Abstract
p53 is a tumor suppressor, which belongs to the p53 family of proteins. The family consists of p53, p63 and p73 proteins, which share similar structure and function. Activation of wild-type p53 or TAp73 in tumors leads to tumor regression, and small molecules restoring the p53 pathway are in clinical development. Protoporphyrin IX (PpIX), a metabolite of aminolevulinic acid, is a clinically approved drug applied in photodynamic diagnosis and therapy. PpIX induces p53-dependent and TAp73-dependent apoptosis and inhibits TAp73/MDM2 and TAp73/MDM4 interactions. Here we demonstrate that PpIX is a dual inhibitor of p53/MDM2 and p53/MDM4 interactions and activates apoptosis in B-cell chronic lymphocytic leukemia cells without illumination and without affecting normal cells. PpIX stabilizes p53 and TAp73 proteins, induces p53-downstream apoptotic targets and provokes cancer cell death at doses non-toxic to normal cells. Our findings open up new opportunities for repurposing PpIX for treating lymphoblastic leukemia with wild-type TP53.
Similar content being viewed by others
Introduction
B-cell chronic lymphocytic leukemia (CLL) is one of the most common forms of blood cancers1,2. The incidence of CLL in the western world is 4.2/100 000 per year. In patients over the age of 80 years, the incidence increases to greater than 30/100 000 per year. CLL develops mostly in patients above the age of 72 years old which is linked to poor prognosis3.
In Sweden, and worldwide, males show a higher prevalence of lymphoid and hematological tissue cancers than women and leukemia incidence in both genders increases above the age of 554.
Common chromosomal aberrations in CLL include deletions of 13q14, trisomy 12, 11q23, and 17p13 deletions or mutations5. Deletions of 17p13 and 11q23 affect p53 pathway and are linked with poor prognosis and acclerated disease progression6. Recent studies point to the positive outcome of the Bcl-2 inhibitor venetoclax in handling 17p-deleted relapsed/refractory CLL7. The clinical trial for the treatment of näive CLL in elderly patients shows a promise for Ibrutinib, a Bruton’s tyrosine kinase (BTK) inhibitor8. However, the development of resistance to targeted therapies poses significant therapeutic constraints.
Introduction of new compounds into clinical practice is both, time constraining and a financial endeavor, which more often than not is subject to failure. Drug repurposing brings a selective advantage to the field of drug discovery as it is easier and more cost-effective to authorize the approved drug for a new indication9.
Protoporphyrin IX (PpIX) is a natural precursor of heme and a metabolite of aminolevulinic acid, a drug clinically used in photodynamic therapy and diagnosis10. In drug repurposing approach PpIX was identified as an activator of p53 and TAp73α tumor suppressors. Recent work demonstrates that PpIX inhibits TAp73/MDM2 and TAp73/MDM4 interactions, which leads to stabilization of TAp73 protein and induction of TAp73-dependent apoptosis in cancers with TP53 gene mutations11,12.
The tumor suppressor p53 is inactivated in the majority of tumors by mutations occurring in the TP53 gene (http://p53.iarc.fr/). In cancers retaining intact TP53 gene, p53 protein is targeted for degradation by the deregulated E3 ubiquitin ligase MDM2. In addition, MDM2 homolog, MDM4 protein binds p53 and inhibits its transcription activity13,14,15.
Activation of wild-type (wt) p53 is a promising therapeutic strategy, and the compounds inhibiting oncogenic MDM2 or modulating p53 post-translational modifications are currently in the clinical development16. However, due to systemic toxicity, highly selective inhibitors of p53/MDM2 interactions including analogs of nutlin, MI, or RG compounds, have not been approved yet17,18. Even though the advancement in the field, these compounds cannot inhibit MDM4 protein and are thus inefficient in targeting tumors that overexpress MDM4 oncogene such as cutaneous melanomas19.
p73 is a tumor suppressor and induces apoptosis and tumor regression in a p53-independent manner20,21,22. TP73 gene is rarely mutated in cancers and p73 protein is often inactivated by binding to oncogenic partners including MDM2, MDM4, ΔNp73, or mutant p5323. Strategies aiming at targeted activation of p73 in cancer are, however, at a very early stage of development.
Here, we applied a fluorescent two-hybrid assay and a yeast-based reporter assay and showed that PpIX inhibits p53/MDM2 and p53/MDM4 interactions. Next, analysis in cancer cells revealed that PpIX induces p53-dependent apoptosis in CLL cells. We demonstrate that PpIX triggers accumulation of p53 and TAp73 and activates cell death at doses not affecting healthy peripheral blood mononuclear cells (PBMCs).
Materials and methods
Reagents and cell lines
PpIX and nutlin were purchased from Sigma-Aldrich (Munich, Germany) and re-constituted in 100% DMSO (Sigma-Aldrich, Munich, Germany) to 2 mg/ml or 10 mM, respectively. PpIX was stored in amber eppendorf tubes at room temperature and nutlin was aliquoted and stored at −20 °C. RITA was purchased from Calbiochem (Solna, Sweden) reconstituted in 100% DMSO to 0.1 M, aliquoted and stored at −20 °C.
Cisplatin (CDDP) (Sigma-Aldrich, Munich, Germany) was prepared in 0.9% NaCl solution to 1 mM, protected from light and stored at −20 °C. MG132 was from Sigma-Aldrich (Munich, Germany) reconstituted in 100% DMSO to 10 mM and stored at −20 °C. IgG and protein A agarose beads were from Santa Cruz Biotechnology (Solna, Sweden), protease inhibitors were prepared from tablets cOmplete® Roche to 100× concentration (Sigma-Aldrich, Munich, Germany), MTT was from Sigma-Aldrich (Munich, Germany).
