Abstract
It is not established whether central blood pressure (BP) evaluated by a radial pulse wave analysis is useful to predict cardiovascular prognoses. We tested the hypothesis that central BP predicts future cardiovascular events in treated hypertensive subjects. We conducted a multicenter, observational cohort study of 3566 hypertensives being treated with antihypertensive medications at 27 institutions in Japan. We performed the radial pulse wave analyses using applanation tonometry in all subjects. The primary outcome was the incidence of any of the following: stroke, myocardial infarction (MI), sudden cardiac death, and acute aortic dissection. The mean age of the subjects was 66.0 ± 10.9 years, and 50.6% were male. The mean brachial SBP and central SBP were 138 ± 18 mm Hg and 128 ± 19 mm Hg, respectively. When the central SBP was divided into quintiles, the number of events was least in the 2nd quintile, and we set it as the reference. In the Cox regression analysis adjusting for age, sex, body mass index, creatinine, diabetes, use of β-blocker, and history of MI/stroke, the patients in the 3rd (hazard ratio (HR) 3.55, 95% confidence interval 1.29–9.78, p = 0.014), 4th (HR 4.12, 95% CI 1.53–11.10, p = 0.005), and 5th quintiles (HR 2.87, 95% CI 1.01–8.18, p = 0.048) had a significantly higher incidence of cardiovascular events compared to the 2nd quintile. The results were essentially unchanged when brachial DBP was additionally adjusted. In conclusion, in treated hypertensives, high central SBP was associated with worse cardiovascular outcomes.
Similar content being viewed by others
Introduction
Whether or not central blood pressure (BP) is superior to brachial BP in predicting future cardiovascular events is a matter of debate. In the Conduit Artery Function Evaluation (CAFE) sub-study of the Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT), central BP was more closely associated with cardiovascular prognosis than brachial BP [1]. In the Strong Heart study, central pulse pressure (PP) values of ≥50 mm Hg predicted adverse cardiovascular disease (CVD) outcomes [2]. We identified 11 prospective cohort studies performed since 2002 that assessed the relationship between central hemodynamics and prognosis (Supplementary Table 1). Among them, 9 studies demonstrated a relationship between central blood pressures and cardiovascular risk, but in the Australian National Blood Pressure Study 2 (ANBP2) [3] and Framingham study [4], central hemodynamics including central systolic blood pressure (SBP) and PP were not predictors of CVD when adjusted by conventional risk factors [4, 5]. A meta-analysis of published and unpublished data suggested that there was a trend for central PP to predict CVD events better than brachial PP [6].
Because central hemodynamic parameters are affected by many modifying factors such as age, body size, heart rate, dietary sodium [7], 24 h urine sodium/potassium ratio [8], and cardiovascular medications, the impact of central BP on cardiovascular disease would vary by the subjects’ characteristics. However, previous studies enrolled varying populations, ranging from the general population to hemodialysis patients. Of note, only four studies reported the prognostic significance of central hemodynamics in treated and/or untreated hypertensive subjects [1, 3, 9, 10]. It has not been established whether central hemodynamics are useful to predict incident cardiovascular events in hypertensive subjects. In the present study, we tested the hypothesis that central hemodynamics parameters such as central BP could be useful to predict future cardiovascular events in treated hypertensive subjects.
Methods
Study design and subjects
This is a main paper of the ABC-J II study, an expanded version of the original “Antihypertensives and Blood pressure of Central artery” in Japan (ABC-J) study. The ABC-J study has enrolled almost 4000 treated hypertensive subjects since 2007. The first report from this study was published in 2010 [11]. Briefly, the ABC-J II study is an observational study being conducted to evaluate the predictive values of central BP for cardiovascular events in Japanese treated hypertensive subjects. The protocol of the ABC-J II study has been registered on the University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR) website (trial no. UMIN000002966). This is a prospective study of retrospectively collected data. All of the subjects in the present study were treated hypertensive subjects enrolled in the ABC-J II study [11]. The subjects had been under stable antihypertensive treatment for at least 3 months. The Institutional Review Board of the Jichi Medical University School of Medicine and each participating institute approved this study, and because of the retrospective nature, no written informed consent was obtained from the participants. In the first part of this study, 1727 subjects were analyzed and by prolonging the study period, we further recruited rest of the subjects in participating institutions who performed radial tonometry as a part of routine clinical practice retrospectively. Between January 2007 and March 2011, the central BP data of 4310 subjects were examined as part of clinical practice, and these subjects were enrolled as part of an observational study by 29 doctors at 27 institutions (13 primary practices, 3 hospital-based outpatient clinics, and 11 specialized university hospitals) throughout Japan. The chart review was performed until May 2013, and event information was collected from each institution and provided to a central study office.
