Abstract
Purpose
Currently, 31 patients with classical-like EDS (clEDS) due to tenascin-X deficiency have been reported in the literature. We report on the clinical and molecular characteristics of 20 additional patients with clEDS to expand knowledge and to enable improved management of this rare genetic disorder.
Methods
Patients diagnosed with clEDS by the national EDS service in the UK (n = 21) and abroad (n = 1) were asked for consent for publication of their clinical and molecular data.
Results
Of 22 patients, 20 consented. All patients had typical features of clEDS: joint hypermobility, easy bruising, and skin hyperextensibility without atrophic scars. Importantly, 3/20 patients experienced gastrointestinal complications consisting of small or large bowel ruptures and one esophageal rupture. Other notable observations included two separate occurrences of spontaneous compartment syndrome, suspicion of nonaccidental injury due to significant bruising, and significant clinical variability regarding the debilitating effect of joint dislocations.
Conclusions
We propose a predisposition to tissue fragility, particularly of the gastrointestinal tract in patients with clEDS. As such, clinical and molecular confirmation of this diagnosis is essential. It is recommended to follow up these patients closely to understand the natural history to develop better recommendations for management.
Similar content being viewed by others
INTRODUCTION
Ehlers–Danlos syndromes (EDS) are a group of heritable connective tissue disorders with key features of joint hypermobility, skin and vascular fragility, and generalized connective tissue friability.1 Currently, 13 types of EDS are recognized.2 A monogenic type of EDS, previously called tenascin-X (TN-X) deficient EDS, was first reported in 1997.3 It is caused by recessive pathogenic variants in TNXB resulting in deficiency of TN-X. The clinical features strongly resemble classical EDS (cEDS) with two key differences: (1) the absence of atrophic scarring and (2) autosomal recessive inheritance. Due to the clinical resemblance with cEDS, recent updated nosology has renamed the condition classical-like EDS (clEDS).4
The major clinical criteria of clEDS are (1) skin hyperextensibility, with velvety skin texture and absence of atrophic scarring; (2) generalized joint hypermobility with or without recurrent dislocations (most commonly shoulder and ankle); and (3) easy or spontaneous bruising of skin. Minor criteria are (1) foot deformities including broad/plump forefoot, brachydactyly with excessive skin, pes planus, hallux valgus, and piezogenic papules; (2) edema in the legs in absence of cardiac failure; (3) mild proximal and distal muscle weakness; (4) axonal polyneuropathy; (5) atrophy of muscles in hands and feet; (6) acrogeric hands, mallet finger(s), clinodactyly, and brachydactyly; and (7) vaginal/uterus/rectal prolapse.2 Minimal criteria suggestive for clEDS consist of all three major criteria.2 A family history compatible with autosomal recessive inheritance is supportive.2
TNXB encodes the trimeric glycoprotein Tenascin-X (TN-X), which is important for structural support of matrix macromolecular complexes, including collagen fibrils and elastic fibers. For example, TN-X regulates spacing and cohesiveness between collagen fibrils through direct interaction with collagen molecules or through extracellular matrix (ECM) components that interact with collagen fibrils. Interestingly, TN-X also resembles a dynamically expressed, nonstructural protein as it regulates cell adhesion–deadhesion processes. Laboratory studies suggest that TN-X regulates the bioavailability of TGF-β in vitro. In adults, TN-X messenger RNA (mRNA) is observed in several organs and tissues, including blood vessels, with a higher level in the digestive tract (pancreas, stomach, jejunum, ileum, and colon), and the highest expression in the heart, the skin, skeletal muscles, ligaments, and tendons.5
