Abstract
Communicating results from genomic sequencing to family members can play an essential role allowing access to surveillance, prevention, treatment or prophylactic measures. Yet, many patients struggle with communication of these results and it is unclear to what extent this is discussed during the consent process. We conducted an online systematic search and used content analysis to explore how consent forms for genomic sequencing address communication of genetic information to family members. Our search yielded 68 consent forms from 11 countries. Although 57 forms alluded to the familial nature of results, forms varied in their discussion of the potential familial implications of results. Only 11 addressed communication of genetic information with family members, with differences in who would be responsible for this process. Several forms offered patients options regarding communication, even in countries where national guidelines and legislation allow for the disclosure of results in the absence of patient consent. These findings are concerning because they show how forms may potentially mislead patients and health care professionals about whether communication is permissible in cases where the patient does not consent. We suggest that providers and health care professionals reconsider how consent forms address communicating genetic information to family members.
Similar content being viewed by others
Introduction
Integration of genomic sequencing (GS), including genome/exome sequencing and gene panels, into healthcare is leading to increased rates of molecular diagnosis of genetic diseases [1, 2]. The fact that GS examines many genes simultaneously, some of which are not related to the genetic condition being investigated, means that it is more likely to identify both causative variants for the condition in question as well as unsolicited findings [3]. While genetic results may be considered very personal, they are also inherently familial [4] since results can have implications for those other than the patient for whom GS was requested. The identification of a certain condition in a patient is likely to indicate that another family member is at risk, either of developing the condition, passing the condition on to their offspring, or both. Informing relatives of these risks may enable them to undergo early screening, prevention, treatment, or utilize reproductive technologies to avoid having an affected child and thus they can potentially have significantly improved health outcomes for themselves and their future children [5].
Despite the benefits that can arise from the communication of genomic results, many patients struggle to initiate these conversations with family members [6,7,8,9,10,11,12,13]. Patients may be hesitant to share information that could cause their relatives distress. Furthermore, patients may experience concern regarding the impact of genetic findings on family relationships, although not communicating can also strain family relations [6, 14, 15]. Although health care professionals (HCPs) may try to explain the medical benefits of disclosure to their patient, not all patients agree to communicate results to family members. In cases where the patient refuses to disclose information to their relatives, HCPs are left with the dilemma of how to balance the patient’s right to privacy with the desire to prevent harm in their patient’s family members who are at risk. While this only occurs in a small minority of families that HCPs see, many HCPs encounter this dilemma over the course of their careers, yet feel that they lack the proper resources to help them make a decision to breach confidentiality [4, 7, 16, 17].
The uncertainty about this decision that HCPs may face is in part due to the fact that there is a lack of harmonization between guidelines and policies, as well as difficulties regarding their interpretation and implementation. While a minority of guidelines hold that the communication of genomic information is never permissible when the patient has not given consent to do so [18], most guidelines allow for the disclosure of results in cases where the genetic condition is considered both severe and actionable regardless of whether consent has been obtained [19,20,21,22,23,24].
Translating these guidelines into practice, however, can be challenging [25,26,27]. One area in which implementing guidelines into practice may be particularly difficult is in the consent process [28]. Consent has typically taken a more individual approach, while in contrast, genetic information is familial and relational [28]. Although it has been recommended that the consent process includes a discussion of the familial implications of results [19, 29,30,31], it is not clear to what degree this is reflected or even possible in practice.
While the consent process entails more than just the consent form, examining these documents can provide helpful insight into how different providers address communication of genetic information with family members. Consent forms can be an important way to document that patients have received certain information [32], and can play a key role in guiding discussions between HCPs and patients [33]. Despite the demonstrated importance of consent forms, previous analyses have observed high degrees of inconsistency between forms and even instances in which forms were not in accordance with guidelines [34].
To date, there has been no systematic analysis focused on how diagnostic consent forms for GS address the issues of communicating genetic information to family members. To address this gap, we used a systematic online search to identify diagnostic consent forms and explored how they address the familial nature, implications, and communication of GS results.