Rabbit polyclonal anti-MDMX was from Imgenex (Cambridge, UK), rabbit polyclonal anti-TAp73 (A300-126A) (Bethyl Laboratories, TX, USA), anti-PUMA (ABC158; Merck, MA, USA), anti-BAX (N-20; Santa Cruz Biotechnology, Germany), anti-BID (FL-195; Santa Cruz Biotechnology, TX, USA), anti-PARP (F-2; Santa Cruz Biotechnology), anti-β-ACTIN (A2228; Sigma-Aldrich), normal mouse IgG (sc-2025) were from Santa Cruz Biotechnology. Anti-mouse HRP and anti-rabbit HRP secondary antibodies were from (Jackson ImmunoResearch Inc., Ely, UK) Reverse transcription iScript cDNA synthesis kit and SSo Advanced Universal SYBR Green kit were from Bio-Rad (Solna, Sweden)24.
Cell lines
EHEB (wt-p53) chronic B cell leukemia cells were kindly provided by Dr. Anders Österborg, Karolinska Institutet (source ATCC). HL-60 (p53-null) acute promyelocytic leukemia cell lines were provided by Dr. Sören Lehmann, Karolinska Institutet (source ATCC). PBMCs were provided by Dr. Noemi Nagy, Karolinska Institutet and separated as described previously25. HCT 116 cells were a kind gift from Dr. Bert Vogelstein, The Johns Hopkins University School of Medicine26.
Leukemic cells and PBMCs were cultured in RPMI-1640 (Roswell Park Memorial Institute) medium (Sigma-Aldrich, Munich, Germany) and HCT 116 cells in DMEM medium with 10% fetal calf serum (Sigma-Aldrich) and penicillin/streptomycin (10 units/ml) (Sigma-Aldrich) at 37 °C in a humidified 5% CO2/95% air atmosphere.
Cell viability assay
The viability of EHEB, HL60 and PBMCs after 72-hour treatment with PpIX was assessed by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay according to manufacturer’s protocol. Briefly, 5 mg/ml MTT solution was prepared in PBS buffer and filter-sterilized. Cells were washed once with RPMI-1640 medium and 1 × 105 cells/ml were transferred to eppendorf tubes and treated with 0.5% DMSO or the investigated compounds. Next, cells were seeded onto 96-well plates at the density of 1 × 104 cells/well and incubated for 72 h at 37 °C. After this time, MTT reagent was added to each well to a final concentration of 10% and the plates were incubated for 3 h at 37 °C in a humidified 5% CO2/95% air atmosphere. The supernatant was removed and 200 μl DMSO/well was added. The plates were incubated at 37 °C for 30 min and the absorbance of the formazan was measured at 560 nm in a Perkin-Elmer (Waltham, MA, USA) microplate reader.
Untreated EHEB cells, RPMI 1640 medium and RPMI 1640 medium with 20 μg/ml PpIX were used as background controls. The experiments were performed in triplicates and in at least three independent repeats.
Immunoprecipitation and western blot
Immunoprecipitation was performed using a modified protocol described previously27. Briefly, HCT 116 cells were seeded in 10 cm dish at 4 × 106 cells, allowed to adhere overnight and treated with the compounds for 8 h followed by 3 h with 20 μM MG132. Cells were washed 2× with ice-cold PBS and solubilized in IP buffer: 25 mM Tris-HCl, pH 8.0, 150 mM NaCl, and 0.5% Nonidet P-40 supplemented with protease inhibitors and lyzed on ice for 30 min and 1 mg of total protein in IP buffer was added to 30 μl mouse protein A agarose beads and 1 μg mouse anti-p53 DO-1 antibody or 1 μg normal mouse IgG and immunoprecipitated for 16 h at 4 °C. The beads were washed three times with buffer 1 (50 mM Tris-HCl, pH 7.5, 5 mM EDTA, 500 mM NaCl, and 0.5% Nonidet P-40) and one time with buffer 2 (50 mM Tris-HCl pH 7.5, 5 mM EDTA, 150 mM NaCl). The beads were resuspended in 15 μl of lysis buffer and 5 μl of 5× Laemmli buffer and boiled prior to western blot analysis.
For western blot, total proteins were transferred to HyBond membrane (GE healthcare), blocked with 5% milk in PBS for 20 min and incubated with the relevant antibodies. After washing in PBS membranes were incubated with secondary antibodies (1:3000 in 5% milk) for 2 h at room temperature. The protein signals were detected using Super Signal West Dura Extended Duration Substrate (Bio-Rad, Solna, Sweden) and ChemiDoc (Bio-Rad).
PBMCs were cultured in RPMI 1640 supplemented with 10% fetal calf serum and penicillin/streptomycin (10 units/ml) without supplementation with growth factors at 37 °C for three days before treatment with compounds.
Quantitative PCR
Quantitative PCR was performed as described previously24,28. Briefly, cells were treated with 2.5 μg/ml PpIX for 8 h. qPCR was performed using following primers pairs:
Gene | Forward primer 5′-3′ | Reverse primer 5′-3′ |
|---|---|---|
GAPDH | TCATTTCCTGGTATGACAACG | ATGTGGGCCATGAGGT |
PUMA | CTCAACGCACAGTACGAG | GTCCCATGAGATTGTACAG |
HMOX-1 | TTCACCTTCCCCAACATTGC | TATCACCCTCTGCCTGACTG |
BAX | GCTGTTGGGCTGGATCCAAG | TCAGCCCATCTTCTTCCAGA |
Fluorescence activated cell sorting (FACS)
Cells were cultured in 6-well plates with 8 × 105 (K562 and HL60) and 1 × 106 PBMC cells and 2 ml media/well and treated with the compounds. Propidium iodide (PI) and FITC-Annexin V (both from BD Biosciences, CA, USA) staining was performed according to the manufacturer’s protocols. For PBMCs, cells were washed and fixed with 500 μl ice-cold 70% ethanol and stored at 4 °C. Next, cells were washed and stained in 300 μl PI solution for 30 min. Cells were then centrifuged at 300g for 5 min and supernatant was removed and re-suspended in 200 μl PBS. FACS was carried out using the CELLQuest software (CELLQuest, NJ, USA) as described previously29.