Currently, 31 patients with clEDS have been reported worldwide. Five patients had no genetic confirmation but were diagnosed because of clinical features and absence of TN-X in serum. The remainder had a confirmed genetic cause including patients with contiguous deletions of TNXB and CYP21A2,3,6,7 whole TNXB gene deletions, and intragenic homozygous and compound heterozygous pathogenic variants.7,8,9,10,11,12,13,14,15 Pathogenic TNXB variants (splice site variants or variants introducing premature stop codon) usually lead to nonsense-mediated decay of the mutant RNA. Molecular analysis is challenging as it is complicated by the presence of an adjacent pseudogene (TNXA), which is more than 97% identical to the 3’ end of TNXB (exons 32–44). The TNXB and CYP21A2 (associated with congenital adrenal hyperplasia) genes are located within the human leukocyte antigen histocompatibility complex at chromosome region 6p23.1 alongside their homologous pseudogenes TNXA and CYP21A1P. This region is prone to meiotic nonhomologous recombination resulting in gene conversions (exchange of DNA sequences between gene and pseudogene). There are three types of TNXA/TNXB gene conversions identified.16 CAH-X chimera 1 (CH-1) has TNXB exons 35–44 replaced with TNXA and is characterized by the presence of the c.11435_11524+30del variant. CAH-X chimera 2 (CH-2) has TNXB exons 40–44 replaced with TNXA and is characterized by the TNXA derived c.12174C>G; p.(Cys4058Trp) variant. CAH-X chimera 3 (CH-3) has TNXB exons 41–44 replaced by TNXA and is characterized by the presence of three TNXA derived missense variants.
Here, we report the clinical and molecular features of 20 additional individuals who have been diagnosed with clEDS.
MATERIALS AND METHODS
Patients
Patients with a molecularly confirmed diagnosis of clEDS were identified through the national complex EDS service in London and Sheffield or through collaborations. This service functions as an expert center (London and Sheffield) for diagnosis and follow-up of monogenic types of EDS. Clinicians with expertise in EDS performed medical and family histories, as well as clinical examinations. Written consent for publication, including photographs, was obtained from all individuals. According to the institutional review board (IRB) no formal research ethics approval or research and development approval was required as stipulated by the UK Policy Framework for Health and Social Care Research and the Health Research Authority decision tool.
Transmission electron microscopy
Ten patients had skin biopsies for transmission electron microscopy (TEM) taken from the upper inner arm using punch biopsy. These samples were processed for TEM as reported previously.17
Molecular genetic analysis
DNA sequence analysis of the COL1A1, COL3A1, and TNXB genes was performed by means of Illumina HiSeq next-generation sequencing (NGS). The pseudogene-homolog region of TNXB was analyzed by Sanger sequencing in a few large multiexon amplicons. Gene dosage analysis of TNXB and COL3A1 was performed using multiplex ligation-dependent probe amplification (MLPA, MRC-Holland kit P155). DNA analysis of COL3A1 and COL1A1 including MLPA of COL3A1 was performed in two patients with esophageal rupture and/or bowel rupture.
Variants were analyzed according to best practice guidelines for the evaluation of pathogenicity and the reporting of sequence variants in clinical molecular genetics (2020, Association for Clinical Genomic Science, https://www.acgs.uk.com/news/uk-practice-guidelines-for-variant-classification-2019/). Confirmation of clinically significant sequence variants by Sanger sequencing was performed as necessary. DNA changes have been described according to NM_019105.7 (TNXB), NM_000090.3 (COL3A1), and NM_000088.3 (COL1A1).
RESULTS
Patients
A total of 21 patients from 17 families were diagnosed with clEDS in our service and 1 patient was diagnosed abroad. Twenty patients consented for publication of their clinical and molecular features (P1–P20). The age at diagnosis varied between 3 and 68 years, with all patients describing onset of symptoms in childhood. The male:female ratio was 1:1.5. Parental consanguinity occurred in four families. Ethnic background was Pakistani in 2 families, Indian in 1 family, and Caucasian in 14 families. An overview of clinical features can be found in Supplementary Table 1. Photographs of common clinical features are presented in Fig. 1.
