Abstract
To investigate whether anemia is an independent risk factor for cardiovascular and renal events in hypertensive outpatients, we performed a subgroup analysis of the ATTEMPT-CVD study based on baseline hemoglobin. The ATTEMPT-CVD study was a multicenter, prospective, randomized study of hypertensive outpatients that compared the efficacy of angiotensin receptor blocker (ARB)-based antihypertensive treatment with non-ARB antihypertensive treatment over 3 years. In the present subanalysis, ATTEMPT-CVD study participants (n = 1213) were categorized into the anemic group and nonanemic group according to their baseline hemoglobin. We compared the anemic and nonanemic groups mainly in regard to the incidence of cardiovascular and renal events and blood pressure. We also performed a multivariable Cox proportional hazards analysis to determine the prognostic factors that were independently associated with cardiovascular and renal events. Of the 1213 patients enrolled in the ATTEMPT-CVD, 194 patients had anemia (mostly mild anemia) and 1019 patients did not. Blood pressure was well-controlled during the 3 years of antihypertensive therapy in both the anemic and nonanemic groups. However, the incidence of cardiovascular and renal events was significantly greater in the anemic group than in the nonanemic group (HR = 1.945: 95%CI 1.208–3.130; P = 0.0062). Even after adjustment, anemia was independently associated with cardiovascular and renal events (HR = 1.816: 95%CI 1.116–2.955; P = 0.0163) in overall hypertensive patients with well-controlled blood pressure. Anemia, even mild anemia, is an independent risk factor for cardiovascular and renal events in hypertensive outpatients whose blood pressure is well-controlled. Thus, anemia may be a novel therapeutic target for cardiovascular and renal diseases in hypertensive outpatients with anemia.
Similar content being viewed by others
Introduction
High blood pressure is the worldwide leading cause of death and disability-adjusted life years [1,2,3]. Hypertension is more responsible for cardiovascular deaths than any other modifiable risk factors [4] and is one of the most important risk factors for end-stage renal disease [5]. Lowering blood pressure is known to considerably reduce cardiovascular or renal morbidity and mortality in hypertensive patients [3]. However, it is important to note that an antihypertensive treatment cannot completely prevent cardiovascular or renal events in hypertensive patients and that residual cardiovascular risk exists even in hypertensive patients with intensive blood pressure control [3]. Little is known about the specifics of the residual cardiovascular risk in hypertensive patients with well-controlled blood pressure. A trial of telmisartan prevention of cardiovascular disease (ATTEMPT-CVD) was a multicenter, prospective, randomized, active-controlled trial that compared the effects of angiotensin II receptor blocker (ARB)-based antihypertensive therapy and those of non-ARB antihypertensive therapy on biomarker level changes and the incidence of cardiovascular and renal events in Japanese hypertensive outpatients who had at least one of several specified cardiovascular risk factors [6, 7]. The ATTEMPT-CVD study found that despite similar reductions in blood pressure, ARB-based antihypertensive therapy reduced urinary albumin excretion more than non-ARB therapy and was more effective at preventing an increase in plasma brain natriuretic peptide (BNP) than non-ARB therapy [6]. However, the incidence of cardiovascular and renal events did not differ between ARB-based antihypertensive therapy and non-ARB antihypertensive therapy [6]. These findings showed that the favorable reduction in urinary albumin excretion or plasma BNP by ARB-based therapy was not associated with cardiovascular and renal events in hypertensive outpatients [6]. Therefore, unknown cardiovascular risk factors other than urinary albumin or plasma BNP seem to be responsible for the cardiovascular and renal outcomes in the cohort of participants in the ATTEMPT-CVD study.
