Abstract
Optical coherence tomography (OCT) is a paragon of success in the translation of biophotonics science to clinical practice. OCT systems have become ubiquitous in eye clinics but access beyond this is limited by their cost, size and the skill required to operate the devices. Remarkable progress has been made in the development of OCT technology to improve the speed of acquisition, the quality of images and into functional extensions of OCT such as OCT angiography. However, more needs to be done to radically improve the access to OCT by addressing its limitations and enable penetration outside of typical clinical settings and into underserved populations. Beyond high-income countries, there are 6.5 billion people with similar eye-care needs, which cannot be met by the current generation of bulky, expensive and complex OCT systems. In addition, advancing the portability of this technology to address opportunities in point-of-care diagnostics, telemedicine and remote monitoring may aid development of personalised medicine. In this review, we discuss the major milestones in OCT hardware development to reach those beyond the eye clinic.
摘要
相干光断层扫描成像技术 (Optical coherence tomography, OCT) 是将生物光子学在临床实践中应用的成功典范 。在眼科门诊中, OCT 系统得到了广泛应用, 但是由于其成本高、体积大以及需要具备操作技能的原因, OCT 系统在门诊之外的应用受到限制。目前, OCT 技术的发展在提高图像获取速度、图片质量以及OCT在血管功能检测的拓展应用 (如OCT血管成像技术) 方面取得了显著进展。然而, 我们还需通过解决OCT技术的局限性问题以从根本上改善OCT的应用范围方面做更多的工作, 将OCT技术在典型的临床环境之外以及医学资源相对匮乏的地区人群中得以应用。在高收入国家之外, 还有65亿人有类似的眼保健需求, 而目前这一代OCT系统因其庞大、昂贵和复杂而无法满足这些需求。此外, 提高OCT设备的便携性可为医疗点诊断、远程医疗以及远程监护提供可能, 这有助于个体化医疗的发展。在这篇综述中, 我们主要讨论了OCT硬件发展中为实现OCT技术在眼科门诊之外应用目标的里程碑式的成绩。
Similar content being viewed by others
Introduction
Optical coherence tomography (OCT) imaging is rapid, safe, non-invasive and is increasingly being recognised as the gold standard for diagnosis of retinal disease [1]. However, OCT machines have several limitations. First, they are costly; the price of a retinal OCT device ranges from ~£30,000–£100,000 [2]. Second, the use of these devices are limited to typical clinical settings such as hospital clinics to optometry practices. They are housed in large tabletop configurations and require alternating current power, constraining their portability. As patients are required to sit upright and stabilise their head on a chinrest whilst maintaining steady fixation, they are only suitable for ambulatory and cooperative patients. Third, a certain level of skill is required to capture optimal images. A technician is often required to align the device, capture the image and perform a quality check to ensure that the image is acceptable. In this review, we discuss several efforts that aim to address these limitations and thus improve the accessibility of this valuable diagnostic tool and enable point-of-care diagnostics, telemedicine and remote monitoring.
Miniature and low-cost OCT
The construction of a portable, handheld, OCT device could increase the ease of access and expand OCT into settings where use is currently prohibited by its cost and size. There are currently three commercial handheld SD-OCT systems available that address this issue. The Envisu C2300 OCT (Leica Microsystems, Germany) became the first handheld OCT scanner to receive clearance from the Food and Drug Administration [3]. The scanner consists of a 1.5 kg imaging probe connected to a console by a 1.3-m flexible cable with the most recent iteration, the C2300 additionally providing a variety of lenses to enable both anterior- and posterior-segment imaging. The iVue system by Optovue Inc. consists of a stand-mounted unit with a 2.2-kg removable scanner [4]. Both systems benefit from rapid acquisition speed facilitating capture in paediatric populations. A third device, the Heidelberg Spectralis Flex Module (Heidelberg Engineering, Heidelberg, Germany) incorporates a flexible ‘boom’ arm, which houses the acquisition lens and can be adjusted up to 100 cm from the main device body (Fig. 1) [5]. The portability of all these devices has allowed for imaging of patients in different positions and beyond the outpatient clinic, from intensive care to the surgical environment [6]. However, these handheld systems are associated with a steep learning curve for operation, are subject to motion artifacts and are still considerably heavy to hold or are attached to much larger systems that restrict use outside of the clinic.