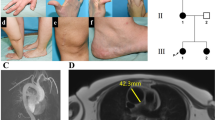
a Skin hyperextensibility. b Easy bruising and spontaneous ecchymoses in a child with TN-X deficiency; a suspicion on nonaccidental injury was raised. c Wrinkled skin with deformation of thumbs and fingers due to joint hypermobility and repetitive dislocations. d Fat pads on soles of feet and piezogenic pedal papules. e Foot deformations with hallux valgus and over and underlapping toes in P3 at the age of 62 years. f Broad forefeet with short toes. g Foot deformations with hallux valgus and over and underlapping toes in P1 at the age of 55 years. h Skin wrinkling on soles of feet. i Subcutaneous spheroids on elbow. j Clinical photographs of patients P3 and P6 showing no specific dysmorphic features but sagging of the skin in 62-year-old P3 and to a milder degree in 68-year-old P6.
Transmission electron microscopy
Six patients with clEDS had a normal biopsy. Other reports concluded (1) reduced collagen in the reticular dermis and some abnormal changes in the elastic fibers (P1); (2) relatively normal biopsy with some excess of elastic fibers (P2); (3) collagen bundle content packing occasionally mildly loose (P18); (4) seven small collagen flowers, mean collagen fibril diameter significantly less than expected for age, 71 nm compared with 85 nm (P15); and (5) moderate numbers of small collagen flowers present (P13).
Molecular analysis
Figure 2 illustrates the identified (likely) pathogenic variants. There were 16 different (likely) pathogenic variants identified of which 11 have not been reported previously (depicted in light grey). The most frequently identified pathogenic variants were (1) c.12174C>G; p.(Cys4058Trp) (14 alleles), (2) pathogenic deletion including exon 35 (c.11381-?_11524+?del) (9 alleles), (3) c.[11431-?_12725+?del] (3 alleles), and (4) c.4129G>T; p.(Glu1377*) (2 alleles). The notation c.11381-?_11524+?del describes a deletion of exon 35 detected via MLPA with the “?” indicating that it is unknown how far into the neighboring introns the deletion extends. All variants were classified as pathogenic apart from c.5279–24_5279–14del, which has been classified as likely pathogenic. DNA analysis of COL3A1 including MLPA and COL1A1 was normal in two patients (P6, P8) with esophageal rupture and/or bowel rupture. In P13 and P15, who had evidence of collagen flowers on skin biopsy, DNA analysis of COL5A1 and COL5A2 was performed, which did not show abnormalities.
DISCUSSION
We describe the largest cohort of newly identified patients so far reported in the literature with a clinical and molecularly confirmed diagnosis of clEDS. The clinical features in our cohort are largely similar to those previously reported in the literature. The most frequent features are hyperextensible skin, easy bruising, joint hypermobility, and velvety/smooth skin. Skin hyperextensibility was the only feature invariably present in our patients and all other reported individuals. A recent large study reassessed 11 patients from 7 families7,9,15 along with 6 new patients from 4 families.8 In addition to the well documented features of soft, velvety, hyperextensible skin; easy bruising; and joint hypermobility, the authors highlighted issues with foot and hand deformities (including brachydactyly, piezogenic pedal papules, hallux valgus, broad feet), fatigue, and muscle pain and weakness. This is observed as well in our series of patients, as Supplementary Table 1 demonstrates, although we have not been able to objectively show muscle weakness in our cohort. However, we also made several important additional observations.
Gastrointestinal fragility
In our cohort 3/20 individuals (P6–8) experienced serious gastrointestinal complications. In addition, a family member of P1 and of P8 with respectively confirmed clEDS and likely clEDS also had serious gastrointestinal complications.
P6 sustained an esophageal perforation at the age of 55 during his third gastroscopy because of complaints of reflux and bloating, a spontaneous small bowel perforation at the age of 56, and one small bowel perforation after nasojejunal barium study at the age of 59. A colonoscopy at the age of 66 years showed pan-colonic diverticulosis. He has a homozygous c.12174C>G; p.(Cys4058Trp) variant.
