Abstract
Background
We aimed to investigate whether the degree of transverse sinus stenosis (TSS) on magnetic resonance venography (MRV) can predict visual outcomes in patients with idiopathic intracranial hypertension (IIH).
Methods
A chart review identified IIH patients followed for at least 6 months. Mean deviation (MD) on visual field (VF) testing at initial and last follow-up visits, best corrected visual acuity (BCVA), patients’ age, body mass index, and lumbar puncture opening pressure were recorded. MRV was used to grade TSS on a scale of 0–8 using the grading scheme proposed by Farb et al., whereby a lower score indicates a greater degree of stenosis. Linear regression analysis was used to test for association between above variables and MD on VF, including change in MD over time.
Results
44 female patients were followed for a median of 26 months. All patients had unilateral or bilateral TSS on MRV. The median TSS score was 3/8. The median BCVA was 0.06 logMAR at baseline. The median VF MD was −3.02 dB at baseline and −1.96 dB at final follow-up. There was no significant association between the degree of TSS and either baseline MD (P-value = 0.34) or the change in MD on VF over the course of follow-up (P-value = 0.54). Baseline BCVA (P = 0.045) and baseline MD (P < 0.001) were the only significant predictors of changes in MD on VF over follow-up.
Conclusions
Baseline BCVA and MD on VF were the only significant predictors of visual outcome in IIH patients. While all patients demonstrated TSS on MRV, the degree of TSS did not correlate with visual prognosis.
Similar content being viewed by others
Introduction
Idiopathic intracranial hypertension (IIH) is a frequent cause of optic neuropathy in women of childbearing age who have increased body mass index (BMI) [1]. The prevalence of IIH has been paralleling the rise of obesity, generating a greater burden on the healthcare system [1]. IIH has previously been shown to cause various degrees of visual loss in about 1/4 of patients, significantly affecting their quality of life [2,3,4]. It is also often misdiagnosed, putting patients at risk of significant morbidity from invasive investigations and treatment [5].
Identifying patients with IIH who are at risk of poor visual outcomes would allow clinicians to guide treatment decisions. Currently, the most sensitive reported predictor of final visual outcome in IIH is visual acuity at presentation [6]. However, identifying another objective marker that can predict poor outcomes would be very useful. Current neuro-imaging findings associated with IIH include reduced pituitary gland height, increased optic nerve sheath diameter, flattening of the posterior globe, low-lying cerebellar tonsils, and transverse sinus stenosis (TSS) [7]. One recent study demonstrated that TSS is the most sensitive neuro-imaging correlate of IIH [8]. Moreover, treatment of TSS with endovascular dural sinus stenting has been consistently proposed as one of the therapeutic modalities for IIH [9, 10]. We thus hypothesized that there might be a relationship between the severity of TSS at presentation and final visual outcome in patients with IIH.
Only one previous study has investigated the relationship between the severity of TSS and visual outcomes. Although the authors did not find a correlation between TSS and VF loss [11], the study categorized VF loss into 3 stages using subjective criteria, rather than mean deviation (MD) on VF testing. It also graded TSS based on percent stenosis, a labor-intensive method which while allowing for increased precision may be difficult to apply in clinical practice. Thus, we designed our study to investigate the relationship between TSS and final visual outcomes in patients with IIH by using MD on VF testing as an indicator of visual function, and graded TSS on imaging using the criteria proposed by Farb et al. [12], which are much easier to apply in practice, in a separate patient cohort.
The primary objective of the study was to evaluate whether the severity of TSS, as classified on contrast-enhanced magnetic resonance venography (MRV) using Farb et al.’s criteria, is correlated with the change in mean deviation (measured in dB) on Humphrey VF (24-2 algorithm) testing from presentation to final follow-up in patients with IIH.
The secondary objective was to determine whether baseline VF predicted visual function at last follow-up, and to test the association between TSS at presentation and other variables of clinical relevance to the management of IIH, such as lumbar puncture opening pressure (LP OP) and BMI.
