Abstract
Purpose
Advances in gene therapy and precision medicine have led to a growing number of novel treatments for rare genetic diseases. Patients/families may lack access to up-to-date, accurate, and relevant information about these treatments. Social media offers one potentially important resource for these communities. Our goal was to understand how patients/families with spinal muscular atrophy (SMA)—a rare genetic condition—used social media to share, consume, and evaluate information about the novel treatment nusinersen (Spinraza) following the drug’s approval.
Methods
We conducted qualitative, semistructured interviews with 20 SMA patients or parents of patients, deriving themes and subthemes through content and thematic network analysis. Participants also completed a demographic survey.
Results
Participants described leveraging social media to learn about nusinersen treatment, make informed treatment decisions, and advocate for/access treatment. They also described critically evaluating the trustworthiness of nusinersen-related information on social media and the privacy risks of social media use.
Conclusion
Patients/families used social media to navigate the new and dynamic landscape of nusinersen treatment for SMA, while attempting to mitigate misinformation and privacy risks. As new treatments become available, providers and patients/families may benefit from proactively discussing social media use, so as to maximize important benefits while minimizing risks.
Similar content being viewed by others
INTRODUCTION
Novel treatments can offer transformative health improvements for patients with rare genetic diseases. Spinal muscular atrophy (SMA), one such disease, is characterized by muscle atrophy and weakness, with an estimated incidence of 1 in 11,000 live births.1,2 The severity of the condition ranges from infantile skeletal muscle and respiratory paralysis resulting in death (type I) to adult-onset weakness with normal life expectancy (type IV).1
In December 2016, the FDA approved the antisense oligonucleotide therapy nusinersen (Spinraza)—at that time, the only disease-modifying treatment for SMA.3,4 Nusinersen increases production of full-length survival of motor neuron (SMN) protein, slowing disease progression and even reversing symptoms in some patients.4,5,6 The treatment course is lifelong, requiring intrathecal administration (via lumbar puncture) six times during the first year of treatment and three times each year thereafter.7 At a cost of $750,000 in the first year and $375,000 in subsequent years, it is among the most expensive medications in the world.8 Nusinersen’s cost, invasive delivery route, and unequal geographic distribution of specialized treatment centers pose practical and ethical challenges for families, who must decide whether and how to seek treatment, and for providers, who aim to ensure equitable access.9,10 (Throughout this article, we refer to nusinersen by its generic name. Participants referred to nusinersen by its trade name, Spinraza; in quotes, we have left this unchanged.)
Social media has been identified as an important source of information and support for patients and families,11,12,13 including those with rare genetic conditions,14,15,16,17 who may feel socially isolated and/or lack access to condition-specific medical expertise.18 To our knowledge, however, no study has specifically investigated the role of social media for patients/families navigating novel treatments for rare genetic diseases. As greater numbers of such treatments become available with advances in gene therapy and precision medicine,19,20 it is important to understand how patients and families share, consume, and evaluate treatment information through social media. Characterizing nusinersen-related social media use for SMA can elucidate risks and potential benefits of social media for rare conditions, allowing providers to tailor patient guidance accordingly.
Our goal was to understand how SMA patients and families used social media, including Facebook, for their nusinersen-related needs. We focused on the early period after nusinersen’s approval, when SMA was transitioning from an untreatable to a potentially treatable disease, in part to describe a phenomenon that may occur with other novel treatments for rare genetic diseases.
MATERIALS AND METHODS
Study design and ethics
We conducted semistructured interviews with SMA patients or parents of patients within 15 months after nusinersen’s approval. Qualitative methods allowed us to characterize patient/family experiences21,22 and reveal potentially unanticipated information.23,24 We collected demographic and basic descriptive information about participants’ social media use through an online survey.
This study was performed in accordance with relevant guidelines and regulations, including those set forth in the Declaration of Helsinki. Informed consent was obtained from all subjects, and de-identified data were used for analysis and reporting. The Stanford University Institutional Review Board approved this study.
