Abstract
Gut dysbiosis is associated with sepsis and necrotizing enterocolitis in preterm infants, which can adversely affect long-term growth and neurodevelopment. We aimed to synthesise evidence for the effect of probiotic supplementation on growth and neurodevelopmental outcomes in preterm infants. MEDLINE, EMBASE, EMCARE, Cochrane CENTRAL, and grey literature were searched in February 2022. Only randomized controlled trials (RCTs) were included. Meta-analysis was performed using random effects model. Effect sizes were expressed as standardized mean difference (SMD), mean difference (MD) or risk ratio (RR) and their corresponding 95% confidence intervals (CI). Risk of Bias (ROB) was assessed using the ROB-2 tool. Certainty of Evidence (CoE) was summarized using GRADE guidelines. Thirty RCTs (n = 4817) were included. Meta-analysis showed that probiotic supplementation was associated with better short-term weight gain [SMD 0.24 (95%CI 0.04, 0.44); 22 RCTs (n = 3721); p = 0.02; I2 = 88%; CoE: low]. However, length [SMD 0.12 (95%CI −0.13, 0.36); 7 RCTs, (n = 899); p = 0.35; I2 = 69%; CoE: low] and head circumference [SMD 0.09 (95%CI −0.15, 0.34); 8 RCTs (n = 1132); p = 0.46; I2 = 76%; CoE: low] were similar between the probiotic and placebo groups. Probiotic supplementation had no effect on neurodevelopmental impairment [RR 0.91 (95%CI 0.76, 1.08); 5 RCTs (n = 1556); p = 0.27; I2 = 0%; CoE: low]. Probiotic supplementation was associated with better short-term weight gain, but did not affect length, head circumference, long-term growth, and neurodevelopmental outcomes of preterm infants. Adequately powered RCTs are needed in this area. Prospero Registration: CRD42020064992.
Similar content being viewed by others
Introduction
Survival of preterm infants has significantly improved due to advances in perinatal and neonatal care [1,2,3]. While there is a trend towards decreased disability rates, a significant proportion of survivors suffer from disabilities. Cheong et al. (Australia) compared the outcomes of extreme premature infants over four longitudinal cohorts: 1991–1992 (n = 422), 1997 (n = 215), 2005 (n = 263), and 2016-2017 (n = 252). Survival to 2 years was 53% vs 70% vs 63% vs 73% indicating that the most recent cohort had highest survival rates. The rates of major neurodevelopmental disability were similar across the study epochs (20% vs 26% vs 15% vs 15%). However, survival free of major disability increased steadily over time: 42% vs 51% vs 53% vs 62% (P < 0.001) [4]. Pierrat et al. (France) reported the neurodevelopmental outcomes at 2 years corrected age for children born at < 34 weeks in 2011 and evaluated changes since 1997. Among 5170 neonates, survival at 2 years corrected age was 51.7% at 22–26 weeks’ gestation, 93.1% at 27–31 weeks’ gestation, and 98.6% at 32–34 weeks’ gestation. Survival without severe or moderate disabilities increased between 1997 and 2011, from 45.5% to 62.3% at 25–26 weeks’ gestation, but no change was observed at 22–24 weeks’ gestation. At 32–34 weeks’ gestation, the proportion of survivors with cerebral palsy (CP) declined [5]. Bell et al. (US-NICHD) reviewed the outcomes at 22–26 months’ corrected age for extremely preterm infants. The study included 10877 extremely preterm infants born between 2013 and 2018. Outcomes were compared with a similar cohort of infants born in 2008–2012. Survival to discharge increased from 76% in 2008–2012 to 78.3% in 2013–2018. Among 2458 fully evaluated infants, 48.7% (1198/2458) had no or mild, 29.3% (709/2419) had moderate, and 21.2% (512/2419) had severe neurodevelopmental impairment [6].
Hence, the focus has shifted to optimising neurodevelopmental outcomes in this population.
Gut dysbiosis, with decreased abundance of Bifidobacteria and Lactobacilli, and increased abundance of pathogenic bacteria (E. coli, Pseudomonas, Staphylococci, Enterobacter, Clostridia) is known to precede necrotizing enterocolitis (NEC) and late onset sepsis (LOS) in very preterm infants [7,8,9,10]. In turn, NEC and LOS are known to be associated with increased mortality and adverse long-term effects on growth and neurodevelopment [11,12,13,14,15,16]. Evidence from randomised as well as non-randomised studies indicates that probiotic supplementation attenuates dysbiosis and thereby reduces the risk of NEC ≥ Stage II, LOS, and mortality and improves feeding tolerance in preterm infants [17,18,19,20,21]. Probiotic supplementation can improve nutrition through its trophic effects on intestinal villi, peristalsis, and by reducing the risk of NEC, sepsis, and feeding intolerance [19,20,21]. Through these beneficial effects, probiotics have the potential to improve growth and neurodevelopmental outcomes of preterm infants. In addition, a healthy gut microbiome is known to play an important role in brain development and a healthy gut brain axis [22, 23]. Overall, probiotics have the potential to be neuroprotective considering their direct (e.g.: regulation of gene expression, synthesis of neurotransmitters, and expression of neurotrophic growth factors in the brain, and reduced neuroinflammation due to anti-inflammatory properties) and indirect effects (e.g., reduced risk of NEC, LOS, improved nutrition, and nutrient absorption) [16, 22, 24].
In their prospective observational study in 17 preterm very low birth weight (VLBW) infants, Beghetti et al. reported that bifidobacterial abundance on day 30 was positively correlated with neurodevelopment at 24-months (p = 0.001). They also reported that B. longum and B. breve were absent in the gut microbiota of infants with neurodevelopmental impairment [23].
Given these data, the importance of assessing long-term growth and neurodevelopmental outcomes in probiotic supplemented preterm infants cannot be overemphasised. Upadhyay et al. reported a meta-analysis of data from randomised controlled trials (RCTs) evaluating the effect of prebiotic and probiotics on neurodevelopment in preterm VLBW ( < 1500 g) infants [25]. A total of 7 RCTs were included, of which six involved preterm infants < 33 weeks of gestation. Long-term outcomes were assessed at ≥18–22 months of corrected age in five RCTs. They reported that probiotic supplementation had no effect on the risk of cognitive and motor impairment, CP, and visual, and hearing impairment. The quality of evidence was deemed to be “low” to “very low.” Subsequently, more RCTs have been published evaluating growth and neurodevelopment and hence, we aimed to update the current evidence in this field.
