Abstract
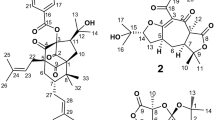
Three new antimalarial compounds, clonocoprogens A, B, and C, were isolated from the static culture of an Okinawan fungus, Clonostachys compactiuscula FKR-0021. These compounds were new analogs of N14-palmitoylcoprogen, reported as a siderophore. They showed moderate antimalarial activity against chloroquine-sensitive and chloroquine-resistant Plasmodium falciparum strains, with IC50 values ranging from 1.7 to 9.9 µM.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
World Health Organization. World Malaria Report 2019. World Health Organization. https://www.who.int/publications-detail/world-malaria-report-2019. 2019.
Blasco B, Leroy D, Fidock DA. Antimalarial drug resistance: linking Plasmodium falciparum parasite biology to the clinic. Nat Med. 2017;23:917–28.
Hayashi Y, Fukasawa W, Hirose T, Iwatsuki M, Hokari R, Ishiyama A, et al. Kozupeptins, antimalarial agents produced by Paracamarosporium species: isolation, structural elucidation, total synthesis, and bioactivity. Org Lett. 2019;21:2180–4.
Iwatsuki M, Takada S, Mori M, Ishiyama A, Namatame M, Nishihara-Tsukashima A, et al. In vitro and in vivo antimalarial activity of puberulic acid and its new analogs, viticolins A–C, produced by Penicillium sp. FKI-4410. J Antibiot. 2011;64:183–8.
Iwatsuki M, Nishihara-Tsukashima A, Ishiyama A, Namatame M, Watanabe Y, Handasah S, et al. Jogyamycin, a new antiprotozoal aminocyclopentitol antibiotic, produced by Streptomyces sp. a-WM-JG16.2. J Antibiot. 2012;65:169–71.
Anke H, Kinn J, Bergquist K, Sterner O. Production of siderophores by strains of the genus Trichoderma. Isolation and characterization of the new lipophilic coprogen derivative, palmitoylcoprogen. Biol Met. 1991;4:176–80.
Musgrave OC, Curvularin. Part I. Isolation and partial characterisation of a metabolic product from a new species of Curvularia. Chem Soc. 1956; 4301–5.
Ricardo FR, James BG, Jinx C, Carol AS. Ophiocerins A-D and ophioceric acid: tetrahydropyran derivatives and an African sesquiterpenoid from the freshwater aquatic fungus Ophioceras venezuelense. J Nat Prod. 2005;68:701–5.
Bao J, Zhang XY, Dong JJ, Xu XY, Nong XH, Qi SH, et al. Cyclopentane-condensed chromones from marine-derived fungus Penicillium oxalicum. Chem Lett. 2014;43:837–9.
Shiina I, Kawakita Y. The effective use of substituted benzoic anhydrides for the synthesis of carboxamides. Tetrahedron. 2004;60:4729–33.
Szymanski W, Zwolinska M, Ostaszewski R. Studies on the application of the Passerini reaction and enzymatic procedures to the synthesis of tripeptide mimetics. Tetrahedron. 2007;63:7647–53.
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl Acids Res. 1997;25:3389–402.
Hawksworth DL, Punithalingam E. New and interesting microfungi from Slapton, South Devonshire: Deuteromycotina II. Trans Br Mycol Soc. 1975;64:89–9.
Clay BF, Michael DB, William S. Structure of triornicin, a new siderophore. Biochemistry. 1981;20:2436–8.
Mahbubul AFJ, Steve KL, der Dickvan H. Siderophore mediated Iron (III) uptake in Gliocladium virens: 1. properties of cis-fusarinine, trans-fusarmine, dimerum acid, and their ferric complexes. J Inorg Biochem. 1986;28:417–30.
Harada K, Fujii K, Hayashi K, Suzuki M, Ikai Y, Oka H. Application of D,L-FDLA derivatization to determination of absolute configuration of constituent amino acids in peptide by advanced Marfey’s method. Tetrahedron Lett. 1996;37:3001–4.