Participant recruitment
There are many SMA-specific Facebook groups that SMA patients and families can use to communicate through written posts, photos, and videos (Box 1). We identified 30 SMA Facebook groups by searching “SMA” and “spinal muscular atrophy” on Facebook (Supplementary Table 1). We invited group moderators to post a recruitment text in their groups. Of the 30 Facebook groups contacted, 15 moderators responded via Facebook Messenger with interest in posting the recruitment text, and 7 viewed the message but did not respond. Because the groups are private, we could not count the exact number of moderators who followed through with the request to post our text. We also recruited participants from the neuromuscular clinic at Lucile Packard Children’s Hospital (Stanford, CA) through direct emails to patients, the clinic’s email list, the clinic’s private Facebook page, and professional contacts of a clinic provider. We used snowball sampling, asking participants to email the text to contacts who used SMA social media resources. We included SMA patients and parents of children with SMA who were at least 18 and English-speaking. Participants provided verbal informed consent and received a $15 Amazon gift card.
Data collection
Each participant completed a 53-item RedCap survey, which included questions about demographics, diagnosis (e.g., type of SMA), and social media use (e.g., platforms used, number of groups used, moderator status). One author (J.R.B.) conducted and audiorecorded all interviews remotely between July 2017 and March 2018 via Zoom videoconference software or phone. We piloted the interview guide with a four-person convenience sample: two students, one parent of a patient with a neuromuscular condition, and one physician/researcher. The guide included questions in four domains: (1) diagnostic odysseys, including the role of social media; (2) SMA-related social media use, including information-gathering and social support; (3) nusinersen-related social media use, including information-gathering, treatment decision-making, and advocacy; and (4) perceptions of misinformation/privacy risks—two frequent concerns about health-related social media use.12 This analysis focuses on the third and fourth domains.
Data analysis
Audio files were transcribed verbatim, and de-identified transcripts were analyzed using content and thematic network analysis.25,26 First, two investigators (J.R.B. and H.K.T.) read and discussed all transcripts, identifying structural codes based on interview questions, and identifying emergent thematic codes through iterative discussion. Three investigators (A.A.I, J.R.B., and H.K.T.) independently coded the transcripts using DeDoose software (version 8.1), reconciling differences by consensus. Two investigators (A.A.I. and H.K.T.) collaboratively generated code summaries, refined them through discussion, and identified overarching themes and subthemes.
RESULTS
We recruited 19 participants through SMA Facebook groups or snowball sampling, and three through the Lucile Packard Children’s Hospital neuromuscular clinic. Because nusinersen was not yet approved internationally, we excluded two interviews with participants receiving health care outside of the United States prior to analysis.
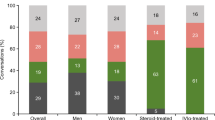
Of the final 20 participants, seven were parents of children with SMA types I–III (parent mean age 37.0 years, SD 5.4 years; child mean age 5.7 years, SD 3.8 years), and 13 were adults with SMA types II–III (mean age 38.2 years, SD 11.4 years). Three were self-identified moderators of an SMA Facebook group. Full demographics are shown in Table 1.
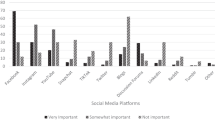
When asked which social media platforms they used for SMA, almost all participants (n = 18) described Facebook as their primary/only platform. When asked why they joined SMA Facebook groups, two participants said the approval of nusinersen and associated clinical trials were specific triggers; 17 other participants had used SMA Facebook groups prior to learning about nusinersen, but the drug’s approval prompted them to join additional nusinersen-specific groups. Three participants volunteered that they first learned about nusinersen and/or its approval through SMA Facebook groups, while other participants first learned about nusinersen through their providers and subsequently sought information about the drug through social media.
Participants described using social media to meet three nusinersen-related needs: (1) learning about nusinersen treatment; (2) making informed treatment decisions; and (3) advocating for/accessing treatment (Table 2). They also critically evaluated the trustworthiness of social media and the privacy risks of using it for SMA (Table 2).