Materials and methods
Search strategy
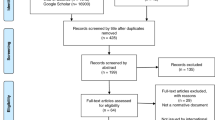
In order to obtain the list of consent forms, we utilized three complementary strategies: (1) an online search to identify consent forms used by clinics and diagnostic laboratories published online (from April 2016 to March 2019); (2) inclusion of forms identified from a previous dataset which utilized the same search strategy (from March and April 2016) [34, 35]; and (3) the use of forms known to the authors to be in use in the clinical setting, either via previous studies and/or professional contacts.
For the online search of consent forms, we used Google as the web search engine. The following search string was used: “(consent form OR informed consent OR consent document) AND (whole exome sequencing OR whole genome sequencing OR next generation sequencing OR genome wide sequencing).” We restricted the search to webpages published after 1 April 2016, the end data of the previous search. Each search was conducted by at least two researchers between January and March 2019. The researchers reviewed the first 10 pages (100 entries) of results, beyond which results were often redundant with no additional forms being identified.
Each form was independently assessed by at least 2 of the researchers using the following inclusion criteria: (1) a consent form requiring the signature of the patient, or their parent/guardian (i.e., not a model form, sample form, or requisition form); (2) explicitly for obtaining consent for large gene panels (20 or more genes tested), exome or genome sequencing; (3) for use in the clinical setting; and (4) in English. Forms in use for only research purposes were excluded. If an identified form was the same as from the previous dataset [34, 35], only the most recent version of the form was included.
Data analysis
The data analysis for this study reflected that of previous consent form content analyses [34, 35]. First, a deductive content analysis was conducted where, prior to the analysis of the consent forms, content categories and codes were developed based on the review of the literature and other relevant normative documents [36]. An inductive analysis of the forms was then conducted to further refine and supplement the codes.
Data for each consent form was coded, organized into subcategories, and compared across forms in an iterative manner whereby the results were compared, discrepancies discussed and definitions of the categories refined. Coding was performed by two researchers (AP and DV) and then verified for consistency with the data by two additional researchers (KK and EN).
Results
In total, 68 consent forms met our inclusion criteria. In total, 23 forms were from the previous dataset and 45 from the more recent search. In total, 12 forms were updated versions of those used in the previous dataset. These forms were from 54 separate institutions (either independent laboratories or laboratories affiliated with a hospital/medical center) from 11 different countries (Table 1: Genetic services characteristics). Three of these forms were specifically for use in adult patients, 5 for use specifically in pediatric patients, and 57 for use in either adult or pediatric patients. For three forms, it was unclear which patient group is addressed. Although forms varied in the type of GS they addressed (i.e., exome, genome, or targeted gene panels), we observed no difference between the forms regarding familial implications of results based on which type of test consent was being sought. Therefore, we have not separated forms on this basis.
The results of the analysis are reported under the following categories (Table 2: Categories addressed within the consent forms): (1) familial nature of GS results; (2) potential familial implications of results; and (3) communication of results with family members. Illustrative quotes from the forms are provided.
Familial nature of GS results
The majority of the forms (57/68) analyzed in this study included a statement indicating that genetics is familial in some way. The remaining forms gave no indication that GS results might have relevance to family members. For all but one of these forms, this is likely to be because the forms had almost no information regarding GS more broadly.
Several forms explicitly stated that GS results are relevant to family members.
“The test results, like the results of other genetic tests, may have implications for your relatives” (form 31).
In other forms, the link drawn between a patient’s genetic information and that of their family members was more implicit, such as inferring the relationship between the patient’s results and their family’s through language choices that include the family, rather than just the individual patient, as the subject of GS. One such form states that the purpose of testing is to detect a condition in “you or your family” (form 13), while another states that it is possible that sequencing “identifies the underlying genetic cause for the disorder in your family [emphasis added]” (form 7).
Potential familial implications of results
Not all forms discussed in detail the concrete implications for family members. Several forms only addressed familial implications in brief with statements such as “[g]enetic test results may impact family members” (form 44) with no further elaboration. Some forms that did address this issue mentioned the potential implications for family members in a separate section dedicated to the risks to family members, while others discussed it in conjunction with risks for patients.