Several research groups have designed portable OCT prototypes that incorporate miniaturised components known as microelectromechanical systems (MEMS) in the spectrometer. MEMS enable technology to be constructed at micrometre scale, and is widely used in numerous consumer products such as mobile phones, video game controllers and accelerometers and gyroscopes for drones. In OCT systems, MEMS scanning mirrors can be used instead of bulky galvanometers to enable a lightweight compact design that can be integrated into probes or handheld devices, whilst simultaneously reducing the cost [7,8,9,10]. However, reductions in size and cost come at a significant sacrifice of signal-to-noise ratio and reduced imaging depth. In addition, OCT systems comprise several components including the light source and the computer processing unit, all of which can still be costly. Units integrating MEMS and vertical cavity surface emitting lasers (VCSELs) are also available, combining the advantage of portability with extended depth range [11]. VCSELs are light sources with large coherence lengths of up to 100 mm, greater than the axial length of the eye [12]. Therefore, with VCSEL it is possible to image both anterior and posterior segments concurrently and without the need for additional attachments. A prototype swept-source OCT (SS-OCT) handheld system incorporating VCSEL was demonstrated by Lu et al., which had an acquisition speed of 350,000 A-scans per second—ten times greater than Envisu system [9]. Speed of image acquisition is an important consideration, particularly in handheld systems, which are prone to artifacts from operator motion, and compounded by subject motion. To alleviate impact of these artifacts on the output image, the system acquires two volumes orthogonal to each other, which can be processed to generate a motion-corrected merged volume. The high speed of the system also enables dense sampling of the retina, producing high definition in vivo retinal images with a field of view up to 10 × 10 mm3 captured in this study. The shape and weight of the device is also a critical design consideration to ensure rapid and steady alignment of the device by the operator. With innovations in 3D printing, optical components can be housed in a lightweight ergonomic casing. The authors evaluated two different designs with identical optical components—their power grip design (0.50 kg) supported ambidextrous operation but was overly sensitive to the operator’s hand movements, whereas the camcorder design (0.42 kg) provided improved balancing but could only be operated using the right hand. The optical design also incorporated an iris camera and fixation target to further aid subject alignment. Although the images produced are comparable to commercially available devices, the cost of the prototype was too high to be viable for use outside of the eye clinic. The majority of the cost was due to the VCSEL and data acquisition electronics. Similarly, Nankivil et al. demonstrated a SS-OCT handheld device that incorporated a MEMS scanner with an iris camera to aid alignment. Uniquely, the system rapidly switches from anterior to posterior-segment imaging, miniaturising whole-eye OCT to a portable system [13].
The cost of OCT can be significantly reduced by designing systems that utilise components that are manufactured at scale for other industries and are therefore readily available and inexpensive. One biomedical engineering group from Duke University has built an OCT system that uses commercial off-the shelf components to design the spectrometer, scanner, optics and electronics, costing a total of $7164 [14]. The system uses a variable focus liquid lens that can be precisely adjusted to the desired focal length, and enabling dynamic focus over multiple depths. An inexpensive microcontroller was used to synchronise the MEMS mirror with the spectrometer. The sensor of the spectrometer was inexpensive and is designed to be more robust to system misalignment due to temperature fluctuations and mechanical stress. A mini PC accelerated using a graphics processing unit (GPU) was used to increase the portability, eliminating the need for an external PC or laptop as is used in most commercial OCT systems. The increased processing power from a GPU enables highly efficient image processing. To synchronise the movement of the MEMS mirror with the detector, an Arduino microcontroller was used to detect incoming signals and subsequently senda a trigger to scan the MEMS mirror without delay. The optical components were housed inside a lightweight 3D-printed casing with a total weight of 2.7 kg (Fig. 1A, B). The system had an axial resolution of 7 μm, an imaging depth of 2.8 mm with a 7.0 × 7.0-mm field of view, and a B-scan acquisition rate (512 × 512 pixels) of 12 frames per second. To demonstrate the imaging capability of the system in relevant biological tissue, ex vivo porcine eyes and live mice retinas were imaged. The low-cost OCT system can clearly resolve the tear film, individual corneal layers, the lens and the iris of the porcine eye. Retinal B-scan imaging of live mice could clearly resolve the individual retinal layers (Fig. 1C). Some limitations of this prototype include the slow acquisition speed and lack of alignment tools such as eye tracking or iris cameras, making it prone to motion artifacts and thus reducing the ability to take advantage of averaging to improve the signal-to-noise ratio.