Methods
Guidelines from the Cochrane handbook (https://training.cochrane.org/handbook/current), and the PRISMA statement were followed for conducting and reporting this systematic review [26]. Ethics approval was not required. Databases MEDLINE via PubMed (www.ncbi.nlm.nih.gov, 1966-February 2022), EMBASE via Ovid (http://ovidsp.tx. ovid.com, 1980 to February 2022), Cochrane Central Register of Controlled Trials (www.thecochranelibrary.com, through February 2022) and EMCARE via Ovid (http://ovidsp.tx.ovid.com, 1980 to February 2022) were searched by two authors. Grey literature was searched using the national technical information services (http://www.ntis.gov/), Open Grey (http://www.opengrey.eu/) and Trove (http://trove.nla.gov.au/). The reference lists of eligible studies and review articles were searched to identify additional studies. No language restriction was applied.
The following terms were used for searching on PubMed: (((“Probiotics”[Mesh]) OR (“Growth and Development”[Mesh] OR “Growth”[Mesh] OR “growth and development” [Subheading])) AND (“Infant, Extremely Premature”[Mesh] OR “Premature Birth”[Mesh] OR “Infant, Premature”[Mesh])) OR “Infant, Small for Gestational Age” OR “Infant, extremely low birth weight”[Mesh] OR Infant, low birth weight”[Mesh] [Majr]. Search was repeated using relevant keywords. Other databases were searched using similar terminologies. Additional studies were identified from the cross references of relevant studies.
RCTs comparing probiotics against placebo/control in preterm infants (<37wk) were included. RCTs assessing prebiotics/synbiotics only were excluded. RCT including probiotic along with prebiotic/synbiotic groups, was included and data from the probiotic group was used for review.
Abstracts of citations from the initial search were read to identify potentially eligible studies. Full-text articles of such identified studies were independently assessed for eligibility by all reviewers. Reference lists from included studies were reviewed to identify additional studies. Following full text review, data were extracted by HP and verified by GAJ. If the included studies had summarised continuous outcomes using median, IQR or range, the formula by Wan et al. [27] was used to convert them into mean and SD.
Outcomes of interest
1) Short-term growth (weight, length, and head circumference) during hospital stay and/or at discharge. 2) Long-term growth and neurodevelopmental outcomes including cognition, language, CP, deafness, blindness, and autism spectrum disorder (ASD). Pre-planned subgroup analyses included: 1) Gestation < 28 weeks or birth weight < 1000 grams; 2) Single vs. multi-strain probiotics.
Assessment of risk of bias
Risk of bias for each outcome within a study was assessed using the Cochrane ‘Risk of Bias Assessment Tool-2 (ROB-2) [28]. The ROB-2 assesses the RCTs on the following five domains: Bias arising from the randomisation process, Bias due to deviations from intended interventions, Bias due to missing outcome data, Bias in measurement of the outcome and Bias in selection of the reported result. Under each domain, 3–7 signalling questions are asked, based on which judgement is made if the ROB was low/high/some concerns. Finally, a judgement is made on the overall bias rating as low/high/some concerns.
Statistical analysis
Meta-analysis was performed using Review Manager (version 5.4.1, Cochrane Collaboration, Nordic Cochrane Centre, Copenhagen, Denmark). Random effects model (REM) was used. For data not suitable for meta-analysis, results have been given in a tabular format. Standardized Mean Difference (SMD), Mean Difference (MD) and 95% confidence interval (CI) were calculated for continuous variables. Relative risk (RR) and 95% CI were used for dichotomous variables.
Statistical heterogeneity across studies was quantified using the I2 statistic. An I2 value of > 50% was considered to indicate substantial heterogeneity [29]. Publication bias was assessed using Stata 16.0 (StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC) by Egger’s test, Begg’s test and by inspecting funnel plots wherever there were more than 10 RCTs in the meta-analysis [30,31,32].
The certainty of evidence (CoE) was rated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) framework [33].
Results
Selection, characteristics, and quality of studies
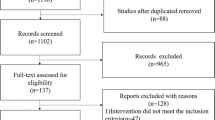
Literature search retrieved 5085 potentially relevant citations. Reviewers HP and GAJ independently completed initial screening of the titles and abstracts, full-text publications of potential studies and published review articles on probiotics. Of the 378 records identified via databases and registers, 208 records were screened of which 98 reports were assessed for eligibility after screening title/abstract. Of the 4707 records identified from other sources such as websites (Google scholar, Open Grey, NTIS, Trove), 471 reports were assessed for eligibility after title/abstract screen. Discrepancies about inclusion or exclusion of studies and interpretation of data were resolved by discussion among all authors. Included study manuscripts were manually reviewed for references to identify key studies to add to final list of eligible studies. After screening the title/abstract, 569 studies were assessed for eligibility, of which 534 were excluded (Fig. 1). Finally, 30 RCTs (n = 4817) with 35 publications were included. Their characteristics are summarized in (Table 1, Supplementary Tables 1 and 2).
Characteristics of included studies
A total of twenty-seven RCTs [34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61] (n = 4018) reported effects of probiotics on short-term growth. Seven RCTs [62,63,64,65,66,67,68] (n = 1982) reported on neurodevelopmental outcomes, of which four [65,66,67,68] (n = 1417) reported on long-term growth. The results of the PROPEL trial [34, 37], Patole et al. [46, 62], Totsu et al. [47, 65], Sari et al. [54, 67] and PROPREMS trial [50, 66] were reported as two separate publications each for different outcomes.
Twenty-two RCTs were single centre [35, 36, 39, 40, 42, 43, 45, 46, 48, 49, 51, 53,54,55,56,57,58, 60,61,62,63,64, 67, 68], while eight were multicentre RCTs [34, 37, 38, 41, 44, 47, 50, 52, 59, 65, 66]. Primary and secondary outcomes in the included studies varied (Supplementary Table 2).
Single-strain probiotic was used in 21 RCTs: Bifidobacteria [36, 44, 46, 47, 56, 57, 60, 62, 65], Lactobacillus [34, 35, 37, 38, 45, 53,54,55, 61, 63, 64, 67] and Saccharomyces [42, 49, 51, 59]. Multi-strain probiotic was used in nine RCTs including one that used two Bifidobacterium strains from same genus [41], whereas eight used a combination of probiotic strains from different genera [39, 40, 43, 48, 50, 52, 58, 66, 68].
