Abstract
Background
Ocular surface squamous neoplasia (OSSN) is a relatively rare disease with a low mortality and highly variable incidence. Despite a high incidence of OSSN in the Southern hemisphere, there is limited epidemiological data for New Zealand. The current study aims to assess the incidence, demographics, and histological grade of OSSN in the Waikato region of New Zealand, home to ~10% of the population of New Zealand.
Methods
Non-interventional retrospective cohort study. All conjunctival biopsy histology reports from 2010 to 2019 in the Waikato region of New Zealand were analysed. Age, sex, and ethnicity were analysed and the incidence of OSSN was calculated. Main outcome measures included histological grade, rate of recurrence, and incidence of OSSN.
Results
A total of 386 patients underwent conjunctival biopsy with histology during the study period. Eighty-three lesions (22%) involving 80 patients (21%) were reported positive for OSSN. Patients with OSSN had a mean age of 68.9 years (SD = 13.2), were predominantly male (76%), and of New Zealand-European ethnicity (53%). Conjunctival intraepithelial neoplasia-1 (30%) was the most frequent diagnosis. Three patients (4%) had recurrent lesions requiring repeat biopsy. The peak annual OSSN incidence rate was 3.81/100,000 population in 2019. The overall ten-year incidence was 2.13/100,000 population/year.
Conclusion
This is the largest study to investigate OSSN incidence in New Zealand. The incidence rate of OSSN is one of the highest rates reported in the literature.
Similar content being viewed by others
Introduction
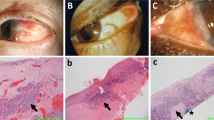
Ocular surface squamous neoplasia (OSSN) constitutes a spectrum of epithelial squamous malignancies of the cornea and conjunctiva [1]. Lesions can be pre-invasive or invasive, depending on the extent of dysplastic invasion of the conjunctival epithelium and surrounding structures. Preinvasive lesion subtypes include conjunctival intraepithelial neoplasia (CIN)-1 with mild dysplasia confined to the lower third, CIN2 with moderate dysplasia extending into the middle third, or CIN3 with severe dysplasia extending into the upper third of the conjunctival epithelium. Full thickness dysplasia of the epithelium is called carcinoma-in-situ (CIS). Invasive squamous cell carcinoma (SCC) lesions show infiltrating cells penetrating the epithelial basement membrane and spread into the conjunctiva stroma and surrounding structures [2, 3]. OSSN is a relatively rare disease with a low mortality and variable incidence according to geographic location [3].
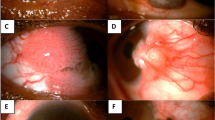
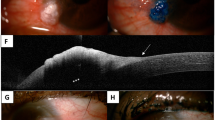
The risk of visual loss, local invasion and metastasis makes timely, accurate diagnosis and effective treatment of OSSN critical [3,4,5]. Clinical presentation includes elevated lesions in the inter-palpebral region involving the cornea and/or bulbar conjunctiva [6]. Macroscopically lesions are described as leukoplakic, gelatinous, papilliform, nodular, or diffuse with characteristic corkscrew shaped blood vessels [3]. Risk factors for OSSN include male sex, age in seventh decade, Caucasian ethnicity and ultraviolet (UV) light exposure [3].
Management options for OSSN include surgical excision and/or topical therapy. Traditional management of OSSN involved surgical excision using a no-touch technique [7], however, incomplete excision was associated with recurrence rates as high as 56% using excision alone [8, 9]. Topical chemotherapeutic agents such as interferon-a2b [10, 11], Mitomycin-C [12, 13], and 5-fluorouracil [14, 15] are used increasingly as alternative or adjuvant treatment to surgical excision and reports suggest this may significantly reduce the rate of recurrence [16,17,18,19].
There is limited epidemiological data for patients with OSSN in New Zealand [20], a country with high levels of sun exposure and high mortality rates of UV-related malignancies [21]. The Waikato region is the fourth largest region by land area in New Zealand. This region is home to over 400,000 people, or 10% of the total population of New Zealand. Approximately 60% of the Waikato population live outside of the main urban centres and spend large amounts of time outdoors with extended periods of sun exposure [22]. The aim of this study is to investigate the incidence of histology-confirmed OSSN cases over a ten-year period within the Waikato region of New Zealand.
Materials and methods
The current study adhered to the tenets of the Declaration of Helsinki [23] and met the criteria for exemption from formal review by the New Zealand Health and Disability Ethics Committee guidelines [24].