Yeast-based reporter assay
The yeast-based functional assay was conducted as previously described30. Briefly, the p53-dependent yeast reporter strain yLFM-PUMA containing the luciferase cDNA cloned at the ADE2 locus and expressed under the control of PUMA promoter31 was transfected with pTSG-p5332, pRB-MDM2 (generously provided by Dr. R. Brachmann, University of California, Irvine, CA, USA), or pTSG-p53 S33/37 mutant and selected on double drop-out media for TRP1 and HIS3. Luciferase activity was measured 16 h after the shift to galactose-containing media31 upon the addition of 2 and 10 μg/ml PpIX or 10 or 50 μM nutlin (Alexis Biochemicals, Sant Diego, CA, USA), or DMSO. Presented are average relative light units and the standard errors obtained from three independent experiments each containing five biological repeats.
F2H®-analysis
The assay was developed and performed as described previously33,34. Briefly, F2H®-analysis (ChromoTek GmbH, Planneg-Martinsried, Germany) was carried out to assess PpIX ability to disrupt p53/MDM2 and p53/MDM4 interactions in U2OS cells, when MDM2 or MDM4 was tethered in the nucleus. U2OS cells were co-transfected with LacI-GFP-MDM2 or MDM4 and RFP-p53 for 8 h and then incubated with 1 or 10 μM of PpIX or nutlin for 16 h. Control interaction values in each independent experiment are normalized to 100%. Averaged interaction values for the treated cells were plotted for p53/MDM2 and p53/MDM4 interactions on the graph. Data is presented as mean ± s.e.m., n = 6, PpIX—p < 0.01, nutlin—p < 0.001, Student’s t-test.
Results
PpIX ablates p53/MDM2 and p53/MDM4 interaction
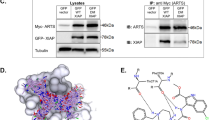
It has been previously shown that PpIX inhibits p53/MDM2 interactions, induces p53-dependent reporter and apoptosis in human cancer cells expressing wild-type p5335,36. Furthermore, PpIX was described to inhibit TAp73/MDM2 and TAp73/MDM4 interactions and to activate the TAp73-dependent apoptosis in cancer cells harboring mutant TP53 gene12,28. The mechanism of inhibition of protein-protein interactions (PPIs) is via binding of PpIX to the N-terminus of TAp7336. Thus, here, we strived to investigate if PpIX, which unlike nutlin, binds to the N-terminus of p5336 and not to MDM2, is also capable of inhibiting the interaction between p53 and MDM4. We engaged the Fluorescent Two-Hybrid (F2H®) analysis performed by ChromoTek GmbH in which the GFP-labeled MDM2 or MDM4 proteins (LacI-GFP-MDM2 or MDM4) were tethered at the nucleus of U2OS cells and examined for the localization of the exogenously expressed RFP-labeled p53 protein before and after drug treatment for 16 h. The data were analysed using fluorescent imaging as described previously34. We observed an inhibitory effect of 10 μM PpIX on both interactions—p53/MDM2 and p53/MDM4. Interaction values dropped from 100% in untreated cells down to 61 ± 8% for p53/MDM2 (mean ± s.e.m, Student’s t-test, p < 0.01, n = 6) and 79 ± 5% for p53/MDM4 (p < 0.05, n = 6). For comparison, the reference compound, 10 μM nutlin-3, induced a similar reduction of the p53/MDM2 interaction (down to 60%), along with reductions of the p53/MDM2 interactions at a lower dose of 1 µM (p < 0.001, Student’s t-test, n = 6). Nutlin-3 did not disrupt the p53/MDM4 interaction (Fig. 1a).
a F2H®-analysis of disruption of interactions of p53/MDM2 and p53/MDM4 by PpIX and nutlin in U2OS cells, when MDM2 or MDM4 is tethered in the nucleus. U2OS cells were co-transfected with LacI-GFP-MDM2 or MDM4 and RFP-p53 for 8 h and then treated with PpIX or nutlin for 16 h. Control interaction values are normalized to 100% and are shown as a red line. Averaged interaction values for the treated cells are plotted for p53/MDM2 interaction (left, green bars) and p53/MDM4 interaction (right, blue bars). Data represented as mean ± s.e.m., n = 5–6, *p < 0.05, **p < 0.01, ***p < 0.001, Student’s t-test. b Rescue of the p53 transcription activity in the presence of MDM4 by PpIX but not nutlin as assessed in the yeast-based reporter assay. The average light units relative to the transactivation activity of p53 alone and the standard errors of at least five biological repeats are presented. The Student’s t-test was performed for statistical analysis with p ≤ 0.05. c PpIX (1 μg/ml) disrupts p53/MDM4 interactions in HCT 116 colon cancer cells as assessed by immunoprecipitation. The data is a representative of two independent experiments. Cropped line represents the site were the membranes were cut. X-sample not relevant to this study
Next, to determine that the inhibition of p53/MDM2 and p53/MDM4 complexes by PpIX is due to the direct interaction between the compound and p53 and not due to post-translational modifications in the p53 protein, we employed a yeast-based reporter assay which allows measuring the transcription activity of human p53 using p53-dependent luciferase reporter11,30. Since p53 is not degraded by the exogenous human MDM2 or MDM4 in yeast cells, the inhibitory effect of MDM2 in this system can be ascribed to the direct interaction with p53 and the interference with the p53-dependent gene transcription37. Co-transfection of MDM2 inhibited the activity of p53 as expressed by the drop from 100 relative light units for p53 itself to 34.02 relative light units in the presence of MDM2 (Fig. 1b). 2 and 10 μg/ml PpIX rescued the wt-p53-mediated activation of luciferase expression to 53.42 and 67.39 relative light units, respectively. The effect on the p53/MDM4 interactions was also significant; PpIX increased the relative light units from 63.48 to 87.79 and 91.42 at 2 and 10 μg/ml, respectively. The reference compound, nutlin-3 did not rescue the transcription activity of p53 in the presence of MDM4 indicating that it does not inhibit p53/MDM4 interactions.
