Abstract
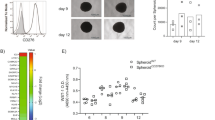
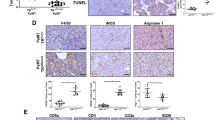
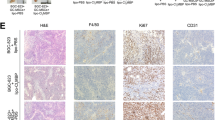
In cancer, myeloid cells have tumor-supporting roles. We reported that the protein GPNMB (glycoprotein nonmetastatic B) was profoundly upregulated in macrophages interacting with tumor cells. Here, using mouse tumor models, we show that macrophage-derived soluble GPNMB increases tumor growth and metastasis in Gpnmb-mutant mice (DBA/2J). GPNMB triggers in the cancer cells the formation of self-renewing spheroids, which are characterized by the expression of cancer stem cell markers, prolonged cell survival and increased tumor-forming ability. Through the CD44 receptor, GPNMB mechanistically activates tumor cells to express the cytokine IL-33 and its receptor IL-1R1L. We also determined that recombinant IL-33 binding to IL-1R1L is sufficient to induce tumor spheroid formation with features of cancer stem cells. Overall, our results reveal a new paracrine axis, GPNMB and IL-33, which is activated during the cross talk of macrophages with tumor cells and eventually promotes cancer cell survival, the expansion of cancer stem cells and the acquisition of a metastatic phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Mantovani, A., Marchesi, F., Malesci, A., Laghi, L. & Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 14, 399–416 (2017).
Belgiovine, C., D’Incalci, M., Allavena, P. & Frapolli, R. Tumor-associated macrophages and anti-tumor therapies: complex links. Cell. Mol. life Sci. 73, 2411–2424 (2016).
Biswas, S. K., Allavena, P. & Mantovani, A. Tumor-associated macrophages: functional diversity, clinical significance, and open questions. Semin. Immunopathol. 35, 585–600 (2013).
Cassetta, L. & Pollard, J. W. Targeting macrophages: therapeutic approaches in cancer. Nat. Rev. Drug Discov. 17, 887–904 (2018).
DeNardo, D. G. & Ruffell, B. Macrophages as regulators of tumour immunity and immunotherapy. Nat. Rev. Immunol. 19, 369–382 (2019).
Solinas, G. et al. Tumor-conditioned macrophages secrete migration-stimulating factor: a new marker for M2-polarization, influencing tumor cell motility. J. Immunol. 185, 642–652 (2010).
Mayi, T. H. et al. Human adipose tissue macrophages display activation of cancer-related pathways. J. Biol. Chem. 287, 21904–21913 (2012).
Rose, A. A. et al. ADAM10 releases a soluble form of the GPNMB/Osteoactivin extracellular domain with angiogenic properties. PLoS ONE 5, e12093 (2010).
Weterman, M. A. et al. nmb, a novel gene, is expressed in low-metastatic human melanoma cell lines and xenografts. Int. J. Cancer 60, 73–81 (1995).
Safadi, F. F. et al. Cloning and characterization of osteoactivin, a novel cDNA expressed in osteoblasts. J. Cell Biochem. 84, 12–26 (2001).
Shikano, S., Bonkobara, M., Zukas, P. K. & Ariizumi, K. Molecular cloning of a dendritic cell-associated transmembrane protein, DC-HIL, that promotes RGD-dependent adhesion of endothelial cells through recognition of heparan sulfate proteoglycans. J. Biol. Chem. 276, 8125–8134 (2001).
Haralanova-Ilieva, B., Ramadori, G. & Armbrust, T. Expression of osteoactivin in rat and human liver and isolated rat liver cells. J. Hepatol. 42, 565–572 (2005).
Ripoll, V. M., Irvine, K. M., Ravasi, T., Sweet, M. J. & Hume, D. A. Gpnmb is induced in macrophages by IFN-gamma and lipopolysaccharide and acts as a feedback regulator of proinflammatory responses. J. Immunol. 178, 6557–6566 (2007).
Abdelmagid, S. M. et al. Osteoactivin, an anabolic factor that regulates osteoblast differentiation and function. Exp. Cell Res. 314, 2334–2351 (2008).