Operating theatre records from all public and private hospitals in the Waikato region that complete ophthalmic surgery were reviewed. Patients who underwent excision biopsy of conjunctival lesions with histological grading of the specimen from 2010 to 2019 were included for analysis. Histology reports from each specimen were reviewed for the presence and grade of OSSN. If more than one specimen was excised from the same eye at any time point then only the more severe histological diagnosis was included. Any specimen removed from the contralateral eye was considered to be a separate occurrence.
Demographic data included age at the time of surgery, sex and ethnicity. Patients of multiple ethnicities could select more than one option for their ethnicity. Clinical data included presence and grade of dysplasia and incidence of recurrence. Recurrence was defined as a second positive biopsy in the same eye at least three months following an initial excision biopsy with reported clear margins and after the initial biopsy site had healed.
The primary outcome measure was the number of patients with a histology positive diagnosis of OSSN, defined as histologically confirmed disease occurring for the first time in a patient. Secondary outcome measures included the incidence of recurrence and patient demographic variables for those with primary or recurrent OSSN.
The number of conjunctival biopsies performed nationally in the public sector from 2009 to 2018 were obtained from New Zealand’s Ministry of Health. Information obtained included patient sex, ethnicity and location of surgery based on the patient’s domiciled district health board. National data for the number of conjunctival excision biopsies was analysed to provide an estimate of disease prevalence nationwide over a ten-year period.
All data analysis was completed in R Version 3.5.2 (R Foundation for Statistical Computing, Vienna, Austria). Regional incidence rates of OSSN were calculated as the number of new cases per year per 100,000 population, estimated using values from the New Zealand 2013 National Census for years 2010–2014 and 2018 National Census for years 2015–2019 [25].
Results
A total of 386 patients had surgical excision of conjunctival lesions with histology in the Waikato during the study period. Eighty-three lesions (22%) from 80 patients (21%) were reported positive for OSSN. Patients with histology-confirmed OSSN had a mean age of 68.9 years (SD 13.2 years) and were predominantly male (n = 61, 76%). Ethnicities of these 80 patients included New Zealand-European (n = 42, 53%), Other/Not stated (n = 31, 39%), Maori (n = 5, 6%), Asian (n = 1, 1%) and Pacific Peoples (n = 1, 1%).
The annual incidence of OSSN is illustrated in Fig. 1. The highest single annual incidence rate was 3.81/100,000 population in 2019 with a ten-year average incidence rate of 2.13/100,000 population.
Histology confirmed CIN1 in 24 (30%), CIN2 in 19 (24%), CIN3 in 13 (16%), CIS in 17 (21%) and invasive SCC in 7 (9%) excised lesions. The mean patient age for each OSSN grade was 63 years (SD 15.8) for CIN1, 67 years (SD 12.9) for CIN2, 75 years (SD 9.8) for CIN3, 73 years (SD 10.4) for CIS and 74 years (SD 6.7) for invasive SCC.
Recurrence was detected in 3 (4%) patients and in all cases excision alone was the only initial treatment provided for the primary lesion with no adjuvant cryotherapy or Mitomycin-C. Of the cases with recurrence, 2 (67%) patients were female and 3 (100%) patients were of New Zealand-European ethnicity. Histological grades of the primary lesions for patients that presented with recurrent disease were CIN1, CIN2 and CIN2. Histological grades of the recurrent lesions were CIN2, CIN2 and CIS, respectively. The shortest time period to positive repeat biopsy was four months.
New Zealand national public-funded surgery data document a total of 8222 conjunctival biopsies completed over the ten-year study duration with 839 (10%) completed in the Waikato region. National data confirm a male predominance (n = 4786, 58%) for conjunctival lesions requiring excision. Patients were mostly of New Zealand-European ethnicity (n = 4386, 53%). The demographics of patients in the Waikato region were similar to those in the national surgery data, as outlined in Table 1.
Discussion
This is the largest study to evaluate patient demographics of histology-confirmed OSSN in New Zealand. In ten years, there were 80 patients newly diagnosed with histology-confirmed OSSN in the Waikato region. The patients were mostly male of Caucasian New Zealand-European ethnicity (53%) and with a mean age of 68.9 years. Globally two patterns of OSSN are reported, Caucasian males in their sixties with cumulative UV light exposure [26, 27] and younger immunocompromised males and females [28, 29]. The current study cohort predominantly fits within the former pattern.
