Abstract
Background/Objectives
We aimed to investigate the effects of short-term hypocaloric diet-induced weight loss on DNA methylation profile in leukocytes from women with severe obesity.
Methods
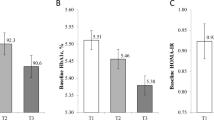
Eleven women with morbid obesity (age: 36.9 ± 10.3 years; BMI: 58.5 ± 10.5 kg/m2) were assessed before and after 6 weeks of a hypocaloric dietary intervention. The participants were compared with women of average weight and the same age (age: 36.9 ± 11.8 years; BMI: 22.5 ± 1.6 kg/m2). Genome-wide DNA methylation analysis was performed in DNA extracted from peripheral blood leukocytes using the Infinium Human Methylation 450 BeadChip assay. Changes (Δβ) in the methylation level of each CpGs were calculated. A threshold with a minimum value of 10%, p < 0.001, for the significant CpG sites based on Δβ and a false discovery rate of <0.05 was set.
Results
Dietary intervention changed the methylation levels at 16,064 CpG sites. These CpGs sites were related to cancer, cell cycle-related, MAPK, Rap1, and Ras signaling pathways. However, regardless of hypocaloric intervention, a group of 878 CpGs (related to 649 genes) remained significantly altered in obese women when compared with normal-weight women. Pathway enrichment analysis identified genes related to the cadherin and Wnt pathway, angiogenesis signaling, and p53 pathways by glucose deprivation.
Conclusion
A short-term hypocaloric intervention in patients with severe obesity partially restored the obesity-related DNA methylation pattern. Thus, the full change of obesity-related DNA methylation patterns could be proportional to the weight-loss rate in these patients after dietary interventions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dyaczynski M, Scanes CG, Koziec H, Koziec H, Pierzchala-Koziec K. Endocrine implications of obesity and bariatric surgery. Endokrynol Pol. 2018;69:574–97.
Locke AE, Kahali B, Berndt SI, Justice AE, Pers TH, Day FR, et al. Genetic studies of body mass index yield new insights for obesity biology. Nature. 2015;518:197–206.
Mansego ML, Milagro FI, Zulet MA, Moreno-Aliaga MJ, Martinez JA. Differential DNA methylation in relation to age and health risks of obesity. Int J Mol Sci. 2015;16:16816–32.
Demerath EW, Guan W, Grove ML, Aslibekyan S, Mendelson M, Zhou YH, et al. Epigenome-wide association study (EWAS) of BMI, BMI change and waist circumference in African American adults identifies multiple replicated loci. Hum Mol Genet. 2015;24:4464–79.
Huang RC, Garratt ES, Pan H, Wu Y, Davis EA, Barton SJ, et al. Genome-wide methylation analysis identifies differentially methylated CpG loci associated with severe obesity in childhood. Epigenetics. 2015;10:995–1005.
Sayols-Baixeras S, Subirana I, Fernandez-Sanles A, Senti M, Lluis-Ganella C, Marrugat J, et al. DNA methylation and obesity traits: An epigenome-wide association study. The REGICOR study. Epigenetics. 2017;12:909–16.
Wahl S, Drong A, Lehne B, Loh M, Scott WR, Kunze S, et al. Epigenome-wide association study of body mass index, and the adverse outcomes of adiposity. Nature. 2017;541:81–86.
Crujeiras AB, Diaz-Lagares A, Sandoval J, Milagro FI, Navas-Carretero S, Carreira MC, et al. DNA methylation map in circulating leukocytes mirrors subcutaneous adipose tissue methylation pattern: a genome-wide analysis from non-obese and obese patients. Sci Rep. 2017;7:41903.
Barres R, Kirchner H, Rasmussen M, Yan J, Kantor FR, Krook A, et al. Weight loss after gastric bypass surgery in human obesity remodels promoter methylation. Cell Rep. 2013;3:1020–7.
Nicoletti CF, Nonino CB, de Oliveira BA, Pinhel MA, Mansego ML, Milagro FI, et al. DNA methylation and hydroxymethylation levels in relation to two weight loss strategies: energy-restricted diet or bariatric surgery. Obes Surg. 2016;26:603–11.
Nicoletti CF, Pinhel MAS, Diaz-Lagares A, Casanueva FF, Jacome A, Pinhanelli VC, et al. DNA methylation screening after roux-en Y gastric bypass reveals the epigenetic signature stems from genes related to the surgery per se. BMC Med Genomics. 2019;12:72.