The study was of treated Japanese essential hypertensive subjects who met all of the following criteria: (1) they had been taking a stable dosage of antihypertensive medication for at least 3 months; (2) radial tonometry data including radial augmentation index (rAI) and central BP data were available; and (3) age ≥35 years. All clinical data were obtained from medical records. Because almost 10% of young men have been reported to have an exaggerated amplification of the arterial pressure wave as it travels out to the periphery [12] (although the clinical significance of this remains controversial [13]), we did not include subjects younger than 35 years.
We excluded: (1) subjects with extremely abnormal BP (i.e., SBP 40 mm Hg or diastolic BP (DBP) 20 mm Hg higher or lower, respectively) compared to usual office BP or home BP during the examination; (2) subjects with arrhythmia; (3) subjects with estimated glomerular filtration rate (eGFR) <15 ml/min/1.73 m2; (4) no follow-up data, or a follow-up period of <100 days; (5) subjects with heart failure history; and (6) rAI ≤50%.
Hypertension was defined as office SBP >140 mm Hg and/or DBP >90 mm Hg, or the subjects being on antihypertensive medication [14]. In the present study, diabetes mellitus (DM) was defined as one or more of the following: self-report of DM; the use of diabetes medication; fasting plasma glucose ≥126 mg/dL; or hemoglobin A1c (HbA1c) (National Glycohemoglobin Standardization Program (NGSP)) ≥6.5% [15, 16]. Dyslipidemia was defined as one or more of the following: self-report; total cholesterol level ≥240 mg/dL; triglycerides ≥150 mg/dL; high-density lipoprotein (HDL) <40 mg/dL; or being under treatment for hyperlipidemia [17]. Chronic kidney disease (CKD) was defined as the presence of overt proteinuria or an eGFR <60 ml/min/1.73 m2, or existing renal disease. Heart failure was diagnosed by the Framingham criteria [18] as is widely accepted. The diagnosis of peripheral artery disease (PAD) was based on the American Heart Association (AHA) guideline [19]. Those with any symptoms or ankle-brachial index <0.9 were defined as having PAD in this study. We identified several cases with histories of aortic regurgitation (n = 7), thoracic aortic aneurism (n = 2), abdominal aortic aneurism (n = 2), and dissecting aneurism (n = 7) among the 3566 cases. Detailed information of these is not available, but all of these patients were stable. Therefore, we determined not to exclude these subjects.
Blood pressure measurements
Brachial BP
Brachial BP was measured by a physician or laboratory technician at each institution based on the hypertension guideline [14] in laboratory circumstances. Briefly, arm circumference was measured and the appropriate cuff size was selected, and brachial BP was measured by an automated BP monitor (HEM-9000AI; Omron Healthcare, Kyoto, Japan) after ≥5 min of rest in the sitting position just before the measurement of the radial artery waveform. The algorithm of BP measurement of HEM-9000AI is the same as that of HEM-907, which is validated in many studies [20, 21].
Assessment of central hemodynamics
Central hemodynamics were assessed based on the rAI. The rAI was measured with the same semi-automatic tonometry device (HEM-9000AI; Omron Healthcare, Kyoto, Japan) as that used for the brachial BP measurements, as previously described [22].
The HEM-9000AI was used to calculate the peripheral augmentation index as (P2−DBP)/(P1−DBP), taking P1 and P2 as the first and second inflection points on the radial pulse waveform [22]. The rAI values are expressed as rAI adjusted for heart rate 75 bpm. Estimated central SBP was calculated from late SBP in the radial artery; i.e., “SBP2” by the equation described [22]. Tonometric radial pressure waveforms were calibrated to the brachial cuff SBP and DBP values. SBP2 was used as a central SBP estimate without any correction for comparison with invasive pressure measurements. SBP2 measured by the HEM-9000AI was almost identical to central SBP estimated by the SphygmoCor system [23]. The high reproducibility of this device has been demonstrated [24].
Assessment of events
We defined an incident cardiovascular (CV) event as the occurrence of any of the following: myocardial infarction (MI), stroke, sudden death, or acute aortic dissection. The definition of each event was based on the COLM (Combination of OLMesartan and calcium channel blocker or diuretic in high-risk elderly hypertensive patients) study [25, 26], but the subjects of the ABC-J II study are completely different from the COLM study. All cardiovascular events and serious adverse events reported by the participating investigators were adjudicated by the Endpoint committee that was blinded to the study group.