Anemia is common among patients with advanced chronic kidney disease (CKD) [8,9,10,11] and those with heart failure [12,13,14,15,16], and anemia is significantly associated with cardiovascular or renal events in these patients, although the benefit of interventions targeting anemia in these patients is controversial [17,18,19,20,21,22]. In contrast with the abundant evidence that anemia poses a significant risk for cardiovascular and renal outcomes in advanced CKD or heart failure, the clinical significance of anemia in hypertension is still unknown. In the present study, to explore the significance of anemia as a residual cardiovascular and renal risk in hypertensive outpatients with well-controlled blood pressure, we performed a subgroup analysis of the patients enrolled in the ATTEMPT-CVD study according to their anemia status (based on baseline hemoglobin levels). We obtained novel evidence to support that anemia, even mild anemia, is an independent risk factor for cardiovascular and renal events in hypertensive outpatients with well-controlled blood pressure.
Methods
Inclusion and exclusion criterion and study protocol of ATTEMPT-CVD
The method used in ATTEMPT-CVD has been described [7]. This trial was registered with ClinicalTrials.gov under number NCT01075698. The major findings of the ATTEMPT-CVD study have already been reported [6]. In brief, ATTEMPT-CVD was a multicenter, prospective, randomized, open-label, controlled trial with a blinded endpoint assessment. Patient enrollment started in July 2009 and was completed in April 2011. Participants were followed up for 3 years at 168 institutions throughout Japan. The inclusion criteria were: outpatients, 40–79 years old, patients with a systolic blood pressure ≥140 mmHg or diastolic blood pressure ≥90 mmHg based on the latest two blood pressure measurements taken in the sitting position or patients who still required treatment for high blood pressure even with a systolic blood pressure <140 mmHg and diastolic blood pressure <90 mmHg, patients with the ability to provide informed consent, and patients who had at least one of several listed cardiovascular risks [7]. The exclusion criteria were: type 1 diabetes, severe renal disorder (serum creatinine level ≥2.0 mg/dL), heart failure (New York Heart Association (NYHA) Classification III or IV), myocardial infarction, percutaneous revascularization and bypass graft of coronary artery/lower-extremity blood vessel, cerebral infarction, cerebral hemorrhage, subarachnoid hemorrhage and transient cerebral ischemic attack within 6 months before the observation period, malignant hypertension, secondary hypertension, pregnant women, clinically relevant allergic disease or past history of hypersensitivity to drugs, past history of significant adverse drug reactions, extremely poor biliary secretion or serious hepatic disorder, patients requiring treatment for a malignant tumor, and patients otherwise judged by the physicians to be unsuitable for the study. The study protocol for the current subanalysis satisfied the guidelines of the ethics committees at our institutions and the study complied with the Declaration of Helsinki. The ATTEMPT-CVD patients were randomly allocated to either the ARB group or the non-ARB group. Stratified randomization was performed according to the following prognostic variables: age ≥65 or age <65, men or women, past history of cardiovascular events, past history of diabetes mellitus, and usage of an ACE inhibitor. Patients allocated to the ARB group were treated with telmisartan. By contrast, patients allocated to the non-ARB group were treated with standard therapy other than ARB. The study physicians obtained relevant survey measurements at the start of the study (at registration), at 3, 6, 12, 24, and 36 months from the start of the study, at any discontinuation/dropout, at the occurrence of any cardiovascular or renal event, and at the occurrence of any adverse event.
Endpoints of ATTEMPT-CVD and the current subanalysis
In ATTEMPT-CVD, the primary endpoints were changes in urinary albumin/creatinine ratio (UACR) and plasma BNP levels from baseline, as described [7]. The prespecified secondary endpoints were time until occurrence of composite cardiovascular and renal events, including cerebral events (cerebral infarction, cerebral hemorrhage, subarachnoid hemorrhage, unknown type of stroke, transient ischemic attack), cardiac events (sudden death, myocardial infarction, angina pectoris, asymptomatic myocardial ischemia, heart failure), aortic/peripheral arterial events (aortic aneurysm, aortic dissection, arteriosclerotic disease), newly occurred or aggravated diabetic complications (diabetic nephropathy, diabetic retinopathy, diabetic neuropathy), and aggravation of renal function (doubling of serum creatinine level, initiation of dialysis, renal transplantation) [7]. The changes in plasma hsCRP (high sensitivity C-reactive protein), urinary 8-OHdG, serum adiponectin, serum high-molecular-weight adiponectin, and estimated glomerular filtration rate (eGFR) were also specified as secondary endpoints.
