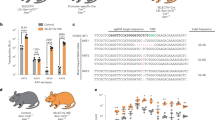
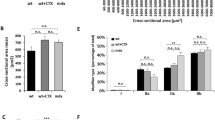
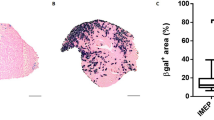
Abstract
The human musculature is a promising and pivotal target for human gene therapy, owing to numerous diseases that affect this tissue and that are often monogenic, making them amenable to treatment and potentially cure on the genetic level. Particularly attractive would be the possibility to deliver clinically relevant DNA to muscle tissue from a minimally invasive, intravenous vector delivery. To date, this aim has been approximated by the use of Adeno-associated viruses (AAV) of different serotypes (rh.74, 8, 9) that are effective, but unfortunately not specific to the muscle and hence not ideal for use in patients. Here, we have thus studied the muscle tropism and activity of another AAV serotype, AAVpo1, that was previously isolated from pigs and found to efficiently transduce muscle following direct intramuscular injection in mice. The new data reported here substantiate the usefulness of AAVpo1 for muscle gene therapies by showing, for the first time, its ability to robustly transduce all major muscle tissues, including heart and diaphragm, from peripheral infusion. Importantly, in stark contrast to AAV9 that forms the basis for ongoing clinical gene therapy trials in the muscle, AAVpo1 is nearly completely detargeted from the liver, making it a very attractive and potentially safer option.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Duan D. Systemic delivery of adeno-associated viral vectors. Curr Opin Virol. 2016;21:16–25.
Muller OJ, Katus HA, Bekeredjian R. Targeting the heart with gene therapy-optimized gene delivery methods. Cardiovasc Res. 2007;73:453–62.
Muller OJ, Leuchs B, Pleger ST, Grimm D, Franz WM, Katus HA, et al. Improved cardiac gene transfer by transcriptional and transductional targeting of adeno-associated viral vectors. Cardiovasc Res. 2006;70:70–8.
Bostick B, Ghosh A, Yue Y, Long C, Duan D. Systemic AAV-9 transduction in mice is influenced by animal age but not by the route of administration. Gene Ther. 2007;14:1605–9.
Yue Y, Ghosh A, Long C, Bostick B, Smith BF, Kornegay JN, et al. A single intravenous injection of adeno-associated virus serotype-9 leads to whole body skeletal muscle transduction in dogs. Mol Ther. 2008;16:1944–52.
Inagaki K, Fuess S, Storm TA, Gibson GA, McTiernan CF, Kay MA, et al. Robust systemic transduction with AAV9 vectors in mice: efficient global cardiac gene transfer superior to that of AAV8. Mol Ther. 2006;14:45–53.
Pacak CA, Mah CS, Thattaliyath BD, Conlon TJ, Lewis MA, Cloutier DE, et al. Recombinant adeno-associated virus serotype 9 leads to preferential cardiac transduction in vivo. Circ Res. 2006;99:e3–9.
Vandendriessche T, Thorrez L, Acosta-Sanchez A, Petrus I, Wang L, Ma L, et al. Efficacy and safety of adeno-associated viral vectors based on serotype 8 and 9 vs. lentiviral vectors for hemophilia B gene therapy. J Thromb Haemost. 2007;5:16–24.
Jones D. Duchenne muscular dystrophy awaits gene therapy. Nat Biotechnol. 2019;37:335–7.
Amoasii L, Long C, Li H, Mireault AA, Shelton JM, Sanchez-Ortiz E, et al. Single-cut genome editing restores dystrophin expression in a new mouse model of muscular dystrophy. Sci Transl Med. 2017;9:eaan8081. https://doi.org/10.1126/scitranslmed.aan8081.
Duan D. Systemic AAV micro-dystrophin gene therapy for duchenne muscular dystrophy. Mol Ther. 2018;26:2337–56.
Childers MK, Joubert R, Poulard K, Moal C, Grange RW, Doering JA, et al. Gene therapy prolongs survival and restores function in murine and canine models of myotubular myopathy. Sci Transl Med. 2014;6:220ra10.
Buj-Bello A, Fougerousse F, Schwab Y, Messaddeq N, Spehner D, Pierson CR, et al. AAV-mediated intramuscular delivery of myotubularin corrects the myotubular myopathy phenotype in targeted murine muscle and suggests a function in plasma membrane homeostasis. Hum Mol Genet. 2008;17:2132–43.
