Abstract
Background
Assessing whole-body skeletal muscle mass (SMM) and fat-free mass (FFM) is essential for the adequate nutritional management and training evaluation of athletes and trained individuals. This study aimed to determine the relationship between SMM assessed using the creatine (methyl‐d3) dilution (D3-creatine) method and SMM estimated by whole-body magnetic resonance imaging (MRI) in healthy young men undergoing exercise training. Additionally, we examined the association between FFM measured using the four-component (4C) method (FFM4C) and the total body protein value estimated using 4C (TBpro4C).
Methods and results
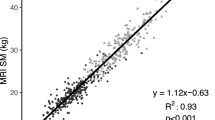
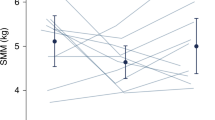
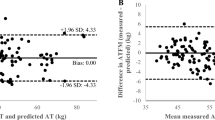
We analyzed the data of 29 males (mean age, 19.9 ± 1.8 years) who exercised regularly. SMM measurements were obtained using the D3-creatine method (SMMD3-creatine) and MRI (SMMMRI). The SMMD3-creatine adjusted to 4.3 g/SMM kg was significantly higher than SMMMRI (p < 0.01). The fit of the creatine pool size compared with SMMMRI was 5.0 g/SMMMRI kg. SMMMRI was significantly correlated with both SMMD3-creatine adjusted to 4.3 g/kg and 5.1 g/kg. TBpro4C was significantly lower than SMMMRI (p < 0.01). Contrastingly, FFM4C was significantly higher than SMMMRI (p < 0.01).
Conclusions
SMMD3-creatine adjusted to 4.3 g/SMM kg—a previously reported value—may differ for athletes and active young males. We believe that a value of 5.0–5.1 g/SMM kg better estimates the total muscle mass in this population. Traditional FFM estimation highly correlates with SMMMRI in well-trained young males, and the relationships appear strong enough for total body protein or SMM to be estimated through the FFM value.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Evans WJ, Hellerstein M, Orwoll E, Cummings S, Cawthon PM. D3 -Creatine dilution and the importance of accuracy in the assessment of skeletal muscle mass. J Cachexia Sarcopenia Muscle. 2019;10:14–21.
Balsom PD, Söderlund K, Ekblom B. Creatine in humans with special reference to creatine supplementation. Sport Med. 1994;18:268–80.
Schaap LA. D3-creatine dilution to assess muscle mass. J Gerontol A Biol Sci Med Sci. 2019;74:842–3.
Cawthon PM, Orwoll ES, Peters KE, Ensrud KE, Cauley JA, Kado DM, et al. Strong relation between muscle mass determined by d3-creatine dilution, physical performance, and incidence of falls and mobility limitations in a prospective cohort of older men. J Gerontol - Ser A Biol Sci Med Sci. 2019;74:844–52.
Evans WJ, Scottoline B, Imam F, Hellerstein M, Garton K, Czerwieniec G, et al. D3-creatine dilution for the noninvasive measurement of skeletal muscle mass in premature infants. Pediatr Res. 2021;89:1508–14.
Evans WJ, Shankaran M, Smith EC, Morris C, Nyangau E, Bizieff A, et al. Profoundly lower muscle mass and rate of contractile protein synthesis in boys with Duchenne muscular dystrophy. J Physiol. 2021;599:5215–27.
Orwoll ES, Peters KE, Hellerstein M, Cummings SR, Evans WJ, Cawthon PM. The importance of muscle versus fat mass in sarcopenic obesity: a re-evaluation using D3-creatine muscle mass versus DXA lean mass measurements. J Gerontol A Biol Sci Med Sci. 2020;75:1362–8.
Rogers-soeder TS, Peters KE, Lane NE, Shikany JM, Judd S, Langsetmo L, et al. Dietary intake, D3Cr muscle mass, and appendicular lean mass in a cohort of older men. J Gerontol Ser A. 2019;75:1353–61.