Methods
Patient identification
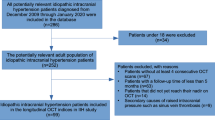
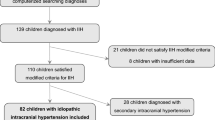
A retrospective chart review was performed to identify patients diagnosed with IIH between February 2014 and May 2019 according to criteria outlined by current diagnostic guidelines [13]. The diagnosis was made by a fellowship-trained neuro-ophthalmologist (EM). Only female patients above the age of 18 years who had a MRV scan of diagnostic quality and performed Humphrey VF testing (24-2 SITA-Fast protocol) at presentation and their last follow-up at least 6 months later were included in the study. Patients whose neuro-imaging was not satisfactory for calculating TSS score and those who completed less than 6 months of follow-up were excluded from the study. Patients with unreliable visual field testing (fixation loss >33% and false-positive errors >15%) were also excluded. Prospective institutional research ethics board approval was obtained from the University of Toronto prior to the initiation of this study.
Data collection
An independent reviewer (AE) used standardized data collection forms to collect the following information from patient records: age at presentation, baseline BMI, baseline Snellen best corrected visual acuity (BCVA), date of baseline and follow-up visits, MD on VF for each eye at baseline and final follow-up, LP OP when available, and prescribed interventions.
A fellowship-trained neuroradiologist (PN) independently evaluated the imaging results (contrast-enhanced MRV) of all patients performed at presentation. MRVs were performed using a previously published, validated contrast-enhanced protocol [14]. In brief, this consists of a 3-D fast spoiled gradient echo sagittal acquisition, obtained 8 seconds following triggering of the scan from contrast arrival in the cavernous carotid. Maximal intensity projection sequences were then created from the MRV data. Advantages of contrast enhanced MRV over non-contrast flow-sensitive techniques include better visualization of vessels (including in-plane vessels) and more accurate estimation of stenosis. This avoids the potential artefacts with flow-sensitive techniques, mainly loss of signal at certain points in the venous system as a result of in-plane spin saturation or vessel tortuosity.
The degree of TSS stenosis was graded based on a previously described classification by Farb et al. [12]. The grading involves identifying the area with the highest degree of stenosis for both the left and right transverse-sigmoid conduit and assigning it a score of 0–4. A score of 0 represents an aplastic or discontinued segment; 1 represents hypoplasia or severe stenosis of less than 25% of the cross-sectional diameter of the lumen of the distal superior sagittal sinus; 2 represents a moderately stenosed segment of the conduit (25–50%); 3 represents a mildly narrowed segment (50–75%); and 4 represents no significant narrowing (75–100%) (Fig. 1). The scores for the left and right conduits are then combined for a total score between 0 and 8.
A Normal (Score 4 bilaterally, 75–100% of the cross-sectional diameter of the lumen of the distal superior sagittal sinus). B Score 3 on the right side (mildly narrowed, 50–75%). Score 4 on the left. C Score 2 on the right side (moderately narrowed, 25–50%). Score 4 on the left. D Score 1 on the right and 1 on the left side (hypoplasia or severe stenosis, <25%). E Score 0 on the right (complete obstruction/aplasia/discontinuity in the sinus with no appreciable filling), 0 on the left side.
Data analysis
For analysis, BCVA was first converted from Snellen notation to logMAR using standardized conversions [15]. The BCVA and MD on VF testing of both eyes were then averaged; this decision was made as the baseline MD, final MD, and change in MD from baseline to final follow-up had high correlation between the left and right eye, indicating symmetric IIH progression (R2 = 0.92, P < 0.0001; R2 = 0.88, P < 0.0001; R2 = 0.85, P < 0.0001). Linear regression analyses were used to determine whether TSS and other clinical factors, such as LP OP, age, baseline BMI, and baseline BCVA were associated with a change in MD on VF testing from baseline to final follow-up, as well as baseline and final MD in separate analyses. Logistic linear regression analysis was used to determine whether any decrease in MD could be predicted by TSS and other clinical factors, as above. Patients with missing data on any clinical variable were excluded from the analysis for that given variable. Given the varying degree of follow-up, a secondary linear regression analysis using change in MD/day was performed. A p-value of 0.05 or lower was considered statistically significant.