As described above, the incidence of composite cardiovascular and renal events was a prespecified secondary endpoint of ATTEMPT-CVD [6, 7]. In our subgroup analysis, the incidence of cardiovascular and renal events was compared between anemic and nonanemic groups of the patients enrolled in ATTEMPT-CVD. In addition, the significance of anemia as a cardiovascular or renal risk factor was evaluated.
Categorization of the patients enrolled in the ATTEMPT-CVD study according to baseline hemoglobin
In the present study, anemia was defined according to hemoglobin concentration, which is a standard indicator for anemia. According to the World Health Organization (WHO) definition, anemia exists when hemoglobin <13 g/dL in men and <12 g/dL in women. The patients enrolled in ATTEMPT-CVD were categorized into anemic group and nonanemic group based on this definition.
Statistical analyses
The sample size and power of ATTEMPT-CVD were established as described by Soejima et al. [7]. All analyses were performed on the intention-to-treat population. With respect to cardiovascular and renal events, time to first event curves was estimated using the Kaplan–Meier method and the log-rank test was used to show the differences between anemic and nonanemic patients. Using the Cox proportional hazards model, the hazard ratio (HR) of the anemic group to the nonanemic group and its 95% confidence interval (CI) were calculated. To estimate the heterogeneity of the HR for the subgroup by sex, the interaction between anemia and sex was assessed using the interaction terms in a stratified Cox proportional hazards model. Repeated measures analysis of variance was used to compare anemic and nonanemic groups on the time course of systolic and diastolic blood pressure during the follow-up period. Multivariable Cox proportional hazards analysis in combination with the backward selection method was performed to determine the independent association of each prognostic factor with the incidence of cardiovascular and renal events. The original model included the following 10 covariates: the presence of anemia, sex, age, presence of baseline cardiovascular diseases, presence of baseline diabetes, eGFR, plasma BNP, UACR, current smoking status, and allocation to ARB-based antihypertensive treatment. Each baseline parameter of the patients was compared between anemic and nonanemic groups using the t-test or Mann–Whitney test for continuous variables and χ2 tests for categorical variables. The software used for the statistical analysis was Windows SAS version 9.2 and subsequent versions (SAS Institute Japan Ltd., Tokyo, Japan). P-values of less than 0.05 were considered statistically significant.
Results
Baseline characteristics of patients with anemia and those without
After excluding 15 of the 1228 patients enrolled in the ATTEMPT-CVD study because of unavailability of baseline hemoglobin levels, 1213 patients were included in the present subanalysis. As shown by the baseline hemoglobin distribution in Fig. 1, most anemic patients had mild anemia, regardless of sex. Only 7 (3.6%) of the 194 anemic patients had hemoglobin levels of less than 10 g/dL. No patient was treated with an erythropoiesis-stimulating agent. Table 1 shows the comparison between the baseline characteristics of patients with anemia (n = 194) and those without (n = 1019). Although MCV, MCH, and MCHC values were slightly lower in the anemic group than in the nonanemic group, the absolute difference in these values between the groups was very small. Compared with the nonanemic group, the anemic group was older and had a lower proportion of males, lower proportion of current smokers, lower diastolic blood pressure, a slightly smaller body mass index, lower eGFR, higher plasma BNP, slightly lower total cholesterol, and slightly lower LDL cholesterol (Table 1). Approximately 60% of the anemic patients had eGFR of 60 mL/min/1.73 m2 or more. Only 8 anemic patients (4.1%) had eGFR of 15–29 mL/min/1.73 m2 (G4), and no anemic patient had eGFR of <15 mL/min/1.73 m2 (G5). On the other hand, there was no significant difference between the anemic and nonanemic groups with respect to the percentage of previous cardiovascular disease, previous diabetes, previous hyperlipidemia, or allocation to ARB-based treatment, baseline systolic blood pressure, and UACR (Table 1). Serum hsCRP levels were significantly lower in the anemic group than in the nonanemic group (P < 0.01) (Table 1).