Placebo was used for comparison in 17 RCTs: maltodextrin [34, 37, 41, 44, 46, 47, 50, 53, 59, 62, 65, 66], medium chain triglyceride (MCT) oil [48], combination of MCT oil and sunflower oil [38], distilled water [45, 51, 60], human milk fortifier [56], milk formula [57, 61] and unspecified formulation [36, 55]. Remaining 13 RCTs used control/no treatment.
Four RCTs used only expressed breast milk (EBM) [43, 45, 63, 68], while 4 used pasteurized donor human milk (PDHM) along with EBM [34, 37, 39, 48, 56]. Fourteen RCTs used a combination of EBM or PDHM with infant formula [36, 40, 41, 44, 46, 47, 49,50,51, 54, 58, 60,61,62, 64,65,66,67]. Seven RCTs used formula only for feeds [35, 38, 42, 53, 55, 57, 59], while one did not specify the type of milk [52].
Thirteen RCTs assessed growth [35, 36, 38, 40,41,42,43, 48, 52, 56, 59, 65, 67] and 7 assessed long-term neurodevelopment as a primary outcome [62,63,64,65,66,67,68].
The 30 included RCTs had significant variation in the dosage of probiotics ranging from 2 million [40] to 10 billion colony forming units per day [47]. The duration of supplementation ranged from 21 days [56] to 6 weeks [41]. Some studies used corrected gestational age (CGA) as an endpoint [37, 46] whereas few continued the supplementation until discharge [51] or achieving weight > 2000g [47].
Probiotic and placebo group data was included from the RCT by Dilli et al. [44] and data from prebiotic/synbiotics group was excluded in the review. Similarly, Data only from the HIV-unexposed group was used from RCT by Van Niekerk et al. [48].
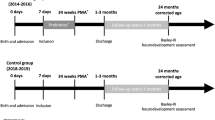
Growth was assessed at different time points (28 RCTs, n = 4311), ranging from day 14 [49, 54] to 6 weeks of age [58], to discharge [46]. The timing of assessment of long-term growth varied from 18 months [65] to 3 years of age [68].
Five RCTs reported on neurodevelopmental outcomes (motor, sensory or cognitive) [63, 65,66,67,68]. Out of them, three assessed children using Bayley Scales for Infant Development-second edition at 18–24 months [63, 67] or at three years [68] post-term. Jacobs et al. [66] assessed children at 2–5 years using Bayley-III, Movement Assessment Battery for Children and Wechsler Preschool and Primary Scale of Intelligence full scale Intelligence Quotient (IQ). Totsu et al. [65] used the Kyoto Scale of Psychological Development 2001 (equivalent to Bayley-III) at 18 months. Follow up rates varied from 48% [66] to 90% [68].
Outcomes
The results of the systematic review including 30 RCTs (35 publications) are summarised in Table 2 and Supplementary Table 3.
Results of meta-analysis are presented in Supplementary Table 4.
Short-term growth
Twenty-two RCTs [35, 37,38,39, 41,42,43,44,45,46,47,48,49,50,51,52, 54, 55, 57,58,59, 61] (n = 3721) reported on weight gain. Meta-analysis showed that probiotic supplemented infants had significantly better weight gain [SMD 0.24 (95%CI 0.04, 0.44); p = 0.02; I2 = 88%; CoE: low] (Fig. 2). Meta-analysis of seven studies [35, 37, 41, 42, 44, 48, 57] (n = 899) showed no difference in length gain between groups [SMD 0.12 (95%CI −0.13, 0.36); p = 0.35; I2 = 69%; CoE: low] (Supplementary Fig. 1). Meta-analysis of eight studies [35, 37, 41, 42, 44, 47, 48, 57] (n = 1132) showed no difference in head circumference gain between groups [SMD 0.09 (95%CI −0.15, 0.34); p = 0.46; I2 = 76%; CoE: low] (Supplementary Fig. 2).
Meta-analysis showed that probiotic supplementation was associated with better short-term weight gain (p = 0.02). Subgroup analysis showed better short-term weight gain (p = 0.04) in single-strain supplemented infants. This analysis was conducted using a random effects model. CI confidence interval, I2 heterogeneity, IV inverse variance, SE standard error.
Long-term growth
Four studies [65,66,67,68] reported on long-term growth. Meta-analysis showed no significant difference in weight [n = 1326; SMD −0.08 (95%CI −0.29, 0.12); p = 0.42; I2 = 68%; CoE: very low], length [n = 1325; SMD −0.03 (95%CI −0.14, 0.07); p = 0.53; I2 = 0%; CoE: very low], and head circumference between the groups [n = 1298; SMD −0.04 (95%CI −0.14, 0.07); p = 0.52; I2 = 0%; CoE: very low] (Supplementary Figs. 3–5).
Long-term neurodevelopmental outcomes
Cognitive outcomes
Meta-analysis of four RCTs [63, 66,67,68] (n = 1388) showed no significant difference in cognitive impairment between probiotic and control group [RR 0.98 (95%CI 0.75, 1.26); p = 0.85; I2 = 0%; CoE: very low] (Supplementary Fig. 6). Meta-analysis of five RCTs [63, 65,66,67,68] (n = 1507) showed no difference in mean cognitive scores between the two groups [MD 0.13 95%CI −1.41, 1.67); p = 0.87; I2 = 0%; CoE: very low] (Supplementary Fig. 7).
Motor outcomes
Meta-analysis of four RCTs [63, 66,67,68] (n = 1388) showed no significant difference in motor impairment between probiotic and placebo groups [RR 1.06 (95%CI 0.79, 1.41); p = 0.71; I2 = 0%; CoE: very low] (Supplementary Fig. 6). Meta-analysis of four RCTs [63, 66,67,68] (n = 1388) showed no difference in mean motor scores between the two groups [MD 1.04 (95%CI −0.43, 2.50); p = 0.16; I2 = 0%; CoE: very low] (Supplementary Fig. 7).