Results
There were 44 patients included in the final analysis. All patients were female with a median age of 29 years (range: 18–49, interquartile range (IQR) = 29–40). The median duration of follow-up was 26 months (IQR: 10–43). Patients had a median BMI of 37.2 kg/m2 (IQR: 31.5–41.6) at presentation and a mean baseline BCVA of 0.06 ± 0.10 logMAR. The median combined TSS score at presentation was 3 (IQR: 2–3), with 86% (38/44) of patients having a TSS score between 1 and 3 (Fig. 2). All patients demonstrated narrowing (defined as 50–75% of the cross-sectional diameter of the lumen of the distal superior sagittal sinus) of the left or right transverse sinus, with 95% (42/44) demonstrating bilateral narrowing. Two patients underwent surgical treatment (1 with a ventriculoperitoneal shunt, 1 with a lumboperitoneal shunt followed by transverse sinus stenting), while the rest underwent medical treatment with acetazolamide (n = 22) or were encouraged to lose weight through dieting and exercise alone (n = 20). Patients receiving acetazolamide were prescribed doses ranging from 250 mg to 2.5 g per day. The median MD was −3.02 dB at baseline (IQR: −4.0 to −1.3) and −1.96 dB (IQR: −3.8 to −0.6) at final follow-up, with a median change in MD of +0.97 (IQR: 0.0 to 2.0) over the course of follow-up (Table 1).
Linear regression analysis revealed no significant relationship between the degree of TSS and the change in MD on VF from baseline to final follow-up (P-value=0.54, R2 = 0.009, Table 2, Fig. 3). This did not change after adjusting for differences in follow-up times between patients (P-value=0.62, R2 = 0.006, Table 2, Figs. 4 and 5). Logistic regression analysis revealed no significant relationship between TSS scores and a decline in MD from baseline to final follow-up (P-value=0.08). Linear regression analysis involving other variables revealed the following significant relationships after adjusting for differences in follow-up times: baseline MD and MD at final follow-up, baseline MD and change in MD, baseline BCVA and MD at final follow-up, baseline BCVA and change in MD. No significant relationship was found between the following variables: TSS and initial or final MD; baseline BMI and TSS; TSS and LP OP; LP OP and initial, final, and change in MD; baseline BMI and initial, final, and change in MD; age and initial, final, and change in MD; age and baseline BMI; age vs. TSS.
The combined transverse sinus stenosis (TSS) score (0–8) is plotted on the x-axis, and the change in mean deviation (MD), in decibels (dB), on visual field testing from baseline to final follow-up, is plotted on the y-axis. A regression line is plotted on the graph with corresponding R2 and P-values.
The combined transverse sinus stenosis (TSS) score (0–8) is plotted on the x-axis, and the average change in mean deviation (MD) per day, in decibels (dB), on visual field testing from baseline to final follow-up is plotted on the y-axis. There is also a regression line plotted on the graph, with corresponding R2 and P-values.
The combined transverse sinus stenosis (TSS) score (0–8) is plotted on the x-axis, and the average change in mean deviation (MD) per day, in decibels (dB), on visual field testing from baseline to final follow-up is plotted on the y-axis. The boxplot shows the first, second, and third quartile values, and the values that are outliers are seen as black circles. There is a dotted line that passes through 0.00 for visual reference.
Further subgroup analysis was performed to see if there were any differences between those who experience extreme VF changes at each end. Five outliers were identified in the cohort, 2 out of 5 had the largest improvements in MD, and 3 out of 5 whom had the largest losses in MD. No observable difference was found between the two groups in terms of baseline BMI, LP OP, age, and prescribed interventions.
Discussion
Previous studies have established a high prevalence of TSS among patients with IIH [12, 16, 17]. This was replicated in our study with all patients demonstrating narrowing of at least one of the two transverse sinuses. However, it is not clear whether TSS causes IIH or is its consequence and whether the severity of TSS influences the course of the disease. Thus, our study investigated the relationship between the degree of TSS on venographic imaging and visual outcomes. Our results were concordant with only one previously published paper on this subject demonstrating that the severity of TSS does not predict visual outcomes in patients with IIH [11].
The visual outcomes of each eye was highly correlated with that of the opposite eye, consistent with a symmetric progression that is more commonly seen in IIH [18]. We also found no relationship between LP OP and TSS, which is also consistent with findings from previous investigations [11, 12]. This suggests the presence of collateral cerebrospinal fluid drainage routes that may limit venous resistance in the cerebral sinuses despite physical restrictions in outflow through the transverse sinuses [11]. These alternative drainage routes include the occipital sinus and vein of Labbe [19, 20], which typically drain into the sigmoid sinus, bypassing the transverse sinus. We also found that age, baseline BMI, and LP OP are all poor predictors of visual outcome in IIH. This data further complicates the management of patients with IIH demonstrating that patients with similar demographics and clinical attributes can experience vastly different outcomes. In our study, the only significant predictors of visual outcomes were BCVA and MD on VF at presentation, which is also aligned with previous literature [21, 22].