Blood pressure of anemic and nonanemic groups during antihypertensive treatment
As shown in Fig. 2, systolic or diastolic blood pressure was well controlled during the 3 years of antihypertensive therapy in both the anemic group and the nonanemic group. Systolic blood pressure (P = 0.0437) and diastolic blood pressure (P < 0.0001) were slightly lower in anemic patients than in nonanemic patients during antihypertensive treatment, although the absolute difference between the groups was very small.
Time course of systolic and diastolic blood pressure of anemic and nonanemic groups during antihypertensive treatment. Anemia (+) represents patients with anemia at baseline; anemia (−) represents patients without anemia at baseline. Values are mean ± SD. The mean values of systolic and diastolic blood pressure at each time point are shown in red for the nonanemic group and in black for the anemic group
Incidence of cardiovascular and renal events in anemic and nonanemic groups during antihypertensive treatment
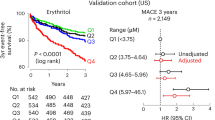
As shown by Kaplan–Meier curves in Fig. 3a, the incidence of cardiovascular and renal events was significantly higher in the anemic group than in the nonanemic group (HR = 1.945: 95%CI 1.208–3.130; P = 0.0062). Among the males, anemic patients had a higher incidence of cardiovascular and renal events than nonanemic patients (HR = 2.246: 95%CI 1.213–4.159; P = 0.0101) (Fig. 3b). Among the females, the incidence of cardiovascular and renal events was also higher in the anemic group than in the nonanemic group, although the difference did not reach statistical significance (P = 0.0749) (Fig. 3c). The interaction between anemia and sex was not significant (P = 0.7875). The details of the cardiovascular and renal events are shown in Table 2.
Kaplan–Meier curves for composite cardiovascular and renal events in the anemic and nonanemic groups of total patients (a), of males (b), and of females (c). Anemia (+) represents patients with anemia at baseline; anemia (−) represents patients without anemia at baseline; HR is hazard ratio; 95%CI is 95% confidence interval. In (a), the number of patients in the anemic and nonanemic groups was 192 and 1012, respectively, and the number of endpoint occurrences was 23 and 65 in the anemic and nonanemic groups, respectively. In (b), the number of males in the anemic and nonanemic groups was 82 and 617, respectively, and the number of endpoint occurrences was 13 and 46 in the anemic and nonanemic groups, respectively. In (c), the number of females in the anemic and nonanemic groups was 110 and 395, respectively, and the number of endpoint occurrences was 10 and 19 in the anemic and nonanemic groups, respectively
Association of prognostic factors with cardiovascular and renal events
Table 3 shows the results of the multivariable Cox regression analysis with the backward selection method starting with the 10 covariates, namely, anemia, sex, age, baseline cardiovascular diseases, baseline diabetes, eGFR, plasma BNP, UACR, current smoking status, and allocation to ARB-based antihypertensive treatment. Of these 10 covariates, age (P =0.8796), eGFR (P = 0.0990), current smoking status (P = 0.4704), and allocation to ARB-based antihypertensive treatment (P = 0.1850) had no significant association with cardiovascular and renal events in total patients, and these covariates were eliminated. On the other hand, anemia was independently associated with cardiovascular and renal events (HR = 1.816: 95%CI 1.116–2.955; P = 0.0163) in total patients. The male sex (P = 0.0242), presence of previous cardiovascular disease (P = 0.0004), presence of previous diabetes (P = 0.0001), plasma BNP ≥19 mg/mL (P = 0.0021), and UACR ≥30 mg/g creatinine (P = 0.0003) were also independently associated with cardiovascular and renal events.
Discussion
Although appropriate blood pressure control significantly reduces cardiovascular and renal morbidity and mortality in hypertensive patients, even strict blood pressure-lowering therapy cannot completely prevent cardiovascular and renal diseases in hypertensive patients, which indicates that significant residual risk for cardiovascular and renal events exists even in hypertensive patients with well-controlled blood pressure [3, 23,24,25]. However, little is known about the specifics of such residual cardiovascular risk in treated hypertension. The main finding of the present study is that anemia, even mild anemia, is an independent risk factor for cardiovascular and renal outcomes in high-risk hypertensive outpatients. This new finding allows us to propose the novel hypothesis that anemia may be a potential target for prevention of cardiovascular and renal diseases in high-risk hypertensive patients with anemia.
