Abstract
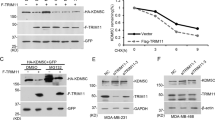
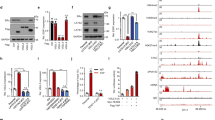
The estrogen receptor alpha (ERα) signaling pathway is a crucial target for ERα-positive breast cancer therapeutic strategies. Co-regulators and other transcription factors cooperate for effective ERα-related enhancer activation. Recent studies demonstrate that the transcription factor CTCF is essential to participate in ERα/E2-induced enhancer transactivation. However, the mechanism of how CTCF is achieved remains unknown. Here, we provided evidence that BAP18 is required for CTCF recruitment on ERα-enriched enhancers, facilitating CTCF-mediated chromatin accessibility to promote enhancer RNAs transcription. Consistently, GRO-seq demonstrates that the enhancer activity is positively correlated with BAP18 enrichment. Furthermore, BAP18 interacts with SMARCA1/BPTF to accelerate the recruitment of CTCF to ERα-related enhancers. Interestingly, BAP18 is involved in chromatin accessibility within enhancer regions, thereby increasing enhancer transactivation and enhancer-promoter looping. BAP18 depletion increases the sensitivity of anti-estrogen and anti-enhancer treatment in MCF7 cells. Collectively, our study indicates that BAP18 coordinates with CTCF to enlarge the transactivation of ERα-related enhancers, providing a better understanding of BAP18/CTCF coupling chromatin remodeling and E-P looping in the regulation of enhancer transcription.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data and material that support the findings of this study are available from the corresponding author upon reasonable request.
References
Husemann Y, Geigl JB, Schubert F, Musiani P, Meyer M, Burghart E, et al. Systemic spread is an early step in breast cancer. Cancer Cell. 2008;13:58–68.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Koren S, Bentires-Alj M. Breast Tumor Heterogeneity: Source of Fitness, Hurdle for Therapy. Mol Cell. 2015;60:537–46.
Cardoso F, Paluch-Shimon S, Senkus E, Curigliano G, Aapro MS, Andre F, et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann Oncol. 2020;31:1623–49.
Harbeck N, Gnant M. Breast cancer. Lancet. 2017;389:1134–50.
Chen S. An “omics” approach to determine the mechanisms of acquired aromatase inhibitor resistance. OMICS. 2011;15:347–52.
Cardoso F, Costa A, Senkus E, Aapro M, Andre F, Barrios CH, et al. 3rd ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 3). Ann Oncol. 2017;28:16–33.
Early Breast Cancer Trialists’ Collaborative G, Davies C, Godwin J, Gray R, Clarke M, Cutter D, et al. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378:771–84.
Toy W, Shen Y, Won H, Green B, Sakr RA, Will M, et al. ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat Genet. 2013;45:1439–45.
Zwart W, Griekspoor A, Berno V, Lakeman K, Jalink K, Mancini M, et al. PKA-induced resistance to tamoxifen is associated with an altered orientation of ERalpha towards co-activator SRC-1. EMBO J. 2007;26:3534–44.
Kastrati I, Semina S, Gordon B, Smart E. Insights into how phosphorylation of estrogen receptor at serine 305 modulates tamoxifen activity in breast cancer. Mol Cell Endocrinol. 2019;483:97–101.
Servetto A, Formisano L, Arteaga CL. FGFR signaling and endocrine resistance in breast cancer: Challenges for the clinical development of FGFR inhibitors. Biochim Biophys Acta Rev Cancer. 2021;1876:188595.
Sklias A, Halaburkova A, Vanzan L, Jimenez NF, Cuenin C, Bouaoun L, et al. Epigenetic remodelling of enhancers in response to estrogen deprivation and re-stimulation. Nucl Acids Res. 2021;49:9738–54.
Escher TE, Dandawate P, Sayed A, Hagan CR, Anant S, Lewis-Wambi J. Enhanced IFNalpha Signaling Promotes Ligand-Independent Activation of ERalpha to Promote Aromatase Inhibitor Resistance in Breast Cancer. Cancers (Basel) 2021;13.
