Abstract
Background
The Royal College of Ophthalmologists has produced guidelines for screening for hydroxychloroquine retinopathy. New imaging modalities had suggested an increased prevalence of retinopathy compared with previous reports. The aim of this study is to identify the real-life prevalence of hydroxychloroquine retinopathy in patients attending Manchester Royal Eye Hospital screening service over a 2-year period using The RCOphth diagnostic criteria.
Methods
Data were collected prospectively from all patients attending Manchester Royal Eye Hospital hydroxychloroquine screening service over the audit period. Results of Humphrey visual field tests, spectral-domain optical coherence tomography and fundus autofluorescence were collected as well as data on dose, indication, duration of treatment, and additional risk factors. Cases were identified as having definite, possible, or no retinopathy based on the 2018 RCOphth criteria. The data are not publicly available due to information that could compromise research participant privacy and confidentiality but are available upon request from the corresponding author.
Results
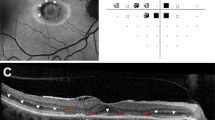
910 patients attended for screening. 566 were identified as being at risk of retinopathy (543 had been on treatment >5 years, 10 had renal impairment, 12 were on doses of >5 mg/kg/day, and one was concurrently on tamoxifen). The prevalence of HCQ retinopathy was 10/910 (1.09%) of all those screened, and 1.76% of those at risk (10/566). Six patients of those deemed at risk were identified as having definite hydroxychloroquine retinopathy, while four had possible retinopathy.
Conclusions
Our results show a prevalence of retinopathy largely consistent with reports from regional audits yet reveal a far lower estimate compared to previously reported figures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The data are not publicly available due to information that could compromise research participant privacy and confidentiality but are available upon request from the corresponding author.
References
Ben-Zvi I, Kivity S, Langevitz P, Shoenfeld Y. Hydroxychloroquine: from malaria to autoimmunity. Clin Rev Allergy Immunol. 2012;42:145–53.
National Guideline Centre (UK). Rheumatoid arthritis in adults: diagnosis and management. London: National Institute for Health and Care Excellence (UK); 2018.
Beksac MS, Donmez HG. Impact of hydroxychloroquine on the gestational outcomes of pregnant women with immune system problems that necessitate the use of the drug. J Obstet Gynaecol Res. 2021;47:570–5.
Melles RB, Marmor MF. Pericentral Retinopathy and Racial Differences in Hydroxychloroquine Toxicity. Ophthalmology. 2015;122:110–6.
Pasadhika S, Fishman GA, Choi D, Shahidi M. Selective thinning of the perifoveal inner retina as an early sign of hydroxychloroquine retinal toxicity. Eye. 2010;24:756–63.
Pasadhika S, Fishman GA. Effects of chronic exposure to hydroxychloroquine or chloroquine on inner retinal structures. Eye. 2010;24:340–6.
Ramsey MS, Fine BS. Chloroquine Toxicity in the Human Eye: Histopathologic Observations by Electron Microscopy. Am J Ophthalmol. 1972;73:229–35.
Mavrikakis I, Sfikakis PP, Mavrikakis E, Rougas K, Nikolaou A, Kostopoulos C, et al. The incidence of irreversible retinal toxicity in patients treated with hydroxychloroquine. Ophthalmology. 2003;110:1321–6.
Melles RB, Marmor MF. The Risk of Toxic Retinopathy in Patients on Long-term Hydroxychloroquine Therapy. JAMA Ophthalmol. 2014;132:1453.
Lee DH, Melles RB, Joe SG, Lee JY, Kim J-G, Lee C-K, et al. Pericentral Hydroxychloroquine Retinopathy in Korean Patients. Ophthalmology. 2015;122:1252–6.
Ahn SJ, Joung J, Lim HW, Lee BR. Optical Coherence Tomography Protocols for Screening of Hydroxychloroquine Retinopathy in Asian Patients. Am J Ophthalmol. 2017;184:11–8.