Nonaka K, Kaifuchi S, Ōmura S, Masuma R. Five new Simplicillium species (Cordycipitaceae) from soils in Tokyo, Japan. Mycoscience. 2013;54:42–53.
Nonaka K, Miyazaki H, Iwatsuki M, Shiomi K, Tomoda H, Omura S, et al. Staphylotrichum boninense, a new hyphomycete (Chaetomiaceae) from soils in the Bonin Islands, Japan. Mycoscience. 2012;53:312–8.
Otoguro K, Kohana A, Manabe C, Ishiyama A, Ui H, Shiomi K, et al. Potent antimalarial activities of polyether antibiotic, X-206. J Antibiot. 2001;54:658–63.
Otoguro K, Ui H, Ishiyama A, Arai N, Kobayashi M, Takahashi Y, et al. In vitro antimalarial activities of the microbial metabolites. J Antibiot. 2003;56:322–4.
Saha M, Sarkar S, Sarkar B, Sharma BK, Bhattacharjee S, Tribedi P. Microbial siderophores and their potential applications: a review. Environ Sci Pollut Res. 2016;23:3984–99.
Anteloa L, Hof C, Welzel K, Eisfeld K, Sterner O, Anke H. Siderophores produced by Magnaporthe grisea in the presence and absence of iron. Z Naturforsch C. 2006;61c:461–4.
Jalal MAF, Van der Helm D. Siderophores of highly phytopathogenic Alternaria lonoipes. Structures of hydroxycoprogens. Biol Met. 1989;2:11–7.
Ferrer P, Vega-Rodriguez J, Tripathi AK, Jacobs-Lorena M, Sullivan DJ Jr. Antimalarial iron chelator FBS0701 blocks transmission by Plasmodium falciparum gametocyte activation inhibition. Antimicrob Agents Chemother. 2015;59:1418–26.
Gordeuk VR, Thuma PE, Brittenham GM, Zulu S, Swimwanza G, Mhangu A, et al. Iron chelation with desferrioxamine B in adults with asymptomatic Plasmodium falciparum parasitemia. Blood. 1992;79:308–12.
Mobarra N, Shanaki M, Ehteram H, Nasiri H, Sahmani M, Saeidi M, et al. A review on iron chelators in treatment of iron overload syndromes. Int J Hematol Stem Cell Res. 2016;10:239–47.
Acknowledgements
We thank Dr K. Nagai and Ms N. Sato (School of Pharmacy, Kitasato University) for various instrumental analyses. We thank Dr T. Nakashima (Kitasato Institute for Life Sciences, Kitasato University) for ESI-MS/MS analysis. We thank Dr A. Také (School of Medicine, Kitasato University) for useful suggestion concerning the experiments. This study was supported by Okinawan Create Leading Projects in Growing Fields 2017–2019, OKINAWA Prefectural Government. This study was also partially supported by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)) from the Japan Agency for Medical Research & Development (AMED) under grant number JP19am0101096. This research was also partially supported by AMED under grant number 18ae0101047.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ouchi, T., Watanabe, Y., Nonaka, K. et al. Clonocoprogens A, B and C, new antimalarial coprogens from the Okinawan fungus Clonostachys compactiuscula FKR-0021. J Antibiot 73, 365–371 (2020). https://doi.org/10.1038/s41429-020-0292-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41429-020-0292-7
This article is cited by
-
New antimalarial iromycin analogs produced by Streptomyces sp. RBL-0292
The Journal of Antibiotics (2024)
-
New antimalarial fusarochromanone analogs produced by the fungal strain Fusarium sp. FKI-9521
The Journal of Antibiotics (2023)
-
Diatretol, an α, α′-dioxo-diketopiperazine, is a potent in vitro and in vivo antimalarial
The Journal of Antibiotics (2021)
-
Discovery and genetic identification of amphiphilic coprogen siderophores from Trichoderm hypoxylon
Applied Microbiology and Biotechnology (2021)