It is well-established that high blood pressure is one of the most important risk factors for cardiovascular and renal diseases [1,2,3,4]. Therefore, it is possible that blood pressure might be responsible for the significant difference in cardiovascular and renal events between anemic and nonanemic groups in our study. However, it is worth noting that, in the present subanalysis, both anemic and nonanemic groups of the ATTEMPT-CVD patients had well-controlled blood pressure during the follow-up period, and the anemic group had slightly lower blood pressure than the nonanemic group, although the absolute difference in blood pressure between the groups was very small. Therefore, it is likely that the mechanism influencing the higher incidence of cardiovascular and renal events in the anemic group as compared with the nonanemic group is independent of blood pressure.
There is convincing evidence that anemia is common among patients with advanced CKD [8,9,10,11] or heart failure [12,13,14,15,16] and is a significant risk factor for cardiovascular or renal events in these patients. Therefore, it is critical to understand whether there might be a close association between our current observations and advanced CKD or severe heart failure. Of note, the exclusion criteria of the ATTEMPT-CVD study [6, 7] included severe renal disorder (defined as serum creatinine level ≥2.0 mg/dL), severe heart failure (NYHA Classification III or IV), and a malignant tumor requiring treatment. The noninclusion of patients with severe renal disease, severe heart failure, or a malignant tumor in the ATTEMPT-CVD study can explain our present findings that approximately 60% of anemic patients had G1 or G2 eGFR stage and only a very small proportion of anemic patients had severe CKD. Thus, nephrogenic anemia was involved in only a portion of the anemic patients in our cohort.
The triad of anemia, renal insufficiency, and heart failure has been called cardio-renal anemia syndrome (CRS) [26,27,28]. The three conditions form a vicious cycle in which each condition exacerbates each of the others, thereby accelerating cardiovascular and renal events or death, although the mechanism is complex and multifactorial [26,27,28]. Therefore, it is possible that the increased cardiovascular and renal events in hypertensive outpatients with anemia could be specifically attributable to renal insufficiency or heart failure. However, as described above, because of the exclusion criteria of the ATTEMPT-CVD study, patients with severe renal dysfunction or severe heart failure were not included in our present cohort. Furthermore, to rule out the possible contribution of renal insufficiency or heart failure to our present results, we selected not only traditional risk factors as covariates but also useful biomarkers of cardiorenal syndrome such as eGFR [29,30,31,32,33], UACR [30,31,32, 34,35,36], and plasma BNP [34, 37,38,39]. After adjustment for these covariates, we found that anemia remained an independent factor associated with cardiovascular and renal events in our cohort of hypertensive outpatients. Therefore, it is likely that the increased cardiovascular and renal risk in high-risk hypertensive patients with anemia might not be sufficiently explained by renal impairment or heart failure at baseline. Rather, anemia seems to be an independent risk factor for cardiovascular and renal events in high-risk hypertensive patients, although further study is required to define our theory.
There are several limitations of this study. First, this study was a post-hoc analysis, and the ATTEMPT-CVD trial was not specifically designed to evaluate the impact of anemia on cardiovascular and renal outcomes. Second, the present study did not allow us to prove the causal relationship between anemia and cardiovascular and renal outcomes. Third, it cannot be excluded that an unmeasured additional confounding factor might be responsible for our observations from the study. Fourth, this study did not address the potential mechanism by which anemia might increase cardiovascular and renal events in hypertensive patients, although various potential mechanisms are proposed that include tissue hypoxia, increased cardiac workload, increased oxidative stress, inflammation, increased fluid retention, and increased sympathetic activity. Fifth, the cause of anemia is multifactorial, including renal dysfunction, iron deficiency, nutritional deficiency, myeloid disease, and chronic inflammation. Only a portion of anemic patients had nephrogenic anemia. However, the present study did not allow us to elucidate the cause of anemia since detailed hematological data were not available. In the present study, anemia was defined according to the WHO definition since that definition is used for the diagnosis of anemia globally, including in Japan [27, 40]. However, there are various definitions of anemia since the normal hemoglobin distribution varies with age, sex, ethnicity, or pathophysiological status, such as renal insufficiency [41]. Finally, the findings of this study were hypothesis-generating because of the small sample size. A future randomized intervention trial is essential to define whether treatment of anemia can reduce the incidence of cardiovascular and renal events in hypertensive patients with comorbid anemia.