These familial implications can be grouped in four categories: (1) clinical implications, (2) nonpaternity and unexpected relationships, (3) psychosocial concerns, and (4) privacy, discrimination, and insurance coverage issues. These categories of implications discussed by the forms also demonstrate the different types of results that may be communicated with family members.
Clinical implications
A substantial portion of the forms (46/68) stated that GS may have direct clinical implications for family members. Many of the forms did not elaborate further than a short statement.
“[t]he results of this test may show that I and/or my family members have an inherited disease or are at an increased risk to be affected by a genetic disease” (form 8).
Nonpaternity and unexpected relationships
The vast majority of the forms (41/68) mentioned the possibility that a genetic test result might identify nonpaternity or unexpected relationships, such as biological relatedness different than those reported by the proband. However, the framing of this issue varied, with some forms emphasizing the impact on the patient alone and others also considering the impact on the family. Most forms stated that they would not disclose if family relationships were not accurately reported by the patient unless it was medically necessary.
“Family relationship findings: …If the genetic analysis reveals a possibility that there is a discrepancy in the provided [familial] relationships, CENTOGENE will not inform you, unless in exceptional cases where this information is absolutely necessary for the completion and correct medical interpretation of the requested analysis” (form 14).
“In some cases, genetic testing can reveal that the true biological relationships in a family are not as they were reported. This includes nonpaternity (the stated father of an individual is not the biological father) and consanguinity (the parents of an individual are related by blood). It may be necessary to report these findings to the health care provider who ordered the test” (form 28).
Others merely stated that discovering unexpected biological relationships was a risk but did not elaborate on what would be done with this information if it was identified during the analysis.
Psychosocial concerns
Some forms (12/68) suggested that results from GS may cause “emotional or psychological distress” (forms 16 and 23) and have “social implications” (forms 1 and 46) for family members. These types of risks to family members are often discussed in conjunction with the similar risks that patients may face.
“The results of this test may have significant medical, psychological, and social implications for you and your family. You and your family members may experience anxiety before, during, and after testing” (form 46).
Privacy and discrimination
A minority of forms (4/68) stated that results may have implications for family members’ privacy and safety from discrimination, particularly related to insurance coverage.
“The test results may show some genetic differences that could affect the patient’s or other family members’ ability to buy life insurance, disability insurance, or long-term care insurance in the future” (form 18).
Communication of results with family members
Approximately one-sixth of forms (11/68) addressed the communication of genetic information with family members with considerable variation in their approaches. First, forms differed on the extent to which they presented family communication as a choice, rather than obligation, for patients. Some forms suggested that patients should communicate the results to family members.
“These secondary findings in risk genes may also have implications for your family. Based on your results we may suggest that you speak to a genetic counselor or discuss genetic testing on your family members. Any such additional testing would be discussed with the appropriate family member and addressed in a different consent” (form 23).
Other forms more clearly articulated family communication as a choice. Two approaches were used with regards to the stage in the process at which patients should make this decision. Some forms prompted patients to make this choice prior to sequencing through use of the opt-in model.
“I agree that the result may also be used, if necessary, to help other family members, for their counseling and diagnosis, without disclosing specific details about the person tested.
□ YES, I consent to results being made known if reasonably indicated to other family members
□ NO, I request that results only be made known to the following people…” (form 54).
"□ I hereby give consent for clinical, laboratory and genetic information that may be relevant to other family members to be made available to relevant health care professionals" (form 55).
These quotes also highlight differences in the framing of who is responsible for communicating genetic information to family members. While in most forms the onus of communication is placed on the patient, some suggest that the HCP could be the one responsible for communication. The forms outline two ways in which HCPs might communicate results with family members: (1) directly with the family members themselves through the contact information provided on the forms (as seen in form 54 above); or (2) by sharing genetic information with family members’ HCPs (as seen in form 55 above).
In contrast to the above examples in which patients are prompted to indicate their choices regarding family communication prior to testing, other forms presented communication as a choice to be discussed with an HCP or genetic counselor following sequencing.