P7 was known with longstanding bowel problems with significant bloating, nausea, vomiting, and constipation and was found to have a slow gut motility. She had a jejunal perforation at the age of 40 years requiring laparotomy and small bowel resection. Histopathology showed the presence of a mesenteric abscess associated with mural inflammation. Many eosinophils were observed surrounding the abscess and in the muscularis propria of the normal tissue. At the age of 42 years she developed an abdominal swelling and was found to have an obstructed incisional hernia for which she had surgery including bowel resection. She developed an abdominal wall abscess following hernia repair which was drained in theater. She has a homozygous c.4129G>T; p.(Glu1377*) variant.
P8 had a spontaneous transverse colon perforation at the age of 51 years followed by a second perforation of the small bowel 3 days postoperatively. She was found to have a c.8488del p.(Gln2830fs) variant in exon 25 and c.10459C>T p.(Gln3487*) variant in exon 31. She had a brother who died at the age of 38 years due to a jejunal perforation followed by a second perforation of the sigmoid colon three days later. He had a history of frequent shoulder dislocations and less frequent patellar dislocations as a child, significant joint hypermobility, and skin hyperextensibility. He had a rectal prolapse at the age of 36 years. Given this clinical history and the mode of inheritance of clEDS, a diagnosis of clEDS is likely, though unproven.
Of note, a sibling of P1 with a molecularly confirmed diagnosis of clEDS experienced a spontaneous large bowel perforation in middle age. Several years later she had a duodenal perforation and eventually was found to have small bowel diverticulosis.
So far, six patients with clEDS and tissue fragility have been reported in the literature, predominantly but not exclusively of the gastrointestinal tract: (1) perforation of colonic diverticulum, development of multiple abscesses requiring partial colectomy, complicated by second small bowel perforation in a 36-year-old man;10 (2) tracheal rupture possibly due to intubation in a 41-year-old woman;6 (3) esophageal rupture possibly due to ultrasound probe in a 57-year-old man[;9 (4) diverticular perforation sigmoid and duodenal perforation after ileus tube insertion in a 45 -year-old woman,13 (5) bowel perforation due to diverticulitis in an individual aged 48 years; and (6) death due to infection following bowel perforation of individual in mid-50s.8
All patients in our cohort and most patients reported in the literature with gastrointestinal complications were middle aged. The youngest patient with gastrointestinal complications was 36 years old;10 the oldest patient was 59 years old (P6). Tracheal rupture during intubation,6 esophageal rupture after use of ultrasound probe,9 and gastroscopy (P6) imply a degree of tissue fragility with complications resulting from invasive procedures. Most patients with clEDS in the literature who had large bowel rupture also had diverticulitis.8,10,13 P6 and the sibling of P1 had respectively pan-colonic diverticulosis and small bowel diverticulosis. It is unknown whether P7 and P8 and her brother had an underlying diverticulosis/diverticulitis. Small bowel perforations have been reported after ileus tube insertion.13 P8 and her brother and the sibling of P1 had a large and a small bowel perforation. P6 and P7 had exclusively small bowel perforations.
Diverticular disease has been noted in patients with clEDS (6/31 patients in the literature) and in our cohort two patients (P6, P17 [terminal ileum, colon]) had proven and one suspected colonic diverticulosis. In contrast to left-sided colonic diverticulosis, small bowel diverticulosis is rare with a prevalence rate of 0.2% to 1.3% at autopsy and 0.3% to 1.9% on small bowel studies.18 Small bowel diverticulosis has been reported in other hereditary connective tissue disorders such as Marfan syndrome.19 We speculate that patients with clEDS may be more prone to have structural defects along the walls of the gastrointestinal tract, which can predispose to diffuse diverticulosis, diverticulitis, and resulting bowel perforation as well as to perforation during invasive procedures and spontaneous perforation.