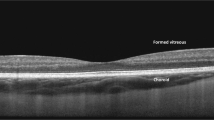
The same group has further refined their low-cost OCT system and successfully applied the device to image the retina of 30 healthy volunteers and 30 patients with retinal pathology [15]. The system offers an axial resolution of 8.0 μm, an imaging depth of 2.7 mm and a 6.6 × 6.6-mm field of view. The prototype OCT images could clearly resolve relevant layers of the retina, comparable to the images from the Heidelberg Spectralis system (Fig. 2). Although the system was mounted on a chinrest for the study to aid stability for comparison with the Heidelberg device, it was also operable as a handheld device. Evaluated in five healthy volunteers, the investigators obtained high-quality images with an operator hand holding the scanner without a chinrest. The superluminescent diode in this system suffered from power fluctuations affecting the image contrast. This engineering step was made to keep cost down, but given the impact on the imaging signal from the retina, the group aims to resolve this in future iterations. To enable complete stand-alone operation of the system, a 7-inch touchscreen was incorporated into the system body to allow controls for data acquisition and display of retinal images (Fig. 3). The total weight of the device was 2.3 kg, with a total cost of materials was $5037. While a productionised version of this system is likely to be more expensive to offset the research and regulatory expenses, it has the potential to offer adequate performance as a retinal screening tool at a fraction of the cost of current commercial systems. The device will be commercialised through the group’s start-up company called Lumedica (Durham, NC), and are currently marketing an OCT microscope constructed using similar technology for research purposes [16, 17]. The OQ-LabScope boasts a low price of 10,000 USD, a compact size (13 × 7.5 × 6 inches) and is lightweight (2.7 kg). The device has an axial resolution of 5 μm in tissue and can acquire B-scan images at a rate of 22 per second. It can be connected to a PC and integrates a graphical user interface for intuitive manipulation of imaging parameters. While this system is affordable and easy to use, it is not yet approved for use in retinal imaging.
One of the most exciting applications is the potential for ‘smart OCT’. The significant advancements in display, sensor and battery technologies together have paved the way for multi-functional devices such as smartphones, tablets and smartwatches. Modern day smart devices incorporate a number of embedded sensors that can be used to measure several health parameters such as cardiovascular health [18], physical activity [19, 20] and sleep quality [21]. Smart devices have the ability to connect to a data network, enabling assessments to be remotely administered and results to be wirelessly transmitted to health providers [22]. Nearly 60% of the world’s population is connected to the internet [23], and nearly half own a smartphone [24], making remote health monitoring a feasible option in large parts of the world. Remote monitoring technologies would not only be valuable in low- and middle-income countries where access to ophthalmologist offices is limited [25], but also for patients with chronic conditions such as age-related macular degeneration, which necessitate ongoing monitoring. Smartphone fundus imaging is already available that utilises the smartphone camera with the addition of a clip-on adaptor that provides the additional optics required [26]. However, integrating OCT into a smart device is more complex due to the need for emission of specialised wavelengths. Advances in silicon photonics have demonstrated the feasibility of integrating optical and electrical circuits onto a chip that can be incorporated into portable devices [27, 28]. OCTCHIP, a European project led by Wolfgang Drexler from the Medical University of Vienna, aims to use this technology to engineer a cost-effective, handheld, wireless, OCT system that is the size of a 1 cent coin [27]. The project endeavours to radically transform OCT towards widespread adoption in point-of-care diagnostics for the early diagnosis of retinal pathologies. In another European-funded project called Handheld OCT, the same group aims to create a handheld system for point-of-care diagnostics with partners from Carl Zeiss, University College Cork, and several other partners in the electronics space [29]. This 5-year project commenced in January 2020.