“Speak with your physician or a genetic counselor about whether you should share your test results with others. If you decide to do this, consider the best way to communicate this information to them” (form 31).
A minority of forms used more directive language encouraging the patient to communicate their results to relevant family members.
“If the test finds a genetic change that may be important to your family’s health, your/your child’s healthcare provider will ask you to tell your family members about it” (form 16).
While these forms still suggest that patients can decide whether or not to communicate results, the information and language used incentivizes patients to inform their family members. None of the forms we identified indicated that family communication may be possible or even obligated in the absence of patient consent.
Discussion
This study is the first to systematically analyze how consent forms for GS address the communication of genetic information with family members. The majority of forms (57/68) stated that genomic information is familial in some way, and most (46/68) stated that GS may have direct clinical implications for family members. However, very few forms (11/68) explicitly discussed communicating genetic information to family members. This raises concerns over whether patients are sufficiently informed about the possibility that there could be a responsibility to disclose that information to their family members. It is important that patients are informed prior to testing that their results can have implications for them and their family members. Furthermore, they should be made aware that they may be asked to inform their family members, and that it may be possible (depending on the region in which testing is taking place) for their HCP to inform relatives. In this case, the patient should be made aware that their family would be informed of their genetic risk and given options regarding testing. The patient should be notified that although none of their personal information would be disclosed, anonymity might not be guaranteed. Informing patients prior to testing is important not only so that patients can make an informed choice, but by preparing patients for this possibility, it could also be an important factor in lowering the obstacles of communicating genetic information to family members. This statement is supported by empirical research, such as that conducted by Young et al. [37], in which HCPs cited cueing patients early to think about potential communication challenges as an important strategy for facilitating communication.
Of the forms that did address family communication, there appeared to be no unified approach among the providers of genetic services. The forms varied in the extent to which they encouraged family communication, some using more directive language than others, although it should be noted that even mentioning family communication in the forms may signal to both HCPs and patients that family communication is an important part of GS.
The forms also varied with regards to the timing of consent for family communication. Some forms indicated that decisions around family communication were to be made following testing with the assistance of a HCP or genetic counselor. Alternatively, other forms took a different approach and used an opt-in model that prompted patients to make decisions prior to testing about whether or not they wanted HCPs to communicate results and to whom. While patients and HCPs may benefit from an indication in the forms of the potential need for communicating genetic information to family members, having patients make decisions about this prior to testing may be problematic. It might be difficult to anticipate the outcomes of a test beforehand. Even with the most thorough consent process, it is not possible to address every possible outcome and its implications, particularly in the case of GS which can yield unexpected results and secondary findings. Patients may generally be in favor of family communication, but they may still find certain results more difficult or even less desirable to share following testing [14]. Although anticipating the discussion about informing family members might be a preferable approach, it raises a potential ethical dilemma in cases where the patient indicates that they do not intend to inform family members. As such, it relocates issues that would normally need to be addressed in the post-test counseling session to the pre-test setting.
Notably, none of the forms analyzed in this study addressed whether genomic information could be communicated to family members in the absence of consent to disclosure of this information. In the forms that discussed family communication, most seemed to indicate that patients could decide whether or not to communicate genetic information with none suggesting that this decision might not be respected in certain cases. This was found even in the countries with clear guidelines or legislation permitting for exceptional breaches in confidentiality. For example, in the UK, the Joint Committee on Genomics in Medicine holds that “the rule of confidentiality is not absolute” [19]. They elaborate, “The assumption that confidentiality is always paramount is as inappropriate as the assumption that disclosure is always permissible, and the decision will need to be tailored to the individual circumstances of the case.” And yet, despite these guidelines, in the forms analyzed from the UK, patients could opt-in to the communication of results with family members. Similarly, this was found in an Australian form where national guidelines and legislation permit HCPs to breach confidentiality [22, 38,39,40]. It is not clear to what extent patient refusal to consent would be respected in these cases, but forms seemed to indicate that the patient’s decision to communicate or not would be upheld, despite the fact that this contradicts guidelines and, in the case of Australia, legislation. Offering patients a choice regarding familial communication seems in direct contrast with guidelines of the UK General Medical Council which states, “You should not ask for consent if you have already decided to disclose information in the public interest but you should tell the patient about your intention to disclose personal information” [21].