Learning about nusinersen treatment
Participants reported having many questions about nusinersen following FDA approval, and seeking information about the drug through Facebook groups. They said that Facebook provided experience-based information and anticipatory guidance from fellow patients/families: “A lot of information [in nusinersen-specific Facebook groups] was very new at the time but it helped me see what I would be going through in the next couple months/years” (P7-Patient, type II).
Participants used Facebook groups to gather information about how nusinersen is administered, including firsthand information about body positioning during the lumbar puncture, the anatomical site and duration of the injection, and details of postprocedural recovery. This information was frequently shared visually, using photos and videos: “People are posting pictures of how the injection is done, what the injection needle looks like, what gauge it is, you know, you eat before or after, or whatever side effects that you face” (P21-Patient, type III). Other participants shared or discovered strategies for mitigating side effects: “Just to lay down…first, be well-hydrated going into it, and then secondly…when they do sit up, go very gradual…” (P8-Parent, type II).
Participants described specific benefits of receiving this anticipatory guidance directly from fellow patients. For example, an adult with SMA said they weighed experience-based information alongside information from traditional medical sources, like doctors or drug companies:
“I want to get the medical opinion and the patient opinion…instead of just what it says on the Spinraza website […I want patients’] opinions, their testimony about how they get the injection, and then that helps me to figure out OK, this person did it this way, so I might be able to do it as well.” (P4-Patient, type III)
Participants also sought information about nusinersen’s effectiveness through Facebook groups. They described viewing videos, photos, and/or written narratives about changes in functional status in response to the drug. These helped one participant, “see more of the actual results and what people are going through.” (P20-Patient, type II) Several participants, inspired by these firsthand narratives, shared their own pre- and post-treatment videos documenting effectiveness:
“I plan to do the same thing that everybody else did for me…to do a little video of what my capabilities are before I start taking it and then maybe once every six months kind of redo that and see if there’s a difference—not only for me personally to see growth, but for others as well because I think that’s the smartest way to share is to document data.” (P22-Patient, type III)
Making informed treatment decisions
Almost half of the participants (n = 8) said information from Facebook groups influenced their decision about whether to seek treatment. One mother described how she changed her mind about pursuing nusinersen treatment for her son: “But now I’ve seen others [in Facebook groups] go through this, get access to this drug, that are [child]’s age, so now I’ve gone to the other side, now I’m pushing for Spinraza” (P5-Parent, type I).
Some adults with SMA expressed concern that the early clinical data from children with SMA type 1 might not generalize to adults with SMA types II–IV. They described seeking and assessing videos and written experiences from clinically similar adults on Facebook before making treatment decisions: “I’m still waiting and seeing how it works with other adults. I’ll let them be the guinea pig…and then go from there” (P6-Patient, type II). Several of these participants noted that because they were geographically isolated from SMA experts, they could not find this information elsewhere.
Facebook groups were also useful for assessing potential treatment burdens. Some participants noticed information about specific challenges that were unique to their situations as adults with disabilities, or as people living far from a treatment site: “You hear some people experience like, ‘Oh the second [injection] was easier,’ so I’m thinking, I want to make sure if I have to drive ten hours back, it’s not crazy exhausting or painful” (P1-Patient, type III).