Multiple-reference OCT (MRO) is a new type of OCT system that has a low-cost and compact configuration, with a comparable footprint to the pick-up head contained within a DVD unit [30]. The foundations of the technology are similar to first-generation time-domain OCT. In time-domain systems, a Michelson-type interferometer is used to divide a broadband light, emitted from a superluminescent diode, into a reference arm of known path length and time-delay, and a sample arm that is focused at the tissue of interest, such as the retina. The two light beams are reflected back to a photodetector using a mirror to generate an interference pattern. By mechanically moving the mirror over the sample, light is reflected from each depth in the tissue producing multiple A-scans that subsequently comprise a B-scan. This involves several moving parts that travel considerable distances, consequently limiting the scan speed and making it prone to motion artifacts. The proposed MRO system reduces the need to mechanically adjust the optical path length with the addition of a partial mirror close to the reference mirror, and thus enabling a more compact design. The partial mirror propels a portion of the backscattered light to travel to the detector, and the remaining portion is reflected back and forth between the partial and reference mirror multiple times. Each successive reflection corresponds to regions deeper within the target tissue. Every interference signal generated by the reflections can be separated out using digital signal processing techniques, and stitched together for each region of the A-scan to form a continuous image. Subhash et al. have demonstrated the implementation of this technology using a smartphone interface [31]. In this work, an analogue front-end receives the optical signals. An analogue-to-digital converter connects the analogue front-end to a smartphone to digitise the received data. The smartphone acts as a signal processor and a graphical user interface for the user to start and stop scan measurements and see a live graph of incoming OCT measurements. These measurements can be transformed into an image and subsequently analysed, saved or transmitted via email. The next steps for this technology will require improving the imaging performance to achieve real-time B-scan images [32]. Although the acquisition speed of MRO cannot compete with the latest Fourier domain systems, the moderate speed of the system does not demand high specification computational platforms therefore may be deployable on mobile systems. This technology will be commercialised by a start-up company called Compact Imaging (Mountain View, CA) in partnership with Novartis (Basel, Switzerland) [33, 34].
Automated OCT
Much of the burden of chronic eye conditions—to both patients and to healthcare systems—may be attributed to the limitations of the current eye examination. Patients with such conditions commonly require long-term monitoring with frequent, time-consuming visits to the eye clinic. The workflow in many eye clinics is inefficient, with patients being asked to wait multiple times, interact with several different healthcare staff and see their doctor on multiple occasions. The need for extensive testing also results in large staffing and equipment costs and extensive floor space requirements. Moreover, the fundamental unit of the eye examination—slit-lamp ophthalmoscopy—has little changed since its original description in 1911 by Gullstrand [35], being time-consuming, subjective, non-quantitative and requiring an experienced clinician. A new form of eye examination has been developed by Envision Diagnostics, Inc. (El Segundo, CA, USA) termed binocular OCT [36], that aims to address the shortcomings of the current eye exam, and adds many unique capabilities.
A prototype of this system has undergone usability testing among patients with chronic eye conditions and healthy volunteers [37]. Unlike other OCT devices, the binocular OCT prototype consists of two oculars that align to the patient’s eyes in an automated manner, enabling a pair of eyes to be imaged simultaneously. This reduces the labour required to align the device, and also reduces the overall scan time. The binocular OCT also utilises a tuneable swept-source laser system with adjustable optics that can switch from anterior eye imaging to lens imaging, to vitreous imaging and to posterior pole imaging, permitting whole-eye OCT without the need for additional attachments. Furthermore, the device incorporates ‘smart technology’, offering more advanced display, input and computing capabilities than conventional OCT. A speaker system is used to deliver audio instructions to guide the automated examination. In addition, the binocular aspect of the device can be exploited so that OCT imaging can be used for novel applications such as diagnostic functional tests including pupillometry [38] (Fig. 4), strabismus measurement [39] and ocular motility [37]. Traditionally these tests have been subjective and required significant clinical expertise to interpret.
A Resting diameters pre-stimulus; B flash presented to the left eye, constriction of both pupils observed; C both pupils dilated to their resting diameter; D flash presented to the right eye. Constriction amplitude of both eyes is less than that observed when the flash was presented to the left eye.
With these features, the binocular OCT aims to incorporate many aspects of the eye examination into a single automated, patient-facing instrument, and has a number of potential benefits if this was adopted in tertiary eye-care clinics, including:
-
Increased efficiency of eye clinics, allowing patients with chronic eye disease to spend less time waiting during routine hospital eye examinations.
-
Reduced costs, through a reduction in the total number of diagnostic instruments required and their associated labour costs.
-
Improved quality of eye care, through the introduction of more, quantitative, standardised ocular measurements and high-resolution imaging.