Ronn T, Volkov P, Davegardh C, Dayeh T, Hall E, Olsson AH, et al. A six months exercise intervention influences the genome-wide DNA methylation pattern in human adipose tissue. PLoS Genet. 2013;9:e1003572.
Morcillo S, Macias-Gonzalez M, Tinahones FJ. The effect of metabolic and bariatric surgery on DNA methylation patterns. Curr Atheroscler Rep. 2017;19:40.
Samblas M, Mansego ML, Zulet MA, Milagro FI, Martinez JA. An integrated transcriptomic and epigenomic analysis identifies CD44 gene as a potential biomarker for weight loss within an energy-restricted program. Eur J Nutr. 2019;58:1971–80.
Li R, Grimm SA, Mav D, Gu H, Djukovic D, Shah R, et al. Transcriptome and DNA methylome analysis in a mouse model of diet-induced obesity predicts increased risk of colorectal cancer. Cell Rep. 2018;22:624–37.
Cortes-Oliveira C, Nicoletti CF, de Souza Pinhel MA, de Oliveira BA, Quinhoneiro DC, Noronha NY, et al. UCP2 expression is associated with weight loss after hypocaloric diet intervention. Eur J Clin Nutr. 2017;71:402–6.
Bibikova M, Barnes B, Tsan C, Ho V, Klotzle B, Le JM, et al. High density DNA methylation array with single CpG site resolution. Genomics. 2011;98:288–95.
Dedeurwaerder S, Defrance M, Calonne E, Denis H, Sotiriou C, Fuks F. Evaluation of the infinium methylation 450K technology. Epigenomics. 2011;3:771–84.
ElGendy K, Malcomson FC, Lara JG, Bradburn DM, Mathers JC. Effects of dietary interventions on DNA methylation in adult humans: systematic review and meta-analysis. Br J Nutr. 2018;120:961–76.
Abete I, Gomez-Uriz AM, Mansego ML, De Arce A, Goyenechea E, Blazquez V, et al. Epigenetic changes in the methylation patterns of KCNQ1 and WT1 after a weight loss intervention program in obese stroke patients. Curr Neurovasc Res. 2015;12:321–33.
Delgado-Cruzata L, Zhang W, McDonald JA, Tsai WY, Valdovinos C, Falci L, et al. Dietary modifications, weight loss, and changes in metabolic markers affect global DNA methylation in Hispanic, African American, and Afro-Caribbean breast cancer survivors. J Nutr. 2015;145:783–90.
Martin-Nunez GM, Cabrera-Mulero R, Rubio-Martin E, Rojo-Martinez G, Olveira G, Valdes S, et al. Methylation levels of the SCD1 gene promoter and LINE-1 repeat region are associated with weight change: an intervention study. Mol Nutr Food Res. 2014;58:1528–36.
Cordero P, Campion J, Milagro FI, Goyenechea E, Steemburgo T, Javierre BM, et al. Leptin and TNF-alpha promoter methylation levels measured by MSP could predict the response to a low-calorie diet. J Physiol Biochem. 2011;67:463–70.
Nilsson EK, Ernst B, Voisin S, Almen MS, Benedict C, Mwinyi J, et al. Roux-en Y gastric bypass surgery induces genome-wide promoter-specific changes in DNA methylation in whole blood of obese patients. PLoS ONE. 2015;10:e0115186.
Bostrom AE, Mwinyi J, Voisin S, Wu W, Schultes B, Zhang K, et al. Longitudinal genome-wide methylation study of Roux-en-Y gastric bypass patients reveals novel CpG sites associated with essential hypertension. BMC Med Genomics. 2016;9:20.
Zhang N. Epigenetic modulation of DNA methylation by nutrition and its mechanisms in animals. Anim Nutr. 2015;1:144–51.
Kadayifci FZ, Zheng S, Pan YX. Molecular mechanisms underlying the link between diet and DNA methylation. Int J Mol Sci. 2018;19:4055.
Li Y, Daniel M, Tollefsbol TO. Epigenetic regulation of caloric restriction in aging. BMC Med. 2011;9:98.
Maegawa S, Lu Y, Tahara T, Lee JT, Madzo J, Liang S, et al. Caloric restriction delays age-related methylation drift. Nat Commun. 2017;8:539.
Drong AW, Lindgren CM, McCarthy MI. The genetic and epigenetic basis of type 2 diabetes and obesity. Clin Pharm Ther. 2012;92:707–15.
