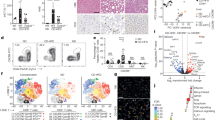
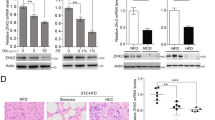
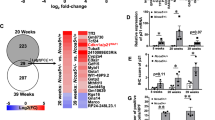
Abstract
Obesity is a major risk factor for cancers including hepatocellular carcinoma (HCC) that develops from a background of non-alcoholic fatty liver disease (NAFLD). Hypercholesterolemia is a common comorbidity of obesity. Although cholesterol biosynthesis mainly occurs in the liver, its role in HCC development of obese people remains obscure. Using high-fat high-carbohydrate diet-associated orthotopic and spontaneous NAFLD-HCC mouse models, we found that hepatic cholesterol accumulation in obesity selectively suppressed natural killer T (NKT) cell-mediated antitumor immunosurveillance. Transcriptome analysis of human liver revealed aberrant cholesterol metabolism and NKT cell dysfunction in NAFLD patients. Notably, cholesterol-lowering rosuvastatin restored NKT expansion and cytotoxicity to prevent obesogenic diet-promoted HCC development. Moreover, suppression of hepatic cholesterol biosynthesis by a mammalian target of rapamycin (mTOR) inhibitor vistusertib preceded tumor regression, which was abolished by NKT inactivation but not CD8+ T cell depletion. Mechanistically, sterol regulatory element-binding protein 2 (SREBP2)-driven excessive cholesterol production from hepatocytes induced lipid peroxide accumulation and deficient cytotoxicity in NKT cells, which were supported by findings in people with obesity, NAFLD and NAFLD-HCC. This study highlights mTORC1/SREBP2/cholesterol-mediated NKT dysfunction in the tumor-promoting NAFLD liver microenvironment, providing intervention strategies that invigorating NKT cells to control HCC in the obesity epidemic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data in this study are available in the text and its Supplementary Information files. The data that support the findings of this study are available from the corresponding authors upon reasonable request. The RNAseq and scRNAseq data analyzed in this study were accessed with the Gene Expression Omnibus (GEO) series accession numbers: GSE135251 and GSE159977.
References
Lauby-Secretanuby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K. Body fatness and cancer-viewpoint of the IARC Working Group. N Engl J Med. 2016;375:794–8. https://doi.org/10.1056/NEJMsr1606602
Quail DF, Dannenberg AJ. The obese adipose tissue microenvironment in cancer development and progression. Nat Rev Endocrinol. 2019;15:139–54. https://doi.org/10.1038/s41574-018-0126-x
Demark-Wahnefriedmark-Wahnefried W, Platz EA, Ligibel JA, Blair CK, Courneya KS, Meyerhardt JA, et al. The role of obesity in cancer survival and recurrence. Cancer Epidemiol Biomark Prev. 2012;21:1244–59. https://doi.org/10.1158/1055-9965.EPI-12-0485
Avgerinos KI, Spyrou N, Mantzoros CS, Dalamaga M. Obesity and cancer risk: emerging biological mechanisms and perspectives. Metabolism. 2019;92:121–35. https://doi.org/10.1016/j.metabol.2018.11.001
Ringelngel AE, Drijvers JM, Baker GJ, Catozzi A, García-Cañaveras JC, Gassaway BM, et al. Obesity shapes metabolism in the tumor microenvironment to suppress anti-tumor immunity. Cell. 2020;183:1848–66 e1826. https://doi.org/10.1016/j.cell.2020.11.009
Wardrd ZJ, Bleich SN, Cradock AL, Barrett JL, Giles CM, Flax C, et al. Projected U.S. state-level prevalence of adult obesity and severe obesity. N Engl J Med. 2019;381:2440–50. https://doi.org/10.1056/NEJMsa1909301