The results of a usability study, and related focus group testing, make it clear that patients are receptive to the concept of an automated eye examination. To be attractive to users, easy to use and effective at performing automated eye examinations, the system will need to be quick, responsive and comfortable. Once established, binocular OCT will offer objective, quantifiable information about almost every aspect of the eye examination and has the potential to supersede many traditional but flawed testing methods. It is unlikely that the automated eye examination will be suitable for use in all patients. However, if such a system can replace some aspects of the eye examination, workflows and waiting times are likely to improve and costs are likely to reduce.
Home OCT
At-home disease monitoring devices are also in development and may be more cost-effective for monitoring patients at high-risk of developing neovascular AMD [40]. Notal Vision (Tel Aviv, Israel) received FDA approval for the ForeSeeHome in 2010, an at-home digital macular visual field and hyperacuity testing device that transmits data directly to the ophthalmologist. The device has since undergone a randomised clinical trial, concluding that individuals at high risk of conversion to neovascular AMD would benefit from a home monitoring strategy for earlier detection of choroidal neovascularisation development [41]. Notal also plans to launch a ‘Home OCT’ device, a self-operated SD-OCT device that could be provided to patients at risk of vision loss from AMD, or between visits to the ophthalmologist to customise appointments to the individual patient’s needs (Fig. 5) [42]. The device acquires images of the central 10 degrees of the macula and subsequently reads the image using the company’s artificial intelligence platform, Notal OCT Analyzer (NOA). In a prospective clinical trial, 90% of 196 patients were able to obtain a gradable image in at least one eye after a 2-min video tutorial [43]. When evaluated on images of AMD taken using the device, NOA demonstrated >97% sensitivity and specificity for detection of intraretinal and subretinal fluid [44]. The device received Breakthrough Device Designation by the FDA in 2018, expediting assessment and review for 510(k) clearance and market authorisation. Though, the usability and validation of the OCT device and software is yet to be determined through peer-reviewed publications.
Self-examination low-cost full-field OCT (SELF-OCT) is another proposed low-cost system that allows the patient to independently examine the disease progression of AMD at home without the presence of a physician [45, 46]. In contrast to commercially available systems, the SELF-OCT system sequentially acquires transversal en face images at different depths instead of cross-sectional images in the axial direction. The SELF-OCT prototype device captures images at an axial resolution of 9.1 µm and a transversal resolution of 6.4 and 12.8 µm, respectively; although, in vivo retinal scans from the system exhibit more artifacts and a greater signal-to-noise ratio than clinical OCT systems. Furthermore, the field of view of the SELF-OCT is 1.4 × 4.8 × 1.5 mm, which is considerably smaller than clinical OCT systems, but may be sufficient for monitoring of wet AMD [47]. This technology has been patented and is being commercialised by Visotec [48].
A proposed novel home-based SD-OCT system termed ‘sparse OCT’ has recently been developed by a Swiss group (Fig. 6) [49]. The portable device, named MIMO-OCT, generates single 5-mm line scans at a resolution of 400 pixels, or small sample 3.8 × 3.8-mm volume scans at a resolution of up to 150 × 150 pixels. The downsampled scanning pattern is less dense than scan protocols available on commercial SD-OCT systems, but reduces the size of the instrument and enables fast scanning. The prototype required the subject’s head to be inclined downwards onto the headrest, enabling stable positioning to reduce moving artifacts. Manually graded central retinal thicknesses from sparse OCT data in 30 AMD patients were compared to automated measurements obtained from the Heidelberg Spectralis device, showing no statistical difference. Sparse OCT may be one method of delivering a portable at-home OCT system with adequate resolution for monitoring.
Conclusion
In only two decades, OCT has evolved from an experimental investigation to redefining the landscape of ophthalmic assessment and management, particularly within the sphere of retinal diseases. While such devices are now prevalent in most ophthalmic units and increasingly in community optometry practices, the next decade will usher a further democratisation of OCT through advances in hardware, cloud-based computing and artificial intelligence. Recent regulatory approvals and feasibility studies highlight the emerging permeation of miniaturised, portable and handheld OCT systems, affording their use as a point-of-care diagnostic tool in non-traditional settings such as intensive care, as well as in the home environment for remote monitoring of chronic conditions such as AMD. The latter in particular could be analogous to continuous monitoring and thus provide opportunities for personalised treatment plans for conditions such as wet AMD that benefit from close monitoring and often require indefinite follow-up. Enabled by the ability for secure wireless information transfer, improvements in the speed of data transmission through 5th generation telecommunication networks [50], and the current dynamism of teleophthalmology, scope also exists for far-reaching impact of a low-cost OCT in low- and middle-income countries. Further efficiencies will come from automated systems, such as the binocular OCT, which have the potential of reducing cost, increasing quality of data and introducing novel applications of OCT that can revolutionise many aspects of the eye exam. Undoubtedly, OCT will continue to transform ophthalmology, and portable, low-cost systems are likely to have their place in the OCT revolution.