Advocating for and accessing treatment
Participants described using social media to learn about which individuals (SMA subtypes, genotypes, and/or functional statuses) were eligible for nusinersen treatment. This information empowered some participants to advocate for their own eligibility. For example, five participants first learned through Facebook groups that SMA patients with spinal fusions were eligible to receive nusinersen:
“So I have like a full spinal fusion and the medicine has to go in your spine, so it was interesting and very helpful to see other people who…weren’t able to do it or people who were able to do it, and what they did and…other resources they use with their doctors, like instead of a regular X-ray they’ll do like a CT scan or something and I was able to like bring that to my doctors and they’re like, oh, that’s interesting, and then they tried it.” (P7-Patient, type II)
Another participant used information from a Facebook group to successfully challenge their physician’s claim that they were not eligible:
“An internal medicine doctor…rejected my treatment because I have three copies of SNM2, and the minimum requirement is [two] SMN2 copies—there has to be at least two. I have three, they rejected it. In fact, just the opposite, the more copies you have, the faster recovery chance you have. I mean this is basic knowledge and they should know this, so I challenged their finding and I asked for a neuromuscular doctor to evaluate my complaint, and got approved in a couple of days.” (P21-Patient, type III)
Social media also facilitated treatment access by allowing patients/families to share practical information about the logistics of access, including which medical centers were administering nusinersen:
“[If someone in an SMA Facebook group] can’t find any hospitals or neurologists that are willing to…give [them] this injection although [they] have been approved […] I will jump in and I will say, hey, you know, this is what you do, this is the doctor name and private message how to go about this.” (P21-Patient, type III)
This information appeared to be especially beneficial to individuals who were located in rural areas, restricted in mobility/transportation options, and/or otherwise isolated from SMA experts or specialized treatment centers. One patient explained, “My local doctors had never heard of [nusinersen] so I had to kind of use social media to learn more about it and find where I could pursue the treatment.” (P20-Patient, type II)
Five participants said they used information from Facebook to educate their providers about administering nusinersen treatment. This was important because many providers/institutions had limited experience with the drug, particularly in the first year after FDA approval:
“[Facebook] was very, very helpful around the country because some of the facilities weren’t even on board and most of the patients already kind of knew what to do, knew what the procedure was going to be like before even their medical staff or their doctor’s office, so they were helping to guide them…because it was all new to everyone and it kind of took everyone by surprise the way it happened so fast. […] A lot of the medical community was not prepared for it.” (P17-Parent, type II)
The experiences shared by patients/families in Facebook groups also revealed heterogeneity in treatment practices across providers/institutions in this early period. These groups provided strategies for navigating these discrepancies. One parent said, “I could go to our medical professional and say, ‘St. Louis is doing this [anesthesia for injections] and Chicago is doing this…that helped me to advocate” (P11-Parent, type I). Other Facebook group members advised families to connect doctors with the drug company to standardize protocols:
“We have found out that each hospital really is doing it very differently, and in some cases, not following [drug company] protocol. And so then we always encourage families, ‘Okay, if they have told you that or done that, you need to speak to…’ You know, we’ll refer them to someone back at Biogen [nusinersen’s manufacturer], you know, ‘Call this person, tell them about it, try and get that straightened out so it doesn’t happen next time.’” (P19-Parent, type I)
When asked about barriers to accessing nusinersen, almost all participants (n = 19) mentioned cost and reimbursement. Nearly all of these participants (n = 18) reported getting useful information from Facebook groups about how to navigate insurance challenges, including whom to contact from insurance companies and what to write in letters requesting reimbursement or appealing denials. This information was often experience-based: “Parents will post their letters from the insurance companies, they’ll also post what they’ve sent to the insurance companies, I’ve used templates and removed their name and put my child’s name in, oh yeah, you name it—definitely has helped” (P17-Parent, type II).
Perceptions of information trustworthiness and privacy risks
When asked, almost all (n = 18) participants said they generally trusted information about nusinersen in SMA Facebook groups, citing two main reasons. First, information was grounded in personal experience: members of the Facebook communities, “are living [with SMA] every day and have more practical experience with the disease than the doctors [do]” (P19-Parent, type I). Second, participants described a “fact-checking” culture within the groups:
“There’s enough moms in the group that are really well-versed…they’re veteran moms. Some of them, you know, that’s all they do all day—this is their world—they have a type 1 child, they don’t have nursing, and this is their connection to the outside world and they’ve kind of taken this on, and they’re pretty spot on. And if [other families] are not [accurate…] they totally call them out.” (P8-Parent, type II)
Despite generally trusting information in social media groups, most participants described critically evaluating its reliability and relevance, as they would with any online information. Three participants explained that savvy Facebook users “take everything with a grain of salt” (P11-Parent, type I; P20-Patient, type II; P22-Patient, type III), and one parent noted, “Just because it’s on [Facebook], does not mean it’s absolute truth or it’s going to be good for my child” (P15-Parent, type III). Over a quarter of participants (n = 6) reported verifying information from social media with providers. These participants described how providers could empower them to speak honestly about their social media use: “Doctors have to be open enough to honestly have a conversation with their patients without being upset…and not just criticize them for looking for different answers” (P19-Parent, type I).