Determining which patients will experience irreversible visual field loss remains a challenge. In our study, 25% (11 out of 44) of patients experienced worsening of MD on VF over follow-up, with only 1 patient experiencing a loss of more than 5 dB. Other retrospective case series have also shown that a minority of patients will experience visual decline over follow-up. Baheti et al. and Riggeal et al. observed a worsening of VF deficits in 11.6% and 16% of patients, respectively [11, 23]. Through lifestyle modifications and medical treatment, most patients with mild to moderate IIH typically stabilize or improve visual function over the course of follow-up, as has been shown in the IIH Treatment Trial and confirmed by our data [24]. The IIH Treatment Trial (IIHTT) evaluated changes in MD on VF for 165 patients with mild visual loss at presentation (−2 to −7 dB) who were randomized to receive dietary interventions alongside either acetazolamide or placebo. In the trial, treatment failure occurred when a participant with a baseline MD up to −3.5 dB had VF worsening of more than 2 dB in either eye, or when a participant with a baseline MD between −3.5 dB and −7 dB had worsening of more than 3 dB in either eye. Over the course of follow-up, only 4.2% of participants were determined to be treatment failures based on a substantial decline in MD on VF [24]. Using the same criteria for treatment failure, our study found a similarly low treatment failure rate of 10.7% (3 out of 28) among all participants with mild visual loss at presentation. When our analysis was restricted to patients with mild baseline visual loss treated with acetazolamide, the treatment failure rate was slightly higher at 20% (3 out of 15). However, the IIHTT initially determined that 25% of participants met the criteria for possible treatment failure, and this was reduced to 4.2% after VF re-testing of participants to control for performance errors. Re-testing was not performed in our study given clinical restraints, which may explain the slight discrepancy in treatment failure rates. Finally, The improvement in MD in our study (0.97 dB) was similar to that observed during long-term follow-up in IIHTT [25].
Our study’s subgroup analysis of patients with the highest and lowest changes in MD revealed no major differences between the groups in LP OP, baseline BMI, age, or TSS scores. Our findings highlight the challenge of patient selection for appropriate intervention in IIH. Although prior work has suggested that asymmetry of papilledema [26], morbid obesity [27], and ethnicity [28] can be associated with visual loss, these variables do not necessarily have prognostic value in predicting which patients will experience progressive worsening of VFs. For instance, the severity of papilledema at presentation does not predict a change in MD over time and worsening of papilledema over time is also not a predictor of VF loss [21]. Although weight gain over the course of follow-up has been shown to predict poor outcomes [29], baseline BMI was not a significant predictor of change in MD on VF in our study.
Although TSS is the most sensitive indirect imaging marker for the presence of IIH [8], its severity does not appear to be important. In patients with TSS, cerebrospinal fluid may drain through collateral conduits, such as the vein of Labbe and occipital sinus [19, 20]. Exploring possible defects or anatomic anomalies in these alternative drainage routes could potentially explain the clinical variability of IIH. Other avenues for exploration include measuring differences in optic nerve morphology on MR imaging or measuring pressure variations in cerebral venous sinuses, although these techniques are not currently feasible in clinical practice [30, 31].
Our study was limited by its relatively small sample size, yet the results mirror those of another study investigating this topic [11]. As some patients were unsure about their weight and were uncomfortable and/or unwilling to have their weight measured in the clinic, exact BMI data were missing in 19 of 44 patients. However, a comparison of patients with and without missing BMI data did not reveal any differences in the overall trend between TSS and visual field mean deviation. While our study measured BMI at baseline only, current evidence suggests that weight loss interventions lead to greater improvements in visual outcomes in IIH [32]. Another limitation of this retrospective review is the lack of data on treatment compliance. Many patients received treatment through prescribed lifestyle modifications, which were difficult to monitor, and patients’ compliance with acetazolamide treatment was also not specifically assessed. Thus, it is unclear if non-compliance could have played a role in outcome variability. Future studies should continue to explore other prognostic markers for visual outcomes in IIH, in order to determine which patients can remain on conservative medical treatments and which would benefit most from the invasive surgical procedures. These studies should be large enough to generate sufficient power, and prospectively performed to control for important variables such as treatment regimen and medication compliance.