References
Rosenfeld PJ. Optical Coherence Tomography and the Development of Antiangiogenic Therapies in Neovascular Age-Related Macular Degeneration. Invest. Ophthalmol. Vis. Sci. 2016;57:OCT14–26.
Olson J, Sharp P, Goatman K, Prescott G, Scotland G, Fleming A, et al. Improving the economic value of photographic screening for optical coherence tomography-detectable macular oedema: a prospective, multicentre, UK study. Health Technol Assess. 2013;17:1–142.
Food and Drug Administration. 510(k) Premarket notification, Envisu Spectral Domain Ophthalmic Imaging System (SDOIS). 2012. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPMN/pmn.cfm?ID=K120057. Accessed 23 Aug 2020.
Maloney R. The Optovue iVue OCT System from Grafton Optical: the possibilities of hand-held OCT devices in ophthalmic practice. J Vis Commun Med. 2012;35:76–81.
Liu X, Kale AU, Capewell N, Talbot N, Ahmed S, Keane PA, et al. Optical coherence tomography (OCT) in unconscious and systemically unwell patients using a mobile OCT device: a pilot study. BMJ Open. 2019;9:e030882.
Dayani PN, Maldonado R, Farsiu S, Toth CA. Intraoperative use of handheld spectral domain optical coherence tomography imaging in macular surgery. Retina. 2009;29:1457–68.
Larocca F, Nankivil D, Farsiu S, Izatt JA. Handheld simultaneous scanning laser ophthalmoscopy and optical coherence tomography system. Biomed Opt Express. 2013;4:2307–21.
Demian D, Duma V-F, Sinescu C, Negrutiu ML, Cernat R, Topala FI, et al. Design and testing of prototype handheld scanning probes for optical coherence tomography. Proc Inst Mech Eng H. 2014;228:743–53.
Lu CD, Kraus MF, Potsaid B, Liu JJ, Choi W, Jayaraman V, et al. Handheld ultrahigh speed swept source optical coherence tomography instrument using a MEMS scanning mirror. Biomed Opt Express. 2013;5:293–311.
Sayegh SI, Nolan RM, Jung W, Kim J, McCormick DT, Chaney EJ, et al. Comparison of a MEMS-based handheld OCT scanner with a commercial desktop OCT system for retinal evaluation. Transl Vis Sci Technol. 2014;3:10.
Potsaid B, Jayaraman V, Fujimoto JG, Jiang J, Heim PJS, Cable AE. MEMS tunable VCSEL light source for ultrahigh speed 60 kHz–1 MHz axial scan rate and long range centimeter class OCT imaging. In: Optical coherence tomography and coherence domain optical methods in biomedicine XVI, vol 8213. San Francisco, California, United States: SPIE BiOS; 2012. p. 82130M.
Grulkowski I, Liu JJ, Potsaid B, Jayaraman V, Lu CD, Jiang J, et al. Retinal, anterior segment and full eye imaging using ultrahigh speed swept source OCT with vertical-cavity surface emitting lasers. Biomed Opt Express. 2012;3:2733–51.
Nankivil D, Waterman G, LaRocca F, Keller B, Kuo AN, Izatt JA. Handheld, rapidly switchable, anterior/posterior segment swept source optical coherence tomography probe. Biomed Opt Express. 2015;6:4516–28.
Kim S, Crose M, Eldridge WJ, Cox B, Brown WJ, Wax A. Design and implementation of a low-cost, portable OCT system. Biomed Opt Express. 2018;9:1232–43.
Song G, Chu KK, Kim S, Crose M, Cox B, Jelly ET, et al. First clinical application of low-cost OCT. Transl Vis Sci Technol. 2019;8:61.
Lumedica. Lumedica. https://www.lumedicasystems.com/. Accessed 29 Aug 2020.
The Engineer. Low-cost OCT scanner promises to save eyesight. 2019. https://www.theengineer.co.uk/oct-scanner-sight-duke-university/. Accessed 29 Aug 2020.