Elverman M, Goddard MA, Mack D, Snyder JM, Lawlor MW, Meng H, et al. Long-term effects of systemic gene therapy in a canine model of myotubular myopathy. Muscle Nerve. 2017;56:943–53.
Manno CS, Pierce GF, Arruda VR, Glader B, Ragni M, Rasko JJ, et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat Med. 2006;12:342–7.
Mingozzi F, High KA. Immune responses to AAV in clinical trials. Curr Gene Ther. 2007;7:316–24.
Mingozzi F, High KA. Therapeutic in vivo gene transfer for genetic disease using AAV: progress and challenges. Nat Rev Genet. 2011;12:341–55.
Mingozzi F, Meulenberg JJ, Hui DJ, Basner-Tschakarjan E, Hasbrouck NC, Edmonson SA, et al. AAV-1-mediated gene transfer to skeletal muscle in humans results in dose-dependent activation of capsid-specific T cells. Blood. 2009;114:2077–86.
Pien GC, Basner-Tschakarjan E, Hui DJ, Mentlik AN, Finn JD, Hasbrouck NC, et al. Capsid antigen presentation flags human hepatocytes for destruction after transduction by adeno-associated viral vectors. J Clin Investig. 2009;119:1688–95.
Martino AT, Basner-Tschakarjan E, Markusic DM, Finn JD, Hinderer C, Zhou S, et al. Engineered AAV vector minimizes in vivo targeting of transduced hepatocytes by capsid-specific CD8+ T cells. Blood. 2013;121:2224–33.
Sun B, Zhang H, Franco LM, Brown T, Bird A, Schneider A, et al. Correction of glycogen storage disease type II by an adeno-associated virus vector containing a muscle-specific promoter. Mol Ther. 2005;11:889–98.
Sun B, Zhang H, Franco LM, Young SP, Schneider A, Bird A, et al. Efficacy of an adeno-associated virus 8-pseudotyped vector in glycogen storage disease type II. Mol Ther. 2005;11:57–65.
Mah C, Pacak CA, Cresawn KO, Deruisseau LR, Germain S, Lewis MA, et al. Physiological correction of Pompe disease by systemic delivery of adeno-associated virus serotype 1 vectors. Mol Ther. 2007;15:501–7.
Han SO, Li S, Brooks ED, Masat E, Leborgne C, Banugaria S, et al. Enhanced efficacy from gene therapy in Pompe disease using coreceptor blockade. Hum Gene Ther. 2015;26:26–35.
Bello A, Tran K, Chand A, Doria M, Allocca M, Hildinger M, et al. Isolation and evaluation of novel adeno-associated virus sequences from porcine tissues. Gene Ther. 2009;16:1320–8.
Bello A, Chand A, Aviles J, Soule G, Auricchio A, Kobinger GP. Novel adeno-associated viruses derived from pig tissues transduce most major organs in mice. Sci Rep. 2014;4:6644.
Puppo A, Bello A, Manfredi A, Cesi G, Marrocco E, Della Corte M, et al. Recombinant vectors based on porcine adeno-associated viral serotypes transduce the murine and pig retina. PLoS One. 2013;8:e59025.
Zincarelli C, Soltys S, Rengo G, Rabinowitz JE. Analysis of AAV serotypes 1-9 mediated gene expression and tropism in mice after systemic injection. Mol Ther. 2008;16:1073–80.
Li X, Eastman EM, Schwartz RJ, Draghia-Akli R. Synthetic muscle promoters: activities exceeding naturally occurring regulatory sequences. Nat Biotechnol. 1999;17:241–5.
Rincon MY, Sarcar S, Danso-Abeam D, Keyaerts M, Matrai J, Samara-Kuko E, et al. Genome-wide computational analysis reveals cardiomyocyte-specific transcriptional Cis-regulatory motifs that enable efficient cardiac gene therapy. Mol Ther. 2015;23:43–52.
Sarcar S, Tulalamba W, Rincon MY, Tipanee J, Pham HQ, Evens H, et al. Next-generation muscle-directed gene therapy by in silico vector design. Nat Commun. 2019;10:492.
Miesbach W, Meijer K, Coppens M, Kampmann P, Klamroth R, Schutgens R, et al. Gene therapy with adeno-associated virus vector 5-human factor IX in adults with hemophilia B. Blood. 2018;131:1022–31.