Frara, N. et al. Transgenic expression of Osteoactivin/gpnmb enhances bone formation in vivo and osteoprogenitor differentiation ex vivo. J. Cell. Physiol. 231, 72–83 (2016).
Li, B. et al. The melanoma-associated transmembrane glycoprotein Gpnmb controls trafficking of cellular debris for degradation and is essential for tissue repair. FASEB J. 24, 4767–4781 (2010).
Nakano, Y. et al. Glycoprotein nonmetastatic melanoma protein B (GPNMB) as a novel neuroprotective factor in cerebral ischemia-reperfusion injury. Neuroscience 277, 123–131 (2014).
Neal, M. L., Boyle, A. M., Budge, K. M., Safadi, F. F. & Richardson, J. R. The glycoprotein GPNMB attenuates astrocyte inflammatory responses through the CD44 receptor. J. Neuroinflamm. 15, 73 (2018).
Abe, H. et al. Transgenic expression of osteoactivin in the liver attenuates hepatic fibrosis in rats. Biochem. Biophys. Res. Commun. 356, 610–615 (2007).
Ogawa, T. et al. Osteoactivin upregulates expression of MMP-3 and MMP-9 in fibroblasts infiltrated into denervated skeletal muscle in mice. Am. J. Physiol. Cell Physiol. 289, C697–C707 (2005).
Chung, J. S., Sato, K., Dougherty, I. I., Cruz, P. D. Jr. & Ariizumi, K. DC-HIL is a negative regulator of T lymphocyte activation. Blood 109, 4320–4327 (2007).
Kobayashi, M. et al. Blocking monocytic myeloid-derived suppressor cell function via Anti-DC-HIL/GPNMB antibody restores the in vitro integrity of t cells from cancer patients. Clin. Cancer Res. 25, 828–838 (2019).
Maric, G. et al. GPNMB augments Wnt-1 mediated breast tumor initiation and growth by enhancing PI3K/AKT/mTOR pathway signaling and beta-catenin activity. Oncogene 38, 5294–5307 (2019).
Maric, G., Rose, A. A., Annis, M. G. & Siegel, P. M. Glycoprotein non-metastatic b (GPNMB): a metastatic mediator and emerging therapeutic target in cancer. OncoTargets Ther. 6, 839–852 (2013).
Ott, P. A. et al. A phase 2 study of glembatumumab vedotin, an antibody-drug conjugate targeting glycoprotein NMB, in patients with advanced melanoma. Cancer 125, 1113–1123 (2019).
Raggi, C. et al. Cholangiocarcinoma stem-like subset shapes tumor-initiating niche by educating associated macrophages. J. Hepatol. 66, 102–115 (2017).
Rose, A. A. et al. Glycoprotein nonmetastatic B is an independent prognostic indicator of recurrence and a novel therapeutic target in breast cancer. Clin. Cancer Res. 16, 2147–2156 (2010).
Fiorentini, C. et al. GPNMB/OA protein increases the invasiveness of human metastatic prostate cancer cell lines DU145 and PC3 through MMP-2 and MMP-9 activity. Exp. Cell Res. 323, 100–111 (2014).
Truong, D. D. et al. A human organotypic microfluidic tumor model permits investigation of the interplay between patient-derived fibroblasts and breast cancer cells. Cancer Res. 79, 3139–3151 (2019).
Germano, G. et al. Role of macrophage targeting in the antitumor activity of trabectedin. Cancer Cell. 23, 249–262 (2013).
Kauffmann, A., Gentleman, R. & Huber, W. arrayQualityMetrics—a bioconductor package for quality assessment of microarray data. Bioinformatics 25, 415–416 (2009).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics 16, 284–287 (2012).
Kanehisa, M. & Goto, S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Anderson, M. G. et al. Mutations in genes encoding melanosomal proteins cause pigmentary glaucoma in DBA/2J mice. Nat. Genet. 30, 81–85 (2002).
Zadra, G., Batista, J. L. & Loda, M. Dissecting the dual role of AMPK in cancer: from experimental to human studies. Mol. Cancer Res. 13, 1059–1072 (2015).
Patel, A., Sabbineni, H., Clarke, A. & Somanath, P. R. Novel roles of Src in cancer cell epithelial-to-mesenchymal transition, vascular permeability, microinvasion and metastasis. Life Sci. 157, 52–61 (2016).