The findings of this study are concerning because they show how forms may potentially mislead both patients and HCPs about whether communication is permissible or not in cases where the patient does not consent to the disclosure of information to family members. The results raise questions about whether there is adequate education and training of providers and/or HCPs who offer GS, especially with regards to the ethical problems such testing raises. Furthermore, a question may be posed as to whether the potential breach of confidentiality in the context of GS raises concerns among the providers, which could be the reason why it has not been addressed in line with guidelines. More research is needed to better understand the underlying cause for the discrepancy between guidelines and consent forms observed in this study.
Limitations
Our sample was limited by language as well as the public internet accessibility of consent forms. As such, although our sample had representation from 11 countries, the majority of the forms were from American genetic services, and thus our findings are not generalizable to all countries. Further research on consent forms in languages other than English would help form a more complete picture of how GS providers address family communication. Nonetheless, our analysis still provides important insight into the practices of over 50 different genetic providers and highlights issues regarding both the lack of harmonization between forms and also inconsistencies between forms and guidelines regarding communicating genetic information to family members.
Another limitation of any analysis of consent forms is that the content of consent forms are only one component of the consent process and we cannot be certain of what else is communicated in the interaction between the patient and the HCP. To better understand this, an analysis of other informational materials and observations of consultations should be conducted. That being said, analyzing consent forms provides valuable insight into the consent process as forms may serve as a guide for discussions in addition to being a means to verifying that patients have received certain information [32]. This claim has been verified by empirical studies in which researchers found that “HCPs saw consent forms as sometimes helpful because they prompted and structured their discussions with patients” [33].
References
Basel D, McCarrier J. Ending a diagnostic odyssey: Family education, Counseling, and response to eventual diagnosis. Vol. 64, Pediatric Clinics of North America. W.B. Saunders; 2017. p. 265–72.
Hall A, Finnegan T, Alberg C, Burton H, Hallowell N, Kroese M, et al. Realising genomics in clinical practice PHG foundation project team [Internet]. 2014 [cited 2019 Nov 8]. www.phgfoundation.org.
Van ElCG, Cornel MC, Borry P, Hastings RJ, Fellmann F, Hodgson SV, et al. Whole-genome sequencing in health care. Eur J Hum Genet [Internet]. 2013;21:580–4. http://www.ncbi.nlm.nih.gov/pubmed/23676617.
Falk MJ, Dugan RB, O’Riordan MA, Matthews AL, Robin NH. Medical geneticists’ duty to warn at-risk relatives for genetic disease. Am J Med Genet A. 2003;120A:374–80.
Godino L, Turchetti D, Jackson L, Hennessy C, Skirton H. Impact of presymptomatic genetic testing on young adults: A systematic review. Eur J Hum Genet Nat Publ Group. 2016;24:496–503.
Black L, McClellan KA, Avard D, Knoppers BM. Intrafamilial disclosure of risk for hereditary breast and ovarian cancer: points to consider. J Community Genet. 2013;4:203–14.
Clarke A, Richards M, Kerzin-Storrar L, Halliday J, Young MA, Simpson SA, et al. Genetic professionals’ reports of nondisclosure of genetic risk information within families. Eur J Hum Genet [Internet]. 2005 May [cited 2019 Nov 6];13:556–62. http://www.ncbi.nlm.nih.gov/pubmed/15770225.
Gaff CL, Clarke AJ, Atkinson P, Sivell S, Elwyn G, Iredale R, et al. Process and outcome in communication of genetic information within families: a systematic review. Eur J Hum Genet. 2007;15:999–1011.
Arribas-Ayllon M, Featherstone K, Atkinson P. The practical ethics of genetic responsibility: Non-disclosure and the autonomy of affect. Soc Theory Heal. 2011;9:3–23. https://doi.org/10.1057/sth.2009.22.