Overall neurodevelopmental, hearing, and visual impairment, CP and ASD
Meta-analysis of five RCTs [63, 65,66,67,68] showed no difference in neurodevelopmental impairment between groups [n = 1556; RR 0.91(95%CI 0.76–1.08); p = 0.27; I2 = 0%; CoE: low]. There was no difference in the incidence of CP [5 studies [63, 65,66,67,68], (n = 1588); RR 1.11(95%CI 0.64–1.91); p = 0.70; I2 = 30%; CoE: very low]. Meta-analysis of four RCTs [63, 66,67,68] (n = 1388) showed no difference in the incidence of hearing impairment [RR 0.7 (95%CI 0.17–2.95; p = 0.62; I2 = 35%; CoE: low] or visual impairment between groups [RR 0.52 (95%CI 0.12–2.21); p = 0.38; I2 = 0%; CoE: very low]. Meta-analysis of effect of probiotics on overall neurodevelopmental impairment, CP, hearing impairment and visual impairment showed no difference between groups (Fig. 3). None of the included studies reported on ASD.
Subgroup analysis: (i) Gestation < 28 weeks or birthweight < 1000g
Three RCTs were eligible [37, 48, 52]. Meta-analysis of three RCTs (n = 335) showed no difference in short term weight gain [SMD 0.05 (95%CI −0.29, 0.38); p = 0.79; I2 = 60%] (Supplementary Fig. 8). Meta-analysis of two RCTs [37, 48] (n = 234) showed no difference in short term length gain [SMD −0.10 (95%CI −0.36, 0.15); p = 0.43; I2 = 0%] or head circumference gain [SMD 0.04 (95%CI −0.24, 0.32); p = 0.77; I2 = 17% (Supplementary Figs. 9–10).
(ii) Single-strain and multi-strain probiotics
Meta-analysis of 15 RCTs [35, 37, 38, 42, 44,45,46,47, 49, 51, 54, 55, 57, 59, 61] using single strain probiotic (n = 1916) showed better short-term weight gain [SMD 0.34 (95%CI 0.02, 0.65); p = 0.04; I2 = 91%]. Meta-analysis of 7 RCTs [39, 41, 43, 48, 50, 52, 58] using multi-strain probiotics (n = 1805) showed no difference in short-term weight gain [SMD 0.08 (95%CI −0.12, 0.27); p = 0.46; I2 = 64%] (Fig. 2).
Summary of outcomes from studies not included in meta-analysis
Three RCTs [36, 56, 60] (n = 195) reported significantly better short-term weight gain, Romeo et al. [64] (n = 249) reported improved neurodevelopment in the probiotic group at 12 months CGA (Table 1).
Risk of bias assessment for studies reporting on growth showed that 15.6% had high risk, 18.8% had some concerns, whereas 65.6% had low risk. Five RCTs seemed to have overall high ROB for assessing growth as outcome, whereas six studies had some concerns. A total of 21 studies were assessed as having low ROB (Supplementary Fig. 11). Risk assessment for studies reporting long-term neurodevelopmental outcomes showed 57.1% had high ROB, 14.3% had some concerns and 28.6% had low ROB (Supplementary Fig. 11). Missing data on long term neurodevelopmental outcomes of participants in the RCTs was a major reason for ‘high ROB.’
Funnel plot for comparison of probiotics for short term weight gain appeared to have no asymmetry (Supplementary Fig. 12) but Egger’s test (p = 0.0217) and Begg’s test (p = 0.0112) result showed that publication bias was likely. Publication bias could not be assessed for other outcomes as there were < 10 studies in the meta-analyses.
Summary of findings (GRADE evidence)
The CoE for short-term growth was deemed ‘low’. The CoE for long-term growth and neurodevelopmental outcomes was deemed ‘low to very low’ in view of significant statistical heterogeneity, differences in timing of assessments of outcomes and definitions, wide CIs and overall moderate to high ROB in the included studies (Table 3).
Discussion
Our systematic review found that preterm infants supplemented with probiotics had better short-term weight gain, but the size of benefit (SMD 0.24) was small. SMD values of 0.2–0.5 are considered small, 0.5–0.8 medium and > 0.8 are considered large [69]. However, probiotics had no significant effect on linear and head growth, as well as long-term growth, and neurodevelopment, CP, hearing, or visual impairment. Subgroup analyses showed improved weight gain with single strain, but not with multi-strain probiotics. It was reassuring that none of the included studies noted adverse effects related to probiotics.
In contrast to our findings, the systematic review by Sun et al. [70] showed lower short-term weight gain (primary outcome) in probiotic supplemented VLBW infants, however, this was statistically non-significant. They suggested that variable timing and methods of weight measurement may explain the lack of benefit in the pooled results. Furthermore, their subgroup analysis showed that the type of feeding may influence the effect of probiotics on weight gain. Probiotic supplemented infants fed either breast milk or formula had better weight gain [(MD: 2.2 g (95% CI: 0.08 to 4.48 g; p < 0.05)] compared to those fed only formula [(MD: −0.89 g (95% CI: −3.97 to 2.18; p = 0.57)].
Totsu et al. reported significantly better short-term weight gain in the probiotic group in their pilot trial [47]. However, their cluster RCT showed no difference in body weight, length, or head circumference at 18 months between probiotic and placebo groups [71]. The improved short-term weight gain following probiotic supplementation could relate to increased absorption of key nutrients in the early postnatal period and reduced gut dysbiosis and nutrient malabsorption by strengthening the gut barrier [72,73,74,75].
The lack of effect on short-term linear or head growth or overall long-term growth could be explained by the interplay between several variables including the degree and complications of prematurity (e.g. LOS, NEC, chronic lung disease), type of feeding, postnatal exposure to steroids, and differences in strategies for neonatal enteral and parenteral nutrition, and post discharge nutrition [70, 76,77,78,79].
Similar to our findings, the recent Cochrane systematic review showed that probiotics may have little or no effect on severe neurodevelopmental impairment (RR: 1.03; 95% CI: 0.84 to 1.26; five RCTs, n = 1518 infants; CoE: low) [20]. There were no differences in CP, visual or hearing impairment and cognitive performance. In their systematic review, Upadhyay et al. reported improvement in mean motor scores (p = 0.07) in probiotic group, but suggested caution given the overall high risk of bias [25]. Romeo et al. reported normal range of optimal scores on Hammersmith Infant Neurological Examination at 12 months in the probiotic group [64]. The authors attributed these findings to reduced Candida infections and gut colonisation with probiotic bacteria [64]. Totsu et al. reported improved developmental quotient scores following B. bifidum OLB 6378 supplementation in neonatal period in female infants at 18 months with reduced rates of CP [65].
