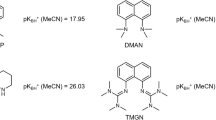
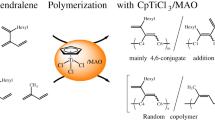
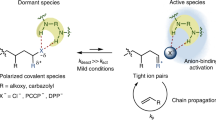
Abstract
Methylaluminoxane (MAO) is known as a versatile activator of transition metal catalysts for coordination polymerization reactions, even though its precise structure and the origin of its activation properties have not been fully investigated. Considering that MAO exists as a counter anion that can interact with cationic metal species in a polymerization medium, the ability to impart structural modifications to MAO would provide a new direction for controlling the polymerization behavior. Here, the modification, the composition control, and a new preparation method of MAO will be summarized, as well as the application of modified MAO for the synthesis of stereoblock polydienes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Resconi L, Cavallo L, Fait A, Piemontesi F. Selectivity in propene polymerization with metallocene catalysts. Chem Rev. 2000;100:1253–346.
Möhring PC, Coville NJ. Group 4 metallocene polymerisation catalysts: quantification of ring substituent steric effects. Coord Chem Rev. 2006;250:18–35.
Nakamura A, Ito S, Nozaki K. Coordination-Insertion copolymerization of fundamental polar monomers. Chem Rev. 2009;109:5215–44.
Chen EY-X. Coordination polymerization of polar vinyl monomers by single-site metal catalysts. Chem Rev. 2009;109:5157–214.
Franssen NMG, Reeka JNH, de Bruin B. Synthesis of functional ‘polyolefins’: state of the art and remaining challenges. Chem Soc Rev. 2013;42:5809–32.
Dong J-Y, Hu Y. Design and synthesis of structurally well-defined functional polyolefins via transition metal-mediated olefin polymerization chemistry. Coord Chem Rev. 2006;250:47–65.
Domskia GJ, Rose JM, Coates GW, Bolig AD, Brookhart M. Living alkene polymerization: new methods for the precision synthesis of polyolefins. Prog Polym. 2007;32:30–92.
Valente A, Mortreux A, Visseaux M, Zinck P. Coordinative chain transfer polymerization. Chem Rev. 2013;113:3836–57.
Chen EY-X, Marks TJ. Cocatalysts for metal-catalyzed olefin polymerization: activators, activation processes, and structure−activity relationships. Chem Rev. 2000;100:1391–434.
Bochmann M. The chemistry of catalyst activation: the case of Group 4 polymerization catalysts. Organometallics. 2010;29:4711–40.
Bryliakov KP, Talsi EP. Frontiers of mechanistic studies of coordination polymerization and oligomerization of α-olefins. Coord Chem Rev. 2012;256:2994–3007.
Chen M-C, Roberts JAS, Marks TJ. Marked counteranion effects on single-site olefin polymerization processes. correlations of ion pair structure and dynamics with polymerization activity, chain transfer, and syndioselectivity. J Am Chem Soc. 2004;126:4605–25.
Roberts JAS, Chen M-C, Seyam AM, Li L, Zuccaccia C, Stahl NG, et al. Diverse stereocontrol effects induced by weakly coordinating anions. Stereospecific olefin polymerization pathways at archetypal C s- and C 1-symmetric metallocenium catalysts using mono- and polynuclear halo-perfluoroarylmetalates as cocatalysts. J Am Chem Soc. 2007;129:12713–33.
Zurek E, Ziegler T. Theoretical studies of the structure and function of MAO (methylaluminoxane). Prog Polym Sci. 2004;29:107–48.
Kaminsky W. Discovery of methylaluminoxane as cocatalyst for olefin polymerization. Macromolecules. 2012;45:3289–97.
Kaminsky W. Methylaluminoxane: key component for new polymerization catalysts. Adv Polym Sci. 2013;258:1–28.
Zijlstra HS, Harder S. Methylalumoxane—history, production, properties, and applications. Eur J Inorg Chem. 2015;2015:19–43.
Tanaka R, Hirose T, Nakayama Y, Shiono T. The preparation of boron-containing aluminoxanes and their application as cocatalysts in the polymerization of olefins. Polym J. 2016;48:67–71.
Tanaka R, Yuuya K, Sato H, Eberhardt P, Nakayama Y, Shiono T. Synthesis of stereodiblock polyisoprene consisting of cis-1,4 and trans-1,4 sequences by neodymium catalyst: change of the stereospecificity triggered by aluminum compound. Polym Chem. 2016;7:1239–43.
Tanaka R, Kawahara T, Shinto Y, Nakayama Y, Shiono T. An alternative method for the preparation of trialkylaluminum-depleted modified-methylaluminoxane (dMMAO). Macromolecules. 2017;50:5989–93.
Tanaka R, Shinto Y, Nakayama Y, Shiono T. Synthesis of stereodiblock polybutadiene using Cp*Nd(BH4)2(thf)2 as a catalyst. Catalysts. 2017;7:284.
Tanaka R, Yamashita T, Tonoko N, Nakayama Y, Shiono T. Effect of added phenols and silanol on the cocatalyst activity of methylaluminoxane. Kobunshi Ronbunshu. 2018;75:551–6.
Okajima Y, Nakayama Y, Shiono T, Tanaka R. Preparation of methylaluminoxane from carbon dioxide. Eur J Inorg Chem. 2019;2019:2392–5.
Crappo CC, Malpass DB. Synthesis of methylaluminoxanes. Eur. Pat. EP0372617. 1990.
Kissin YV, Brandolini AJ. An alternative route to methylalumoxane: synthesis, structure, and the use of model methylalumoxanes as cocatalysts for transition metal complexes in polymerization reactions. Macromolecules. 2003;36:18–26.
