients delivered their babies at Showa University Hospital between June 2017 and December 2019. Participants were classified into high- and low-risk groups according to the FMF Bayes theorem-based model. Frequencies of PE were compared between groups. The screening performance of the model was validated using the area under receiver operating characteristic (AUROC) curve. A total of 26 patients (2.8%) developed PE, including 11 patients (1.2%) with preterm PE (delivery at <37 weeks). The frequency of preterm PE was significantly higher in the high-risk group than in the low-risk group (3.8% vs. 0.2%, p < 0.05). This population model achieved a 91% detection rate for the prediction of preterm PE at a screen-positive rate of 10% by a combination of maternal characteristics, MAP, and PlGF. The AUROC curve for the prediction of preterm PE was 0.962 (0.927–0.981). In conclusion, the prediction of preterm PE using the FMF Bayes theorem-based model is feasible in the Japanese population.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
23 December 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41440-020-00593-y
08 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41440-021-00622-4
09 April 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41440-021-00653-x
References
Bujold E, Roberge S, Lacasse Y, Bureau M, Audibert F, Marcoux S, et al. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: a meta-analysis. Obstet Gynecol. 2010;116:402–414.
Rolnik DL, Wright D, Poon LC, O’Gorman N, Syngelaki A, de Paco Matallana C, et al. Aspirin versus placebo in pregnancies at hight risk for preterm preeclampsia. N. Engl J Med. 2017;377:613–622.
Wright D, Rolnik DL, Syngelaki A, de Paco Matallana C, Machuca M, de Alvarado M, et al. Aspirin for evidence-based preeclampsia prevention trial: effect of aspirin on length of stay in the neonatal intensive care unit. Am J Obstet Gynecol. 2018;218:612.e1–612.e6.
O’Gorman N, Wright D, Syngelaki A, Akolekar R, Wright A, Poon LC, et al. Competing risks model in screening for preeclampsia by maternal factors and biomarkers at 11–13 weeks gestation. Am J Obstet Gynecol. 2016;214:103.e1–103.e12.
Krantz DA, Hallahan TW. Incorporating the probability of competing event(s) into the preeclampsia competing risk algorithm. Am J Obstet Gynecol. 2019;221:533–4.
Chaemsaithong P, Pooh RK, Zheng M, Ma R, Chaiyasit N, Tokunaka M, et al. Prospective evaluation of screening performance of first trimester prediction models for preterm preeclampsia in an Asian population. Am J Obstet Gynecol. 2019;221:650.e1–650.e16.
Wright D, Syngelaki A, Akolekar R, Poon LC, Nicolaides KH. Competing risks model in screening for preeclampsia by maternal characteristics and medical history. Am J Obstet Gynecol. 2015;213:62.e1–62.e10.
Poon LC, Zymeri NA, Zamprakou A, Syngelaki A, Nicolaides KH. Protocol for measurement of mean arterial pressure at 11–13 weeks gestation. Fetal Diagn Ther. 2012;31:42–48.
Khalil A, Nicolaides KH. How to record uterine artery Doppler in the first trimester. Ultrasound Obstet Gynecol. 2013;42:478–479.
Rolnik DL, da Silva Costa F, Sahota D, Hyett J, McLennan A. Quality assessment of uterine artery Doppler measurement in first-trimester combined screening for pre-eclampsia. Ultrasound Obstet Gynecol. 2019;53:245–50.
Sato K. A proposal for a new definition and classification of “Pregnancy induced Hypertension (PIH)” (2004). In: Japan Society for the Study of Toxemia of Pregnancy (ed), Historical Perspective of Study of Pregnancy-Induced Hypertension in Japan Tokyo. Tokyo, Japan: Medical View Co; 2005. p. 54–87.
Watanabe K, Naruse K, Tanaka K, Metoki H, Suzuki Y. Outline of definition and classification of “Pregnancy induced Hypertension (PIH)”. Hypertens Res Pregnancy. 2013;1:3–4.
O’Gorman N, Wright D, Poon LC, Rolnik DL, Syngelaki A, de Alvarado M, et al. Multicenter screening for pre-eclampsia by maternal factors and biomarkers at 11–13 weeks gestation: comparison with NICE guidelines and ACOG recommendations. Ultrasound Obstet Gynecol. 2017;49:756–760.
Wright A, Wright D, Syngelaki A, Georgantis A, Nicolaides KH. Two-stage screening for preterm preeclampsia at 11–13 weeks gestation. Am J Obstet Gynecol. 2019;220:197.E1–197.E11.
Tong J, Niu Y, Chen ZJ, Zhang C. Comparison of the transcriptional profile in the decidua of early-onset and late-onset pre-eclampsia. J Obstet Gynecol Res. 2020;46:1055–1066.
Roberts JM, Hubel CA. The two stage model of preeclampsia: variations on the theme. Placenta. 2009;30:S32–S37.
Brunelli E, Seidenari A, Germano C, Prefumo F, Cavoretto P, Di Martino D, et al. External validation of a simple risk score based on the ASPRE trial algorithm for preterm pre-eclampsia considering maternal characteristics in nulliparous pregnant women: a multicenter retrospective cohort study. BJOG. 2020;127:1210–5.
Di Martino D, Masturzo B, Paracchini S, Bracco B, Cavoretto P, Prefumo F, et al. Comparison of two “a priori” risk assessment algorithms for preeclampsia in Italy: a prospective multicenter study. Arch Gynecol Obstet. 2019;299:1587–96.
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research (C) (No. 19K18653) from the Japan Society for the Promotion of Science. We kindly thank all the participants, obstetricians, nurses, and research assistants employed in this study. This work was supported by the PerkinElmer Company (Waltham, MA, USA), which provided the database. Supplementary information is available at the Hypertension Research website.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
PCL has received speaker fees and consultancy payments from Roche Diagnostics, Ferring Pharmaceuticals, and GE Healthcare and in-kind contributions from Roche Diagnostics, PerkinElmer, Thermo Fisher Scientific, and GE Healthcare. The other authors report no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: plus the same explanatory text of the problem as in the erratum/correction article.
Supplementary information
Rights and permissions
About this article
Cite this article
Goto, M., Koide, K., Tokunaka, M. et al. Accuracy of the FMF Bayes theorem-based model for predicting preeclampsia at 11–13 weeks of gestation in a Japanese population. Hypertens Res 44, 685–691 (2021). https://doi.org/10.1038/s41440-020-00571-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-020-00571-4
Keywords
This article is cited by
-
Prediction of preterm preeclampsia risk in Asians using a simple two-item assessment in early pregnancy
Hypertension Research (2024)
-
Automated electrochemiluminescence immunoassay for serum PlGF levels in women with singleton pregnancy at 9–13 weeks of gestation predicts preterm preeclampsia: a retrospective cohort study
Hypertension Research (2023)
-
Annual reports on hypertension research 2020
Hypertension Research (2022)