In addition to their anti-neuroinflammatory effects, probiotics may influence neurodevelopment through short-chain fatty acids (SCFA) production. SCFAs play an important role in ‘gut-brain’ axis by regulating central nervous system processes (e.g., cell-cell interaction, neurotransmitter synthesis and release, microglia activation, mitochondrial function, and gene expression) [80,81,82,83]. SCFAs promote neurosphere growth from human neural stem cells and differentiation of embryonic stem cells into neural cells [84].
Upadhyay et al. [25] reported significantly lower risk of hearing impairment (RR 0.25; 95% CI: 0.07, 0.88) (n = 838; I2 = 15.2%; p = 0.03) in a subgroup where multi-strain probiotics were commenced within first week of life. The ProPrems RCT also reported reduced hearing impairment in the probiotic group. Reduced cochlear and outer hair cell injury was pointed out as a possible mechanism for this benefit of probiotics [66].
The lack of effect of probiotics on neurodevelopment in our review could relate to variations in probiotic strain, dose and duration in different studies and ages and methods of neuro-developmental assessment. Considering only seven RCTs reported neurodevelopmental data, inadequate sample size is a concern. Furthermore, it is difficult to comment on the role played parental education and socio-economic status, factors known to influence neurodevelopmental outcomes [85, 86]. Our subgroup analyses (gestation < 28 week and single vs. multistrain probiotics) were limited due to lack of suitable data from included studies. Furthermore, individual participant details would be needed for a detailed gestational age wise analysis, which is beyond the scope of our review. One of the major limitations of our study includes small sample size and number of included studies for comparisons of long-term growth and neurodevelopmental outcomes. For example, hearing, and visual impairment had only four studies (n = 1388) to compare.
Despite its limitations, we believe that our comprehensive systematic review with robust methodology focussing specifically on growth and neurodevelopment adds useful data to guide research and clinical practice in this field. Emerging evidence supports the importance of early postnatal enteral nutrient intake in brain development and maturation, and role of gut-microbiota in long-term neurodevelopment and neuropsychiatric disorders [18, 22, 87,88,89,90]. Given these data, and our results, adequately powered well designed RCTs are justified to assess the long-term effects of probiotics in preterm infants. Such RCTs should stratify infants based on gestational age (<28w, 29–32w and > 32w). Adequate information needs to be provided to parents regarding the controversies about the short-term benefits such as reduction in NEC, mortality and LOS to enable them to consent for participating in a placebo-controlled trial. In units where there is no equipoise, routine probiotic supplementation could be considered if a suitable product is available. Well-designed and adequately powered prospective observational studies will be helpful to assess the effects of probiotic supplementation on long-term growth and neurodevelopment in preterm infants in such real-life circumstances.
Conclusions
This systematic review and meta-analysis showed that preterm infants supplemented with probiotics had better weight gain during initial hospitalisation. However, probiotics had no significant effect on linear and head growth, as well as long-term growth, and neurodevelopment.
Data availability
Data described in the manuscript, code book, and analytic code will be made available upon reasonable request sent via email to gayatri.jape@health.wa.gov.au.
Change history
22 March 2023
Missing Open Access funding information has been added in the Funding Note.
References
Cheong JLY, Lee KJ, Boland RA, Spittle AJ, Opie GF, Burnett AC, et al. Changes in long-term prognosis with increasing postnatal survival and the occurrence of postnatal morbidities in extremely preterm infants offered intensive care: a prospective observational study. Lancet Child Adolesc Health. 2018;2:872–9.
Patel RM. Short- and Long-Term Outcomes for Extremely Preterm Infants. Am J Perinatol. 2016;33:318–28.
Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Trends in Care Practices, Morbidity, and Mortality of Extremely Preterm Neonates, 1993-2012. Jama. 2015;314:1039–51.
Cheong JLY, Olsen JE, Lee KJ, Spittle AJ, Opie GF, Clark M, et al. Temporal Trends in Neurodevelopmental Outcomes to 2 Years After Extremely Preterm Birth. JAMA Pediatr. 2021;175:1035–42.
Pierrat V, Marchand-Martin L, Arnaud C, Kaminski M, Resche-Rigon M, Lebeaux C, et al. Neurodevelopmental outcome at 2 years for preterm children born at 22 to 34 weeks’ gestation in France in 2011: EPIPAGE-2 cohort study. BMJ. 2017;358:j3448.
Bell EF, Hintz SR, Hansen NI, Bann CM, Wyckoff MH, DeMauro SB, et al. Mortality, In-Hospital Morbidity, Care Practices, and 2-Year Outcomes for Extremely Preterm Infants in the US, 2013-2018. Jama. 2022;327:248–63.
Marißen J, Haiß A, Meyer C, Van Rossum T, Bünte LM, Frommhold D, et al. Efficacy of Bifidobacterium longum, B. infantis and Lactobacillus acidophilus probiotics to prevent gut dysbiosis in preterm infants of 28+0-32+6 weeks of gestation: a randomised, placebo-controlled, double-blind, multicentre trial: the PRIMAL Clinical Study protocol. BMJ Open. 2019;9:e032617.
Pammi M, Cope J, Tarr PI, Warner BB, Morrow AL, Mai V, et al. Intestinal dysbiosis in preterm infants preceding necrotizing enterocolitis: a systematic review and meta-analysis. Microbiome. 2017;5:31.
El Manouni El Hassani S, Niemarkt HJ, Berkhout DJC, Peeters CFW, Hulzebos CV, van Kaam AH, et al. Profound Pathogen-Specific Alterations in Intestinal Microbiota Composition Precede Late-Onset Sepsis in Preterm Infants: A Longitudinal, Multicenter, Case-Control Study. Clin Infect Dis. 2021;73:e224–e32.
Lee CC, Feng Y, Yeh YM, Lien R, Chen CL, Zhou YL, et al. Gut Dysbiosis, Bacterial Colonization and Translocation, and Neonatal Sepsis in Very-Low-Birth-Weight Preterm Infants. Front Microbiol. 2021;12:746111.
Luu TM, Rehman Mian MO, Nuyt AM. Long-Term Impact of Preterm Birth: Neurodevelopmental and Physical Health Outcomes. Clin Perinatol. 2017;44:305–14.