Participants expressed awareness of social media privacy risks, but generally felt that social media’s benefits outweighed these risks. To minimize privacy risks, participants filtered what they shared based on the size and privacy level of the specific Facebook group: “Whenever there’s been like an emergency or something very scary, I wouldn’t post about that on [son’s] public Facebook page, but I might, just for support, post about it on an SMA type 1 family group…because they’ve been through that, and that’s private and closed” (P11-Parent, type I).
DISCUSSION
To our knowledge, this is the first study describing the intersection of two phenomena in precision medicine. The first is the increasing prevalence of novel treatments for rare genetic diseases,19,20 which can present challenges around cost, access, and informed decision-making.10 The second phenomenon is the increased use of social media for information-sharing and social support among rare genetic disease communities.14,15,16,17 This intersection is especially important for patients/families who already feel socially isolated and often struggle to receive adequate medical information.18 Participants in this study leveraged social media to navigate the new and dynamic landscape of nusinersen treatment for SMA: learning about nusinersen, making informed treatment decisions, and advocating for/accessing treatment. Participants also critically evaluated the trustworthiness of social media and the privacy risks of using it for SMA.
These results suggest that social media may offer important benefits for patients with rare conditions and their families. First, condition-specific Facebook groups may constitute modern-day disease advocacy communities, assuming some roles of more traditional advocacy organizations,27,28 i.e., information dissemination, fact-checking, emotional support, and advocacy around treatment access. Because rare disease patients/families are few in number, widely dispersed, and may have mobility restrictions, social media offers a unique way to connect these communities and facilitate information exchange. In contrast, traditional advocacy group websites may not offer the same levels of community, patient/family-driven interaction, or responsiveness available in social media groups.12
Second, social media may “democratize” treatment-related information that is otherwise difficult to obtain. Indeed, even basic information about some rare genetic conditions is available only through experts at specific academic medical centers.18,29 Our participants described sharing videos of the nusinersen injection procedure, technical information about treatment eligibility, copies of letters requesting insurance coverage, and experiences of treatment effectiveness. In a previous study, SMA patients/families who were grappling with decisions about nusinersen treatment said they wanted this kind of experience-based information, and had struggled to find it;9 our results suggest that social media helps meet this need. The information patients/families access on social media may, in turn, help them make more informed treatment decisions in collaboration with providers. This is particularly relevant for novel gene/gene-targeted therapies, since some providers may initially lack information about these treatments.18,29
However, there are also known risks to social media use, some of which may be heightened for patients with rare genetic diseases and their families. First, social media could spread and amplify misinformation,30,31 potentially misguiding patients/families.31 Second, tension between physicians with professional expertise and patients with social media–derived information could weaken therapeutic relationships.32 Third, privacy violations33 could be a particular concern for rare and ultrarare disease communities, since small patient numbers make individuals more identifiable and social media platforms may be vulnerable to breaches and loopholes.34 Finally, condition-specific social media communities could become a forum for third parties (e.g., pharmaceutical companies) to manipulate patients’ and families’ opinions and/or behavior, or for bad actors to cause direct harm.35 Our participants sought to minimize these risks through critical judgment and self-monitoring, and nearly all of them felt that the potential benefits outweighed the risks.
Implications
In light of these risks and potential benefits, how should providers anticipate and respond to treatment-related social media use? It has been reported that some providers are unaware of, or hostile to, patients’ and families’ use of the Internet for medical information.36 Rather than ignoring, discouraging, or condemning treatment-related social media use, we suggest a proactive approach that attempts to maximize the potential benefits of social media while minimizing its risks.