This study identified a peak annual incidence of 3.81/100,000 population in 2019 and a ten year average of 2.13/100,000 population, one of the highest reported incidence rates of OSSN globally. An increased number of cases was identified in 2019. A contributing factor may be the addition of a second corneal subspecialist in the region thereby changing the threshold for adding patients to the surgical waitlist. Global annual incidence rates of OSSN vary from 2.8/100,000 in Brisbane, Australia [1], 0.92/100,000 in Malawi [29], 0.3-0.61/100,000 in the United States [30, 31], 0.064/100,000 in Canada [32], to 0.053/100,000 in the United Kingdom [33]. New Zealand and Australia have comparable UV indices and comparably high rates of UV-related melanoma cases compared to countries in the northern hemisphere [21, 34]. The higher incidence rate in the southern hemisphere is potentially related to the higher UV index [3, 35].
All histological grades of OSSN were identified during the ten-year study period, with CIN1 being the most frequently reported grade. Patients with CIN1 and CIN2 were, on average, 6–10 years younger than patients with CIN3, CIS or invasive SCC. Patients with CIN3, CIS and invasive SCC were of similar ages. This is in contrast to a previous study reporting the average age of CIS patients to be 5–9 years younger than those with invasive SCC [9]. The reason for the relationship between age at presentation and grade remains unclear. It is possible that, in some cases, low-grade lesions may have been present for some time prior to presentation. Slow transformation to higher grade lesions would be consistent with previous reports that up to 9.8% of all pterygia have dysplasia on histology [36].
Recurrence of OSSN was noted in 4% of cases in the current study. The rate of recurrence is low compared to other studies reporting rates as high as 56% with excision alone [8, 9]. The ten-year duration of the current study increased the likelihood of identifying recurrent cases. Other studies report recurrence rates ranging from 13 to 56% [3, 8, 9, 37]. In general, longer follow up periods are associated with higher recurrence rates [8]. Risk factors for recurrence include higher initial histological grade of the primary lesion [5, 30], incomplete surgical excisions [38], no adjuvant topical treatment [12, 27, 39], or short duration of follow up. In this cohort, OSSN recurrence was not associated with a higher pathologic grade or postoperative Mitomycin-C use; however, due to the low numbers of recurrent lesions in the study cohort it is difficult to draw any firm conclusions regarding the risk of recurrence.
The intervention rate for conjunctival lesion excisions performed nationally was similar to the intervention rate in the Waikato region over the duration of the current study. The national dataset included all excised conjunctival lesions, including pterygia. Similar to OSSN, pterygium risk factors include older age, male gender, outdoors occupation, and residing in rural environments [40]. UV indices in New Zealand are relatively similar nationwide, with marginally higher UV indices in the northern parts of the country [41]. These data suggest the results of the current study could be reasonably extrapolated to other parts of New Zealand. The national dataset did not include clinical or histological information, so it is not possible to test this assumption.
Little is known about the incidence rate of OSSN in New Zealand. A previous audit of public-funded conjunctival biopsies performed in Auckland, New Zealand over a six year period identified 62 (47%) cases of OSSN out of 132 conjunctival specimens from 115 patients. Four patients required an exenteration, 27 biopsies involved the margins and 12 patients required more than one surgical procedure. The current study was performed over a ten-year period in the Waikato region, with nearly one-third the population of Auckland. Histology reports were reviewed for all conjunctival lesions excised at either the public-funded hospital or at either of the two regional private Ophthalmology practices. This study identified 83 (22%) cases of OSSN in 80 patients out of 386 specimens over a ten-year period. These two studies provide evidence for a high incidence rate of OSSN in New Zealand [20].
There are some limitations in this retrospective audit of histology reports. The retrospective study design may underestimate the true incidence as not all conjunctival lesion excisions were sent for histology analysis. A prospective audit is required where all excised lesions are sent for histology analysis. Additional OSSN risk factors, including immunocompromised status or allergic eye disease, were not reviewed. Not all clinical notes were complete and legible so it is possible that additional cases may have been missed despite the best efforts of the investigators.
This is the first published study to analyse the demographics and histology of OSSN over a ten-year period in New Zealand’s Waikato region. This study includes all patients managed with surgical excision of conjunctival lesions within the Waikato region during the study period. The peak annual incidence of 3.81/100,000 population is the highest reported rate for OSSN as far as the authors are aware. Ophthalmologists should maintain a high level of suspicion for OSSN in conjunctival and corneal lesions. Routine histological analysis of all conjunctival specimens is important to accurately identify, diagnose and treat OSSN.
Summary
What was known before
-
Ocular surface squamous neoplasia (OSSN) is the most common ocular malignancy.
-
OSSN has variable incidence according to geographic location.
What this study adds
-
This is the first published study to analyse the demographics and histology of OSSN over a ten-year period in New Zealand.
-
The peak annual incidence of 3.81/100,000 population is the highest reported rate for OSSN as far as the authors are aware.