Younossiunossi Z, Anstee QM, Marietti M, Hardy T, Henry L, Eslam M, et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2018;15:11–20. https://doi.org/10.1038/nrgastro.2017.109
Bhaskaranaskaran K, Douglas I, Forbes H, dos-Santos-Silva I, Leon DA, Smeeth L. Body-mass index and risk of 22 specific cancers: a population-based cohort study of 5.24 million UK adults. Lancet. 2014;384:755–65. https://doi.org/10.1016/S0140-6736(14)60892-8
Powell EE, Wong VW, Rinella M. Non-alcoholic fatty liver disease. Lancet. 2021;397:2212–24. https://doi.org/10.1016/S0140-6736(20)32511-3
Grohmannrohmann M, Wiede F, Dodd GT, Gurzov EN, Ooi GJ, Butt T, et al. Obesity drives STAT-1-dependent NASH and STAT-3-dependent HCC. Cell. 2018;175:1289–306 e1220. https://doi.org/10.1016/j.cell.2018.09.053
Masaroneasarone M, Rosato V, Dallio M, Gravina AG, Aglitti A, Loguercio C, et al. Role of oxidative stress in pathophysiology of nonalcoholic fatty liver disease. Oxid Med Cell Longev. 2018;2018:9547613. https://doi.org/10.1155/2018/9547613
Anstee QM, Reeves HL, Kotsiliti E, Govaere O, Heikenwalder M. From NASH to HCC: current concepts and future challenges. Nat Rev Gastroenterol Hepatol. 2019;16:411–28. https://doi.org/10.1038/s41575-019-0145-7
Ioannou GN. The role of cholesterol in the pathogenesis of NASH. Trends Endocrinol Metab. 2016;27:84–95. https://doi.org/10.1016/j.tem.2015.11.008
Ståhlberg D, Rudling M, Angelin B, Björkhem I, Forsell P, Nilsell K, et al. Hepatic cholesterol metabolism in human obesity. Hepatology. 1997;25:1447–50. https://doi.org/10.1002/hep.510250623
Min HK, Kapoor A, Fuchs M, Mirshahi F, Zhou H, Maher J, et al. Increased hepatic synthesis and dysregulation of cholesterol metabolism is associated with the severity of nonalcoholic fatty liver disease. Cell Metab. 2012;15:665–74. https://doi.org/10.1016/j.cmet.2012.04.004
Huang DQ, El-Serag HB, Loomba R. Global epidemiology of NAFLD-related HCC: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2021;18:223–38. https://doi.org/10.1038/s41575-020-00381-6
Hsiang JC, Wong GL-H, Tse Y-K, Wong VW-S, Yip TC-F, Chan HL-Y. Statin and the risk of hepatocellular carcinoma and death in a hospital-based hepatitis B-infected population: a propensity score landmark analysis. J Hepatol. 2015;63:1190–7. https://doi.org/10.1016/j.jhep.2015.07.009
McEwen-Smith RM, Salio M, Cerundolo V. The regulatory role of invariant NKT cells in tumor immunity. Cancer Immunol Res. 2015;3:425–35. https://doi.org/10.1158/2326-6066.CIR-15-0062
Gao B, Radaeva S, Park O. Liver natural killer and natural killer T cells: immunobiology and emerging roles in liver diseases. J Leukoc Biol. 2009;86:513–28. https://doi.org/10.1189/JLB.0309135
Bandyopadhyay K, Marrero I, Kumar V. NKT cell subsets as key participants in liver physiology and pathology. Cell Mol Immunol. 2016;13:337–46. https://doi.org/10.1038/cmi.2015.115
Ma C, Han M, Heinrich B, Fu Q, Zhang Q, Sandhu M, et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells. Science. 2018;360. https://doi.org/10.1126/science.aan5931.
Yang W, Feng Y, Zhou J, Cheung OK-W, Cao J, Wang J, et al. A selective HDAC8 inhibitor potentiates antitumor immunity and efficacy of immune checkpoint blockade in hepatocellular carcinoma. Sci Transl Med. 2021;13. https://doi.org/10.1126/scitranslmed.aaz6804.