For example, providers could encourage transparency about social media use, offer to help evaluate information from social media, and remind patients/families to exercise caution and critical judgment. They could also warn patients/families of privacy risks and even compile a set of vetted social media resources for distribution. This may be particularly important, as Facebook has received scrutiny for its allegedly incomplete response to a privacy loophole allowing third parties to discover the names of members in private groups.34
Importantly, many groups are patient/family-only spaces. Health-care professionals should respect membership guidelines and may not be able to vet content themselves, but could instead partner with patient-experts to amass reliable resources. It is unlikely that patients and families will stop using social media to learn and communicate about novel treatments, and they often derive emotional support or empowerment from what they learn online. Therefore, our proposed strategies could mitigate the harms of online misinformation while enhancing shared decision-making surrounding novel treatments.
More research is needed in this area. For example, studies could examine the content of condition-specific social media groups and evaluate reliability, bias, sources, and the possible influence of third parties (e.g., pharmaceutical companies). Assessing these biases may be especially important when competing treatments are available. Indeed, since these interviews were conducted, nusinersen has become more widely available in the United States, the SMA gene therapy onasemnogene abeparvovec-xioi (Zolgensma) has received FDA approval,37 and other treatments have advanced in clinical development.38 At a cost of $2.1 million, Zolgensma has—like nusinersen—posed practical and ethical challenges surrounding affordability and access, including questions about unconventional rationing approaches (e.g., lottery) where the gene therapy is not yet available.39 As more data become available about these novel treatments and the barriers to accessing them, social media may become even more important in helping patients/families gather information and make treatment decisions.
Future research on rare genetic diseases may benefit from partnership with condition-specific social media groups. For example, social media content could provide information about patients’ preferences and unmet needs.40 Researchers and providers could partner with social media groups to rapidly administer surveys, recruit patients for clinical trials, and otherwise engage inaccessible stakeholders.41 In this way, social media could augment partnerships between researchers and traditional patient advocacy groups.
Limitations
This study has several limitations. First, because we recruited most of our participants through social media, they may have been more positive about its use than non–social media users. Second, our participants were English-speaking and primarily white. Our results may not reflect the perspectives of diverse social media users for SMA, and may also indicate demographic disparities in access to or use of SMA-specific social media resources.
Third, our results are based on patient/parent interviews describing interactions with social media content and not on analysis of the content itself. Many condition-specific social media groups are private, making a comprehensive content analysis difficult to conduct. Analyzing a subset of open groups could help familiarize providers with the content their patients produce and consume online.
Conclusion
As novel treatments for rare genetic diseases become more common with advances in gene therapy and precision medicine, many patients and families will likely turn to social media to share and consume information about these treatments. Participants in this study leveraged social media to navigate the new and dynamic landscape of nusinersen treatment for SMA: learning about nusinersen, making informed treatment decisions, and advocating for/accessing the treatment itself. They also critically evaluated the trustworthiness of social media and the privacy risks of using it for SMA. As new treatments become available, providers and patients/families may benefit from proactively discussing social media use, so as to maximize important benefits while minimizing risks.
References
Lunn MR, Wang CH. Spinal muscular atrophy. Lancet. 2008;371:2120–2133.
Sugarman EA, Nagan N, Zhu H, et al. Pan-ethnic carrier screening and prenatal diagnosis for spinal muscular atrophy: clinical laboratory analysis of >72 400 specimens. Eur J Hum Genet. 2012;20:27–32.
Hoy SM. Nusinersen: first global approval. Drugs. 2017;77:473–479.
Corey DR. Nusinersen, an antisense oligonucleotide drug for spinal muscular atrophy. Nat Neurosci. 2017;20:497–499.
Finkel RS, Mercuri E, Darras BT, et al. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med. 2017;377:1723–1732.