To determine that PpIX inhibits p53/MDM4 interactions also in cancer cells, we treated HCT 116 human colon cancer cells with PpIX for 16 h. To stabilize MDM4 that is otherwise driven to degradation by MDM2 released from the complex with p53 by PpIX, cells were treated with MG132. Immunoprecipitation with monoclonal anti-p53 DO-1 antibody was performed, and the membrane was blotted with the polyclonal anti-p53 FL-393. The results demonstrate that PpIX readily inhibited the interaction between p53 and MDM4 (Fig. 1c). We detected unspecific binding of MDM4 to mouse IgG, however, the signal was much weaker than the one obtained for the untreated cells (Fig. 1c). Thus the background binding does not affect the conclusion that PpIX readily inhibits p53/MDM4 interactions in cancer cells.
From the data presented, F2H® yeast-based reporter assay and pull down demonstrated that the binding of PpIX to p53 results in the inhibition of both p53/MDM2 and p53/MDM4 interactions.
PpIX inhibits proliferation and induces apoptosis in B-CLL leukemia cells
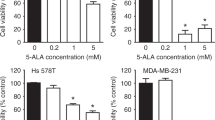
PpIX was shown to induce apoptosis38, p53-dependent, p73-dependent cell death in several cancer cell lines and to shrink tumors in vivo11,35. To estimate the cytoxicity of PpIX in CLL cells, we conducted a 72 h cell viability assay using the wt-p53 EHEB B-CLL cell line and the p53-null HL60 acute promyelocytic leukemia cells. 1 μM RITA (Reactivation of p53 and Induction of tumor cell apoptosis) was used for comparison between two compounds known to activate the p53 pathway. Cisplatin (CDDP) was used as a reference compound. The viability of EHEB and HL-60 cells was significantly reduced with the increasing concentrations of PpIX (Fig. 2a, b). Of note, PpIX did not inhibit the proliferation of PBMCs at the concentrations effective in cancer cells (Fig. 2c). Based on the above and the IC50 values, which are 2.5 μg/ml for EHEB and 2.4 μg/ml for HL60, we next investigated the mechanism of growth inhibition of cancer cells by PpIX and applied 2.5 μg/ml dose in all further experiments.
a Viability test (MTT) shows inhibition of proliferation of EHEB cells by PpIX (red bars), RITA (1 μM) (dark yellow bar) and CDDP (50 μM) (yellow bar) after 72 h treatment. DMSO was used at 0.5%. Error bars represent SD values. n = 3, C—untreated control. b Viability assay (MTT) in HL60 cells treated with increasing PpIX doses (red bars) or RITA (1 μM) (dark yellow bars) for 72 h. DMSO was used at 0.5%. Error bars represent SD values. n = 3, C—untreated control. c Viability of PBMCs treated with PpIX for 72 h (red bars). DMSO was used at 0.5%. The error bars represent SD values. n = 3, C—untreated control. d PpIX (2.5 μg/ml) activates late (upper panel) and early apoptosis (lower panel) in EHEB cells after 24 h and 48 h treatment (e). C—control treated with DMSO
To assess the induction of cancer cell death, cells were treated with PpIX and stained with propidium iodide (PI) or Annexin V. Compared with untreated control cells, PpIX-treated EHEB cells showed a significant increase in the PI positive population (late apoptosis), from 20.73 to 94.45% and in the Annexin V positive population (early apoptosis), from 21.74 to 39.59% (Fig. 2d) after 24 h. After 48 h treatment with PpIX, the increase in the PI positive population was from 22.98 to 94.94% and in the Annexin V positive population from 32.28 to 52.13% (Fig. 2e). We did not detect any increase of PBMCs in subG1 phase after treatment with 5 or 10 μg/ml PpIX, corroborating the lack of toxicity induction by PpIX in normal cells (Supplementary Fig. 1a, b). The results in PBMCs correlated with the lack of induction of growth inhibition when treated with PpIX as assessed by MTT assay suggesting a redundant role of PpIX in healthy PBMCs (Fig. 2c).
PpIX induces p53- and TAp73-dependent apoptosis in wt-p53 CLL cells but not in PBMCs
To determine the mechanism of induction of apoptosis by PpIX in CLL cells, EHEB cells were treated with 2.5 μg/ml PpIX for 8 h and analysed for the expression of p53 and TAp73 apoptotic target genes BAX and PUMA by quantitative PCR (qPCR). Induction of BAX and PUMA on mRNA levels was detected (Fig. 3a) and this corresponded to the accumulation of BAX and PUMA protein levels as assessed by western blots (Fig. 3b, c). As reported previously, consistently with the induction of BAX and PUMA, PpIX induced p53 and TAp73 levels in EHEB cells in a time-dependent and dose-dependent manner (Fig. 3b, c). Interestingly, p53 accumulated more rapidly comparing to TAp73, suggesting preferential binding of PpIX to p53 in EHEB cells (Fig. 3c). Induction of p53 preceded the accumulation of the apoptotic protein BID and cleaved PARP. In healthy PMBCs, 2.5 μg/ml PpIX induced p53 levels. However, it did not correlate with the induction of apoptotic p53 targets PUMA and BID (Fig. 3d). In cancer cells, in addition to PUMA and BAX, PpIX induced the expression of heme oxygenase HMOX-1 (Fig. 3a), a stress response gene. This is in agreement with the previous studies showing that PpIX induces antioxidant response in cancer cells12. PpIX is an inhibitor of thioredoxin reductase (TrxR), a selenoprotein that plays a critical role in the oxidoreductase system and is often overexpressed in cancers. Inhibition of TrxR by PpIX induces reactive oxygen species (ROS) in cancer cells, triggers antioxidant response and contributes to cancer cell death12,39. Thus, we speculate that PpIX induces p53- and TAp73- dependent apoptosis and ROS in B-CLL cells without affecting non-transformed cells. The putative mechanism of leukemia cell death is via stabilization of p53 and TAp73 resulting from the inhibition of their interactions with oncogenic MDM2 and MDM4, the parallel accumulation of ROS resulting from inhibition of TrxR and induction of potent apoptosis (Fig. 3e).