Bright HR, Babata K, Allred EN, Erdei C, Kuban KCK, Joseph RM, et al. Neurocognitive Outcomes at 10 Years of Age in Extremely Preterm Newborns with Late-Onset Bacteremia. J Pediatr. 2017;187:43–9 e1.
Sewell E, Roberts J, Mukhopadhyay S. Association of Infection in Neonates and Long-Term Neurodevelopmental Outcome. Clin Perinatol. 2021;48:251–61.
Matei A, Montalva L, Goodbaum A, Lauriti G, Zani A. Neurodevelopmental impairment in necrotising enterocolitis survivors: systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2020;105:432–9.
Mondal A, Misra D, Al-Jabir A, Hubail D, Ward T, Patel B. Necrotizing Enterocolitis in Neonates: Has the Brain Taken a Hit 10 Years Later? J Pediatr Neurosci. 2021;16:30–4.
Keunen K, van Elburg RM, van Bel F, Benders MJ. Impact of nutrition on brain development and its neuroprotective implications following preterm birth. Pediatr Res. 2015;77:148–55.
Chi C, Li C, Buys N, Wang W, Yin C, Sun J. Effects of Probiotics in Preterm Infants: A Network Meta-analysis. Pediatrics. 2021;147:e20200706.
Boscarino G, Di Chiara M, Cellitti R, De Nardo MC, Conti MG, Parisi P, et al. Effects of early energy intake on neonatal cerebral growth of preterm newborn: an observational study. Sci Rep. 2021;11:18457.
Athalye-Jape G, Deshpande G, Rao S, Patole S. Benefits of probiotics on enteral nutrition in preterm neonates: a systematic review. Am J Clin Nutr. 2014;100:1508–19.
Sharif S, Meader N, Oddie SJ, Rojas-Reyes MX, McGuire W. Probiotics to prevent necrotising enterocolitis in very preterm or very low birth weight infants. Cochrane Database Syst Rev. 2020;10:Cd005496.
Morgan RL, Preidis GA, Kashyap PC, Weizman AV, Sadeghirad B. Probiotics Reduce Mortality and Morbidity in Preterm, Low-Birth-Weight Infants: A Systematic Review and Network Meta-analysis of Randomized Trials. Gastroenterology. 2020;159:467–80.
Lu J, Claud EC. Connection between gut microbiome and brain development in preterm infants. Dev Psychobiol. 2019;61:739–51.
Beghetti I, Barone M, Turroni S, Biagi E, Sansavini A, Brigidi P, et al. Early-life gut microbiota and neurodevelopment in preterm infants: any role for Bifidobacterium? Eur J Pediatr. 2022;181:1773–7.
Buffet-Bataillon S, Bellanger A, Boudry G, Gangneux JP, Yverneau M, Beuchee A, et al. New Insights Into Microbiota Modulation-Based Nutritional Interventions for Neurodevelopmental Outcomes in Preterm Infants. Front Microbiol. 2021;12:676622.
Upadhyay RP, Taneja S, Chowdhury R, Strand TA, Bhandari N. Effect of prebiotic and probiotic supplementation on neurodevelopment in preterm very low birth weight infants: findings from a meta-analysis. Pediatr Res. 2020;87:811–22.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535.
Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135.
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898.
Higgins J, Thomas J, Chandler J, Cumpston M, Li T, Page M, et al. Cochrane Handbook for Systematic Reviews of Interventions. 2nd Edition. Chichester (UK): John Wiley & Sons.; 2019.0
Sterne JA, Sutton AJ, Ioannidis JP, Terrin N, Jones DR, Lau J, et al. Recommendations for examining and interpreting 12funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ. 2011;343:d4002.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101.
Guyatt GH, Oxman AD, Santesso N, Helfand M, Vist G, Kunz R, et al. GRADE guidelines: 12. Preparing summary of findings tables-binary outcomes. J Clin Epidemiol. 2013;66:158–72.
Spreckels JE, Wejryd E, Marchini G, Jonsson B, de Vries DH, Jenmalm MC, et al. Lactobacillus reuteri Colonisation of Extremely Preterm Infants in a Randomised Placebo-Controlled Trial. Microorganisms. 2021;9:915.
Cui X, Shi Y, Gao S, Xue X, Fu J. Effects of Lactobacillus reuteri DSM 17938 in preterm infants: a double-blinded randomized controlled study. Ital J Pediatr. 2019;45:140.
Oshiro T, Nagata S, Wang C, Takahashi T, Tsuji H, Asahara T, et al. Bifidobacterium Supplementation of Colostrum and Breast Milk Enhances Weight Gain and Metabolic Responses Associated with Microbiota Establishment in Very-Preterm Infants. Biomed Hub. 2019;4:1–10.
Wejryd E, Marchini G, Frimmel V, Jonsson B, Abrahamsson T. Probiotics promoted head growth in extremely low birthweight infants in a double-blind placebo-controlled trial. Acta Paediatr. 2019;108:62–9.
Indrio F, Riezzo G, Tafuri S, Ficarella M, Carlucci B, Bisceglia M, et al. Probiotic Supplementation in Preterm: Feeding Intolerance and Hospital Cost. Nutrients 2017;9:965.
Shashidhar A, Suman Rao PN, Nesargi S, Bhat S, Chandrakala BS. Probiotics for Promoting Feed Tolerance in Very Low Birth Weight Neonates - A Randomized Controlled Trial. Indian Pediatr. 2017;54:363–7.
Sukanyaa S, Vinoth S, Ramesh S. Role of probiotics in preterm infants: a randomized controlled trial. Int J Contemp Pediatr. 2017;4:447–9.
Hays S, Jacquot A, Gauthier H, Kempf C, Beissel A, Pidoux O, et al. Probiotics and growth in preterm infants: A randomized controlled trial, PREMAPRO study. Clin Nutr. 2016;35:802–11.
Xu L, Wang Y, Wang Y, Fu J, Sun M, Mao Z, et al. A double-blinded randomized trial on growth and feeding tolerance with Saccharomyces boulardii CNCM I-745 in formula-fed preterm infants. J Pediatr. 2016;92:296–301.
Choudhury Moni S, Shahidullah M, Saha D, Dey AC, Dey SK, Mannan MA. Probiotic Supplementation and Its Effect on Weight and Feed tolerance in Preterm Low Birth Weight Newborns: A Clinical Trial. Bangladesh J Child Health. 2017;39:82–7.