Statistical analysis
All statistical analyses were carried out with SAS system ver. 9.4 software (SAS, Cary, NC, USA). All analyses were performed using the final dataset of the ABC-J II study. This is a prespecified post-hoc analysis of the ABC-J II study. The main analyses were performed by quintiles of central SBP (SBP2) because the relationship between central BP and cardiovascular events was not linear, but J-curved. Based on the Wald test, marginally significant linear or non-linear relationship exists between central SBP and CV outcomes. By comparison, the Wald chi-square test for linear SBP2 model was also marginally significant (χ2 = 3.4969, p = 0.0615), while that for SBP2_max140 (predictor in the 3rd model) was statistically significant (χ2 = 5.9708, p = 0.0145). In summary, we conclude from these additional analyses that central SBP is associated with CVD risk, and that the relationship is non-linear, but is not clearly non-monotonic. The subjects were classified by quintiles (Q) of central SBP (Q1: 73.0–111.7; Q2: 112.0–122.6; Q3: 122.7–131.0; Q4: 132.0–142.5; Q5: 143.0–215.0 mm Hg).
We performed multivariable Cox regression analyses to analyze factors associated with the study endpoint. Factors associated with central SBP in the bivariate analysis or confirmed associating factors were entered as independent variables in the models. As a first step, we performed a preliminary analysis using age, sex, body mass index (BMI), presence of DM, use of β-blockers, history of MI and/or stroke, serum creatinine, CKD, the use of an α-blocker, diuretics, and the number of antihypertensive medications as independent variables. Then, as a second step, we entered significant covariates in the preliminary analysis of the Cox model. Thus, in multivariable Cox regression model, sex, history of MI and/or stroke, and number of hypertensive drugs were used as independent variables, and incident CV events were set as a dependent variable. The variance inflation factor (VIF) in serum creatinine was 1.226 and that in eGFR was 1.122, both of which are far below VIF 5 in this multivariable model. Therefore, there is no collinearity between serum creatinine or eGFR and other covariates, but we prefer serum creatinine because serum creatinine was not associated with age in our dataset (r = 0.0046, p = 0.79). We performed receiver operating characteristic (ROC) analysis to determine the cutoff value of central BP. We made a spline curve to characterize the functional form of the association between central BP and outcome. Comparative analyses between linear and non-linear model were based on comparing the two log likelihood chi-squares (the higher one is better) or comparing the two Akaike information criteria (AIC) (the lower one is better) [27]. Probability values (p) < 0.05 were considered significant.
Results
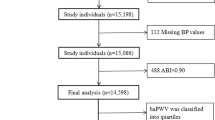
There were 1806 females and 1758 males; the mean age was 66.0 ± 10.9 years, and 939 subjects (26.3%) had DM. The flow of the subjects is shown in Fig. 1. A total of 4023 treated hypertensives were initially enrolled. After excluding subjects with eGFR <15 ml/min/1.73 m2 (n = 106), no follow-up data (n = 31), or a follow-up period <100 days (n = 44), the number of subjects was reduced to 3842. We then further excluded subjects with a heart failure history (n = 101), atrial fibrillation (n = 111), or rAI ≤50% (n = 64). Thus, a final total of 3566 subjects was analyzed.
Table 1 shows the characteristics of the subjects divided by quintiles of central SBP. The lower central BP groups tended to have more females, be younger, have a longer history of hypertension, and more frequently have DM. The Q5 tended to have more males, be thinner, less frequently have DM, have a lower rate of high uric acid, and a higher rate of PAD. Otherwise, there were no significant differences in clinical characteristics among the groups.
Table 2 shows the BP parameters among the groups. As defined, SBP2 increased from Q1 to Q5 as did all of the other BP parameters including brachial BP, mean arterial pressure (MAP), central pulse pressure (PP2), rAI, and deltaSBP2 (brachial SBP minus central SBP). However, heart rates tended to decrease from Q1 to Q5.
Table 3 shows the laboratory data among the groups. Serum creatinine and serum uric acid tended to be lower, whereas total cholesterol, HDL cholesterol, and low-density lipoprotein (LDL) cholesterol tended to be higher from Q1 to Q5. Table 4 shows the cardiovascular medications used in each group. Diuretics, calcium channel blockers (CCBs), and α-blockers tended to be used more in the low central BP groups, but no other drugs including β-blockers differed significantly among the groups. The number of BP medications used was higher in the lower central BP groups than the higher groups.