It is advised in the literature that intubation and endoscopic studies should be performed carefully to prevent rupture of trachea and esophagus.4 Based on the new information presented here, we would additionally suggest that there is a low threshold to refer patients with clEDS and gastrointestinal complaints to a gastroenterologist. It is important that patients are actively informed about reported gastrointestinal fragility and the possible link with diverticulosis and diverticulitis. We suggest caution when considering invasive gastrointestinal procedures and to avoid these interventions if possible. Furthermore, we suggest careful selection of analgesic medications in view of elevated risks of diverticulitis and diverticular bleeding in users of aspirin or nonsteroidal anti-inflammatory drugs (NSAIDs).20 Carrying a medic alert card with information about clEDS including reported gastrointestinal fragility is important.
Cardiovascular and other
D’hondt et al.21 reported 10/19 clEDS patients with hematomas in their systematic literature review of vascular phenotypes in nonvascular subtypes of EDS. In line with this, our cohort demonstrated easy bruising in 19/20 individuals and hematomas in 10/19. In P13, the only child in our cohort, a suspicion on nonaccidental injury had been raised previously because of excessive bruising. P17 developed a spontaneous left calf hematoma that had to be drained surgically and developed a right arm cephalic vein thrombosis and pulmonary embolism during admission for adrenal crisis. She was subsequently started on anticoagulant therapy and shortly after required a hospital admission for spontaneous subcutaneous hematoma of the lower half of the body, causing acute anemia and requiring blood transfusion. P15 required surgery for two separate incidences of spontaneous compartment syndrome in his right arm at the age of 30 and in the left arm at the age of 31, suggesting vascular fragility. Compartment syndrome has been reported to occur in vascular EDS as well.22
Of 31 patients in the clEDS literature, 5 had experienced frequent subconjunctival hemorrhages and 4/19 patients reported this in our cohort. Demirdas et al. reported a 58-year-old man with a thoracoabdominal aortic aneurysm and aneurysm of both common ileac artery and superior mesenteric artery.8 In addition, aneurysmal abdominal arteries were detected on postmortem of a patient who died in his mid-50s due to a bowel rupture. We did not identify any examples of vascular fragility leading to arterial aneurysm and/or dissection in our cohort, but the patients do not undergo routine vascular imaging. For medical professionals it would be important to know that arterial fragility has been observed but thus far does not seem to be a key feature of this condition.
Valvular abnormalities have been reported in individuals with TN-X deficiency.8,23 Demirdas et al. reported valvular abnormalities in 24% and recommended to perform an echocardiogram at first presentation. P8 reported mild aortic valve regurgitation at the age of 55 years. P6 was diagnosed with dilated cardiomyopathy at the age of 62. He did not have risk factors for this condition. P20 was diagnosed with a mild mitral valve prolapse at the age of 24 years.
Interestingly, at the age of 47 years P20 developed extensive surgical emphysema within the subcutaneous tissues of her face and computed tomography (CT) scans of the sinuses revealed a defect of the left nasal cartilages anteriorly allowing air to track into the soft tissues. It was felt most likely that vigorous nose blowing had been the cause of the emphysema. This may point again to tissue fragility in people with clEDS.