a PpIX (2.5 μg/ml) induces expression of apoptotic genes BAX, PUMA, and antioxidant response gene HMOX-1 in EHEB leukemia cells. b Western blot analysis of EHEB cells treated with increasing doses of PpIX for 24 h demonstrates accumulation of p53 and TAp73 and p53-downstream apoptotic proteins. C—untreated control. c PpIX (2.5 μg/ml) induces p53 and TAp73 and p53-downstream apoptotic targets in EHEB cells in a time dependent manner. C—untreated control. d Western blot analysis of PBMCs treated with increasing doses of PpIX for 24 h. C—untreated control. e A scheme representing the mechanism of induction of cell death in CLL cells by PpIX. PpIX simultaneously disrupts interactions between p53 or TAp73 and oncogenic MDM2 and MDM4. This results in p53 and TAp73 accumulation, induction of p53-downstream apoptotic proteins and cancer cell death
Discussion
Genetic or pharmacological restoration of wt-p53 activity leads to regression of tumors in vivo40,41,42. Several therapeutic strategies have been developed to activate the p53 protein in tumors depending on the TP53 gene status. Thus, in cancers, harboring missense TP53 gene mutations, small molecules belonging to the group of Michael acceptors that target reactive cysteine groups in the p53 core domain were found to be highly effective. These compounds refold mutant p53 protein to wild-type like conformation and induce p53-dependent apoptosis both in cancer cells in vitro and in vivo43,44,45,46. The most promising compound, a small molecule APR-246, a pro-drug converted to methylene quinuclidinone (MQ) is currently tested in Phase II trial in TP53-mutated high-grade serous ovarian cancer in combination with carboplatin and pegylated liposomal doxorubicin hydrochloride (PLD) (Clinical trial identifier: NCT02098343).
In cancers expressing wt-p53 protein, p53 pathway is often inhibited by the protein–protein interactions. MDM2 protein is a major E3 ubiquitin ligase of p53, which binds to p53 in the absence of cellular stress. The binding is via the N-terminal transactivation domain of p53 and the central domain of MDM2, inhibits p53 transcription activity and drives p53 to ubiquitin-dependent degradation. In addition, MDM2 promotes its self-degradation as well as of its homolog, MDM4 protein. MDM4, unlike MDM2, does not degrade p53 but binds to p53 N-terminus and inhibits its transcription activity. MDM2 and MDM4 are often overexpressed in cancers and therefore serve as promising therapeutic targets19,42,47. A large data of evidence shows that small-molecule antagonists of MDM2 restore the activity of wt-p53 in cells and thus are promising candidates for improved therapy of wt-TP53 cancers.
Several compounds that bind to MDM2 and inhibit p53/MDM2 interactions are in pre-clinical and clinical development48,49,50. However, the high-affinity inhibitors of MDM2 might not be applicable in the clinical practice due to the development of resistant mutations. A recent study showed that prolonged exposure of wt-p53 harboring cancer cells to RG7388 (idasanutlin) leads to the development of resistant mutations in the TP53 gene51. Several clinical studies with a small molecule MDM2 inhibitor, AMG-232, which binds with picomolar affinity to MDM252 are on-going; however, the compound is not approved yet and targets only MDM2 oncoprotein, without displaying an inhibitory effect towards MDM4.
Studies have shown that PpIX, induces apoptosis in cancer cells, binds to p53 and TAp73 and induces p53-downstream apoptotic genes11,35,36. Cancer cells harboring wt-TP53 gene, undergo cell death via a p53-dependent mechanism as PpIX inhibits p53/MDM2 complexes which leads to p53 stabilization and a subsequent induction of apoptosis35. Next, PpIX inhibits TAp73/MDM2 and TAp73/MDM4 interactions, which induces TAp73 accumulation and TAp73-dependent apoptosis in cancers with TP53 gene deletions or mutations12,28. This is in agreement with previous studies showing that colon cancer cells harboring wt-TP53 gene are more sensitive to photodynamic therapy with Photofrin®, a derivative of hematoporphyrin and, a close structural analog of PpIX53. Based on the above and our observations, we speculated that the pre-treatment of cancer cells with porphyrin-like photosensitizers activates the p53 pathway via p53 stabilization which sensitizes cancer cells to photodynamic reaction induced by light matching the absorption spectrum of the compound. Here we show, using fluorescent two-hybrid technology, yeast-based p53 reporter assay and pull-down, that PpIX inhibits p53/MDM2 and p53/MDM4 interactions. Thus, PpIX, which unlike nutlin binds to p53 and not to MDM2, is a novel dual inhibitor of p53/MDM2 and p53/MDM4 interactions. It is particularly interesting since it has been demonstrated that the combined inhibition of MDM2 and MDM4 led to the enhanced p53 response and tumor regression in virus-positive Merkel cell carcinoma54.