There were 64 incident CV events: 39 cerebral infarctions, 11 myocardial infarctions, 6 cerebral bleedings, 4 sudden deaths, 3 aortic dissections, and 1 undetermined stroke. The median follow-up duration was 5.0 years (interquartile range: 3.1 to 6.2 years). The event rate of incident CV events was 3.92/1000 person-year. In multivariable Cox regression analysis, central BP (SBP2) as a continuous variable was associated with incident CV events (hazard ratio (HR) 1.02, 95% confidence interval (CI) 1.00–1.03, p = 0.02) adjusting for the same covariates used in Fig. 2.
Multivariable Cox regression analysis of central SBP quintiles for incident CV events. Incident CV events defined as myocardial infarction, stroke, sudden death, or acute aortic diseases were set as the outcome. As a first step, we performed a preliminary analysis using age, sex, body mass index (BMI), presence of DM, use of β-blockers, history of MI and/or stroke, serum creatinine, chronic kidney disease (CKD), the use of an α-blocker, diuretics, and the number of antihypertensive medications as independent variables, and CV events as the dependent variable. As a result, sex, history of MI and/or stroke, and number of hypertensive drugs were selected as significant variables. Then, as a second step, we entered these as covariates in the Cox model. Thick bars indicate HRs and thin bars 95% CIs
Supplementary Figure 1 shows the Kaplan–Meier analysis of the subjects by quintiles of central SBP. Q4 had the highest event rate, followed by Q3, Q1, and Q5, and Q2 had the lowest event rate among the groups. The results of the multivariable Cox regression analysis are shown in Fig. 2. When the patients in Q2 were set as a reference, the patients in Q3 (HR 3.55, 95% CI 1.29–9.78, p = 0.014), Q4 (HR 4.12, 95% CI 1.53–11.10, p = 0.005), and Q5 (HR 2.87, 95% CI 1.01–8.18, p = 0.048) had a significantly higher incidence of incident CV events. We created a spline curve to characterize the functional form of the association between central BP and outcome (Supplementary Figure 2). Based on the result of functional form of the association between central BP and outcome, we have run a “3rd model” using the following predictor: SBP2_max140 = min (140, SBP2). This model tests the hypothesis that the effect of BP increases up to 140 mm Hg, but then is flat above that. AIC for the “3rd model” suggests that this model is a better model (916.597 − 913.381 > 2.0) than the linear model. AIC for the “3rd model” suggests that this model is also better model (915.621 − 913.381 > 2.0) than the spline model. Therefore, the relationship between central BP and CV outcome was non-linear, but linear increase in risk up to central SBP = 140, and constant risk thereafter.
The patients in Q1 tended to have more events compared to Q2 (HR 2.23, 95% CI 0.77–6.44, p = 0.138), but this was not significant. The results did not change when the use of statin and aspirin were added in the same model.
When the brachial SBP was divided by quintiles, only Q4 had a higher event rate (Supplementary Figure 3) compared to the others. In the multivariable Cox regression analysis, when Q2 was set as a reference, only Q4 had a significantly higher event rate (Supplementary Figure 4). However, same quintile analyses by brachial PP did not show significant results. The comparison of CV risk among brachial DBP quintiles (Supplementary Figure 5) or quartiles (data not shown) failed to show significant inter-group differences. Of note, we looked at the proportion of resistant hypertension, defined by brachial BP level ≥140 and/or ≥90 mm Hg and the use of 3 or more antihypertensive medications. As shown in supplementary Figure 6, subjects with higher central BP tended to have more proportion of resistant hypertension.
We ran ROC analyses to determine the appropriate cutoff value of central BP to predict cardiovascular events. As shown in Fig. 3, the cutoff value of central SBP in which the Youden Index become maximal was 122.1 mm Hg for the follow-up period 1000 days, and 123 mm Hg for the follow-up period 2000 days.
Finally, we compared AICs of the linear model, spline model, and 3rd model in central SBP and brachial SBP, respectively, to compare which model is better than the other model based on the paper of Snipes and Taylor [27] (Supplementary Table 2). As a result, AIC for the brachial spline models was much better than the spline model of central BP (915.621 − 912.364 > 2.0) and 3rd model of central BP. Therefore, for the comparison between central vs. brachial BP, spline model of brachial SBP is better than spline model of central BP.