Skin
Three patients presented with mild atrophic scarring. It was noted that scarring was not to the extent usually seen in classical EDS (cEDS). Four patients have been described as having atrophic scarring in the clEDS literature.24 This could be due to subjective recording of atrophic scarring or may represent a subset of patients who are vulnerable to this scarring. Interestingly, murine studies suggest that TN-X is involved in the later stages of wound healing when matrix remodeling enhances biomechanical strength of the dermis, which could explain the mild skin fragility noted in these patients.25
Pregnancy
There were 19 pregnancies in our cohort. Five post- or peripartum hemorrhages were reported with three occurring in one patient (P7); five miscarriages were reported, one premature rupture of membranes (PROM) and one uterus prolapse. Egging et al.26 advise based on mouse models with TN-X deficiency to monitor clEDS patients closely during pregnancy, as they are likely to have weaker genitourinary (GU) connective tissue and could therefore be at risk of GU complications.4 We were able to identify 22 pregnancies in the literature with 6 reports of postpartum hemorrhage, as well as vaginal (2/22), rectal (6/22) and uterine prolapse (1/22); 3 miscarriages and 3 perineal tears; 1 case of PROM; and 1 report of pelvic instability. Given these data and the fact that the literature about pregnancy in clEDS is sparse, we advise gynecological follow-up during pregnancy and specialist delivery with careful handling as recommended in the literature.4
Musculoskeletal disability
In our cohort we observed significant variability in the severity of musculoskeletal symptoms and their effect on day-to-day function between the different probands and even within the same family. For example, P1 was referred by the rheumatologist and was in an assisted wheelchair since the age of 41 years due to her severe and painful foot deformities, joint dislocations, and fatigue. She is under the care of multiple medical specialists including a rheumatologist, orthopedic surgeon, gastroenterologist, and consultant in pain management. P2 was walking with two sticks since the age of approximately 32 years and used a mobility scooter since the age of 45 years because of joint pains, dislocations, and severe fatigue and is under the care of a rheumatologist. However, P4 was only diagnosed because his son was referred by the pediatrician because of significant joint hypermobility. He was noted to have a very stretchy skin and joint hypermobility. He does not have any complaints and is not requiring multidisciplinary input. P5 was diagnosed because of an impressive history of post-traumatic hematomas but currently does not have joint problems. Given the high frequency of musculoskeletal problems in individuals with clEDS (Supplementary Table 1), we propose that coordination of care for patients with clEDS lies with (pediatric) rheumatology and EDS services if available.
Electron microscopy and molecular analysis
Zweers et al.27 investigated five patients with clEDS and histological investigations reported “gross abnormalities of the elastic fibers and microfibrils in the dermis of these patients and reduced dermal collagen content.... At the ultrastructural level, irregular and immature elastin fibers and fibers devoid of microfibrils were observed.” These findings suggested that “both elastic fiber abnormalities and reduced collagen content contribute to the clinical features seen in patients with clEDS.” There were no consistent electron microscopy (EM) findings in the ten patients in our cohort who had EM performed. Five patients had no abnormalities and in two patients, collagen flowers were noted. These are seen in several other types of EDS as well, typically in classical EDS.16 Interestingly, one patient had some abnormal changes in the elastic fibers and another patient had some excess of elastic fibers.
clEDS is caused by homozygous or compound heterozygous pathogenic variants in the TNXB gene, which leads to nonsense-mediated mRNA decay of TNXB and result in complete absence of TN-X or in misfolding of the protein8. We noted considerable genetic heterogeneity in our cohort with 16 different pathogenic variants identified of which 11 have not been reported previously (Fig. 2). Pathogenic variants derived from gene conversion events with the TNXA pseudogene were most commonly identified, namely c.12174C>G; p.(Cys4058Trp) (14 alleles), pathogenic deletion including exon 35 (c.11381-?_11524+?del) (9 alleles), and c.11431-?_12725+?del (3 alleles). We assume that the exon 35 deletions with notation c.11381-?_11524+?del, detected via MLPA, and the c.11435_11524+30del variant, detected via NGS,8 are likely the same variant. The frequency of the exon 35 deletion and the p.(Cys4058Trp) variant are comparable in our patients and the patients reported by Demirdas et al.8 The exact pathogenic mechanism of the p.(Cys4058Trp) is currently unknown but the variant affects a cysteine residue that is predicted to form a disulfide bond, stabilizing tertiary protein structure.8 It seems, reviewing the published literature and the LOVD (www.lovd.nl), and HGMD (www.hgmd.cf.ac.uk) databases, that clEDS is often caused by gene conversion events and this is the most common recurrent mechanism of pathogenicity. No genotype–phenotype correlation was observed in our patients.