Le Romancer M, Poulard C, Cohen P, Sentis S, Renoir JM, Corbo L. Cracking the estrogen receptor’s posttranslational code in breast tumors. Endocr Rev. 2011;32:597–622.
Jeselsohn R, De Angelis C, Brown M, Schiff R. The Evolving Role of the Estrogen Receptor Mutations in Endocrine Therapy-Resistant Breast Cancer. Curr Oncol Rep. 2017;19:35.
Zhou Y, Gerrard DL, Wang J, Li T, Yang Y, Fritz AJ, et al. Temporal dynamic reorganization of 3D chromatin architecture in hormone-induced breast cancer and endocrine resistance. Nat Commun. 2019;10:1522.
Fridriksdottir AJ, Kim J, Villadsen R, Klitgaard MC, Hopkinson BM, Petersen OW, et al. Propagation of oestrogen receptor-positive and oestrogen-responsive normal human breast cells in culture. Nat Commun. 2015;6:8786.
Holding AN, Cullen AE, Markowetz F. Genome-wide Estrogen Receptor-alpha activation is sustained, not cyclical. Elife 2018;7.
Li W, Notani D, Ma Q, Tanasa B, Nunez E, Chen AY, et al. Functional roles of enhancer RNAs for oestrogen-dependent transcriptional activation. Nature. 2013;498:516–20.
Pnueli L, Rudnizky S, Yosefzon Y, Melamed P. RNA transcribed from a distal enhancer is required for activating the chromatin at the promoter of the gonadotropin alpha-subunit gene. Proc Natl Acad Sci. 2015;112:4369–74.
Liu Z, Merkurjev D, Yang F, Li W, Oh S, Friedman MJ, et al. Enhancer activation requires trans-recruitment of a mega transcription factor complex. Cell. 2014;159:358–73.
Zhu C, Li L, Zhang Z, Bi M, Wang H, Su W, et al. A Non-canonical Role of YAP/TEAD Is Required for Activation of Estrogen-Regulated Enhancers in Breast Cancer. Mol Cell. 2019;75:791–806.e798.
Achinger-Kawecka J, Valdes-Mora F, Luu PL, Giles KA, Caldon CE, Qu W, et al. Epigenetic reprogramming at estrogen-receptor binding sites alters 3D chromatin landscape in endocrine-resistant breast cancer. Nat Commun. 2020;11:320.
Watson G, Ronai ZA, Lau E. ATF2, a paradigm of the multifaceted regulation of transcription factors in biology and disease. Pharm Res. 2017;119:347–57.
Huebner K, Prochazka J, Monteiro AC, Mahadevan V, Schneider-Stock R. The activating transcription factor 2: an influencer of cancer progression. Mutagenesis. 2019;34:375–89.
Pugacheva EM, Kubo N, Loukinov D, Tajmul M, Kang S, Kovalchuk AL, et al. CTCF mediates chromatin looping via N-terminal domain-dependent cohesin retention. Proc Natl Acad Sci . 2020;117:2020–31.
Quintin J, Le Peron C, Palierne G, Bizot M, Cunha S, Serandour AA, et al. Dynamic estrogen receptor interactomes control estrogen-responsive trefoil Factor (TFF) locus cell-specific activities. Mol Cell Biol. 2014;34:2418–36.
Ross-Innes CS, Brown GD, Carroll JS. A co-ordinated interaction between CTCF and ER in breast cancer cells. BMC Genomics. 2011;12:593.
Korkmaz G, Manber Z, Lopes R, Prekovic S, Schuurman K, Kim Y, et al. A CRISPR-Cas9 screen identifies essential CTCF anchor sites for estrogen receptor-driven breast cancer cell proliferation. Nucl Acids Res. 2019;47:9557–72.
Razavi P, Chang MT, Xu G, Bandlamudi C, Ross DS, Vasan N, et al. The Genomic Landscape of Endocrine-Resistant Advanced Breast Cancers. Cancer Cell. 2018;34:427–38.e426.
Fiorito E, Sharma Y, Gilfillan S, Wang S, Singh SK, Satheesh SV, et al. CTCF modulates Estrogen Receptor function through specific chromatin and nuclear matrix interactions. Nucl Acids Res. 2016;44:10588–602.