Discussion
In the present study, in treated hypertensive subjects, high central BP was associated with a higher incidence of cardiovascular events independently of various covariates. This is one of the largest studies to demonstrate a positive association between central BP and incident CV events in treated hypertensive subjects.
Central BP and prognosis
We observed that high central BP was associated with a higher incidence of CV events. Compared to the Q2 patients, the patients in Q3, Q4, and Q5 had a significantly higher incidence of incident CV events (Fig. 2). Because these results were unchanged after additional adjustment for DBP, it is apparent that higher central SBP can be used as a prognostic indicator of incident CV events in treated hypertensives.
These results are in line with most of the previous studies’ finding that central BP was associated with CV outcomes—ANBP2 [3] and two sub-studies from the Framingham study are exceptions (Supplementary Table 1) [5].
Because central BP directly affects strain vessels in the brain, heart, and kidney, central BP could be a better marker than brachial BP, as shown in the present study (especially in treated individuals). Additionally, in light of our present findings, we propose 123 mm Hg as the threshold of central SBP for the prediction of CV events based on the ROC curves (Fig. 3).
On the other hand, brachial SBP and DBP were not predictors of CV events. Therefore, in stably treated hypertensives, brachial SBP, which is largely modulated by pulse pressure amplification due to pulse wave propagation and reflection in the upper limb arteries, or DBP, which is nearly unaltered throughout a conduit artery from the central to the periphery but with less variance due to antihypertensive treatment, may not be a very useful hemodynamic markers in predicting future CV events.
Linear vs. non-linear association
In the present study, the model which most parsimoniously explains the association between central BP and outcome was the “3rd model” in which the relationship was linear up to SBP2 of 140 mm Hg, but was flat above 140 mm Hg. The spline model was the 2nd best model, with the linear model showing a weak positive association with CV risk, but providing the poorest overall fit to the data. Accordingly, we conclude that CV risk increases as the central SBP increases up to a central SBP level of around 140 mm Hg. This is consistent with the observation from the Framingham study that increased aortic stiffness does not always reflect increased wave reflection. Age-associated wave amplitude changed minimally with advancing age, despite a steady increase in forward wave amplitude [28]. This is in contrast to the steady increase in aortic stiffness assessed by carotid femoral pulse wave velocity. In summary, the relationship between central BP and CV events is not simple, but central SBP would be predictive in the treated hypertensive range (<140 mm Hg).
Is low central BP a risk?
In the present study, insignificantly higher incidence of CV events was observed in Q1. Although central BP as a continuous variable was weakly associated with incident CV events, we set Q2 as the reference, because the relationship between central BP and CV events was non-linear. As shown in Tables 1 and 4, the Q1 patients tended to show more CKD (p = 0.001 for trend) and history of MI, and their uses of α-blocker and diuretics were high, and the number of antihypertensive medications used was highest among the groups. Further adjustment using these variables did not change the main result of this study. However, the clinical significance of a very low level of central SBP (e.g., <112 mm Hg) cannot be clarified from this study.
Technical aspects of central BP measurement
We estimate central SBP as SBP2, the second peak of the radial artery pressure waveform determined by the HEM-9000AI [22], which has been established as almost identical to the generalized transfer function-derived central aortic SBP estimate by the use of a SphygmoCor [29], which is regarded as a standard method of evaluating central hemodynamics noninvasively. However, the SBP2-based central BP estimation method was reported to underestimate central SBP in patients with lower BP or lower augmentation [30]. Especially in individuals with a central AI value as low as in Murgo type C beat, radial SBP2 can no longer reflect central peak pressure [31]. Hence, we excluded subjects with rAI values ≤50%, which was determined based on the relationship between central and rAI as previously reported [32].
Study limitations
This study has several limitations. Because the subjects in this study are all treated for their hypertension, the event number was small at 3.92/1000 person-year for the combined endpoints. Therefore, the different cutoff values of central SBP by the quintile analysis (Fig. 2) and quartile analysis (data not shown) did not show consistent results. Based on the goodness-of-fit analysis, we cannot say that central BP is superior to brachial BP (or vice versa) in predicting incident CV events, and the spline model of brachial SBP was better than spline model of central BP. Because of the high collinearity (variance inflation factor >5) when both central SBP and brachial SBP are entered in the same model, adjustment by brachial SBP cannot be performed in the Cox model in Fig. 2. Because the endpoint of this study was based on the COLM study, heart failure was not included as a CV event. As a prospective study of retrospectively collected data, brachial and central BP data were picked up from examination records at each institution, and the BP data were taken only once, not as the composite of baseline and follow-up. Finally, to explore cutoff limits of central BP, a population study might be more appropriate. However, in the study, we aimed to explore the cutoff value of treated individuals.