Conclusion
Currently, 31 patients with clEDS due to tenascin-X deficiency have been reported. Here we report on 20 patients who fulfill the new diagnostic criteria for clEDS and have a molecular diagnosis of clEDS. This increases the number of reported cases from 31 to 51 and broadens the phenotype of this condition, especially with the observation of tissue fragility of the gastrointestinal tract. We propose a predisposition to tissue fragility in patients with clEDS, in particular of the gastrointestinal tract and emphasize the importance of counseling with regard to the risk of gastrointestinal complications. We suggest caution when considering invasive gastrointestinal procedures and to avoid these interventions if possible. We recommend careful selection of analgesic medication in view of elevated risks of diverticulitis and diverticular bleeding in users of aspirin or NSAIDs. We advise carrying a medic alert card with information about clEDS including reported gastrointestinal fragility. We recommend following up these patients closely to develop better recommendations for management based on long-term outcome data for example by cardiovascular screening every 2–5 years and assessments of quality of life by questionnaires. Given the high frequency of musculoskeletal problems in individuals with clEDS (Supplementary Table 1), we propose that coordination of care and follow-up for patients with clEDS lie with (pediatric) rheumatology and if available with specialized Ehlers–Danlos services.
References
Malfait F. Vascular aspects of the Ehlers–Danlos syndromes. Matrix Biol. 2018;71-72:380–395.
Malfait F, Francomano C, Byers P, et al. The 2017 international classification of the Ehlers–Danlos syndromes. Am J Med Genet C Semin Med Genet. 2017;175:8–26.
Burch GH, Gong Y, Liu W, et al. Tenascin-X deficiency is associated with Ehlers–Danlos syndrome. Nat Genet. 1997;17:104–108.
Brady AF, Demirdas S, Fournel-Gigleux S, et al. The Ehlers–Danlos syndromes, rare types. Am J Med Genet C Semin Med Genet. 2017;175:70–115.
Valcourt U, Alcaraz LB, Exposito JY, et al. Tenascin-X: beyond the architectural function. Cell Adh Migr. 2015;9:154–165.
Besselink-Lobanova A, Maandag NJ, Voermans NC, et al. Trachea rupture in tenascin-X-deficient type Ehlers–Danlos syndrome. Anesthesiology. 2010;113:746–749.
Schalkwijk J, Zweers MC, Steijlen PM, et al. A recessive form of the Ehlers–Danlos syndrome caused by tenascin-X deficiency. N Engl J Med. 2001;345:1167–1175.
Demirdas S, Dulfer E, Robert L, et al. Recognizing the tenascin-X deficient type of Ehlers–Danlos syndrome: a cross-sectional study in 17 patients. Clin Genet. 2017;91:411–425.
Hendriks AG, Voermans NC, Schalkwijk J, et al. Well-defined clinical presentation of Ehlers–Danlos syndrome in patients with tenascin-X deficiency: a report of four cases. Clin Dysmorphol. 2012;21:15–18.
Lindor NM, Bristow J. Tenascin-X deficiency in autosomal recessive Ehlers–Danlos syndrome. Am J Med Genet A. 2005;135:75–80.
O’Connell M, Burrows NP, van Vlijmen-Willems MJ, et al. Tenascin-X deficiency and Ehlers–Danlos syndrome: a case report and review of the literature. Br J Dermatol. 2010;163:1340–1345.
Penisson-Besnier I, Allamand V, Beurrier P, et al. Compound heterozygous mutations of the TNXB gene cause primary myopathy. Neuromuscul Disord. 2013;23:664–669.
Sakiyama T, Kubo A, Sasaki T, et al. Recurrent gastrointestinal perforation in a patient with Ehlers–Danlos syndrome due to tenascin-X deficiency. J Dermatol. 2015;42:511–514.