Dilli D, Aydin B, Fettah ND, Ozyazici E, Beken S, Zenciroglu A, et al. The propre-save study: effects of probiotics and prebiotics alone or combined on necrotizing enterocolitis in very low birth weight infants. J Pediatr. 2015;166:545–51.e1.
Shadkam MN, Fatemeh J, Khadijeh N. Effects of Probiotic Lactobacillus Reuteri (DSM 17938) on the Incidence of Necrotizing Enterocolitis in Very Low Birth Weight Premature Infants. Iran J Neonatol. 2015;6:15–20.
Patole S, Keil AD, Chang A, Nathan E, Doherty D, Simmer K, et al. Effect of Bifidobacterium breve M-16V supplementation on fecal bifidobacteria in preterm neonates–a randomised double blind placebo controlled trial. PLoS One. 2014;9:e89511.
Totsu S, Yamasaki C, Terahara M, Uchiyama A, Kusuda S, Probiotics Study Group in J. Bifidobacterium and enteral feeding in preterm infants: cluster-randomized trial. Pediatr Int. 2014;56:714–9.
Van Niekerk E, Kirsten GF, Nel DG, Blaauw R. Probiotics, feeding tolerance, and growth: a comparison between HIV-exposed and unexposed very low birth weight infants. Nutrition 2014;30:645–53.
Demirel G, Erdeve O, Celik IH, Dilmen U. Saccharomyces boulardii for prevention of necrotizing enterocolitis in preterm infants: a randomized, controlled study. Acta Paediatr. 2013;102:e560–5.
Jacobs SE, Tobin JM, Opie GF, Donath S, Tabrizi SN, Pirotta M, et al. Probiotic effects on late-onset sepsis in very preterm infants: a randomized controlled trial. Pediatrics 2013;132:1055–62.
Serce O, Benzer D, Gursoy T, Karatekin G, Ovali F. Efficacy of Saccharomyces boulardii on necrotizing enterocolitis or sepsis in very low birth weight infants: a randomised controlled trial. Early Hum Dev. 2013;89:1033–6.
Al-Hosni M, Duenas M, Hawk M, Stewart LA, Borghese RA, Cahoon M, et al. Probiotics-supplemented feeding in extremely low-birth-weight infants. J Perinatol. 2012;32:253–9.
Chrzanowska-Liszewska D, Seliga-Siwecka J, Kornacka MK. The effect of Lactobacillus rhamnosus GG supplemented enteral feeding on the microbiotic flora of preterm infants-double blinded randomized control trial. Early Hum Dev. 2012;88:57–60.
Sari FN, Dizdar EA, Oguz S, Erdeve O, Uras N, Dilmen U. Oral probiotics: Lactobacillus sporogenes for prevention of necrotizing enterocolitis in very low-birth weight infants: a randomized, controlled trial. Eur J Clin Nutr. 2011;65:434–9.
Indrio F, Riezzo G, Raimondi F, Bisceglia M, Cavallo L, Francavilla R. The effects of probiotics on feeding tolerance, bowel habits, and gastrointestinal motility in preterm newborns. J Pediatr. 2008;152:801–6.
Mohan R, Koebnick C, Schildt J, Mueller M, Radke M, Blaut M. Effects of Bifidobacterium lactis Bb12 supplementation on body weight, fecal pH, acetate, lactate, calprotectin, and IgA in preterm infants. Pediatr Res. 2008;64:418–22.
Stratiki Z, Costalos C, Sevastiadou S, Kastanidou O, Skouroliakou M, Giakoumatou A, et al. The effect of a bifidobacter supplemented bovine milk on intestinal permeability of preterm infants. Early Hum Dev. 2007;83:575–9.
Bin-Nun A, Bromiker R, Wilschanski M, Kaplan M, Rudensky B, Caplan M, et al. Oral probiotics prevent necrotizing enterocolitis in very low birth weight neonates. J Pediatr. 2005;147:192–6.
Costalos C, Skouteri V, Gounaris A, Sevastiadou S, Triandafilidou A, Ekonomidou C, et al. Enteral feeding of premature infants with Saccharomyces boulardii. Early Hum Dev. 2003;74:89–96.
Kitajima H, Sumida Y, Tanaka R, Yuki N, Takayama H, Fujimura M. Early administration of Bifidobacterium breve to preterm infants: randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 1997;76:F101–7.
Reuman PD, Duckworth DH, Smith KL, Kagan R, Bucciarelli RL, Ayoub EM. Lack of effect of Lactobacillus on gastrointestinal bacterial colonization in premature infants. Pediatr Infect Dis. 1986;5:663–8.
Agrawal S, Pestell CF, Granich J, Rao S, Nathan E, Wray JA, et al. Difficulties in developmental follow-up of preterm neonates in a randomised-controlled trial of Bifidobacterium breve M16-V - Experience from Western Australia. Early Hum Dev. 2020;151:105165.
Akar M, Eras Z, Oncel MY, Arayici S, Guzoglu N, Canpolat FE, et al. Impact of oral probiotics on neurodevelopmental outcomes in preterm infants. J Matern Fetal Neonatal Med. 2017;30:411–5.
Romeo MG, Romeo DM, Trovato L, Oliveri S, Palermo F, Cota F, et al. Role of probiotics in the prevention of the enteric colonization by Candida in preterm newborns: incidence of late-onset sepsis and neurological outcome. J Perinatol. 2011;31:63–9.
Totsu S, Terahara M, Kusuda S. Probiotics and the development of very low birthweight infants: follow-up study of a randomised trial. BMJ Paediatr Open. 2018;2:e000256.
Jacobs SE, Hickey L, Donath S, Opie GF, Anderson PJ, Garland SM, et al. Probiotics, prematurity and neurodevelopment: follow-up of a randomised trial. BMJ Paediatr Open. 2017;1:e000176.
Sari FN, Eras Z, Dizdar EA, Erdeve O, Oguz SS, Uras N, et al. Do oral probiotics affect growth and neurodevelopmental outcomes in very low-birth-weight preterm infants? Am J Perinatol. 2012;29:579–86.
Chou IC, Kuo HT, Chang JS, Wu SF, Chiu HY, Su BH, et al. Lack of effects of oral probiotics on growth and neurodevelopmental outcomes in preterm very low birth weight infants. J Pediatr. 2010;156:393–6.