Perspectives
With regard to central BP, although a number of important findings that clarified the physiological mechanisms of arterial stiffness have been reported, the clinical applications of these methods are still under way. Several studies have reported normal values or reference values of central BP [10, 33, 34]. The normal value of central SBP was proposed as 130 mm Hg based on data from Asia [10]. Although it is slightly lower, we propose 123 mm Hg as a possible target level of central SBP in antihypertensive therapy, based on our present findings. In treated individuals, a very low value of central BP is not always a sign of low risk, but the confirmation of the waveform (which determines central BP) is necessary. These issues should be evaluated in further prospective studies.
Conclusion
In treated hypertensives, higher central SBP was associated with an increased risk of incident cardiovascular events, especially among those with central SBP ≥123 mm Hg.
References
The CAFE Investigators for the Anglo-Scandinavian Cardiac Outcomes Trial Investigators CAFE Steering Committee and Writing Committee, Williams B, Lacy PS, Thom SM, Cruickshank K, Stanton A, Collier D, et al. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes: principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation. 2006;113:1213–25.
Roman MJ, Devereux RB, Kizer JR, Okin PM, Lee ET, Wang W, et al. High central pulse pressure is independently associated with adverse cardiovascular outcome the strong heart study. J Am Coll Cardiol. 2009;54:1730–4.
Dart AM, Gatzka CD, Kingwell BA, Willson K, Cameron JD, Liang Y-L, et al. Brachial blood pressure but not carotid arterial waveforms predict cardiovascular events in elderly female hypertensives. Hypertension. 2006;47:785–90.
Mitchell GF, Hwang S-J, Vasan RS, Larson MG, Pencina MJ, Hamburg NM, et al. Arterial stiffness and cardiovascular events: the Framingham Heart Study. Circulation. 2010;121:505–11.
Mitchell GF, Hwang SJ, Larson MG, Hamburg NM, Benjamin EJ, Vasan RS, et al. Transfer function-derived central pressure and cardiovascular disease events: the Framingham Heart Study. J Hypertens. 2016;34:1528–34.
Vlachopoulos C, Aznaouridis K, O’Rourke MF, Safar ME, Baou K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: a systematic review and meta-analysis. Eur Heart J. 2010;31:1865–71.
Han W, Han X, Sun N, Chen Y, Jiang S, Li M. Relationships between urinary electrolytes excretion and central hemodynamics, and arterial stiffness in hypertensive patients. Hypertens Res. 2017;40:746–51.
Rhee M-Y, Shin S-J, Gu N, Nah D-Y, Kim B-K, Hong K-S, et al. Relationship between 24-h urine sodium/potassium ratio and central aortic systolic blood pressure in hypertensive patients. Hypertens Res. 2016;40:405–10.
Wang K-L, Cheng H-M, Chuang S-Y, Spurgeon HA, Ting C-T, Lakatta EG, et al. Central or peripheral systolic or pulse pressure: which best relates to target organs and future mortality? J Hypertens. 2009;27:461–7.
Cheng H-M, Chuang S-Y, Sung S-H, Yu W-C, Pearson A, Lakatta EG, et al. Derivation and validation of diagnostic thresholds for central blood pressure measurements based on long-term cardiovascular risks. J Am Coll Cardiol. 2013;62:1780–7.
Miyashita H, Aizawa A, Hashimoto J, Hirooka Y, Imai Y, Kawano Y, et al. Cross-sectional characterization of all classes of antihypertensives in terms of central blood pressure in Japanese hypertensive patients. Am J Hypertens. 2010;23:260–8.
O’Rourke MF, Vlachopoulos C, Graham RM. Spurious systolic hypertension in youth. Vasc Med. 2000;5:141–5.
Saladini F, Palatini P. Isolated systolic hypertension in young individuals: pathophysiological mechanisms, prognostic significance, and clinical implications.High Blood Press Cardiovasc Prev. 2017;24:133–9.
Shimamoto K, Ando K, Fujita T, Hasebe N, Higaki J, Horiuchi M, et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2014). Hypertens Res. 2014;37:253–390.
The Committee of the Japan Diabetes Society on the Diagnostic Criteria of Diabetes Mellitus, Seino Y, Nanjo K, Tajima N, Kadowaki T, Kashiwagi A, Araki E, et al. Report of the committee on the classification and diagnostic criteria of diabetes mellitus. J Diabetes Investig. 2010;1:212–28.