Arnesen S, Blanchard Z, Williams MM, Berrett KC, Li Z, Oesterreich S, et al. Estrogen Receptor Alpha Mutations in Breast Cancer Cells Cause Gene Expression Changes through Constant Activity and Secondary Effects. Cancer Res. 2021;81:539–51.
Barbaric S, Reinke H, Horz W. Multiple mechanistically distinct functions of SAGA at the PHO5 promoter. Mol Cell Biol. 2003;23:3468–76.
Boyer LA, Langer MR, Crowley KA, Tan S, Denu JM, Peterson CL. Essential role for the SANT domain in the functioning of multiple chromatin remodeling enzymes. Mol Cell. 2002;10:935–42.
Sterner DE, Wang X, Bloom MH, Simon GM, Berger SL. The SANT domain of Ada2 is required for normal acetylation of histones by the yeast SAGA complex. J Biol Chem. 2002;277:8178–86.
Vermeulen M, Eberl HC, Matarese F, Marks H, Denissov S, Butter F, et al. Quantitative interaction proteomics and genome-wide profiling of epigenetic histone marks and their readers. Cell. 2010;142:967–80.
Sun S, Zhong X, Wang C, Sun H, Wang S, Zhou T, et al. BAP18 coactivates androgen receptor action and promotes prostate cancer progression. Nucl Acids Res. 2016;44:8112–28.
Wang X, Wang C, Yan G, Kang Y, Sun G, Wang S, et al. BAP18 is involved in upregulation of CCND1/2 transcription to promote cell growth in oral squamous cell carcinoma. EBioMedicine. 2020;53:102685.
Sun G, Wang C, Wang S, Sun H, Zeng K, Zou R, et al. An H3K4me3 reader, BAP18 as an adaptor of COMPASS-like core subunits co-activates ERalpha action and associates with the sensitivity of antiestrogen in breast cancer. Nucl Acids Res. 2020;48:10768–84.
Tang T, Jiao J, Li D, Sun G, Lin L, Wang C, et al. The function of BAP18 on modulation of androgen receptor action in luteinized granulosa cells from normal weight women with and without PCOS. Mol Cell Endocrinol. 2021;527:111228.
Wei SJ, Li J, Wang JY, Zhang LX, Zhang HB, Du DJ, et al. BAP18 induces growth of non-small-cell lung carcinoma through upregulating transcriptional level of CCND1/2. Eur Rev Med Pharm Sci. 2022;26:3074–82.
Zhang YL, Deng L, Liao L, Yang SY, Hu SY, Ning Y, et al. Chromatin complexes subunit BAP18 promotes triple-negative breast cancer progression through transcriptional activation of oncogene S100A9. Cell Death Dis. 2022;13:408.
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339:819–23.
Wang C, Sun H, Zou R, Zhou T, Wang S, Sun S, et al. MDC1 functionally identified as an androgen receptor co-activator participates in suppression of prostate cancer. Nucl Acids Res. 2015;43:4893–908.
Nelson JD, Denisenko O, Bomsztyk K. Protocol for the fast chromatin immunoprecipitation (ChIP) method. Nat Protoc. 2006;1:179–85.
Muthurajan U, Mattiroli F, Bergeron S, Zhou K, Gu Y, Chakravarthy S, et al. In Vitro Chromatin Assembly: Strategies and Quality Control. Methods Enzymol. 2016;573:3–41.
Hagege H, Klous P, Braem C, Splinter E, Dekker J, Cathala G, et al. Quantitative analysis of chromosome conformation capture assays (3C-qPCR). Nat Protoc. 2007;2:1722–33.
Lee KM, Guerrero-Zotano AL, Servetto A, Sudhan DR, Lin CC, Formisano L, et al. Proline rich 11 (PRR11) overexpression amplifies PI3K signaling and promotes antiestrogen resistance in breast cancer. Nat Commun. 2020;11:5488.
Kong SL, Li G, Loh SL, Sung WK, Liu ET. Cellular reprogramming by the conjoint action of ERalpha, FOXA1, and GATA3 to a ligand-inducible growth state. Mol Syst Biol. 2011;7:526.