Geelen E, Van Hoyweghen I, Horstman K. Making genetics not so important: family work in dealing with familial hypertrophic cardiomyopathy. Soc Sci Med. 2011;72:1752–9.
McClellan KA, Kleiderman E, Black L, Bouchard K, Dorval M, Simard J, et al. Exploring resources for intrafamilial communication of cancer genetic risk: we still need to talk. Eur J Hum Genet. 2013;21:903–10.
Wauters A, Van Hoyweghen I. Global trends on fears and concerns of genetic discrimination: a systematic literature review. J Hum Genet. 2016;61:275–82.
Suthers GK, Armstrong J, McCormack J, Trott D. Letting the family know: balancing ethics and effectiveness when notifying relatives about genetic testing for a familial disorder. J Med Genet. 2006;43:665–70. http://www.ncbi.nlm.nih.gov/pubmed/16371501.
Derbez B. Is there a “right time” for bad news? Kairos in familial communication on hereditary breast and ovarian cancer risk. Soc Sci Med. 2018;202:13–9.
Chivers Seymour K, Addington-Hall J, Lucassen AM, Foster CL. What facilitates or impedes family communication following genetic testing for cancer risk? A systematic review and meta-synthesis of primary qualitative research. J Genet Couns. 2010;19:330–42.
Dheensa S, Fenwick A, Lucassen A. Approaching confidentiality at a familial level in genomic medicine: a focus group study with healthcare professionals. BMJ Open. 2017;7:e012443. http://bmjopen.bmj.com/content/7/2/e012443.abstract.
D’Audiffret Van Haecke D, de Montgolfier S. Genetic test results and disclosure to family members: qualitative interviews of healthcare professionals’ perceptions of ethical and professional issues in France. J Genet Couns. 2016;25:483–94.
Institute of Medicine (US) Committee on Assessing Genetic Risks; Andrews LB, Fullarton JE, Holtzman NA, Motulsky AG, editors. Assessing Genetic Risks: Implications for Health and Social Policy. Washington (DC): National Academies Press; 1994.
Royal College of Physicians. Consent and confidentiality in genomic medicine Guidance on the use of genetic and genomic information in the clinic. Report of the Joint Committee on Medical Genetics. 2019; 3rd edition.
ASHG. ASHG statement. Professional disclosure of familial genetic information. The American Society of Human Genetics Social Issues Subcommittee on Familial Disclosure. Am J Hum Genet. 1998;62:474–83.
General Medical Council. Confidentiality: good practice in handling patient information. General Medical Council, 2017. https://www.gmc-uk.org/Confidentiality_good_practice_in_handling_patient_information___English_0417.pdf_70080105.pdf.
National Health and Medical Research Council, Office of the Privacy Commissioner. Use and disclosure of genetic information to a patient's genetic relatives under section 95AA of the Privacy Act 1988 (Cth). Guidelines for health practitioners in the private sector. Canberra: NHMRC; 2009. https://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/e96.pdf.
Menko FH, Aalfs CM, Henneman L, Stol Y, Wijdenes M, Otten E, et al. Informing family members of individuals with Lynch syndrome: a guideline for clinical geneticists. Fam Cancer 2013;12:319–24.
Forrest LE, Delatycki MB, Skene L, Aitken MA. Communicating genetic information in families—A review of guidelines and position papers. Eur J Hum Genet. 2007;15:612–8.
Dove ES, Chico V, Fay M, Laurie G, Lucassen AM, Postan E. Familial genetic risks: how can we better navigate patient confidentiality and appropriate risk disclosure to relatives? J Med Ethics. 2019 Aug 1 [cited 2019 Nov 6];45:504 LP – 507. http://jme.bmj.com/content/45/8/504.abstract.
Lucassen A, Gilbar R. Disclosure of genetic information to relatives: balancing confidentiality and relatives’ interests. J Med Genet. 2018;55:285–6.
Dheensa S, Fenwick A, Shkedi-Rafid S, Crawford G, Lucassen A. Health-care professionals’ responsibility to patients’ relatives in genetic medicine: a systematic review and synthesis of empirical research. Genet Med [Internet]. 2016 [cited 2019 Nov 6];18:290–301. http://www.ncbi.nlm.nih.gov/pubmed/26110233.