References
Lee GA, Hirst LW. Incidence of ocular surface epithelial dysplasia in metropolitan Brisbane. A 10-year survey. Arch Ophthalmol. 1992;110:525–7.
Lee GA, Hirst LW. Retrospective study of ocular surface squamous neoplasia. Aust NZ J Ophthalmol. 1997;25:269–76.
Basti S, Macsai MS. Ocular surface squamous neoplasia: a review. Cornea. 2003;22:687–704.
Kaliki S, Jajapuram SD, Maniar A, Taneja S, Mishra DK. Ocular surface squamous neoplasia with intraocular tumour extension: a study of 23 patients. Eye. 2020;34:319–26.
Murillo JC, Galor A, Wu MC, Kye NK, Wong J, Ahmed IO, et al. Intracorneal and intraocular invasion of ocular surface squamous neoplasia after intraocular surgery: report of two cases and review of the literature. Ocul Oncol Pathol. 2017;3:66–72.
Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39:429–50.
Shields JA, Shields CL, De, Potter P. Surgical management of conjunctival tumors. The 1994 Lynn B. McMahan Lecture. Arch Ophthalmol. 1997;115:808–15.
Tabin G, Levin S, Snibson G, Loughnan M, Taylor H. Late recurrences and the necessity for long-term follow-up in corneal and conjunctival intraepithelial neoplasia. Ophthalmology. 1997;104:485–92.
Erie JC, Campbell RJ, Liesegang TJ. Conjunctival and corneal intraepithelial and invasive neoplasia. Ophthalmology. 1986;93:176–83.
Karp CL, Galor A, Chhabra S, et al. Subconjunctival/perilesional recombinant interferon alpha2b for ocular surface squamous neoplasia: a 10-year review. Ophthalmology. 2010;117:2241–6.
Galor A, Karp CL, Chhabra S, Barnes S, Alfonso EC. Topical interferon alpha 2b eye-drops for treatment of ocular surface squamous neoplasia: a dose comparison study. Br J Ophthalmol. 2010;94:551–4.
Birkholz ES, Goins KM, Sutphin JE, Kitzmann AS, Wagoner MD. Treatment of ocular surface squamous cell intraepithelial neoplasia with and without mitomycin C. Cornea. 2011;30:37–41.
Gupta M, Muecke J. Treatment of ocular surface squamous neoplasia with mitomycin C. Br. J Ophthalmol. 2010;94:555–8.
Joag MG, Sise A, Murillo JC, Sayed-Ahmed IO, Wong JR, Mercado C, et al. Topical 5-fluorouracil 1% as primary treatment for ocular surface squamous neoplasia. Ophthalmology. 2016;123:1442–8.
Rudkin AK, Muecke JS. Adjuvant 5-fluorouracil in the treatment of localised ocular surface squamous neoplasia. Br J Ophthalmol. 2011;95:947–50.
Bahrami B, Greenwell T, Muecke JS. Long-term outcomes after adjunctive topical 5-flurouracil or mitomycin C for the treatment of surgically excised, localized ocular surface squamous neoplasia. Clin Exp Ophthalmol. 2014;42:317–22.
Nanji AA, Moon CS, Galor A, Sein J, Oellers P, Karp CL. Surgical versus medical treatment of ocular surface squamous neoplasia: a comparison of recurrences and complications. Ophthalmology. 2014;121:994–1000.
Adler E, Turner JR, Stone DU. Ocular surface squamous neoplasia: a survey of changes in the standard of care from 2003 to 2012. Cornea. 2013;32:1558–61.
Venkateswaran N, Mercado C, Galor A, Karp CL. Comparison of topical 5-fluorouracil and interferon Alfa-2b as primary treatment modalities for ocular surface squamous neoplasia. Am J Ophthalmol. 2019;199:216–22.
Mathew A, Stumpf T, McGhee C. Impression cytology: implications for ocular surface squamous neoplasia. Br J Ophthalmol. 2008;92:157–8.
Bell KJL, Cust AE. Beyond country-specific incidence and mortality: the global burden of melanoma. Br J Dermatol 2018;178:315–6.
Waikato District Health Board. Mental Health & Addictions Strategy 2016-21. 2016. Available at: https://www.waikatodhb.health.nz/assets/Docs/About-Us/Key-Publications/Strategies/76275ab51b/MHAS-Strategic-Plan-2016.pdf.
New Zealand Ministry of Health. Ethical Guidelines for Observational Studies: Observational research, audits and related activities. 2012.