Mingozzi F, Maus MV, Hui DJ, Sabatino DE, Murphy SL, Rasko JE, et al. CD8(+) T-cell responses to adeno-associated virus capsid in humans. Nat Med. 2007;13:419–22.
George LA, Sullivan SK, Giermasz A, Rasko JEJ, Samelson-Jones BJ, Ducore J, et al. Hemophilia B gene therapy with a high-specific-activity factor IX variant. N Engl J Med. 2017;377:2215–27.
Mendell JR, Al-Zaidy S, Shell R, Arnold WD, Rodino-Klapac LR, Prior TW, et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med. 2017;377:1713–22.
Nathwani AC, Tuddenham EG, Rangarajan S, Rosales C, McIntosh J, Linch DC, et al. Adenovirus-associated virus vector-mediated gene transfer in hemophilia B. N Engl J Med. 2011;365:2357–65.
Grimm D, Zolotukhin S. E pluribus unum: 50 years of research, millions of viruses, and one goal-tailored acceleration of AAV evolution. Mol Ther. 2015;23:1819–31.
Weinmann J, Grimm D. Next-generation AAV vectors for clinical use: an ever-accelerating race. Virus Genes. 2017;53:707–13.
Grimm D, Lee JS, Wang L, Desai T, Akache B, Storm TA, et al. In vitro and in vivo gene therapy vector evolution via multispecies interbreeding and retargeting of adeno-associated viruses. J Virol. 2008;82:5887–911.
Herrmann AK, Bender C, Kienle E, Grosse S, El Andari J, Botta J, et al. A robust and all-inclusive pipeline for shuffling of adeno-associated viruses. ACS Synth Biol. 2019;8:194–206.
Zinn E, Pacouret S, Khaychuk V, Turunen HT, Carvalho LS, Andres-Mateos E, et al. In silico reconstruction of the viral evolutionary lineage yields a potent gene therapy vector. Cell Rep. 2015;12:1056–68.
Fakhiri J, Schneider MA, Puschhof J, Stanifer M, Schildgen V, Holderbach S, et al. Novel chimeric gene therapy vectors based on adeno-associated virus and four different mammalian bocaviruses. Mol Ther Methods Clin Dev. 2019;12:202–22.
Acknowledgements
WT, QHP, JEA, TV, MKC, and DG are very grateful for funding and other support from the MYOCURE project. MYOCURE has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement no. 667751. DG is thankful for support by the German Center for Infection Research (DZIF, BMBF; TTU-HIV 04.803 and TTU-HIV 04.815). DG acknowledges additional funding by the German Research Foundation (DFG) through the Cluster of Excellence CellNetworks (EXC81) and the Collaborative Research Centers SFB1129 (Projektnummer 240245660) and TRR179 (Projektnummer 272983813). TV and MKC obtained funding from the Fonds Wetenschappelijk Onderzoek (FWO), VUB Industrieel Onderzoeksfonds (IOF), Koning Boudewijn Stichting (Creemers-Opdebeek) and Association Belge contre les Maladies Neuromusculaires (ABMM). We are grateful to Alexander Bello and Gary Kobinger for initially supplying the po1 sequence. The authors thank Julia Fakhiri for critical reading of the manuscript as well as Ermira Samara for her help with AAV vector production.
Author information
Authors and Affiliations
Contributions
TV, MKC, and DG conceived and designed the experiments. WT, JW, QHP, and JEA generated constructs and performed experiments. WT, JEA, TV, MKC, and DG wrote the manuscript. All authors read the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
DG is a co-founder and shareholder of AaviGen GmbH. JW is currently an employee of Boehringer Ingelheim Pharma GmbH & Co. KG. All other authors declare that they have no conflict of interest.
Ethical approval
All animal procedures were approved by the institutional animal ethics committee of the Free University of Brussels (VUB) (Brussels, Belgium). Husbandry was carried out in individually ventilated Thoren cages that contained Hygienic Animal Bedding (Lignocel). Temperature was maintained at ~21 °C with 50–60% humidity. Animals were fed SsniFF laboratory animal food (ABEDD Vertriebs GmbH, Vienna, Austria) ad libitum.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tulalamba, W., Weinmann, J., Pham, Q.H. et al. Distinct transduction of muscle tissue in mice after systemic delivery of AAVpo1 vectors. Gene Ther 27, 170–179 (2020). https://doi.org/10.1038/s41434-019-0106-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-019-0106-3