Dettoraki M, Moschos MM. The role of multifocal electroretinography in the assessment of drug-induced retinopathy: a review of the literature. Ophthalmic Res. 2016;56:169–77.
Marmor MF, Kellner U, Lai TYY, Melles RB, Mieler WF. Recommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy (2016 Revision). Ophthalmology. 2016;123:1386–94.
Yusuf IH, Foot B, Galloway J, Ardern-Jones MR, Watson S-L, Yelf C, et al. The Royal College of Ophthalmologists recommendations on screening for hydroxychloroquine and chloroquine users in the United Kingdom: executive summary. Eye. 2018;32:1168–73.
Marshall E, Robertson M, Kam S, Penwarden A, Riga P, Davies N. Correction: prevalence of hydroxychloroquine retinopathy using 2018 Royal College of Ophthalmologists diagnostic criteria. Eye. 2021;35:358–358.
Gobbett A, Kotagiri A, Bracewell C, Smith J. Two years’ experience of screening for hydroxychloroquine retinopathy. Eye. 2021;35:1171–7.
Yusuf IH, Sharma S, Luqmani R, Downes SM. Hydroxychloroquine retinopathy. Eye. 2017;31:828–45.
Schrezenmeier E, Dörner T. Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol. 2020;16:155–66.
Ledingham J, Gullick N, Irving K, Gorodkin R, Aris M, Burke J, et al. BSR and BHPR guideline for the prescription and monitoring of non-biologic disease-modifying anti-rheumatic drugs. Rheumatology. 2017;56:2257–2257.
Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin PE, Ksiazek TG, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69.
Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020;71:732–9.
Wolfe F, Marmor MF. Rates and predictors of hydroxychloroquine retinal toxicity in patients with rheumatoid arthritis and systemic lupus erythematosus. Arthritis Care Res. 2010;62:775–84.
della Porta A, Bornstein K, Coye A, Montrief T, Long B, Parris MA. Acute chloroquine and hydroxychloroquine toxicity: a review for emergency clinicians. Am J Emerg Med. 2020;38:2209–17.
Eo D, Lee MG, Ham D-I, Kang SW, Lee J, Cha HS, et al. Frequency and Clinical Characteristics of Hydroxychloroquine Retinopathy in Korean Patients with Rheumatologic Diseases. J Korean Med Sci. 2017;32:522.
Manoj M, Sahoo RR, Singh A, Hazarika K, Bafna P, Kaur A, et al. Prevalence of hydroxychloroquine retinopathy with long-term use in a cohort of Indian patients with rheumatic diseases. Rheumatol Int. 2021;41:929–37.
al Adel F, Shoughy S, Tabbara K. Hydroxychloroquine dosing and toxicity: a real-world experience in Saudi Arabia of 63 patients. Saudi J Ophthalmol. 2020;34:151.
Fung AT, Lu V, Mack HG. Screening for hydroxychloroquine retinopathy in Australia. Med J Aust. 2021;215:434–434.
Yusuf IH, Foot B, Lotery AJ. The Royal College of Ophthalmologists recommendations on monitoring for hydroxychloroquine and chloroquine users in the United Kingdom (2020 revision): executive summary. Eye. 2021;35:1532–7.
Dadhaniya NV, Sood I, Patil A, Mallaiah U, Upadhyaya S, Handa R, et al. Screening for Hydroxychloroquine Retinal Toxicity in Indian Patients. J Clin Rheumatol. 2021;27:e395–e398.
Author information
Authors and Affiliations
Contributions
RA designed and registered the audit, interpreted the results, and drafted the paper. NB collected the data prospectively and revised and edited the paper. HS supervised the work, reviewed the data and study protocol, and revised the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alieldin, R.A., Boonarpha, N. & Saedon, H. Outcomes of screening for hydroxychloroquine retinopathy at the Manchester Royal Eye Hospital: 2 years’ audit. Eye 37, 1410–1415 (2023). https://doi.org/10.1038/s41433-022-02159-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-02159-3
This article is cited by
-
Hydroxychloroquine
Reactions Weekly (2023)