Zeng X, Zhou J, Xiong Z, Sun H, Yang W, Mok MTS, et al. Cell cycle-related kinase reprograms the liver immune microenvironment to promote cancer metastasis. Cell Mol Immunol. 2021;18:1005–15. https://doi.org/10.1038/s41423-020-00534-2
Tian Y, Wong VW, Wong GL, Yang W, Sun H, Shen J, et al. Histone deacetylase HDAC8 promotes insulin resistance and beta-catenin activation in NAFLD-associated hepatocellular carcinoma. Cancer Res. 2015;75:4803–16. https://doi.org/10.1158/0008-5472.CAN-14-3786
Sun H, Yang W, Tian Y, Zeng X, Zhou J, Mok M, et al. An inflammatory-CCRK circuitry drives mTORC1-dependent metabolic and immunosuppressive reprogramming in obesity-associated hepatocellular carcinoma. Nat Commun. 2018;9:5214. https://doi.org/10.1038/s41467-018-07402-8
Krenkel O, Hundertmark J, Abdallah AT, Kohlhepp M, Puengel T, Roth T, et al. Myeloid cells in liver and bone marrow acquire a functionally distinct inflammatory phenotype during obesity-related steatohepatitis. Gut. 2020;69:551–63. https://doi.org/10.1136/gutjnl-2019-318382
Yao L, Abe M, Kawasaki K, Akbar SMF, Matsuura B, Onji M, et al. Characterization of liver monocytic myeloid-derived suppressor cells and their role in a murine model of non-alcoholic fatty liver disease. PLoS ONE. 2016;11:e0149948. https://doi.org/10.1371/journal.pone.0149948
Govaere O, Cockell S, Tiniakos D, Queen R, Younes R, Vacca M, et al. Transcriptomic profiling across the nonalcoholic fatty liver disease spectrum reveals gene signatures for steatohepatitis and fibrosis. Sci Transl Med. 2020;12:150. https://doi.org/10.1126/scitranslmed.aba4448
Luo J, Yang H, Song BL. Mechanisms and regulation of cholesterol homeostasis. Nat Rev Mol Cell Biol. 2020;21:225–45. https://doi.org/10.1038/s41580-019-0190-7
Pfister D, Nunez NG, Pinyol R, Govaere O, Pinter M, Szydlowska M, et al. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature. 2021;592:450–6. https://doi.org/10.1038/s41586-021-03362-0
Engel I, Seumois G, Chavez L, Samaniego-Castruita D, White B, Chawla A, et al. Innate-like functions of natural killer T cell subsets result from highly divergent gene programs. Nat Immunol. 2016;17:728–39. https://doi.org/10.1038/ni.3437
Baranek T, Lebrigand K, de Amat Herbozo C, Gonzalez L, Bogard G, Dietrich C, et al. High dimensional single-cell analysis reveals iNKT cell developmental trajectories and effector fate decision. Cell Rep. 2020;32:108116. https://doi.org/10.1016/j.celrep.2020.108116
Kapanadze T, Gamrekelashvili J, Ma C, Chan C, Zhao F, Hewitt S, et al. Regulation of accumulation and function of myeloid derived suppressor cells in different murine models of hepatocellular carcinoma. J Hepatol. 2013;59:1007–13. https://doi.org/10.1016/j.jhep.2013.06.010
Kawachi Y, Fujishima Y, Nishizawa H, Nakamura T, Akari S, Murase T, et al. Increased plasma XOR activity induced by NAFLD/NASH and its possible involvement in vascular neointimal proliferation. JCI Insight. 2021;6:1. https://doi.org/10.1172/jci.insight.144762
Zhang X, Coker OO, Chu ES, Fu K, Lau HCH, Wang YX, et al. Dietary cholesterol drives fatty liver-associated liver cancer by modulating gut microbiota and metabolites. Gut. 2021;70:761–74. https://doi.org/10.1136/gutjnl-2019-319664
Liu D, Wong CC, Zhou Y, Li C, Chen H, Ji F, et al. Squalene epoxidase induces nonalcoholic steatohepatitis via binding to carbonic anhydrase III and is a therapeutic target. Gastroenterology. 2021;160:2467–82 e2463. https://doi.org/10.1053/j.gastro.2021.02.051
Basu B, Dean E, Puglisi M, Greystoke A, Ong M, Burke W, et al. First-in-human pharmacokinetic and pharmacodynamic study of the dual m-TORC 1/2 inhibitor AZD2014. Clin Cancer Res. 2015;21:3412–9. https://doi.org/10.1158/1078-0432.CCR-14-2422
Janku F, Yap TA, Meric-Bernstam F. Targeting the PI3K pathway in cancer: are we making headway? Nat Rev Clin Oncol. 2018;15:273–91. https://doi.org/10.1038/nrclinonc.2018.28
Liscum L, Faust JR. The intracellular transport of low density lipoprotein-derived cholesterol is inhibited in Chinese hamster ovary cells cultured with 3-beta-[2-(diethylamino)ethoxy]androst-5-en-17-one. J Biol Chem. 1989;264:11796–806.