In conclusion, our present subanalysis provided evidence to suggest that anemia, even mild anemia, is an independent risk factor for cardiovascular and renal morbidity in high-risk hypertensive outpatients with anemia, independently of antihypertensive treatment or traditional cardiovascular risk factors. It is critical to investigate whether drug treatment of anemia can prevent cardiovascular or renal events in high-risk hypertensive patients.
References
Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marczak L, et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mmHg, 1990–2015. JAMA. 2017;317:165–82.
Lim SS, Vos T, Flaxman AD, Danaei G, Shibuya K, Adair-Rohani H, et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2224–60.
Whelton PK, Carey RM, Aronow WS, Casey Jr DE, Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2017;71:1269–324.
Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJ, et al. The preventable causes of death in the United States: comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med. 2009;6:e1000058.
Saran R, Li Y, Robinson B, Ayanian J, Balkrishnan R, Bragg-Gresham J, et al. US renal data system 2014 annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2015;66:S1–305.
Ogawa H, Soejima H, Matsui K, Kim-Mitsuyama S, Yasuda O, Node K, et al. A trial of telmisartan prevention of cardiovascular diseases (ATTEMPT-CVD): biomarker study. Eur J Prev Cardiol. 2016;23:913–21.
Soejima H, Ogawa H, Yasuda O, Kim-Mitsuyama S, Matsui K, Node K, et al. The changes of biomarkers by telmisartan and their significance in cardiovascular outcomes: design of a Trial of Telmisartan Prevention of Cardiovascular Diseases (ATTEMPT-CVD). J Clin Trials. 2014;4:162.
Clement FM, Klarenbach S, Tonelli M, Johnson JA, Manns BJ. The impact of selecting a high hemoglobin target level on health-related quality of life for patients with chronic kidney disease: a systematic review and meta-analysis. Arch Intern Med. 2009;169:1104–12.
Mohanram A, Zhang Z, Shahinfar S, Keane WF, Brenner BM, Toto RD. Anemia and end-stage renal disease in patients with type 2 diabetes and nephropathy. Kidney Int. 2004;66:1131–8.
Vlagopoulos PT, Tighiouart H, Weiner DE, Griffith J, Pettitt D, Salem DN, et al. Anemia as a risk factor for cardiovascular disease and all-cause mortality in diabetes: the impact of chronic kidney disease. J Am Soc Nephrol. 2005;16:3403–10.
Weiner DE, Tighiouart H, Vlagopoulos PT, Griffith JL, Salem DN, Levey AS, et al. Effects of anemia and left ventricular hypertrophy on cardiovascular disease in patients with chronic kidney disease. J Am Soc Nephrol. 2005;16:1803–10.
Anand I, McMurray JJ, Whitmore J, Warren M, Pham A, McCamish MA, et al. Anemia and its relationship to clinical outcome in heart failure. Circulation. 2004;110:149–54.
Felker GM, Adams KF Jr, Gattis WA, O’Connor CM. Anemia as a risk factor and therapeutic target in heart failure. J Am Coll Cardiol. 2004;44:959–66.
O’Meara E, Clayton T, McEntegart MB, McMurray JJ, Lang CC, Roger SD, et al. Clinical correlates and consequences of anemia in a broad spectrum of patients with heart failure: results of the Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity (CHARM) program. Circulation. 2006;113:986–94.
Sharma R, Francis DP, Pitt B, Poole-Wilson PA, Coats AJ, Anker SD. Haemoglobin predicts survival in patients with chronic heart failure: a substudy of the ELITE II trial. Eur Heart J. 2004;25:1021–8.