Ribeiro AS, Avelar A, Kassiano W, Nunes JP, Schoenfeld BJ, Aguiar AF, et al. Creatine supplementation does not influence the ratio between intracellular water and skeletal muscle mass in resistance-trained men. Int J Sport Nutr Exerc Metab. 2020;11:1–7. https://doi.org/10.1123/ijsnem.2020-0080.
Clark RV, Walker AC, Connor-semmes RLO, Leonard MS, Miller RR, Stimpson SA, et al. Total body skeletal muscle mass: estimation by creatine (methyl-d3) dilution in humans. J Appl Physiol. 2014;116:1605–13.
Clark RV, Walker AC, Miller RR, O’Connor-Semmes RL, Ravussin E, Cefalu WT, et al. Creatine (methyl-d3) dilution in urine for estimation of total body skeletal muscle mass: Accuracy and variability vs. MRI and DXA. J Appl Physiol. 2018;124:1–9.
Weber MA, Krakowski-Roosen H, Schröder L, Kinscherf R, Krix M, Kopp-Schneider A, et al. Morphology, metabolism, microcirculation, and strength of skeletal muscles in cancer-related cachexia. Acta Oncol. 2009;48:116–24.
Willer B, Stucki G, Hoppeler H, Brühlmann P, Krähenbühl S. Effects of creatine supplementation on muscle weakness in patients with rheumatoid arthritis. Rheumatology 2000;39:293–8.
Brault JJ, Terjung RL. Creatine uptake and creatine transporter expression among rat skeletal muscle fiber types. Am J Physiol-Cell Physiol 2003;284:1481–9.
Morris-Paterson TE, Stimpson SA, Miller RR, Barton ME, Leonard MS, Carmichael O, et al. Total body skeletal muscle mass estimated by magnetic resonance imaging and creatine (methyl-d3) dilution in athletes. Scand J Med Sci Sport. 2020;30:421–8.
Harris RC, Söderlund K, Hultman E. Elevation of creatine in resting and exercised muscle of normal subjects by creatine supplementation. Clin Sci. 1992;83:367–74.
Prior BM, Modlesky CM, Evans EM, Sloniger MA, Saunders MJ, Lewis RD, et al. Muscularity and the density of the fat-free mass in athletes. J Appl Physiol. 2001;90:1523–31.
Jensen B, Braun W, Geisler C, Both M, Klückmann K, Müller MJ, et al. Limitations of fat-free mass for the assessment of muscle mass in obesity. Obes Facts. 2019;12:307–15.
Sagayama H, Yamada Y, Tanabe Y, Kondo E, Ohnishi T, Takahashi H. Validation of skeletal muscle mass estimation equations in active young adults: A preliminary study. Scand J Med Sci Sport. 2021;31:1897–907.
McKay AKA, Stellingwerff T, Smith ES, Martin DT, Mujika I, Goosey-Tolfrey VL, et al. Defining training and performance caliber: a participant classification framework. Int J Sports Physiol Perform. 2022;17:317–31.
Snyder W, Cook M, Nasset E, Karhansen L, Howells G, Tipton I Report of the task group on reference men. Oxford, United Kingdom Pergamon Press. 1975;23.
Shankaran M, Czerwieniec G, Fessler C, Wong PA, Killion S, Turner SM, et al. Dilution of oral D3 -Creatine to measure creatine pool size and estimate skeletal muscle mass: development of a correction algorithm. J Cachexia Sarcopenia Muscle. 2018;9:540–6.
Heymsfield SB, Ebbeling CB, Zheng J, Pietrobelli A, Strauss BJ, Silva AM, et al. Multi-component molecular-level body composition reference methods: Evolving concepts and future directions. Obes Rev. 2015;16:282–94.
Kondo E, Shiose K, Yamada Y, Osawa T, Sagayama H, Motonaga K, et al. Effect of thoracic gas volume changes on body composition assessed by air displacement plethysmography after rapid weight loss and regain in elite collegiate wrestlers. Sport. 2019;7.
Sagayama H, Kondo E, Tanabe Y, Ohnishi T, Yamada Y, Takahashi H. Bone mineral density in male weight-classified athletes is higher than that in male endurance-athletes and non-athletes. Clin Nutr ESPEN. 2020;36:106–10.