Previous studies showed that PpIX induces apoptosis in human lung and pancreatic cancer cells by activating TAp73. However, the potency of the compound against hematological tissue cancers has not been tested. We applied B-CLL cell line EHEB, harboring wt-TP53 gene and showed the induction of apoptosis by PpIX at concentrations that have no effect on healthy PBMCs. The p53-null HL60 cells were also sensitive at the tested doses; however, the induction of apoptosis has not been unequivocally studied. Real-time PCR and western blot analysis revealed that PpIX induces expression of p53-downstream apoptotic PUMA and BAX. This was in agreement with simultaneous induction of p53 and TAp73 levels and accumulation of cleaved PARP. Thus, we conclude that PpIX stabilizes both p53 and TAp73 by targeting their interaction with oncogenic MDM2 and MDM4. In addition, induction of HMOX-1 stress response gene, suggests induction of ROS in EHEB cells. We, alongside others, showed that PpIX is an inhibitor of thioredoxin reductase, a key enzyme of the thioredoxin antioxidant defense system12,39,55. Inhibition of TrxR leads to induction of ROS, thus we speculate that PpIX induces HMOX-1 in EHEB cells by activating stress response pathway due to inhibition of TrxR. Of note, several p53 targeting compounds have already been shown to inhibit TrxR which resulted in potent cancer cells eradication56,57. Thus, simultaneous targeting of cancer cell vulnerabilities by PpIX; namely, inactive tumor suppressors and oncogenic TrxR might bring a selective advantage over compounds already in clinical development. This is due to the fact that targeting several apoptosis-promoting pathways by PpIX might drastically reduce the risk of development of treatment resistance, as evidenced for the approved targeted therapies. Next, it has become apparent from our study that PpIX does not induce apoptosis in healthy PBMCs. Even though p53 was upregulated after 24 h, we did not detect accumulation of apoptotic proteins PUMA and BID. This was in agreement with the lack of growth inhibition of PBMCs by PpIX as assessed by the MTT assay. Taken together, PpIX, unlike approved modalities, might have very little effect on healthy bone marrow cells, making the compound particularly attractive for the management of pediatric and elderly blood cancers. The best clinical outcome in the management of leukemia is achieved in patients undergoing haematopoietic stem cell transplantation (HSCT). However, conditioning with busulfan alone or in combination with other myeloablative drugs is aggressive58, has a low therapeutic window and despite good protective effects of N-acetyl-l-cysteine (NAC)59 the side effects, particularly in older patients, result in low survival rates. In addition, busulfan has been demonstrated to affect fertility in female survivors of childhood cancers60. Infants and very young children undergoing haematopoietic stem cells transplantation (HSCT) are a vulnerable group of patients and are particularly sensitive to HSCT-related morbidities61. Moreover, late cardio-toxicities of anthracycline and anthraquinone frequently used in HSCT is often a significant health burden in childhood cancer survivors62. Thus, new treatment strategies are needed to treat pediatric leukemia patients.
PM2, a stapled peptide that binds to MDM2 and MDM4 and stabilizes p53 has been recently described63 and a small molecule, LEM2, inhibiting both TAp73/MDM2 and TAp73/MDM4 interactions has been discovered in a yeast-based reporter assay64. However, in comparison to PpIX, the capacity of these compounds to activate both p53 and TAp73 has not been unequivocally tested.
The exact mechanism of how PpIX inhibits p53 and p73 interactions with MDM2 and MDM4 remains to be elucidated. We speculate that by binding to p53 or p73 N-terminus, PpIX might alter the conformation of the α-helix spanning the MDM2 binding residues F19, W23, and L26, which might in turn prevent the interaction between p53/MDM2 and p53/MDM4.
Summing up, our study demonstrates that PpIX is a potent activator of p53 and TAp73 in B-CLL. Our findings might speed up repurposing of PpIX in treating cancers containing wt-p53 and TAp73 and with high expression of MDM2 and MDM4 oncogenes.
References
Diez, P., Gongora, R., Orfao, A. & Fuentes, M. Functional proteomic insights in B-cell chronic lymphocytic leukemia. Expert. Rev. Proteom. 14, 137–146 (2017).
Hallek, M. Chronic lymphocytic leukemia: 2017 update on diagnosis, risk stratification, and treatment. Am. J. Hematol. 92, 946–965 (2017).
Eichhorst, B. et al. Chronic lymphocytic leukemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 22(Suppl 6), vi50–vi54 (2011).
Socialstyrelsen, The National Board of Health and Welfare, Statistics on Cancer Incidence 2017. Health and Medical Care, ISSN 1401–0216 (2018).
Hallek, M. Signaling the end of chronic lymphocytic leukemia: new frontline treatment strategies. Hematology 2013, 138–150 (2013).
Alonso, R. et al. Forodesine has high antitumor activity in chronic lymphocytic leukemia and activates p53-independent mitochondrial apoptosis by induction of p73 and BIM. Blood 114, 1563–1575 (2009).
Mato, A. R. et al. Real-world outcomes and management strategies for venetoclax-treated chronic lymphocytic leukemia patients in the United States. Haematologica 103, 1511–1517 (2018).
Moreno, C. et al. Ibrutinib plus obinutuzumab versus chlorambucil plus obinutuzumab in first-line treatment of chronic lymphocytic leukaemia (iLLUMINATE): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 20, 43–56 (2019).
Peters, S., Clezardin, P., Marquez-Rodas, I., Niepel, D. & Gedye, C. The RANK-RANKL axis: an opportunity for drug repurposing in cancer? Clin. Transl. Oncol. https://doi.org/10.1007/s12094-018-02023-5 (2019).
Acedo, P. & Zawacka-Pankau, J. p53 family members—important messengers in cell death signaling in photodynamic therapy of cancer? Photochem. Photobiol. Sci. 14, 1390–1396 (2015).
Sznarkowska, A. et al. Reactivation of TAp73 tumor suppressor by protoporphyrin IX, a metabolite of aminolevulinic acid, induces apoptosis in TP53-deficient cancer cells. Cell Div. 13:10 https://doi.org/10.1186/s13008-018-0043-3 (2018).
Acedo, P., Fernandes, A. & Zawacka-Pankau, J. Activation of TAp73 and inhibition of thioredoxin reductase for improved cancer therapy in TP53 mutant pancreatic tumors. Future Sci. OA https://doi.org/10.4155/fsoa-2018-0082 (2018).
Vousden, K. H. & Prives, C. Blinded by the light: the growing complexity of p53. Cell 137, 413–431 (2009).
Joerger, A. C. & Fersht, A. R. The p53 pathway: origins, inactivation in cancer, and emerging therapeutic approaches. Annu. Rev. Biochem. 85, 375–404 (2016).