Schmid H, Schneidawind C, Jahnke S, Kettemann F, Secker KA, Duerr-Stoerzer S, et al. Culture-expanded human invariant natural killer T cells suppress T-cell alloreactivity and eradicate leukemia. Front Immunol. 2018;9:1817. https://doi.org/10.3389/fimmu.2018.01817
Girotti AW, Korytowski W. Cholesterol hydroperoxide generation, translocation, and reductive turnover in biological systems. Cell Biochem Biophys. 2017;75:413–9. https://doi.org/10.1007/s12013-017-0799-0
Forcina GC, Dixon SJ. GPX4 at the crossroads of lipid homeostasis and ferroptosis. Proteomics. 2019;19:e1800311. https://doi.org/10.1002/pmic.201800311
Girotti AW, Korytowski W. Cholesterol peroxidation as a special type of lipid oxidation in photodynamic systems. Photochem Photobiol. 2019;95:73–82. https://doi.org/10.1111/php.12969
Xu S, Chaudhary O, Rodríguez-Morales P, Sun X, Chen D, Zappasodi R, et al. Uptake of oxidized lipids by the scavenger receptor CD36 promotes lipid peroxidation and dysfunction in CD8(+) T cells in tumors. Immunity. 2021;54:1561–77 e1567. https://doi.org/10.1016/j.immuni.2021.05.003
Khan AA, Agarwal H, Reddy SS, Arige V, Natarajan B, Gupta V, et al. MicroRNA 27a Is a Key Modulator of Cholesterol Biosynthesis. Mol Cell Biol. 2020;40. https://doi.org/10.1128/MCB.00470-19.
Ma X, Xiao L, Liu L, Ye L, Su P, Bi E, et al. CD36-mediated ferroptosis dampens intratumoral CD8(+) T cell effector function and impairs their antitumor ability. Cell Metab. 2021;33:1001–12 e1005. https://doi.org/10.1016/j.cmet.2021.02.015
Neuschwander-Tetri BA. Therapeutic landscape for NAFLD in 2020. Gastroenterology. 2020;158:1984–98 e1983. https://doi.org/10.1053/j.gastro.2020.01.051
Liu D, Wong CC, Zhou Y, Li C, Chen H, Ji F, et al. Squalene epoxidase induces nonalcoholic steatohepatitis via binding to carbonic anhydrase 3 and is a therapeutic target. Gastroenterology. 2021;160:2467–82. https://doi.org/10.1053/j.gastro.2021.02.051
El-Serag HB, Johnson ML, Hachem C, Morgana RO. Statins are associated with a reduced risk of hepatocellular carcinoma in a large cohort of patients with diabetes. Gastroenterology. 2009;136:1601–8. https://doi.org/10.1053/j.gastro.2009.01.053
McGlynn KA, Divine GW, Sahasrabuddhe VV, Engel LS, VanSlooten A, Wells K, et al. Statin use and risk of hepatocellular carcinoma in a U.S. population. Cancer Epidemiol. 2014;38:523–7. https://doi.org/10.1016/j.canep.2014.06.009
Kaplan DE, Serper MA, Mehta R, Fox R, John B, Aytaman A, et al. Effects of hypercholesterolemia and statin exposure on survival in a large national cohort of patients with cirrhosis. Gastroenterology. 2019;156:1693–706 e1612. https://doi.org/10.1053/j.gastro.2019.01.026
LaMarche NM, Kane H, Kohlgruber AC, Dong H, Lynch L, Brenner MB. Distinct iNKT cell populations use IFNgamma or ER stress-induced IL-10 to control adipose tissue homeostasis. Cell Metab. 2020;32:243–58 e246. https://doi.org/10.1016/j.cmet.2020.05.017
Wolf MJ, Adili A, Piotrowitz K, Abdullah Z, Boege Y, Stemmer K, et al. Metabolic activation of intrahepatic CD8+ T cells and NKT cells causes nonalcoholic steatohepatitis and liver cancer via cross-talk with hepatocytes. Cancer Cell. 2014;26:549–64. https://doi.org/10.1016/j.ccell.2014.09.003
Dudek M, Pfister D, Donakonda S, Filpe P, Schneider A, Laschinger M, et al. Auto-aggressive CXCR6(+) CD8 T cells cause liver immune pathology in NASH. Nature. 2021;592:444–9. https://doi.org/10.1038/s41586-021-03233-8
Huang B, Song BL, Xu C. Cholesterol metabolism in cancer: mechanisms and therapeutic opportunities. Nat Metab. 2020;2:132–41. https://doi.org/10.1038/s42255-020-0174-0
Ma X, Bi E, Lu Y, Su P, Huang C, Liu L, et al. Cholesterol induces CD8(+) T cell exhaustion in the tumor microenvironment. Cell Metab. 2019;30:143–56 e145. https://doi.org/10.1016/j.cmet.2019.04.002