New Zealand Ministry of Health. Standard Operating Procedures for Health and Disability Ethics Committees. In: Ministry of Health, ed. Standard Operating Procedures for Health and Disability Ethics Committees. Wellington, NZ: New Zealand Ministry of Health; 2014.
Stats N. Z. New Zealand Census. Stats NZ. Available at: https://www.stats.govt.nz/topics/census.
Yousef YA, Finger PT. Squamous carcinoma and dysplasia of the conjunctiva and cornea: an analysis of 101 cases. Ophthalmology. 2012;119:233–40.
Maudgil A, Patel T, Rundle P, Rennie IG, Mudhar HS. Ocular surface squamous neoplasia: analysis of 78 cases from a UK ocular oncology centre. Br J Ophthalmol. 2013;97:1520–4.
Gichuhi S, Sagoo MS, Weiss HA, Burton MJ. Epidemiology of ocular surface squamous neoplasia in Africa. Trop Med Int Health. 2013;18:1424–43.
Tiong T, Borooah S, Msosa J, Dean W, Smith C, Kambewa E, et al. Clinicopathological review of ocular surface squamous neoplasia in Malawi. Br J Ophthalmol. 2013;97:961–4.
Sun EC, Fears TR, Goedert JJ. Epidemiology of squamous cell conjunctival cancer. Cancer Epidemiol Biomark Prev. 1997;6:73–77.
Dalvin LA, Salomão DR, Patel SV. Population-based incidence of conjunctival tumours in Olmsted County. Minn Br J Ophthalmol. 2018;102:1728–34.
Darwich R, Ghazawi FM, Le M, Rahme E, Alghazawi N, Zubarev A, et al. Epidemiology of invasive ocular surface squamous neoplasia in Canada during 1992-2010. Br J Ophthalmol. 2020;104:1368–72.
Kiire CA, Stewart RMK, Srinivasan S, Heimann H, Kaye SB, Dhillon B. A prospective study of the incidence, associations and outcomes of ocular surface squamous neoplasia in the United Kingdom. Eye. 2019;33:283–94.
Whiteman DC, Green AC, Olsen CM. The Growing Burden of Invasive Melanoma: Projections of Incidence Rates and Numbers of New Cases in Six Susceptible Populations through 2031. J Invest Dermatol. 2016;136:1161–71.
Fioletov V, Kerr JB, Fergusson A. The UV index: definition, distribution and factors affecting it. Can J Public Health. 2010;101:I5–9.
Hirst LW, Axelsen RA, Schwab I. Pterygium and associated ocular surface squamous neoplasia. Arch Ophthalmol. 2009;127:31–32.
Pizzarello LD, Jakobiec FA. Bowen’s disease of the conjunctiva: a misnomer. In: Jakobiec FA, ed. Ocular and Adnexal Tumors. Birmingham, AL: Aesculapius; 1978. pp. 553–71.
Galor A, Karp CL, Oellers P, Kao AA, Abdelaziz A, Feuer W, et al. Predictors of ocular surface squamous neoplasia recurrence after excisional surgery. Ophthalmology. 2012;119:1974–81.
Blasi MA, Maceroni M, Sammarco MG, Pagliara MM. Mitomycin C or interferon as adjuvant therapy to surgery for ocular surface squamous neoplasia: comparative study. Eur J Ophthalmol. 2018;28:204–9.
Rezvan F, Khabazkhoob M, Hooshmand E, Yekta A, Saatchi M, Hashemi H. Prevalence and risk factors of pterygium: a systematic review and meta-analysis. Surv Ophthalmol. 2018;63:719–35.
McKenzie R. A climatology of UVI for New Zealand. Report commissioned by the Cancer Society of NZ. Lauder, New Zealand: NIWA; 2008.
Acknowledgements
We would like to thanks the following consultants for providing access to their data: Dr Chris Murphy at Flagstaff Eye Care and Drs John Dickson, Stephen Guest, Benjamin Hoy, Selma Matloob, Michael Merriman, Stephen Ng, Hussain Patel, Bheema Patil, Rohan Weerekoon and David Worsley at Hamilton Eye Clinic. An additional thank you to The Ministry of Health for providing the national data.
Author contributions
RH: data acquisition, analysis, manuscript drafting, final approval, accountable for accuracy and integrity. JM: conception and design, analysis and interpretation, revising draft and approval of final draft, accountable for accuracy and integrity.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hossain, R.R., McKelvie, J. Ocular surface squamous neoplasia in New Zealand: a ten-year review of incidence in the Waikato region. Eye 36, 1567–1570 (2022). https://doi.org/10.1038/s41433-021-01662-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01662-3