Theodorou V, Stark R, Menon S, Carroll JS. GATA3 acts upstream of FOXA1 in mediating ESR1 binding by shaping enhancer accessibility. Genome Res. 2013;23:12–22.
Tan Y, Jin C, Ma W, Hu Y, Tanasa B, Oh S, et al. Dismissal of RNA Polymerase II Underlies a Large Ligand-Induced Enhancer Decommissioning Program. Mol Cell. 2018;71:526–39.e528.
Miranda F, Prazeres H, Mendes F, Martins D, Schmitt F. Resistance to endocrine therapy in HR + and/or HER2 + breast cancer: the most promising predictive biomarkers. Mol Biol Rep. 2022;49:717–33.
Fu X, De Angelis C, Schiff R. Interferon Signaling in Estrogen Receptor-positive Breast Cancer: A Revitalized Topic. Endocrinology 2022;163.
Xu XQ, Pan XH, Wang TT, Wang J, Yang B, He QJ, et al. Intrinsic and acquired resistance to CDK4/6 inhibitors and potential overcoming strategies. Acta Pharm Sin. 2021;42:171–8.
Xu G, Chhangawala S, Cocco E, Razavi P, Cai Y, Otto JE, et al. ARID1A determines luminal identity and therapeutic response in estrogen-receptor-positive breast cancer. Nat Genet. 2020;52:198–207.
Nagarajan S, Rao SV, Sutton J, Cheeseman D, Dunn S, Papachristou EK, et al. ARID1A influences HDAC1/BRD4 activity, intrinsic proliferative capacity and breast cancer treatment response. Nat Genet. 2020;52:187–97.
Qiu Z, Song C, Malakouti N, Murray D, Hariz A, Zimmerman M, et al. Functional interactions between NURF and Ctcf regulate gene expression. Mol Cell Biol. 2015;35:224–37.
Wysocka J, Swigut T, Xiao H, Milne TA, Kwon SY, Landry J, et al. A PHD finger of NURF couples histone H3 lysine 4 trimethylation with chromatin remodelling. Nature. 2006;442:86–90.
Zahid H, Buchholz CR, Singh M, Ciccone MF, Chan A, Nithianantham S, et al. New Design Rules for Developing Potent Cell-Active Inhibitors of the Nucleosome Remodeling Factor (NURF) via BPTF Bromodomain Inhibition. J Med Chem. 2021;64:13902–17.
Guerrero-Zotano AL, Stricker TP, Formisano L, Hutchinson KE, Stover DG, Lee KM, et al. ER(+) Breast Cancers Resistant to Prolonged Neoadjuvant Letrozole Exhibit an E2F4 Transcriptional Program Sensitive to CDK4/6 Inhibitors. Clin Cancer Res. 2018;24:2517–29.
Griffiths JI, Chen J, Cosgrove PA, O’Dea A, Sharma P, Ma C, et al. Serial single-cell genomics reveals convergent subclonal evolution of resistance as early-stage breast cancer patients progress on endocrine plus CDK4/6 therapy. Nat Cancer. 2021;2:658–71.
Walker RR, Patel JR, Gupta A, Davidson AM, Williams CC, Payton-Stewart F, et al. Glyceollins Trigger Anti-Proliferative Effects in Hormone-Dependent Aromatase-Inhibitor-Resistant Breast Cancer Cells through the Induction of Apoptosis. Int J Mol Sci. 2022;23.
Sharifi MN, Anandan A, Grogan P, O’Regan RM. Therapy after cyclin-dependent kinase inhibition in metastatic hormone receptor-positive breast cancer: Resistance mechanisms and novel treatment strategies. Cancer. 2020;126:3400–16.
Jansen VM, Bhola NE, Bauer JA, Formisano L, Lee KM, Hutchinson KE, et al. Kinome-Wide RNA Interference Screen Reveals a Role for PDK1 in Acquired Resistance to CDK4/6 Inhibition in ER-Positive Breast Cancer. Cancer Res. 2017;77:2488–99.
Miller CA, Gindin Y, Lu C, Griffith OL, Griffith M, Shen D, et al. Aromatase inhibition remodels the clonal architecture of estrogen-receptor-positive breast cancers. Nat Commun. 2016;7:12498.