Voermans NC, Jenniskens GJ, Hamel BC, et al. Ehlers–Danlos syndrome due to tenascin-X deficiency: muscle weakness and contractures support overlap with collagen VI myopathies. Am J Med Genet A. 2007;143A:2215–2219.
Voermans NC, van Alfen N, Pillen S, et al. Neuromuscular involvement in various types of Ehlers–Danlos syndrome. Ann Neurol. 2009;65:687–697.
Morissette R, Chen W, Perritt AF, et al. Broadening the spectrum of Ehlers Danlos syndrome in patients with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2015;100:E1143–E1152.
Angwin C, Ghali N, Baker D. et al. Electron Microscopy in the diagnosis of Ehlers–Danlos syndromes: correlation with clinical and genetic investigations. Br J Dermatol. 2020;182:698–707.
Karas L, Asif M, Chun V, Khan FA. Complicated small bowel diverticular disease: a case series. BMJ Case Rep. 2017;2017:bcr2017219699.
Robey BS, Peery AF, Dellon ES. Small bowel diverticulosis and jejunal perforation in Marfan syndrome. ACG Case Rep J. 2018;5:e5.
Strate LL, Liu YL, Huang ES, Giovannucci EL, Chan AT. Use of aspirin or nonsteroidal anti-inflammatory drugs increases risk for diverticulitis and diverticular bleeding. Gastroenterology. 2011;140:1427–1433.
D’Hondt S, Van Damme T, Malfait F. Vascular phenotypes in nonvascular subtypes of the Ehlers–Danlos syndrome: a systematic review. Genet Med. 2018;20:562–573.
Barajas BD, Sun A, Rimoin DL, Reinstein E. Recurrent compartment syndrome in a patient with clinical features of a connective tissue disorder. Am J Med Genet A. 2013;161:1442–1446.
Peeters AC, Kucharekova M, Timmermans J, et al. A clinical and cardiovascular survey of Ehlers–Danlos syndrome patients with complete deficiency of tenascin-X. Neth J Med. 2004;62:160–162.
Chen W, Perritt AF, Morissette R, et al. Ehlers–Danlos syndrome caused by biallelic TNXB variants in patients with congenital adrenal hyperplasia. Hum Mutat. 2016;37:893–897.
Egging D, van Vlijmen-Willems I, van Tongeren T, et al. Wound healing in tenascin-X deficient mice suggests that tenascin-X is involved in matrix maturation rather than matrix deposition. Connect Tissue Res. 2007;48:93–98.
Egging DF, van Vlijmen-Willems I, Choi J, et al. Analysis of obstetric complications and uterine connective tissue in tenascin-X-deficient humans and mice. Cell Tissue Res. 2008;332:523–532.
Zweers MC, van Vlijmen-Willems IM, van Kuppevelt TH, et al. Deficiency of tenascin-X causes abnormalities in dermal elastic fiber morphology. J Invest Dermatol. 2004;122:885–891.
Yates AD, Achuthan P, Akanni W, et al. Ensembl 2020. Nucleic Acids Res. 2020;48:D682–D688.
Acknowledgements
We thank the participants for their kind cooperation, and we are grateful to Dr Cristopher Record for his clinical involvement.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosure
The authors declare no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Green, C., Ghali, N., Akilapa, R. et al. Classical-like Ehlers–Danlos syndrome: a clinical description of 20 newly identified individuals with evidence of tissue fragility. Genet Med 22, 1576–1582 (2020). https://doi.org/10.1038/s41436-020-0850-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41436-020-0850-1
Keywords
This article is cited by
-
An exemplary model of genetic counselling for highly specialised services
Journal of Community Genetics (2023)
-
Detection of the Copy Number Variants of Genes in Patients with Familial Cardiac Diseases by Massively Parallel Sequencing
Molecular Diagnosis & Therapy (2023)