Swedberg K, Young JB, Anand IS, Cheng S, Desai AS, Diaz AR. et al. Treatment of anemia with darbepoetin alfa in systolic heart failure. N Engl J Med. 2013;368:1210–9.
Anker SD, Comin Colet J, Filippatos G, Willenheimer R, Dickstein K, Drexler H, et al. Ferric carboxymaltose in patients with heart failure and iron deficiency. N Engl J Med. 2009;361:2436–48.
Drueke TB, Locatelli F, Clyne N, Eckardt KU, Macdougall IC, Tsakiris D, et al. Normalization of hemoglobin level in patients with chronic kidney disease and anemia. N Engl J Med. 2006;355:2071–84.
Ghali JK, Anand IS, Abraham WT, Fonarow GC, Greenberg B, Krum H, et al. Randomized double-blind trial of darbepoetin alfa in patients with symptomatic heart failure and anemia. Circulation. 2008;117:526–35.
Johansen KL, Finkelstein FO, Revicki DA, Evans C, Wan S, Gitlin M, et al. Systematic review of the impact of erythropoiesis-stimulating agents on fatigue in dialysis patients. Nephrol Dial Transplant. 2012;27:2418–25.
Silverberg DS, Wexler D, Blum M, Keren G, Sheps D, Leibovitch E, et al. The use of subcutaneous erythropoietin and intravenous iron for the treatment of the anemia of severe, resistant congestive heart failure improves cardiac and renal function and functional cardiac class, and markedly reduces hospitalizations. J Am Coll Cardiol. 2000;35:1737–44.
Singh AK, Szczech L, Tang KL, Barnhart H, Sapp S, Wolfson M, et al. Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med. 2006;355:2085–98.
Blacher J, Evans A, Arveiler D, Amouyel P, Ferrieres J, Bingham A, et al. Residual cardiovascular risk in treated hypertension and hyperlipidaemia: the PRIME Study. J Hum Hypertens. 2010;24:19–26.
Ford ES. Trends in mortality from all causes and cardiovascular disease among hypertensive and nonhypertensive adults in the United States. Circulation. 2011;123:1737–44.
Struthers AD. A new approach to residual risk in treated hypertension—3P screening. Hypertension. 2013;62:236–9.
Kazory A, Ross EA. Anemia: the point of convergence or divergence for kidney disease and heart failure? J Am Coll Cardiol. 2009;53:639–47.
Otaki Y, Watanabe T, Takahashi H, Narumi T, Kadowaki S, Honda Y, et al. Association of renal tubular damage with cardio-renal anemia syndrome in patients with heart failure. Int J Cardiol. 2014;173:222–8.
Silverberg D, Wexler D, Blum M, Wollman Y, Iaina A. The cardio-renal anaemia syndrome: does it exist? Nephrol Dial Transplant. 2003;18:7–12.
Astor BC, Matsushita K, Gansevoort RT, van der Velde M, Woodward M, Levey AS, et al. Lower estimated glomerular filtration rate and higher albuminuria are associated with mortality and end-stage renal disease. A collaborative meta-analysis of kidney disease population cohorts. Kidney Int. 2011;79:1331–40.
Chronic Kidney Disease Prognosis Consortium, Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, et al. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet. 2010;375:2073–81.
Gansevoort RT, Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, et al. Lower estimated GFR and higher albuminuria are associated with adverse kidney outcomes. A collaborative meta-analysis of general and high-risk population cohorts. Kidney Int. 2011;80:93–104.
Levey AS, Coresh J. Chronic kidney disease. Lancet. 2012;379:165–80.
Levey AS, de Jong PE, Coresh J, El Nahas M, Astor BC, Matsushita K, et al. The definition, classification, and prognosis of chronic kidney disease: a KDIGO Controversies Conference report. Kidney Int. 2011;80:17–28.
Blankenberg S, McQueen MJ, Smieja M, Pogue J, Balion C, Lonn E, et al. Comparative impact of multiple biomarkers and N-terminal pro-brain natriuretic peptide in the context of conventional risk factors for the prediction of recurrent cardiovascular events in the Heart Outcomes Prevention Evaluation (HOPE) Study. Circulation. 2006;114:201–8.