Lee CH, Lee SJ, Park JW, Kim KH, Lee BY, Oh JS. Preparation of Al(C6F5)3 and its use for the modification of methylalumoxane. J Mol Catal A. 1998;132:231–239.
Hasan T, Ioku A, Nishii K, Shiono T, Ikeda T. Syndiospecificliving polymerization of propene with [t-BuNSiMe2Flu]TiMe2 using MAO as cocatalyst. Macromolecules. 2001;34:3142–5.
Babushkin DE, Semikolenova NV, Panchenko VN, Sobolev AP, Zakharov VA, Talsi EP. Multinuclear NMR investigation of methylaluminoxane. Macromol Chem Phys. 1997;198:3845–54.
Resconi L, Bossi S, Abis L. Study on the role of methylalumoxane in homogeneous olefin polymerization. Macromolecules. 1990;23:4489–91.
Ehm C, Cipullo R, Budzelaar PHM, Busico V. Role(s) of TMA in polymerization. Dalton Trans. 2016;45:6847–55.
Busico V, Cipullo R, Cutillo F, Friederichs N, Ronca S, Wang B. Improving the performance of methylalumoxane: a facile and efficient method to trap “free” trimethylaluminum. J Am Chem Soc. 2003;125:12402–3.
Romano D, Andablo-Reyes EA, Ronca S, Rastogi S. Effect of a cocatalyst modifier in the synthesis of ultrahigh molecular weight polyethylene having reduced number of entanglements. J Polym Sci Part A. 2013;51:1630–5.
Tanaka R, Kamei I, Cai Z, Nakayama Y, Shiono T. Ethylene-propylene copolymerization behavior of ansa-dimethylsilylene(fluorenyl)(amido)dimethyltitanium complex: application to ethylene-propylene-diene or ethylene-propylene-norbornene terpolymers. J Polym Sci Part A. 2015;53:685–91.
Rocchigiani L, Busico V, Pastore A, Macchioni A. Probing the interactions between all components of the catalytic pool for homogeneous olefin polymerisation by diffusion NMR spectroscopy. Dalton Trans. 2013;42:9104.
Zaccaria F, Zuccaccia C, Cipullo R, Budzelaar PHM, Macchioni A, Busico V, et al. On the nature of the lewis acidic sites in “TMA-Free” phenol-modified methylaluminoxane. Eur J Inorg Chem. https://doi.org/10.1002/ejic.201901035.
Hagimoto H, Shiono T, Ikeda T. Supporting effects of methylaluminoxane on the living polymerization of propylene with a chelating (diamide)dimethyltitanium complex. Macromol Chem Phys. 2004;205:19–26.
dos Santos JHZ, Krug C, da Rosa MB, Stedile FC, Dupont J, de Camargo, et al. The effect of silica dehydroxylation temperature on the activity of SiO2-supported zirconocene catalysts. J Mol Catal A. 1999;139:199–207.
Scollard JD, McConville DH, Payne NC, Vittal JJ. Polymerization of α-olefins by chelating diamide complexes of titanium. Macromolecules. 1996;29:5241–3.
Hagimoto H, Shiono T, Ikeda T. Living polymerization of propene with a chelating diamide complex of titanium using dried methylaluminoxane. Macromol Rapid Commun. 2002;23:72–78.
Maiwald S, Weissenborn H, Sommer C, Taube R. Komplexkatalyse: LIX. Die Katalyse der 1,4-trans-polymerisation des butadiens mit tris(allyl)neodym(III) Nd(η 3-C3H5)3 als Einkomponentenkatalysator—Kinetik und Reaktionsmechanismus. J Organomet Chem. 2001;640:1–9.
Bonnet F, Visseaux M, Pereira A, Barbier-Baudry D. Highly trans-stereospecific isoprene polymerization by neodymium borohydrido catalysts. Macromolecules. 2005;38:3162–9.
Maiwald S, Taube R, Hemling H, Schumann HLII. Synthese und Charakterisierung von bis- und mono-(η 3-allyl)neodym(III)-chlorid, Nd(η 3-C3H5)2Cl·1.5 THF und Nd(η 3-C3H5)Cl2·2THF, zur Gewinnung hoch aktiver und selektiver Komplexkatalysatoren für die 1,4-cis-Polymerisation des Butadiens. J Organomet Chem. 1998;522:195–204.
Visseaux M, Mainil M, Terrier M, Mortreux A, Roussel P, Mathivet T, et al. Cationic borohydrido–neodymium complex: Synthesis, characterization and its application as an efficient pre-catalyst for isoprene polymerization. Dalton Trans. 2008;37:4558–61.
Tanaka R, Shinto Y, Matsuzaki R, Nakayama Y, Shiono T. Stereospecific polymerization of conjugated dienes using neodymium alkylborohydride complexes. Dalton Trans. 2019;48:7267–73.
Dalet T, Cramail H, Deffieux A. Non-hydrolytic route to aluminoxane—type derivative for metallocene activation towards olefin polymerisation. Macromol Chem Phys. 2004;205:1394–401.
Acknowledgements
This work was financially supported by the Grant-in-Aid for Young Scientists (B) (No. 26810070) and the Grant-in-Aid for Young Scientists (18K14276) from the Japan Society for the Promotion of Science (JSPS), Japan. The generous donation of MMAO and MAO from Tosoh-Finechem Co. (Japan) is greatly appreciated. The authors wish to thank Prof. Takeshi Shiono for the kind support as well as Takaaki Hirose, Kaede Yuuya, Yuto Shinto, Naoki Tonoko, Ryusei Matsuzaki, and Yuuya Okajima for their contributions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tanaka, R. Precise control of coordination polymerization via the modification of methylaluminoxane (MAO). Polym J 52, 661–670 (2020). https://doi.org/10.1038/s41428-020-0325-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41428-020-0325-x