American Diabetes Association. Standards of medical care in diabetes—2014. Diabetes Care. 2014;37:S14–80.
Teramoto T, Sasaki J, Ishibashi S, Birou S, Daida H, Dohi S, et al. Executive summary of the Japan Atherosclerosis Society (JAS) guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan -2012 version. J Atheroscler Thromb. 2013;20:517–23.
McKee PA, Castelli WP, McNamara PM, Kannel WB. The natural history of congestive heart failure: the Framingham Study. N Engl J Med. 1971;285:1441–6.
Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, et al. ACC/AHA 2005 practice guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients with Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; Transatlantic Inter-Society Consensus; and Vascular Disease Foundation. Circulation. 2006;113:e463–5.
White WB, Anwar YA. Evaluation of the overall efficacy of the Omron office digital blood pressure HEM-907 monitor in adults. Blood Press Monit. 2001;6:107–10.
El Assaad MA, Topouchian JA, Darné BM, Asmar RG. Validation of the Omron HEM-907 device for blood pressure measurement. Blood Press Monit. 2002;7:237–41.
Takazawa K, Kobayashi H, Shindo N, Tanaka N, Yamashina A. Relationship between radial and central arterial pulse wave and evaluation of central aortic pressure using the radial arterial pulse wave. Hypertens Res. 2007;30:219–28.
Hirata K, Kojima I, Momomura S. Noninvasive estimation of central blood pressure and the augmentation index in the seated position: a validation study of two commercially available methods. J Hypertens. 2013;31:508–15.
Tomiyama H, Yamazaki M, Sagawa Y, Teraoka K, Shirota T, Miyawaki Y, et al. Synergistic effect of smoking and blood pressure on augmentation index in men, but not in women. Hypertens Res. 2009;32:122–6.
Ogihara T, Saruta T, Rakugi H, Shimamoto K, Ito S, Matsuoka H, et al. Rationale, study design and implementation of the COLM study: the combination of OLMesartan and calcium channel blocker or diuretic in high-risk elderly hypertensive patients. Hypertens Res. 2009;32:163–7.
Saruta T, Ogihara T, Saito I, Rakugi H, Shimamoto K, Matsuoka H, et al. Comparison of olmesartan combined with a calcium channel blocker or a diuretic in elderly hypertensive patients (COLM study): safety and tolerability. Hypertens Res. 2015;38:132–6.
Snipes M, Taylor DC. Model selection and Akaike information criteria: an example from wine ratings and prices. Wine Econ Policy. 2014;3:3–9.
Mitchell GF, Parise H, Benjamin EJ, Larson MG, Keyes MJ, Vita JA, et al. Changes in arterial stiffness and wave reflection with advancing age in healthy men and women: the Framingham Heart Study. Hypertension. 2004;43:1239–45.
Kips JG, Schutte AE, Vermeersch SJ, Huisman HW, Van Rooyen JM, Glyn MC, et al. Comparison of central pressure estimates obtained from Sphygmocor, Omron HEM-9000AI and carotid applanation tonometry. J Hypertens. 2011;29:1115–20.
Hickson SS, Butlin M, Mir FA, Graggaber J, Cheriyan J, Khan F, et al. The accuracy of central SBP determined from the second systolic peak of the peripheral pressure waveform. J Hypertens. 2009;27:1784–8.
Lin M-M, Cheng H-M, Sung S-H, Liao C-F, Chen Y-H, Huang P-H, et al. Estimation of central aortic systolic pressure from the second systolic peak of the peripheral upper limb pulse depends on central aortic pressure waveform morphology. J Hypertens. 2012;30:581–6.
Millasseau SC, Patel SJ, Redwood SR, Ritter JM, Chowienczyk PJ. Pressure wave reflection assessed from the peripheral pulse. Hypertension. 2003;41:1016.
Takase H, Dohi Y, Kimura G. Distribution of central blood pressure values estimated by Omron HEM-9000AI in the japanese general population. Hypertens Res. 2013;36:50–7.
Herbert A, Cruickshank JK, Laurent S, Boutouyrie P. Establishing reference values for central blood pressure and its amplification in a general healthy population and according to cardiovascular risk factors. Eur Heart J. 2014;35:3122–33.