Strik M, Caillol T, Ramirez FD, Abu-Alrub S, Marchand H, Welte N, et al. Validating QT-interval measurement using the Apple watch ECG to enable remote monitoring during the COVID-19 pandemic. Circulation. 2020;142:416–8.
Case MA, Burwick HA, Volpp KG, Patel MS. Accuracy of smartphone applications and wearable devices for tracking physical activity data. JAMA. 2015;313:625–6.
Degroote L, De Bourdeaudhuij I, Verloigne M, Poppe L, Crombez G. The accuracy of smart devices for measuring physical activity in daily life: validation study. JMIR Mhealth Uhealth. 2018;6:e10972.
Guillodo E, Lemey C, Simonnet M, Walter M, Baca-García E, Masetti V, et al. Clinical applications of mobile health wearable–based sleep monitoring: systematic review. JMIR Mhealth Uhealth. 2020;8:e10733.
Perez MV, Mahaffey KW, Hedlin H, Rumsfeld JS, Garcia A, Ferris T, et al. Large-scale assessment of a smartwatch to identify atrial fibrillation. N Engl J Med. 2019;381:1909–17.
Statista. Global digital population as of July 2020. 2020. https://www.statista.com/statistics/617136/digital-population-worldwide/. Accessed 22 Aug 2020.
Statista. Smartphone users worldwide 2020. 2019. https://www.statista.com/statistics/330695/number-of-smartphone-users-worldwide/. Accessed 22 Aug 2020.
Sommer A, Taylor HR, Ravilla TD, West S, Lietman TM, Keenan JD, et al. Challenges of ophthalmic care in the developing world. JAMA Ophthalmol. 2014;132:640–4.
Bastawrous A, Giardini ME, Bolster NM, Peto T, Shah N, Livingstone IAT, et al. Clinical validation of a smartphone-based adapter for optic disc imaging in Kenya. JAMA Ophthalmol. 2016;134:151–8.
Drexler W. OCTChip. http://www.octchip.researchproject.at/. Accessed 1 Aug 2018.
Cordis. Ophthalmic OCT on a Chip. European Commission; 2016. https://cordis.europa.eu/project/id/688173. Accessed 31 Aug 2020.
Cordis. Handheld optical coherence tomography. European Commission; 2020. https://cordis.europa.eu/project/id/871312. Accessed 31 Aug 2020.
Leahy MJ, Wilson C, Hogan J, O’Brien P, Dsouza R, Neuhaus K, et al. The how and why of a $10 optical coherence tomography system. In: Optical coherence tomography and coherence domain optical methods in biomedicine XX. San Francisco, California, United States: SPIE BiOS 2016; 2016. https://doi.org/10.1117/12.2213465.
Subhash HM, Neuhaus K, Dsouza R, Hogan J, Wilson C, Leahy MJ. Smartphone-based Multiple Reference Optical coherence tomography (MROTM) system. In: Miami, Florida United States:Biomedical optics 2014; 2014. https://doi.org/10.1364/biomed.2014.bt3a.72.
McNamara PM, Dsouza R, O’Riordan C, Collins S, O’Brien P, Wilson C, et al. Development of a first-generation miniature multiple reference optical coherence tomography imaging device. J Biomed Opt. 2016;21:126020.
Compact Imaging. Compact imaging. https://compactimaging.com/. Accessed 29 Aug 2020.
Novartis. Novartis signs collaboration agreement to fund Medtech Development of low cost eye monitor. 2019. https://www.novartis.ie/stories/hope/novartis-signs-collaboration-agreement-fund-medtech-development-low-cost-eye-monitor. Accessed 3 Sep 2020.
Timoney PJ, Breathnach CS. Allvar Gullstrand and the slit lamp 1911. Ir J Med Sci. 2013;182:301–5.
Walsh AC. Binocular optical coherence tomography. Ophthalmic Surg Lasers Imaging. 2011;42(Suppl):S95–105.
Chopra R, Mulholland PJ, Dubis AM, Anderson RS, Keane PA. Human factor and usability testing of a binocular optical coherence tomography system. Transl Vis Sci Technol. 2017;6:16.
Chopra R, Mulholland PJ, Petzold A, Ogunbowale L, Gazzard G, Bremner F, et al. Automated pupillometry using a prototype binocular optical coherence tomography system. Am J Ophthalmol. 2020. https://doi.org/10.1016/j.ajo.2020.02.013.