Wardell SE, Ellis MJ, Alley HM, Eisele K, VanArsdale T, Dann SG, et al. Efficacy of SERD/SERM Hybrid-CDK4/6 Inhibitor Combinations in Models of Endocrine Therapy-Resistant Breast Cancer. Clin Cancer Res. 2015;21:5121–30.
Kirkegaard T, Witton CJ, McGlynn LM, Tovey SM, Dunne B, Lyon A, et al. AKT activation predicts outcome in breast cancer patients treated with tamoxifen. J Pathol. 2005;207:139–46.
McDermott MSJ, Sharko AC, Munie J, Kassler S, Melendez T, Lim CU, et al. CDK7 Inhibition is Effective in all the Subtypes of Breast Cancer: Determinants of Response and Synergy with EGFR Inhibition. Cells 2020;9.
Attia YM, Shouman SA, Salama SA, Ivan C, Elsayed AM, Amero P, et al. Blockade of CDK7 Reverses Endocrine Therapy Resistance in Breast Cancer. Int J Mol Sci. 2020;21.
Sahni JM, Keri RA. Targeting bromodomain and extraterminal proteins in breast cancer. Pharm Res. 2018;129:156–76.
Akhtar MS, Heidemann M, Tietjen JR, Zhang DW, Chapman RD, Eick D, et al. TFIIH kinase places bivalent marks on the carboxy-terminal domain of RNA polymerase II. Mol Cell. 2009;34:387–93.
Larochelle S, Amat R, Glover-Cutter K, Sanso M, Zhang C, Allen JJ, et al. Cyclin-dependent kinase control of the initiation-to-elongation switch of RNA polymerase II. Nat Struct Mol Biol. 2012;19:1108–15.
Zeng M, Kwiatkowski NP, Zhang T, Nabet B, Xu M, Liang Y, et al. Targeting MYC dependency in ovarian cancer through inhibition of CDK7 and CDK12/13. Elife 2018;7.
Loven J, Hoke HA, Lin CY, Lau A, Orlando DA, Vakoc CR, et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell. 2013;153:320–34.
Chapuy B, McKeown MR, Lin CY, Monti S, Roemer MG, Qi J, et al. Discovery and characterization of super-enhancer-associated dependencies in diffuse large B cell lymphoma. Cancer Cell. 2013;24:777–90.
Acknowledgements
We thank Professor Kato Shigeaki from center of regional cooperation in Iwaki Meisei University for his important discussion and support. We appreciate Dr. Yunlong Huo and Dr. Fang Dong for their excellent technical assistance. We thank SeqHealth Tech Co., Ltd Wuhan, China, for ChIP-seq and ATAC-seq analysis.
Funding
This study was supported by the National Natural Science Foundation of China (32170603, 31871286 for YZ, 81872015, 82273123 for CW, 32100440 for GS); China Postdoctoral Science Foundation (276066) for GS; Foundation of Liaoning Province of China (LJKZ0756 for Shengli Wang); Local projects supported by the central government (2022JH6/100100035 for YZ); Foreign expert project of Ministry of Science and Technology (G2022006007L for YZ).
Author information
Authors and Affiliations
Contributions
Conceptualization: GS and YZ. Methodology: GS. Software: YB, HL and MH. Validation: CW and YZ. Formal analysis: GS and YZ. Investigation: GS, BZ, MW, RL, DZ and SW. Resources: YW, SW, KZ, SL, and LL. Writing: GS and YZ. Supervision: QZ and YZ.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
Our study makes use of publicly available sequencing datasets as cited in the results section. As such, prior ethics approval has been obtained for these studies and no additional approval is required. All primary breast cancer tissues and adjacent tissues of patients were procured from the Liaoning Cancer Hospital of China Medical University, all of which received permission from patients already.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, G., Wei, Y., Zhou, B. et al. BAP18 facilitates CTCF-mediated chromatin accessible to regulate enhancer activity in breast cancer. Cell Death Differ 30, 1260–1278 (2023). https://doi.org/10.1038/s41418-023-01135-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41418-023-01135-y