Ma X, Bi E, Huang C, Lu Y, Xue G, Guo X, et al. Cholesterol negatively regulates IL-9-producing CD8(+) T cell differentiation and antitumor activity. J Exp Med. 2018;215:1555–69. https://doi.org/10.1084/jem.20171576
Matter MS, Decaens T, Andersen JB, Thorgeirsson SS. Targeting the mTOR pathway in hepatocellular carcinoma: current state and future trends. J Hepatol. 2014;60:855–65. https://doi.org/10.1016/j.jhep.2013.11.031
Xue L, Qi H, Zhang H, Ding L, Huang Q, Zhao D, et al. Targeting SREBP-2-regulated mevalonate metabolism for cancer therapy. Front Oncol. 2020;10:1510 https://doi.org/10.3389/fonc.2020.01510
Moon SH, Huang CH, Houlihan SL, Regunath K, Freed-Pastor WA, Morris JP, et al. p53 represses the mevalonate pathway to mediate tumor suppression. Cell. 2019;176:564–80 e519. https://doi.org/10.1016/j.cell.2018.11.011
Welte T, Kim IS, Tian L, Gao X, Wang H, Li J, et al. Oncogenic mTOR signalling recruits myeloid-derived suppressor cells to promote tumour initiation. Nat Cell Biol. 2016;18:632–44. https://doi.org/10.1038/ncb3355
Marengo A, Rosso C, Bugianesi E. Liver cancer: connections with obesity, fatty liver, and cirrhosis. Annu Rev Med. 2016;67:103–17. https://doi.org/10.1146/annurev-med-090514-013832
Tian D, Hong H, Shang W, Ho CC, Dong J, Tian XY. Deletion of Ppard in CD11c(+) cells attenuates atherosclerosis in ApoE knockout mice. FASEB J. 2020;34:3367–78. https://doi.org/10.1096/fj.201902069R
Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012;16:284–7. https://doi.org/10.1089/omi.2011.0118
Acknowledgements
We acknowledge Prof. Lars Zender and Prof. Tim F. Greten for their kind gift of the RIL-175 HCC cell line. This project is supported by the University Grants Committee through the Collaborative Research Fund (C4045-18W), AstraZeneca Research Program (2017), General Research Fund (14105419, 14104820), the Li Ka Shing Foundation, and the Terry Fox Foundation.
Author information
Authors and Affiliations
Contributions
WT, JZ, and ASLC conceived the study. WT and JZ conducted experiments, analyzed, and interpreted data. HW assisted with bioinformatics analysis. WY, YF, HW, MTSM, LZ, ZL, XL, ZX, XZ, J.Wang, J.Lu., J.Li., and XT assisted with animal experiments. HS, PCY, YH, HML, CCHL, HHWL, AWHC, KFT, J.Wong, PBSL, KKCN, SKHW, VWSW, and APSK. provided patient materials, acquired clinical data, assisted in analyzing NKT cell frequency in patient specimens and interpreted with clinical data. WT, JZ, and ASLC wrote the manuscript. JZ, PBSL, KKCN, VWSW, APSK, JJYS, and ASLC reviewed and edited the manuscript. JZ, JJYS, and ASLC acquired funding for the study. JZ and ASLC supervised the study. All authors read and provided feedback on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
JZ and ASLC received the funding support and drug (vistusertib) from AstraZeneca. VWSW has consultancy in 3V-BIO, AbbVie, Allergan, Boehringer Ingelheim, Center for Outcomes Research in Liver Diseases, Echosens, Gilead Sciences, Hanmi Pharmaceutical, Intercept, Inventiva, Merck, Novartis, Novo Nordisk, Perspectum Diagnostics, Pfizer, ProSciento, Sagimet Biosciences, TARGET PharmaSolutions and Terns; has lectures in Abbott, AbbVie, Bristol-Myers Squibb, Echosens, Gilead Sciences and Novo Nordisk; received research grants from Gilead Sciences; and holds stock as co-founder of Illuminatio Medical Technology Limited. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Tang, W., Zhou, J., Yang, W. et al. Aberrant cholesterol metabolic signaling impairs antitumor immunosurveillance through natural killer T cell dysfunction in obese liver. Cell Mol Immunol 19, 834–847 (2022). https://doi.org/10.1038/s41423-022-00872-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-022-00872-3