Hu, H. et al. Osteoactivin inhibits dexamethasone-induced osteoporosis through up-regulating integrin β1 and activate ERK pathway. Biomed. Pharmacother. 105, 66–72 (2018).
Moussa, F. M. et al. Osteoactivin promotes osteoblast adhesion through HSPG and αvβ1 integrin. J. Cell. Biochem. 115, 1243–1253 (2014).
Sondag, G. R. et al. Osteoactivin inhibition of osteoclastogenesis is mediated through CD44-ERK signaling. Exp. Mol. Med. 48, e257 (2016).
Yu, B., Sondag, G. R., Malcuit, C., Kim, M. H. & Safadi, F. F. Macrophage-associated Osteoactivin/GPNMB mediates mesenchymal stem cell survival, proliferation, and migration via a CD44-dependent mechanism. J. Cell Biochem. 117, 1511–1521 (2016).
Maric, G. et al. GPNMB cooperates with neuropilin-1 to promote mammary tumor growth and engages integrin alpha5beta1 for efficient breast cancer metastasis. Oncogene 34, 5494–5504 (2015).
Furochi, H. et al. Osteoactivin fragments produced by ectodomain shedding induce MMP-3 expression via ERK pathway in mouse NIH-3T3 fibroblasts. FEBS Lett. 581, 5743–5750 (2007).
Bhattacharyya, S., Feferman, L. & Tobacman, J. K. Inhibition of phosphatase activity follows decline in sulfatase activity and leads to transcriptional effects through sustained phosphorylation of transcription factor MITF. PLoS ONE 11, e0153463 (2016).
Mezzapelle, R. et al. Human malignant mesothelioma is recapitulated in immunocompetent BALB/c mice injected with murine AB cells. Sci. Rep. 6, 22850 (2016).
Ge, Y. & Fuchs, E. Stretching the limits: from homeostasis to stem cell plasticity in wound healing and cancer. Nat. Rev. Genet. 19, 311–325 (2018).
Shibue, T. & Weinberg, R. A. EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat. Rev. Clin. Oncol. 14, 611–629 (2017).
Bolli, E., Movahedi, K., Laoui, D. & Van Ginderachter, J. A. Novel insights in the regulation and function of macrophages in the tumor microenvironment. Curr. Opin. Oncol. 29, 55–61 (2017).
Sica, A., Porta, C., Amadori, A. & Pasto, A. Tumor-associated myeloid cells as guiding forces of cancer cell stemness. Cancer Immunol. Immunother. 66, 1025–1036 (2017).
Jinushi, M. et al. Tumor-associated macrophages regulate tumorigenicity and anticancer drug responses of cancer stem/initiating cells. Proc. Natl Acad. Sci. USA 108, 12425–12430 (2011).
Lu, H. et al. A breast cancer stem cell niche supported by juxtacrine signalling from monocytes and macrophages. Nat. Cell Biol. 16, 1105–1117 (2014).
Hardie, D. G. AMPK—Sensing energy while talking to other signaling pathways. Cell Metab. 20, 939–952 (2014).
Qiao, M., Sheng, S. & Pardee, A. B. Metastasis and AKT activation. Cell Cycle 7, 2991–2996 (2014).
Choi, K. & Kim Park, L. Targeting heat shock protein 27 in cancer: a druggable target for cancer treatment? Cancers 11, 1195 (2019).
Wang, L., Zuo, X., Xie, K. & Wei, D. The role of CD44 and cancer. Stem Cells 1692, 31–42 (2018).
Abdelmagid, S. M. et al. Mutation in osteoactivin decreases bone formation in vivo and osteoblast differentiation in vitro. Am. J. Pathol. 184, 697–713 (2014).
Rose, A. A. et al. Osteoactivin promotes breast cancer metastasis to bone. Mol. Cancer Res. 5, 1001–1014 (2007).
Chen, C. et al. Glycoprotein nmb Is exposed on the surface of dormant breast cancer cells and induces stem cell-like properties. Cancer Res. 78, 6424–6435 (2018).