Sagayama H, Yamada Y, Ichikawa M, Kondo E, Yasukata J, Tanabe Y, et al. Evaluation of fat-free mass hydration in athletes and non-athletes. Eur J Appl Physiol. 2020;120:1179–88.
Sagayama H, Yoshimura E, Yamada Y, Ichikawa M, Ebine N, Higaki Y, et al. Effects of rapid weight loss and regain on body composition and energy expenditure. Appl Physiol Nutr Metab. 2014;39:21–7.
Sagayama H, Shizuma K, Toguchi M, Mizuhara H, Machida Y, Yamada Y, et al. Effect of the Health Tourism weight loss programme on body composition and health outcomes in healthy and excess-weight adults. Br J Nutr. 2018;119:1133–1141.
Sagayama H, Racine NM, Shriver TC, Schoeller DA. Comparison of isotope ratio mass spectrometry and cavity ring-down spectroscopy procedures and precision of the doubly labeled water method in different physiological specimens. Rapid Commun Mass Spectrom. 2021;35:1–9.
Hill DK. The location of creatine phosphate in frog’s striated muscle. J Physiol. 1962;164:31–50.
Edström L, Hultman E, Sahlin K, Sjöholm H. The contents of high-energy phosphates in different fibre types in skeletal muscles from rat, guinea-pig and man. J Physiol. 1982;332:47–58.
Kondo E, Sagayama H, Yamada Y, Shiose K, Osawa T, Motonaga K, et al. Energy deficit required for rapid weight loss in elite collegiate wrestlers. Nutrients. 2018;10.
Miyauchi S, Oshima S, Asaka M, Kawano H, Torii S, Higuchi M. Organ size increases with weight gain in power-trained athletes. Int J Sport Nutr Exerc Metab. 2013;23:617–23.
Müller MJ, Bosy-Westphal A, Braun W, Wong MC, Shepherd JA, Heymsfield SB. What Is a 2021 reference body? Nutrients 2022;14:1–18.
Duchowny KA, Peters KE, Cummings SR, Orwoll ES, Hoffman AR, Ensrud KE, et al. Association of change in muscle mass assessed by D3-creatine dilution with changes in grip strength and walking speed. J Cachexia Sarcopenia Muscle. 2020;11:55–61.
Watt KKO, Garnham AP, Snow RJ. Skeletal muscle total creatine content and creatine transporter gene expression in vegetarians prior to and following creatine supplementation. Int J Sport Nutr Exerc Metab. 2004;14:517–31.
Acknowledgements
We wish to thank the volunteers who participated in this study and their coaches. The authors also thank Mr. Tsuyoshi Sakamoto for his advice on the MRI analysis.
Funding
This investigation was mainly supported by the Research Fellowships from the JSPS KAKENHI (16J11877 and 20K19563 to HS, 19H04017 to HT, and 18H03164 to YY). This data analysis and publication were supported by a grant 2022(I)1 from the Advanced Research Initiative for Human High Performance (ARIHHP), University of Tsukuba.
Author information
Authors and Affiliations
Contributions
HS, YY, and HT planned the study. HS, EK, TY, TO, and HT collected data. MS, EN, WE, MH, JY, and HY performed specimen and data analyses. HS, AU, and YY conducted statistical analyses. HS prepared illustrations. HS, EK, AU, and YY drafted the manuscript. All authors interpreted the results and revised and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
WJE and MH are listed as co-inventors on patents for the D3-creatine dilution method. However, they do not control the IP nor do they derive any income from the use of this method. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sagayama, H., Yamada, Y., Kondo, E. et al. Skeletal muscle mass can be estimated by creatine (methyl‐d3) dilution and is correlated with fat-free mass in active young males. Eur J Clin Nutr 77, 393–399 (2023). https://doi.org/10.1038/s41430-022-01237-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-022-01237-9
This article is cited by
-
D3Creatine Dilution as a Direct, Non-invasive and Accurate Measurement of Muscle Mass for Aging Research
Calcified Tissue International (2023)