Andrade C. Mean Difference, Standardized Mean Difference (SMD), and Their Use in Meta-Analysis: As Simple as It Gets. J Clin Psychiatry. 2020;81:20f13681.
Sun J, Marwah G, Westgarth M, Buys N, Ellwood D, Gray PH. Effects of Probiotics on Necrotizing Enterocolitis, Sepsis, Intraventricular Hemorrhage, Mortality, Length of Hospital Stay, and Weight Gain in Very Preterm Infants: A Meta-Analysis. Adv Nutr. 2017;8:749–63.
Yamasaki C, Totsu S, Uchiyama A, Nakanishi H, Masumoto K, Washio Y, et al. Effect of Bifidobacterium administration on very-low-birthweight infants. Pediatr Int. 2012;54:651–6.
Jäger R, Mohr AE, Carpenter KC, Kerksick CM, Purpura M, Moussa A, et al. International Society of Sports Nutrition Position Stand: Probiotics. J Int Soc Sports Nutr. 2019;16:62–44.
Jäger R, Purpura M, Farmer S, Cash HA, Keller D. Probiotic Bacillus coagulans GBI-30, 6086 Improves Protein Absorption and Utilization. Probiotics Antimicrob Proteins. 2018;10:611–5.
Ballini A, Gnoni A, De Vito D, Dipalma G, Cantore S, Gargiulo Isacco C, et al. Effect of probiotics on the occurrence of nutrition absorption capacities in healthy children: a randomized double-blinded placebo-controlled pilot study. Eur Rev Med Pharm Sci. 2019;23:8645–57.
Judkins TC, Archer DL, Kramer DC, Solch RJ. Probiotics, Nutrition, and the Small Intestine. Curr Gastroenterol Rep. 2020;22:2.
Embleton ND, Wood CL, Pearce MS, Brunskill G, Grahame V. Early diet in preterm infants and later cognition: 10-year follow-up of a randomized controlled trial. Pediatr Res. 2021;89:1442–6.
Jasper EA, Cho H, Breheny PJ, Bao W, Dagle JM, Ryckman KK. Perinatal determinants of growth trajectories in children born preterm. PLoS One. 2021;16:e0245387–e.
Power VA, Spittle AJ, Lee KJ, Anderson PJ, Thompson DK, Doyle LW, et al. Nutrition, Growth, Brain Volume, and Neurodevelopment in Very Preterm Children. J Pediatr. 2019;215:50–5.e3.
Zozaya C, Avila-Alvarez A, Arruza L, García-Muñoz Rodrigo F, Fernandez-Perez C, Castro A, et al. The Effect of Morbidity and Sex on Postnatal Growth of Very Preterm Infants: A Multicenter Cohort Study. Neonatology 2019;115:348–54.
Yoo DY, Kim W, Nam SM, Kim DW, Chung JY, Choi SY, et al. Synergistic effects of sodium butyrate, a histone deacetylase inhibitor, on increase of neurogenesis induced by pyridoxine and increase of neural proliferation in the mouse dentate gyrus. Neurochem Res. 2011;36:1850–7.
Wei Y, Melas PA, Wegener G, Mathé AA, Lavebratt C. Antidepressant-like effect of sodium butyrate is associated with an increase in TET1 and in 5-hydroxymethylation levels in the Bdnf gene. Int J Neuropsychopharmacol. 2014;18:pyu032.
Hsiao EY, McBride SW, Hsien S, Sharon G, Hyde ER, McCue T, et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 2013;155:1451–63.
Cryan JF, O’Riordan KJ, Cowan CSM, Sandhu KV, Bastiaanssen TFS, Boehme M, et al. The Microbiota-Gut-Brain Axis. Physiol Rev. 2019;99:1877–2013.
Yang LL, Millischer V, Rodin S, MacFabe DF, Villaescusa JC, Lavebratt C. Enteric short-chain fatty acids promote proliferation of human neural progenitor cells. J Neurochem. 2020;154:635–46.
Benavente-Fernández I, Siddiqi A, Miller SP. Socioeconomic status and brain injury in children born preterm: modifying neurodevelopmental outcome. Pediatr Res. 2020;87:391–8.
ElHassan NO, Bai S, Gibson N, Holland G, Robbins JM, Kaiser JR. The impact of prematurity and maternal socioeconomic status and education level on achievement-test scores up to 8th grade. PLOS ONE. 2018;13:e0198083.
Doenyas C. Gut Microbiota, Inflammation, and Probiotics on Neural Development in Autism Spectrum Disorder. Neuroscience 2018;374:271–86.
Abdellatif B, McVeigh C, Bendriss G, Chaari A. The Promising Role of Probiotics in Managing the Altered Gut in Autism Spectrum Disorders. Int J Mol Sci. 2020;21:4159.
Checa-Ros A, Jeréz-Calero A, Molina-Carballo A, Campoy C, Muñoz-Hoyos A. Current Evidence on the Role of the Gut Microbiome in ADHD Pathophysiology and Therapeutic Implications. Nutrients 2021;13:249.
Laue HE, Coker MO, Madan JC. The Developing Microbiome From Birth to 3 Years: The Gut-Brain Axis and Neurodevelopmental Outcomes. Front Pediatr. 2022;10:815885.
Acknowledgements
Authors would like to thank Dr Erik Wejryd and Dr Evette Van Niekerk for providing additional study data for meta-analysis.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
(1) HP: Independent literature search and writing the first and final draft of the manuscript, data interpretation with supervision from authors GAJ and SP. (2) GAJ: Rechecking the literature search results and interpretation of data, handling the meta-analysis software, co-authoring and supervising the first and final draft. (3) SR: Rechecking and interpreting data, conducting Egger’s test and Begg’s test for publication bias, acting as a referee author in case of discrepancy between other authors and supervising the first and the final draft of the manuscript. (4) SP: Concept and design, rechecking and interpreting data, acting as a referee author in case of discrepancy between other authors and supervising the first and the final draft of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Panchal, H., Athalye-Jape, G., Rao, S. et al. Growth and neuro-developmental outcomes of probiotic supplemented preterm infants—a systematic review and meta-analysis. Eur J Clin Nutr 77, 855–871 (2023). https://doi.org/10.1038/s41430-023-01270-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-023-01270-2
This article is cited by
-
Probiotics for Preterm Infants—Update 2024
Current Treatment Options in Pediatrics (2024)