Cayrol, C. & Girard, J. P. IL-33: an alarmin cytokine with crucial roles in innate immunity, inflammation and allergy. Curr. Opin. Immunol. 31, 31–37 (2014).
Liew, F. Y., Girard, J. P. & Turnquist, H. R. Interleukin-33 in health and disease. Nat. Rev. Immunol. 16, 676–689 (2016).
Mantovani, A., Dinarello, C. A., Molgora, M. & Garlanda, C. Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity 50, 778–795 (2019).
Choi, Y. S. et al. Interleukin-33 induces angiogenesis and vascular permeability through ST2/TRAF6-mediated endothelial nitric oxide production. Blood 114, 3117–3126 (2009).
Kenswil, K. J. G. et al. Characterization of endothelial cells associated with hematopoietic niche formation in humans identifies IL-33 As an anabolic factor. Cell Rep. 22, 666–678 (2018).
Afferni, C. et al. The pleiotropic immunomodulatory functions of IL-33 and its implications in tumor immunity. Front. Immunol. 9, 2601 (2018).
Shen, J. X., Liu, J. & Zhang, G. J. Interleukin-33 in malignancies: friends or foes? Front. Immunol. 9, 3051 (2018).
Fang, M. et al. IL33 promotes colon cancer cell stemness via JNK activation and macrophage recruitment. Cancer Res. 77, 2735–2745 (2017).
Hu, H. et al. IL-33 facilitates endocrine resistance of breast cancer by inducing cancer stem cell properties. Biochem. Biophys. Res. Commun. 485, 643–650 (2017).
Xie, C. et al. Tobacco smoke induced hepatic cancer stem cell-like properties through IL-33/p38 pathway. J. Exp. Clin. Cancer Res. 38, 39 (2019).
Mager, L. F. et al. IL-33 signaling contributes to the pathogenesis of myeloproliferative neoplasms. J. Clin. Investig. 125, 2579–2591 (2015).
Wang, Y., Richter, L., Becker, M., Amador, C. & Hyde, R. K. IL1RL1 is dynamically expressed on Cbfb-MYH11(+) leukemia stem cells and promotes cell survival. Sci. Rep. 9, 1729 (2019).
Acknowledgements
This work was supported by IG grants from the Italian Association for Cancer Research (AIRC) to P.A. and grants from the Italian Ministry of Health (GR-2013-02356521) to E.M.B. In addition, E.D. is a recipient of a fellowship from AIRC. We thank Fabio Pasqualini for his help in immunohistochemistry experiments and Marina Sironi and Roberta Migliore for their contributions to the in vivo experiments.
Author information
Authors and Affiliations
Contributions
Supervising, researching and designing the study and writing the manuscript: C.B., P.A.; performing in vitro experiments and acquiring data: C.B., M.L., E.D., A.V., F.S.C., V.R., N.P., E.E., M.T., F.M.F.; performing phosphoproteomic assays and protein profiling: E.M.B., S.M., A.C.; performing in vivo experiment and acquiring data: C.B., R.A., E.D., M.L.; performing immunohistochemistry experiments: E.D., M.L.; performing gene expression analysis: L.M., I.C., S.M.; analyzing and interpreting data (e.g., statistical analysis, figures): E.D., C.B.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Liguori, M., Digifico, E., Vacchini, A. et al. The soluble glycoprotein NMB (GPNMB) produced by macrophages induces cancer stemness and metastasis via CD44 and IL-33. Cell Mol Immunol 18, 711–722 (2021). https://doi.org/10.1038/s41423-020-0501-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41423-020-0501-0
Keywords
This article is cited by
-
Tumor associated microglia/macrophages utilize GPNMB to promote tumor growth and alter immune cell infiltration in glioma
Acta Neuropathologica Communications (2024)
-
Emerging roles of MITF as a crucial regulator of immunity
Experimental & Molecular Medicine (2024)
-
Progranulin and GPNMB: interactions in endo-lysosome function and inflammation in neurodegenerative disease
Journal of Neuroinflammation (2023)
-
Supervised discovery of interpretable gene programs from single-cell data
Nature Biotechnology (2023)
-
A variational algorithm to detect the clonal copy number substructure of tumors from scRNA-seq data
Nature Communications (2023)