Ronn T, Volkov P, Gillberg L, Kokosar M, Perfilyev A, Jacobsen AL, et al. Impact of age, BMI and HbA1c levels on the genome-wide DNA methylation and mRNA expression patterns in human adipose tissue and identification of epigenetic biomarkers in blood. Hum Mol Genet. 2015;24:3792–813.
Aronica L, Levine AJ, Brennan K, Mi J, Gardner C, Haile RW, et al. A systematic review of studies of DNA methylation in the context of a weight loss intervention. Epigenomics. 2017;9:769–87.
Crujeiras AB, Casanueva FF. Obesity and the reproductive system disorders: epigenetics as a potential bridge. Hum Reprod Update. 2015;21:249–61.
Crujeiras AB, Diaz-Lagares A, Stefansson OA, Macias-Gonzalez M, Sandoval J, Cueva J, et al. Obesity and menopause modify the epigenomic profile of breast cancer. Endocr Relat Cancer. 2017;24:351–63.
Crujeiras AB, Morcillo S, Diaz-Lagares A, Sandoval J, Castellano-Castillo D, Torres E, et al. Identification of an episignature of human colorectal cancer associated with obesity by genome-wide DNA methylation analysis. Int J Obes. 2019;43:176–88.
Parrillo L, Spinelli R, Nicolo A, Longo M, Mirra P, Raciti GA, et al. Nutritional factors, DNA methylation, and risk of type 2 diabetes and obesity: perspectives and challenges. Int J Mol Sci. 2019;20.
Gillberg L, Perfilyev A, Brons C, Thomasen M, Grunnet LG, Volkov P, et al. Adipose tissue transcriptomics and epigenomics in low birthweight men and controls: role of high-fat overfeeding. Diabetologia. 2016;59:799–812.
Jacobsen SC, Brons C, Bork-Jensen J, Ribel-Madsen R, Yang B, Lara E, et al. Effects of short-term high-fat overfeeding on genome-wide DNA methylation in the skeletal muscle of healthy young men. Diabetologia. 2012;55:3341–9.
Jacobsen SC, Gillberg L, Bork-Jensen J, Ribel-Madsen R, Lara E, Calvanese V, et al. Young men with low birthweight exhibit decreased plasticity of genome-wide muscle DNA methylation by high-fat overfeeding. Diabetologia. 2014;57:1154–8.
Sziraki A, Tyshkovskiy A, Gladyshev VN. Global remodeling of the mouse DNA methylome during aging and in response to calorie restriction. Aging Cell. 2018;17:e12738.
Simons MJ, Koch W, Verhulst S. Dietary restriction of rodents decreases aging rate without affecting initial mortality rate - a meta-analysis. Aging Cell. 2013;12:410–4.
Funding
This study was supported by São Paulo Research Foundation (FAPESP) (grants #2016/06470-7, #2016/05638-1, and #2015/18669-0), Centro de Investigacion Biomedica En Red (CIBERobn) and grants (PI17/01287) from the Instituto de Salud Carlos III (ISCIII), Spain, co-financed by the European Regional Development Fund (FEDER). This research/work has also been supported by MINECO grants MTM2014-52876-R and MTM2017-82724-R, and by the Xunta de Galicia (Grupos de Referencia Competitiva ED431C-2016-015 and Centro Singular de Investigación de Galicia ED431G/01), all of them through the ERDF. ABC is funded by a research contract, “Miguel Servet” (CP17/00088), from the ISCIII, co-financed by the European Regional Development Fund.
Author information
Authors and Affiliations
Contributions
The authors’ responsibilities were as follows: CFN, MASP, and CBN contributed to the conception and design of the study. CC-O and JSM carried out the recruitment of the patients in the study. CFN, NYN, and MASP performed the laboratory analyses. CFN, AJ, and ABC analyzed the data, performed the statistical analysis, and drafted the manuscript. CFN and CBN wrote the manuscript. WSD, BG, and ABC critically revised the manuscript. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statement
This study was approved by the Hospital Ethics Committee of the School of Medicine of the University of São Paulo (CAAE:19442213.3.0000.5440) and was conducted in agreement with the Declaration of Helsinki. All participants gave informed written consent before their participation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Nicoletti, C.F., Cortes-Oliveira, C., Noronha, N.Y. et al. DNA methylation pattern changes following a short-term hypocaloric diet in women with obesity. Eur J Clin Nutr 74, 1345–1353 (2020). https://doi.org/10.1038/s41430-020-0660-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-020-0660-1