Abstract
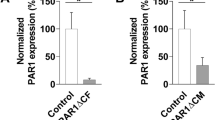
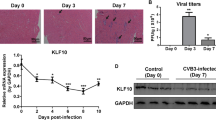
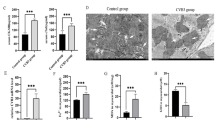
Group B coxsackieviruses (CVBs) are the main cause of virus-induced myocarditis. CVBs use coxsackievirus and adenovirus receptor (CAR) for infection and targeting CAR has been shown to ameliorate CVBs-induced myocarditis. Ligand-of-Numb protein X1 (LNX1) is an E3 ubiquitin ligase that was shown to interact with CAR. However, the precise effect of LNX1 on CAR and the roles of LNX1 on CVBs-induced myocarditis remain unknown. In the present study, we generated mice deficient in LNX1 in the heart and evaluated the symptoms of myocarditis after CVB3 infection. We also monitored the expression and ubiquitination of CAR in LNX1-deficient cardiomyocytes after CVBs infection. We found that CVBs infection decreased CAR expression while promoted the expression of LNX1. Mice with deficiency of LNX1 in the heart had normal myocardial development while had deteriorated myocarditis symptoms after CVB3 infection. In LNX1-deficient cardiomyocytes, decreased ubiquitination of CAR and upregulation of CAR were observed after CVB3 infection. In summary, LNX1 controls CVB3-induced myocarditis by regulating the expression of CAR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baughman KL. Diagnosis of myocarditis: death of Dallas criteria. Circulation. 2006;113:593–5.
Blauwet LA, Cooper LT. Myocarditis. Prog Cardiovasc Dis. 2010;52:274–88.
Bowles NE, Ni J, Kearney DL, Pauschinger M, Schultheiss HP, McCarthy R, et al. Detection of viruses in myocardial tissues by polymerase chain reaction. evidence of adenovirus as a common cause of myocarditis in children and adults. J Am Coll Cardiol. 2003;42:466–72.
Maginnis MS. Virus-receptor interactions: the key to cellular invasion. J Mol Biol. 2018;430:2590–611.
Kallewaard NL, Zhang L, Chen JW, Guttenberg M, Sanchez MD, Bergelson JM. Tissue-specific deletion of the coxsackievirus and adenovirus receptor protects mice from virus-induced pancreatitis and myocarditis. Cell Host Microbe. 2009;6:91–8.
Excoffon K. The coxsackievirus and adenovirus receptor: virological and biological beauty. FEBS Lett. 2020;594:1828–37.
Coyne CB, Bergelson JM. Virus-induced Abl and Fyn kinase signals permit coxsackievirus entry through epithelial tight junctions. Cell. 2006;124:119–31.
Zautner AE, Korner U, Henke A, Badorff C, Schmidtke M. Heparan sulfates and coxsackievirus-adenovirus receptor: each one mediates coxsackievirus B3 PD infection. J Virol. 2003;77:10071–7.
Nie J, McGill MA, Dermer M, Dho SE, Wolting CD, McGlade CJ. LNX functions as a RING type E3 ubiquitin ligase that targets the cell fate determinant Numb for ubiquitin-dependent degradation. EMBO J. 2002;21:93–102.
Sollerbrant K, Raschperger E, Mirza M, Engstrom U, Philipson L, Ljungdahl PO, et al. The Coxsackievirus and adenovirus receptor (CAR) forms a complex with the PDZ domain-containing protein ligand-of-numb protein-X (LNX). J Biol Chem. 2003;278:7439–44.
Zhang NH, Song LB, Wu XJ, Li RP, Zeng MS, Zhu XF, et al. Proteasome inhibitor MG-132 modifies coxsackie and adenovirus receptor expression in colon cancer cell line lovo. Cell Cycle. 2008;7:925–33.
Lei L, Bandola-Simon J, Roche PA. Ubiquitin-conjugating enzyme E2 D1 (Ube2D1) mediates lysine-independent ubiquitination of the E3 ubiquitin ligase March-I. J Biol Chem. 2018;293:3904–12.
Orthopoulos G, Triantafilou K, Triantafilou M. Coxsackie B viruses use multiple receptors to infect human cardiac cells. J Med Virol. 2004;74:291–9.
Pinkert S, Westermann D, Wang X, Klingel K, Dörner A, Savvatis K, et al. Prevention of cardiac dysfunction in acute coxsackievirus B3 cardiomyopathy by inducible expression of a soluble coxsackievirus-adenovirus receptor. Circulation. 2009;120:2358–66.
Pinkert S, Dieringer B, Klopfleisch R, Savvatis K, Van Linthout S, Pryshliak M, et al. Early treatment of coxsackievirus B3-infected animals with soluble coxsackievirus-adenovirus receptor inhibits development of chronic coxsackievirus B3 cardiomyopathy. Circ Heart Fail. 2019;12:e005250.
Lee DH, Goldberg AL. Proteasome inhibitors: valuable new tools for cell biologists. Trends Cell Biol. 1998;8:397–403.
Callis J. The ubiquitination machinery of the ubiquitin system. Arabidopsis Book. 2014;12:e0174.
Pickart CM. Mechanisms underlying ubiquitination. Annu Rev Biochem. 2001;70:503–33.
Metzger MB, Pruneda JN, Klevit RE, Weissman AM. RING-type E3 ligases: master manipulators of E2 ubiquitin-conjugating enzymes and ubiquitination. Biochim Biophys Acta. 2014;1843:47–60.
Mirza M, Hreinsson J, Strand ML, Hovatta O, Söder O, Philipson L, et al. Coxsackievirus and adenovirus receptor (CAR) is expressed in male germ cells and forms a complex with the differentiation factor JAM-C in mouse testis. Exp Cell Res. 2006;312:817–30.
Fu B, Xue W, Zhang H, Zhang R, Feldman K, Zhao Q, et al. MicroRNA-325-3p facilitates immune escape of Mycobacterium tuberculosis through targeting LNX1 via NEK6 accumulation to promote anti-apoptotic STAT3 signaling. mBio. 2020;11:e00557–20.
Author information
Authors and Affiliations
Contributions
TZ, CW, JW, ZZ, XW, and CS did the experiments, analyzed the data, wrote the paper; CS supervised the study; TZ and CS conceived the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, T., Wang, C., Wei, J. et al. Ligand-of-Numb protein X1 controls the coxsackievirus B3-induced myocarditis via regulating the stability of coxsackievirus and adenovirus receptor. Genes Immun 23, 42–46 (2022). https://doi.org/10.1038/s41435-022-00163-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41435-022-00163-x