In conclusion, although TSS is the most sensitive neuro-imaging finding in patients with IIH, the degree of TSS does not appear to be a useful predictor of visual outcomes. Although our study was limited by its sample size, our results mirrored those from the only other study that investigated this subject. This is despite the fact that our study was different in the way visual loss was judged and in the method used to estimate the degree of TSS on imaging. The degree of visual loss at presentation measured through MD on VF and BCVA remain the most reliable predictors of final visual outcome in patients with IIH.
Summary
What was known before
-
Idiopathic intracranial hypertension (IIH) in most cases is associated with indirect neuro-imaging features of increased intracranial pressure such as reduced pituitary gland height (the so-called “empty sella”), increase in diameter of arachnoid sheath around optic nerves, posterior flattening of the globes, and stenosis of brain dural sinuses, most commonly involving transverse sinus. Transverse sinus stenosis (TSS) is the most sensitive neuro-imaging feature of IIH, present in almost 100% of patients.
What this study adds
-
While all patients demonstrated TSS on magnetic resonance venography, the degree of transverse sinus stenosis did not correlate with visual prognosis. Future studies should explore other objective prognostic factors for visual outcomes in IIH, in order to determine which patients can remain on conservative medical treatment and which patients should be treated more aggressively and would most benefit from surgical interventions.
References
Mollan SP, Aguiar M, Evison F, Frew E, Sinclair AJ. The expanding burden of idiopathic intracranial hypertension. Eye 2019;33:478–85.
Wall M, George D. Idiopathic intracranial hypertension a prospective study of 50 patients. Brain 1991;114:155–80.
Corbett JJ, Savino PJ, Thompson HS, Kansu T, Schatz NJ, Orr LS, et al. Visual loss in pseudotumor cerebri: follow-up of 57 patients from five to 41 years and a profile of 14 patients with permanent severe visual loss. Arch Neurol 1982;39:461–74.
Digre KB, Bruce BB, McDermott MP, Galetta KM, Balcer LJ, Wall M. Quality of life in idiopathic intracranial hypertension at diagnosis IIH Treatment Trial results. Neurology 2015;84:2449–56.
Fisayo A, Bruce BB, Newman NJ, Biousse V. Overdiagnosis of idiopathic intracranial hypertension. Neurology 2016;86:341–50.
Wall M, Falardeau J, Fletcher WA, Granadier RJ, Lam BL, Longmuir RA, et al. Risk factors for poor visual outcome in patients with idiopathic intracranial hypertension. Neurology 2015;85:799–805.
Mallery RM, Rehmani OF, Woo JH, Chen YJ, Reddi S, Salzman KL, et al. Utility of magnetic resonance imaging features for improving the diagnosis of idiopathic intracranial hypertension without papilledema. J Neuro-Ophthalmol. 2019;39:299–307.
Morris PP, Black DF, Port J, Campeau N. Transverse sinus stenosis is the most sensitive MR imaging correlate of idiopathic intracranial hypertension. Am J Neuroradiol 2017;38:471–7.
Gurney SP, Ramalingam S, Thomas A, Sinclair AJ, Mollan SP. Exploring the current management idiopathic intracranial hypertension, and understanding the role of dural venous sinus stenting. Eye Brain. 2020;12:1–13.
Liu KC, Starke RM, Durst CR, Wang TR, Ding D, Crowley RW, et al. Venous sinus stenting for reduction of intracranial pressure in IIH: a prospective pilot study. J Neurosurg 2017;127:1126–33.
Riggeal BD, Bruce BB, Saindane AM, Ridha MA, Kelly LP, Newman NJ, et al. Clinical course of idiopathic intracranial hypertension with transverse sinus stenosis. Neurology 2013;80:289–95.
Farb RI, Vanek I, Scott JN, Mikulis DJ, Willinsky RA, Tomlinson G, et al. Idiopathic intracranial hypertension: The prevalence and morphology of sinovenous stenosis. Neurology 2003;60:1418–24.
Friedman DI, Liu GT, Digre KB. Revised diagnostic criteria for the pseudotumor cerebri syndrome in adults and children. Neurology 2013;81:1159–65.
Farb RI, Scott JN, Willinsky RA, Montanera WJ, Wright GA, TerBrugge KG. Intracranial venous system: Gadolinium-enhanced three-dimensional MR venography with auto-triggered elliptic centric-ordered sequence - Initial experience. Radiology 2003;226:203–9.