Dove ES, Kelly SE, Lucivero F, Machirori M, Dheensa S, Prainsack B. Beyond individualism: is there a place for relational autonomy in clinical practice and research? Clin Ethics. 2017 Sep 13 [cited 2019 Nov 8];12:150–65. http://journals.sagepub.com/10.1177/1477750917704156.
Niemiec E, Howard HC. Consenting patients to genome sequencing. In: Clinical genome sequencing: psychological considerations. Academic Press; 2019.
Ayuso C, Millán JM, Mancheño M, Dal-Ré R. Informed consent for whole-genome sequencing studies in the clinical setting. Proposed recommendations on essential content and process. Eur J Hum Genet. 2013;21:1054–9.
Jamal SM, Yu JH, Chong JX, Dent KM, Conta JH, Tabor HK, et al. Practices and Policies of Clinical Exome Sequencing Providers: Analysis and Implications. Am J Med Genet Part A. 2013;161:935–50.
Cattapan AR. Good eggs? Evaluating consent forms for egg donation. J Med Ethics. 2016;42:455–9.
Samuel GN, Dheensa S, Farsides B, Fenwick A, Lucassen A. Healthcare professionals’ and patients’ perspectives on consent to clinical genetic testing: moving towards a more relational approach. BMC Med Ethics. 2017;18:47.
Vears DF, Niemiec E, Howard HC, Borry P. How do consent forms for diagnostic high-throughput sequencing address unsolicited and secondary findings? A content analysis. Clin Genet. 2018;94:321–9.
Vears DF, Niemiec E, Howard HC, Borry P. Analysis of VUS reporting, variant reinterpretation and recontact policies in clinical genomic sequencing consent forms. Eur J Hum Genet. 2018;26:1743–51.
Krippendorff K. Reliability in content analysis. Hum Commun Res. 2004 Jul [cited 2019 Nov 5];30:411–33. https://academic.oup.com/hcr/article/30/3/411-433/4331534.
Young AL, Butow PN, Tucker KM, Wakefield CE, Healey E, Williams R. Challenges and strategies proposed by genetic health professionals to assist with family communication. Eur J Hum Genet. 2019;27:1630–8.
Otlowski MFA. Disclosing genetic information to at-risk relatives: new Australian privacy principles, but uniformity still elusive. Med J Aust. 2015;202:335–7.
McWhirter R, Johnston C, Burke J. Disclosure of genetic results to at-risk relatives without consent: issues for Health Care Professionals in Australia. J Law Med. 2019 Oct [cited 2019 Nov 6];27:108–21. http://www.ncbi.nlm.nih.gov/pubmed/31682345.
Privacy Legislation Amendment Act 2006.
Acknowledgements
We would like to thank Janneke Kuiper and Ine Van Hoyweghen from the KU Leuven Faculty of Social Sciences for their assistance with the refinement of this manuscript.
Funding
DV is a Postdoctoral Research Fellow of the Research Foundation–Flanders (FWO Vlaanderen) and acknowledges the infrastructure funding received from the Victorian State Government through the Operational Infrastructure Support (OIS) Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Phillips, A., Niemiec, E., Howard, H.C. et al. Communicating genetic information to family members: analysis of consent forms for diagnostic genomic sequencing. Eur J Hum Genet 28, 1160–1167 (2020). https://doi.org/10.1038/s41431-020-0627-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41431-020-0627-7
This article is cited by
-
Genetic Testing for Cancer Risk and Perceived Importance of Genetic Information Among US Population by Race and Ethnicity: a Cross-sectional Study
Journal of Racial and Ethnic Health Disparities (2024)
-
Genetic counselling and testing for neurodegenerative disorders using a proposed standard of practice for ALS/MND: diagnostic testing comes first
European Journal of Human Genetics (2022)
-
Comparing the attitudes of physicians and non-physicians toward communicating a patient’s BRCA1 mutation to a first-degree relative against a patient’s wishes
Journal of Community Genetics (2022)