Marine, J. C. & Jochemsen, A. G. MDMX (MDM4), a promising target for p53 reactivation therapy and beyond. Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a026237 (2016).
Ladds, M. & Lain, S. Small molecule activators of the p53 response. J. Mol. Cell Biol. https://doi.org/10.1093/jmcb/mjz006 (2019).
Shangary, S. et al. Reactivation of p53 by a specific MDM2 antagonist (MI-43) leads to p21-mediated cell cycle arrest and selective cell death in colon cancer. Mol. Cancer Ther. 7, 1533–1542 (2008).
Zhao, Y. et al. Diastereomeric spirooxindoles as highly potent and efficacious MDM2 inhibitors. J. Am. Chem. Soc. 135, 7223–7234 (2013).
Gembarska, A. et al. MDM4 is a key therapeutic target in cutaneous melanoma. Nat. Med. 18, 1239–1247 (2012).
Venkatanarayan, A. et al. IAPP-driven metabolic reprogramming induces regression of p53-deficient tumours in vivo. Nature 517, 626–630 (2015).
Rada, M. et al. BTK blocks the inhibitory effects of MDM2 on p53 activity. Oncotarget 8, 106639–106647 (2017).
Tomasini, R. et al. TAp73 knockout shows genomic instability with infertility and tumor suppressor functions. Genes Dev. 22, 2677–2691 (2008).
Rufini, A. et al. p73 in Cancer. Genes Cancer 2, 491–502 (2011).
Kostecka, A. et al. JNK-NQO1 axis drives TAp73-mediated tumor suppression upon oxidative and proteasomal stress. Cell Death Dis. 5, e1484 (2014).
Madapura, H. S. et al. cMyc-p53 feedback mechanism regulates the dynamics of T lymphocytes in the immune response. Cell Cycle 15, 1267–1275 (2016).
Sur, S. et al. A panel of isogenic human cancer cells suggests a therapeutic approach for cancers with inactivated p53. Proc. Natl Acad. Sci. USA 106, 3964–3969 (2009).
Stommel, J. M. & Wahl, G. M. Accelerated MDM2 auto-degradation induced by DNA-damage kinases is required for p53 activation. EMBO J. 23, 1547–1556 (2004).
Sznarkowska, A. et al. Reactivation of TAp73 tumor suppressor by protoporphyrin IX, a metabolite of aminolevulinic acid, induces apoptosis in TP53-deficient cancer cells. Cell Div. 13, 10 (2018).
Shi, Y. et al. ROS-dependent activation of JNK converts p53 into an efficient inhibitor of oncogenes leading to robust apoptosis. Cell Death Differ. 21, 612–623 (2014).
Tomso, D. J. et al. Functionally distinct polymorphic sequences in the human genome that are targets for p53 transactivation. Proc. Natl Acad. Sci. USA 102, 6431–6436 (2005).
Inga, A., Storici, F., Darden, T. A. & Resnick, M. A. Differential transactivation by the p53 transcription factor is highly dependent on p53 level and promoter target sequence. Mol. Cell. Biol. 22, 8612–8625 (2002).
Resnick, M. A. & Inga, A. Functional mutants of the sequence-specific transcription factor p53 and implications for master genes of diversity. Proc. Natl Acad. Sci. USA 100, 9934–9939 (2003).
Zolghadr, K., Rothbauer, U. & Leonhardt, H. The fluorescent two-hybrid (F2H) assay for direct analysis of protein–protein interactions in living cells. Methods Mol. Biol. 812, 275–282 (2012).
Yurlova, L. et al. The fluorescent two-hybrid assay to screen for protein-protein interaction inhibitors in live cells: targeting the interaction of p53 with Mdm2 and Mdm4. J. Biomol. Screen. 19, 516–525 (2014).
Zawacka-Pankau, J. et al. Protoporphyrin IX interacts with wild-type p53 protein in vitro and induces cell death of human colon cancer cells in a p53-dependent and -independent manner. J. Biol. Chem. 282, 2466–2472 (2007).
Sznarkowska, A. et al. Targeting of p53 and its homolog p73 by protoporphyrin IX. FEBS Lett. 585, 255–260 (2011).
Wang, T. et al. hADA3 is required for p53 activity. EMBO J. 20, 6404–6413 (2001).
Bednarz, N., Zawacka-Pankau, J. & Kowalska, A. Protoporphyrin IX induces apoptosis in HeLa cells prior to photodynamic treatment. Pharmacol. Rep. 59, 474–479 (2007).
Prast-Nielsen, S. et al. Inhibition of thioredoxin reductase 1 by porphyrins and other small molecules identified by a high-throughput screening assay. Free Radic. Biol. Med. 50, 1114–1123 (2011).
Junttila, M. R. et al. Selective activation of p53-mediated tumour suppression in high-grade tumours. Nature 468, 567–571 (2010).
Bykov, V. J. N., Eriksson, S. E., Bianchi, J. & Wiman, K. G. Targeting mutant p53 for efficient cancer therapy. Nat. Rev. Cancer 18, 89–102 (2018).
Cheok, C. F. & Lane, D. P. Exploiting the p53 pathway for therapy. Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a026310 (2017).
Mohell, N. et al. APR-246 overcomes resistance to cisplatin and doxorubicin in ovarian cancer cells. Cell Death Dis. 6, e1794 (2015).
Madan, E. et al. The curcumin analog HO-3867 selectively kills cancer cells by converting mutant p53 protein to transcriptionally active wildtype p53. J. Biol. Chem. 293, 4262–4276 (2018).
Zhang, Q., Bykov, V. J. N., Wiman, K. G. & Zawacka-Pankau, J. APR-246 reactivates mutant p53 by targeting cysteines 124 and 277. Cell Death Dis. 9, 439 (2018).
Zhang, Q., Bergman, J., Wiman, K. G. & Bykov, V. J. N. Role of thiol reactivity for targeting mutant p53. Cell Chem. Biol. 25, 1219–1230 e1213 (2018).