Darras BT, Chiriboga CA, Iannaccone ST, et al. Nusinersen in later-onset spinal muscular atrophy: long-term results from the phase 1/2 studies. Neurology. 2019;92:e2492–e2506.
Haché M, Swoboda KJ, Sethna N, et al. Intrathecal injections in children with spinal muscular atrophy: nusinersen clinical trial experience. J Child Neurol. 2016;31:899–906.
Thomas K. Costly drug for fatal muscular disease wins F.D.A. approval. The New York Times. 30 December 2016. https://www.nytimes.com/2016/12/30/business/spinraza-price.html. Accessed 16 December 2019.
Pacione M, Siskind CE, Day JW, Tabor HK. Perspectives on spinraza (nusinersen) treatment study: views of individuals and parents of children diagnosed with spinal muscular atrophy. J Neuromuscul Dis. 2019;6:119–131.
Burgart AM, Magnus D, Tabor HK, et al. Ethical challenges confronted when providing nusinersen treatment for spinal muscular atrophy. JAMA Pediatr. 2018;172:188–192.
Smailhodzic E, Hooijsma W, Boonstra A, Langley DJ. Social media use in healthcare: a systematic review of effects on patients and on their relationship with healthcare professionals. BMC Health Serv Res. 2016;16:442.
Moorhead SA, Hazlett DE, Harrison L, Carroll JK, Irwin A, Hoving C. A new dimension of health care: systematic review of the uses, benefits, and limitations of social media for health communication. J Med Internet Res. 2013;15:e85.
Pew Research Center. Peer-to-peer health care. February 2011. https://www.pewresearch.org/internet/2011/02/28/peer-to-peer-health-care-2/. Accessed 4 March 2020.
Khouri JS, McCheyne MJ, Morrison CS. #Cleft: the use of social media amongst parents of infants with clefts. Cleft Palate Craniofac J. 2018;55:974–976.
Jacobs R, Boyd L, Brennan K, Sinha CK, Giuliani S. The importance of social media for patients and families affected by congenital anomalies: a Facebook cross-sectional analysis and user survey. J Pediatr Surg. 2016;51:1766–1771.
Gundersen T. “One wants to know what a chromosome is”: the internet as a coping resource when adjusting to life parenting a child with a rare genetic disorder. Sociol Health Illn. 2011;33:81–95.
Barton KS, Wingerson A, Barzilay JR, Tabor HK. “Before Facebook and before social media…we did not know anybody else that had this”: parent perspectives on internet and social media use during the pediatric clinical genetic testing process. J Community Genet. 2019;10:375–383.
Pelentsov LJ, Laws TA, Esterman AJ. The supportive care needs of parents caring for a child with a rare disease: a scoping review. Disabil Health J. 2015;8:475–491.
High KA, Roncarolo MG. Gene therapy. N Engl J Med. 2019;381:455–464.
Anguela XM, High KA. Entering the modern era of gene therapy. Annu Rev Med. 2019;70:273–288.
Denzin NK, Lincoln YS, editors. The SAGE handbook of qualitative research. Thousand Oaks, CA: SAGE; 2011.
Silverman D. Doing qualitative research: a practical handbook. 4th ed. Thousand Oaks, CA: SAGE; 2013.
Creswell JW, Clark VLP. Designing and conducting mixed methods research. Thousand Oaks, CA: SAGE; 2017.
Seidman I. Interviewing as qualitative research: a guide for researchers in education and the social sciences. New York: Teachers College Press; 2006.
Hsieh H-F, Shannon SE. Three approaches to qualitative content analysis. Qual Health Res. 2005;15:1277–1288.
Attride-Stirling J. Thematic networks: an analytic tool for qualitative research. Qual Res. 2001;1:385–405.
Epstein SG. Patient groups and health movements. In: Hackett EJ, Amsterdamska O, Lynch M, Wajcman J, editors. The handbook of science and technology studies. Cambridge: MIT Press; 2008. p. 499–539.
National Organization for Rare Disorders. History of leadership. https://rarediseases.org/about/what-we-do/history-leadership/. Accessed 5 March 2020.