Acknowledgements
We thank the numerous study investigators, fellows, nurses, laboratory technicians, and research coordinators who participated in the ABC-J II study. Especially, we thank Dr. Joseph E. Schwartz and Tetsuo Takeuchi for statistical assistance. Contributors and details of the study investigators of the ABC-J II study are described in the Supplementary file (Appendices). The University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR): the trial number is UMIN000002966.
Funding
The study was supported by funding for data collection by Omron Healthcare Inc. (Kyoto, Japan). Omron Healthcare Inc. was not involved in any significant processes of this study such as the design, conduct, monitor, supervision, data analysis, and publication of the study.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Appendices
Participants and participating centers and Members of the Endpoint Committee
Kazuyuki Shimada: Shin-Oyama City Hospital; Kazuo Eguchi: Jichi Medical University and International University of Health and Welfare Hospital; Hiroshi Miyashita: Jichi Medical University and Ishibashi General Hospital; Tsuneo Takenaka: International University of Health and Welfare and Saitama Medical University Hospital; Yasuharu Tabara: Kyoto University and Ehime University Hospital; Hirofumi Tomiyama: Tokyo Medical University; Yasuaki Dohi: Nagoya Gakuin University and Nagoya City University Hospital; Junichiro Hashimoto: Miyagi University of Education and Tohoku University Graduate School of Medicine; Takayoshi Ohkubo: Teikyo University School of Medicine; Yuko Ohta: Kyushu Dental Univ. Hospital; Yoshitaka Hirooka: Kyushu University; Katsuhiko Kohara: Ehime Medical Center and Ehime University Hospital; Sadayoshi Ito: Tohoku University Graduate School of Medicine; Yuhei Kawano: Teikyo University and National Cerebral and Cardiovascular Center; Kenji Sunagawa: Kyushu University; Hiromichi Suzuki: Musashino Tokushukai Hospital and Saitama Medical University Hospital; Yutaka Imai: Tohoku University Graduate School of Pharmaceutical Science; Kazuomi Kario: Jichi Medical University; Kenji Takazawa: Health Surveillance and Preventive Medicine, Tokyo Medical University Hospital and Tokyo Medical University Hachioji Medical Center; Akira Yamashina:Tokyo Medical University;Mari Odaira:Tokyo Medical University; Motoki Fukutomi: Miwa Municipal Hospital; Takahiro Komori: International University of Health and Welfare Shioya Hospital; Hideaki Takata: National Cerebral and Cardiovascular Center; Ken Oyama: Oyama Medical Clinic; Masaaki Miyakawa: Miyakawa Medical Clinic; Hisao Mori: Yokohama Sotetsu Bldg Clinic; Hiro Yamakawa: Yamakawa Medical Clinic; Hareaki Yamamoto: Yamamoto Clinic; Naoto Yagi: Yagi Clinic; Yoshikazu Aoka: Hitotsubashi Hospital; Toru Awaya: Awaya clinic; Toshiro Iketani: Iketani Medical Clinic; Mitsutoshi Kato: Kato Clinic; Tsuguhisa Hatano: Hatano Clinic; Kiyoshi Uchiba: Nagano City Ooka Clinic; Takeshi Takemi: Jingumae Clinic.
Members of the Endpoint committee
Junichi Yamazaki: Toho University; Shinji Hisatake: Toho University Medical Center Omori Hospital; Uichi Ikeda: Nagano Municipal Hospital; Yasuhisa Kitagawa: Tokai University Hachioji Hospital; Hirohisa Okuma: Tokai University Hachioji Hospital; Kentaro Tokuoka: Tokai University Hachioji Hospital; Koichi Hayashi: Keio University; Shu Wakino: Keio University; Hirobumi Tokuyama: Keio University.
Rights and permissions
About this article
Cite this article
Eguchi, K., Miyashita, H., Takenaka, T. et al. High central blood pressure is associated with incident cardiovascular events in treated hypertensives: the ABC-J II Study. Hypertens Res 41, 947–956 (2018). https://doi.org/10.1038/s41440-018-0075-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-018-0075-8
This article is cited by
-
The effect of non-invasively obtained central blood pressure on cardiovascular outcome in diabetic patients in Assiut University Hospitals
The Egyptian Journal of Internal Medicine (2022)
-
Deterioration of sexual function is associated with central hemodynamics in adult Japanese men
Hypertension Research (2020)
-
Central blood pressure predicts the development of hypertension in the general population
Hypertension Research (2020)
-
The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2019)
Hypertension Research (2019)
-
Gathering evidence on the prognostic role of central blood pressure in hypertension
Hypertension Research (2018)