Chopra R, Mulholland PJ, Tailor VK, Anderson RS, Keane PA. Use of a binocular optical coherence tomography system to evaluate strabismus in primary position. JAMA Ophthalmol. 2018;136:811–7.
Wittenborn JS, Clemons T, Regillo C, Rayess N, Liffmann Kruger D, Rein D. Economic evaluation of a home-based age-related macular degeneration monitoring system. JAMA Ophthalmol. 2017;135:452–9.
AREDS2-HOME Study Research Group, Chew EY, Clemons TE, Bressler SB, Elman MJ, Danis RP, et al. Randomized trial of a home monitoring system for early detection of choroidal neovascularization home monitoring of the Eye (HOME) study. Ophthalmology. 2014;121:535–44.
Notal Vision. Home OCT longitudinal home-based study with patient self-operated device has begun. GlobalNewsWire. 2020. https://www.globenewswire.com/news-release/2020/06/17/2049409/0/en/Home-OCT-longitudinal-home-based-study-with-patient-self-operated-device-has-begun.html. Accessed 20 Aug 2020.
Healio. Patients with AMD can successfully self-operate OCT device at home. https://www.healio.com/news/ophthalmology/20191111/patients-with-amd-can-successfully-selfoperate-oct-device-at-home. Accessed 15 Sep 2020.
Lally D, Kim JE, Elman MJ, Tomkins-Netzer O, Alon Y, Bergman E, et al. Performance of a novel deep learning algorithm for Automatic Retinal Fluid Quantification in Home OCT Images. Invest Ophthalmol Vis Sci. 2020;61:2571.
Sudkamp H, Koch P, Spahr H, Hillmann D, Franke G, Münst M, et al. In-vivo retinal imaging with off-axis full-field time-domain optical coherence tomography. Opt Lett. 2016;41:4987–90.
Sudkamp H, Hillmann D, Koch P, Endt MV, Spahr H, Münst M, et al. Simple approach for aberration-corrected OCT imaging of the human retina. Opt Lett. 2018;43:4224–7.
von der Burchard CC, Tode J, Ehlken C, Roider J. 2 mm Central macular volume scan is sufficient to detect exudative age-related macular degeneration activity in optical coherence tomography. Invest Ophthalmol Vis Sci. 2017;58:374.
Visotec. Visotec. https://visotec.health/. Accessed 20 August 2020.
Maloca P, Hasler PW, Barthelmes D, Arnold P, Matthias M, Scholl HPN, et al. Safety and feasibility of a novel sparse optical coherence tomography device for patient-delivered retina home monitoring. Transl Vis Sci Technol. 2018;7:8.
Li J-PO, Liu H, Ting DSJ, Jeon S, Chan RVP, Kim JE, et al. Digital technology, tele-medicine and artificial intelligence in ophthalmology: A global perspective. Prog. Retin. Eye Res. 2020: 100900.
Acknowledgements
The authors would like to thank Dr Adam Dubis for reviewing the manuscript.
Funding
PAK is supported by a Moorfields Eye Charity Career Development Award (R190028A) and a UK Research & Innovation Future Leaders Fellowship (MR/T019050/1). RC receives studentship support from the College of Optometrists, United Kingdom.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
PAK has acted as a consultant for DeepMind, Roche, Novartis and Apellis and is an equity owner in Big Picture Medical. He has received speaker fees from Heidelberg Engineering, Topcon, Allergan and Bayer. RC is an employee of Google LLC and owns Alphabet stock.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chopra, R., Wagner, S.K. & Keane, P.A. Optical coherence tomography in the 2020s—outside the eye clinic. Eye 35, 236–243 (2021). https://doi.org/10.1038/s41433-020-01263-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-020-01263-6
This article is cited by
-
The value of virtual glaucoma clinics: a review
Eye (2024)
-
Teleophthalmology and retina: a review of current tools, pathways and services
International Journal of Retina and Vitreous (2023)
-
Home vision monitoring in patients with maculopathy: current and future options for digital technologies
Eye (2023)
-
Transforming ophthalmology in the digital century—new care models with added value for patients
Eye (2023)
-
Highly sensitive V-shaped SPR PCF biosensor for cancer detection
Optical and Quantum Electronics (2023)