Vandeborne L, van Overbeeke E, Dooms M, De Beleyr B, Huys I. Information needs of physicians regarding the diagnosis of rare diseases: a questionnaire-based study in Belgium. Orphanet J Rare Dis. 2019;14:99.
Vosoughi S, Roy D, Aral S. The spread of true and false news online. Science. 2018;359:1146–1151.
Chou W-YS, Oh A, Klein WMP. Addressing health-related misinformation on social media. JAMA. 2018;320:2417–2418.
Rupert DJ, Moultrie RR, Read JG, et al. Perceived healthcare provider reactions to patient and caregiver use of online health communities. Patient Educ Couns. 2014;96:320–326.
Fazzini K, Farr C. Facebook recently closed a loophole that allowed third parties to discover the names of people in private, “closed” Facebook groups. CNBC. 12 July 2018. https://www.cnbc.com/2018/07/11/facebook-private-groups-breast-cancer-privacy-loophole.html. Accessed 5 March 2020.
Trotter F. SicGRL vulnerability is still not fixed. Light Collective. 12 September 2019. https://lightcollective.org/2019/09/12/sicgrl-update/. Accessed 23 May 2020.
Shead S. People with epilepsy targeted in Twitter attack. BBC News. 18 December 2019. https://www.bbc.com/news/technology-50835214.
Nicholl H, Tracey C, Begley T, King C, Lynch AM. Internet use by parents of children with rare conditions: findings from a study on parents’ web information needs. J Med Internet Res. 2017;19:e51.
Novartis. AveXis receives FDA approval for Zolgensma®, the first and only gene therapy for pediatric patients with spinal muscular atrophy (SMA). 24 May 2019. https://www.novartis.com/news/media-releases/avexis-receives-fda-approval-zolgensma-first-and-only-gene-therapy-pediatric-patients-spinal-muscular-atrophy-sma. Accessed 5 March 2020.
Roche. FDA grants priority review to Roche’s risdiplam for spinal muscular atrophy. 25 November 2019. https://www.roche.com/media/releases/med-cor-2019-11-25.htm. Accessed 5 March 2020.
Joseph A, Silverman E. Lottery like no other offers a cutting-edge medicine — with lives on the line. STAT News. 7 February 2020. https://www.statnews.com/2020/02/07/a-lottery-like-no-other-offers-up-a-cutting-edge-medicine-with-lives-on-the-line/. Accessed 23 May 2020.
Jamison J, Sutton S, Mant J, Simoni AD. Online stroke forum as source of data for qualitative research: insights from a comparison with patients’ interviews. BMJ Open. 2018;8:e020133.
Schumacher KR, Stringer KA, Donohue JE, et al. Social media methods for studying rare diseases. Pediatrics. 2014;133:e1345–e1353.
Facebook. Help center. https://www.facebook.com/help/?helpref=hc_global_nav. Accessed 25 March 2020.
Acknowledgements
The authors thank the participants. They also thank Alyssa Burgart, Skye Miner, Meghan Halley, Carly Siskind, and Saskia Hendriks for helpful feedback. This study was supported by the Stanford Center for Biomedical Ethics, the Stanford Medical Scholars Research Program, and the Intramural Research Program of the National Institutes of Health (NIH), Clinical Center Department of Bioethics.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors declare no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Disclaimer
The views expressed by A.A.I. are his own and do not necessarily reflect those of the National Institutes of Health, the Department of Health and Human Services, or the United States Government.
Supplementary information
Rights and permissions
About this article
Cite this article
Iyer, A.A., Barzilay, J.R. & Tabor, H.K. Patient and family social media use surrounding a novel treatment for a rare genetic disease: a qualitative interview study. Genet Med 22, 1830–1837 (2020). https://doi.org/10.1038/s41436-020-0890-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41436-020-0890-6
Keywords
This article is cited by
-
Opportunities and pitfalls of social media research in rare genetic diseases: a systematic review
Genetics in Medicine (2021)