Zawacka-Pankau, J. & Selivanova, G. Pharmacological reactivation of p53 as a strategy to treat cancer. J. Intern. Med. 277, 248–259 (2015).
Wade, M., Li, Y. C. & Wahl, G. M. MDM2, MDMX and p53 in oncogenesis and cancer therapy. Nat. Rev. Cancer 13, 83–96 (2013).
Skalniak, L. et al. A fluorinated indole-based MDM2 antagonist selectively inhibits the growth ofp53(wt) osteosarcoma cells. FEBS J. https://doi.org/10.1111/febs.14774 (2019).
Sun, D. et al. Discovery of AMG 232, a potent, selective, and orally bioavailable MDM2-p53 inhibitor in clinical development. J. Med. Chem. 57, 1454–1472 (2014).
Skalniak, L. et al. Prolonged idasanutlin (RG7388) treatment leads to the generation of p53-mutated cells. Cancers https://doi.org/10.3390/cancers10110396 (2018).
Canon, J. et al. The MDM2 inhibitor AMG 232 demonstrates robust antitumor efficacy and potentiates the activity of p53-inducing cytotoxic agents. Mol. Cancer Ther. 14, 649–658 (2015).
Fisher, A. M., Rucker, N., Wong, S. & Gomer, C. J. Differential photosensitivity in wild-type and mutant p53 human colon carcinoma cell lines. J. Photochem. Photobiol. B 42, 104–107 (1998).
Park, D. E. et al. Dual inhibition of MDM2 and MDM4 in virus-positive Merkel cell carcinoma enhances the p53 response. Proc. Natl Acad. Sci. USA 116, 1027–1032 (2019).
Zhang, L. et al. Serum thioredoxin reductase is highly increased in mice with hepatocellular carcinoma and its activity is restrained by several mechanisms. Free Radic. Biol. Med. 99, 426–435 (2016).
Peng, X. et al. APR-246/PRIMA-1MET inhibits thioredoxin reductase 1 and converts the enzyme to a dedicated NADPH oxidase. Cell Death Dis. 4, e881 (2013).
Sachweh, M. C. et al. Redox effects and cytotoxic profiles of MJ25 and auranofin towards malignant melanoma cells. Oncotarget 6, 16488–16506 (2015).
Masson, E. & Zamboni, W. C. Pharmacokinetic optimisation of cancer chemotherapy. Clin. Pharmacokinet. 32, 324–343 (1997).
El-Serafi, I. et al. The effect of N-acetyl-l-cysteine (NAC) on liver toxicity and clinical outcome after hematopoietic stem cell transplantation. Sci. Rep. 8, 8293 (2018).
Chow, E. J. et al. Pregnancy after chemotherapy in male and female survivors of childhood cancer treated between 1970 and 1999: a report from the Childhood Cancer Survivor Study cohort. Lancet Oncol. 17, 567–576 (2016).
Vrooman, L. M. et al. Survival and late effects after allogeneic hematopoietic cell transplantation for hematologic malignancy at less than three years of age. Biol. Blood Marrow Transplant. 23, 1327–1334 (2017).
Feijen, E. A. M. et al. Derivation of anthracycline and anthraquinone equivalence ratios to doxorubicin for late-onset cardiotoxicity. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2018.6634 (2019).
Spiegelberg, D. et al. The MDM2/MDMX-p53 antagonist PM2 radiosensitizes wild-type p53 tumors. Cancer Res. 78, 5084–5093 (2018).
Gomes, S. et al. New inhibitor of the TAp73 interaction with MDM2 and mutant p53 with promising antitumor activity against neuroblastoma. Cancer Lett. 446, 90–102 (2019).
Acknowledgements
This work was supported by grants to J.Z.P. from Stockholm Läns Landsting, Karolinska Institutet, Karolinska Institutet/MD Anderson Cancer Center Sister Institution Network Fund (SINF), The Strategic Research Program in Cancer Karolinska Institutet (StratCan) and by Amgen Scholars Program (NM). We thank Sir David Lane and Dr. Galina Selivanova for helpful discussions and suggestions. Special thanks are addressed to Dr. Alberto Inga and Dr. Virginia Andreotti. We would also like to thank Dr. Alicja Sznarkowska, Dr. Elisabeth Hedström and Dr. Suhas Dereker for helpful feedback. The authors are grateful to Yari Ciribilli for technical assistance and to Dr. Kourosh Zolghadr from ChromoTek GmbH for helpful feedback on F2H® studies. We are greatly indebted to all our colleagues who shared with us their reagents and cells.
Authors contributions
L.J., N.M., and P.A., performed experimental work and drafted figures except from Fig. 1a–c and L.J. and N.M. drafted the manuscript, J.Z.P. initiated the project, supervised the study, selected the methods and conditions, obtained the funding, prepared final figures, drafted the manuscript, prepared final version and was responsible for the contact with the Journal. All authors read and approved the final version.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by I. Harris
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiang, L., Malik, N., Acedo, P. et al. Protoporphyrin IX is a dual inhibitor of p53/MDM2 and p53/MDM4 interactions and induces apoptosis in B-cell chronic lymphocytic leukemia cells. Cell Death Discov. 5, 77 (2019). https://doi.org/10.1038/s41420-019-0157-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-019-0157-7
This article is cited by
-
p53 biology and reactivation for improved therapy in MDS and AML
Biomarker Research (2024)
-
A PPIX-binding probe facilitates discovery of PPIX-induced cell death modulation by peroxiredoxin
Communications Biology (2023)
-
Regulated cell death (RCD) in cancer: key pathways and targeted therapies
Signal Transduction and Targeted Therapy (2022)
-
A role for bioinorganic chemistry in the reactivation of mutant p53 in cancer
JBIC Journal of Biological Inorganic Chemistry (2022)
-
The p53/MDM2/MDMX-targeted therapies—a clinical synopsis
Cell Death & Disease (2020)