Khoshnood B, Mesbah M, Jeanbat V, Lafuma A, Berdeaux G. Transforming scales of measurement of visual acuity at the group level. Ophthalmic Physiol Opt 2010;30:816–23.
Higgins JNP, Gillard JH, Owler BK, Harkness K, Pickard JD. MR venography in idiopathic intracranial hypertension: Unappreciated and misunderstood. J Neurol Neurosurg Psychiatry. 2004;75:621–5.
Fera F, Bono F, Messina D, Gallo O, Lanza PL, Auteri W, et al. Comparison of different MR venography techniques for detecting transverse sinus stenosis in idiopathic intracranial hypertension. J Neurol 2005;252:1021–5.
Bidot S, Bruce BB, Saindane AM, Newman NJ, Biousse V. Asymmetric papilledema in idiopathic intracranial hypertension. J Neuro-Ophthalmol. 2015;35:31–6.
Kanno T, Kasama A, Suzuki H. Safety of ablation of the sigmoid and transverse sinuses: an experimental study. Skull Base Surg 1993;3:146–51.
Kobayashi K, Suzuki M, Ueda F, Matsui O. Anatomical study of the occipital sinus using contrast-enhanced magnetic resonance venography. Neuroradiology 2006;48:373–9.
Takkar A, Goyal MK, Bansal R, Lal V. Clinical and neuro-ophthalmologic predictors of visual outcome in idiopathic intracranial hypertension. Neuro-Ophthalmol. 2018;42:201–8.
Friedman DI. Idiopathic intracranial hypertension. Curr Pain Headache Rep. 2007;11:62–8.
Baheti N, Nair M, Thomas S. Long-term visual outcome in idiopathic intracranial hypertension. Ann Indian Acad Neurol. 2011;14:19–22.
Smith SV, Friedman DI. The idiopathic intracranial hypertension treatment trial: a review of the outcomes. Headache 2017;57:1303–10.
Wall M, Kupersmith MJ, Thurtell MJ, Moss HE, Moss EA, Auinger P. The longitudinal idiopathic intracranial hypertension trial: outcomes from months 6–12. Am J Ophthalmol. 2017;176:102–7.
Wall M, White WN. Asymmetric papilledema in idiopathic intracranial hypertension: prospective interocular comparison of sensory visual function. Investig Ophthalmol Vis Sci 1998;39:134–42.
Szewka AJ, Bruce BB, Newman NJ, Biousse V. Idiopathic intracranial hypertension: relation between obesity and visual outcomes. J Neuro-Ophthalmol. 2013;33:4–8.
Bruce BB, Preechawat P, Newman NJ, Lynn MJ, Biousse V. Racial differences in idiopathic intracranial hypertension. Neurology 2008;70:861–7.
Chagot C, Blonski M, Machu JL, Bracard S, Lacour JC, Richard S. Idiopathic intracranial hypertension: prognostic factors and multidisciplinary management. J Obes 2017;2017:5348928.
Schmidt C, Wiener E, Lüdemann L, Kunte H, Kreutz KM, Becker N, et al. Does IIH alter brain microstructures? – A DTI-based approach. Headache 2017;57:746–55.
Boddu SR, Gobin P, Oliveria C, Dinkin M, Patsalides A. Pressure variations in cerebral venous sinuses of idiopathic intracranial hypertension patients. J Vasc Inter Neurol 2018;10:25–30.
Koc F, Isik MR, Sefi-Yurdakul N. Weight reduction for a better visual outcome in idiopathic intracranial hypertension. Arq Bras Oftalmol 2018;81:18–23.
Author information
Authors and Affiliations
Contributions
All authors were involved in designing the study protocol. AE and PN collected data from patient charts. AE and KZ analysed the data and created summary tables and figures. AE, KZ, PN, and EM were all involved in writing and revision of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eshtiaghi, A., Zaslavsky, K., Nicholson, P. et al. Extent of transverse sinus stenosis does not predict visual outcomes in idiopathic intracranial hypertension. Eye 36, 1390–1395 (2022). https://doi.org/10.1038/s41433-021-01651-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01651-6
This article is cited by
-
MRI features of idiopathic intracranial hypertension are not prognostic of visual and headache outcome
The Journal of Headache and Pain (2023)