Brantsma AH, Bakker SJ, de Zeeuw D, de Jong PE, Gansevoort RT, Group PS. Extended prognostic value of urinary albumin excretion for cardiovascular events. J Am Soc Nephrol. 2008;19:1785–91.
Gerstein HC, Mann JF, Yi Q, Zinman B, Dinneen SF, Hoogwerf B, et al. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA. 2001;286:421–6.
Lubien E, DeMaria A, Krishnaswamy P, Clopton P, Koon J, Kazanegra R, et al. Utility of B-natriuretic peptide in detecting diastolic dysfunction: comparison with Doppler velocity recordings. Circulation. 2002;105:595–601.
Maisel AS, Krishnaswamy P, Nowak RM, McCord J, Hollander JE, Duc P, et al. Breathing Not Properly Multinational Study I. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med. 2002;347:161–7.
Wang TJ, Larson MG, Levy D, Benjamin EJ, Leip EP, Omland T, et al. Plasma natriuretic peptide levels and the risk of cardiovascular events and death. N Engl J Med. 2004;350:655–63.
Hamaguchi S, Tsuchihashi-Makaya M, Kinugawa S, Yokota T, Takeshita A, Yokoshiki H, et al. Anemia is an independent predictor of long-term adverse outcomes in patients hospitalized with heart failure in Japan. A report from the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD). Circ J. 2009;73:1901–8.
Yamamoto H, Nishi S, Tomo T, Masakane I, Saito K, Nangaku M, et al. 2015 Japanese Society for Dialysis Therapy: guidelines for renal anemia in chronic kidney disease. Ren Replace Ther. 2017;3:36.
Acknowledgements
The authors thank all of the ATTEMPT-CVD investigators who contributed to the data collection and the patients, physicians, and medical staff who supported this clinical study. The authors also thank Kazuko Noda and Tomoko Moriyama for their excellent support during this study. Parts of the data in this manuscript were presented at Nephro Update Europe 2018 in Budapest.
Funding
This work was supported by a grant from the Japan Foundation for Aging and Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Shokei Kim-Mitsuyama has received consultancy fees/honoraria/research grants from Astellas, Daiichi-Sankyo, and Takeda. Osamu Yasuda has received consultancy fees/honoraria/research grants from Otsuka Pharmaceutical Co., Ltd. and Sanwa Kagaku Kenkyusho Co., Ltd. Koichi Node has received consultancy fees/honoraria/research grants from Boehringer Ingelheim. Hideaki Jinnouchi has received consultancy fees/honoraria/research grants from AstraZeneca Pharmaceuticals, Astellas Pharma, Boehringer Ingelheim, Daiichi Sankyo, Eli Lilly, Takeda, Novartis Pharmaceuticals, Novo Nordisk, and Sanofi. Kunihiko Matsui has received consultancy fees/honoraria/research grants from Daiichi Sankyo and Boehringer Ingelheim Japan. All other authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Trial registration: This trial was registered with ClinicalTrials.gov, number NCT01075698, on February 24, 2010.
Supplementary information
Rights and permissions
About this article
Cite this article
Kim-Mitsuyama, S., Soejima, H., Yasuda, O. et al. Anemia is an independent risk factor for cardiovascular and renal events in hypertensive outpatients with well-controlled blood pressure: a subgroup analysis of the ATTEMPT-CVD randomized trial. Hypertens Res 42, 883–891 (2019). https://doi.org/10.1038/s41440-019-0210-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-019-0210-1
Keywords
This article is cited by
-
Sex-Specific Association Between Iron Status and the Predicted 10-Year Risk for Atherosclerotic Cardiovascular Disease in Hypertensive Patients
Biological Trace Element Research (2022)
-
Association between metabolic parameters and risks of anemia and electrolyte disturbances among stages 3–5 chronic kidney disease patients in Taiwan
BMC Nephrology (2021)
-
Burden of Anemia in Chronic Kidney Disease: Beyond Erythropoietin
Advances in Therapy (2021)