Abstract
Chimeric antigen receptor (CAR) T cells are considered a potentially disruptive cancer therapy, showing highly promising results. Their recent success and regulatory approval (both in the USA and Europe) are likely to generate a rapidly increasing demand and a need for the design of robust and scalable manufacturing and distribution models that will ensure timely and cost-effective delivery of the therapy to the patient. However, there are challenging tasks as these therapies are accompanied by a series of constraints and particularities that need to be taken into consideration in the decision-making process. Here, we present an overview of the current state of the art in the CAR T cell market and present novel concepts that can debottleneck key elements of the current supply chain model and, we believe, help this technology achieve its long-term potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sadelain M, Brentjens R, Rivière I, Park J. CD19 CAR therapy acute lymphoblastic Leukemia. Am Soc Clin Oncol Educ B. 2015;35:e360–e363. https://doi.org/10.14694/EdBook_AM.2015.35.e360.
Jackson HJ, Rafiq S, Brentjens RJ. Driving CAR T-cells forward. Nat Rev Clin Oncol 2016;13:370–383. https://doi.org/10.1038/nrclinonc.2016.36.
Maude SL, Laetsch TW, Buechner J, Rives S, Boyer M, Bittencourt H, et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl J Med 2018;378:439–448. https://doi.org/10.1056/NEJMoa1709866.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl J Med 2017;377:NEJMoa1707447. https://doi.org/10.1056/NEJMoa1707447.
EMA. First two CAR-T cell medicines recommended for approval in the European Union [WWW Document]. 2018. http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2018/06/news_detail_002983.jsp&mid=WC0b01ac058004d5c1. Accessed 10 Sept 18.
FDA. Kymriah, Approved Risk Evaluation and Mitigation Strategies (REMS) [WWW Document]. 2015. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=indvremsdetails.page&rems=368. Accessed 13 Jun 2018.
FDA. Yescarta, Approved Risk Evaluation and Mitigation Strategies (REMS) [WWW Document]. 2015. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=375. Accessed 13 Jun 2018.
Uk, C.R. Worldwide cancer statistics [WWW Document]. Cancer Res. UK. 2014. http://www.cancerresearchuk.org/health-professional/cancer-statistics/worldwide-cancer#heading-Zero. Accessed 12 Jun 2018).
Gill S, Maus MV, Porter DL. Chimeric antigen receptor T cell therapy: 25 years in the making. Blood Rev 2016;30:157–167. https://doi.org/10.1016/j.blre.2015.10.003.
HMRN (Haematological Malignancy Research Network). Statistics [WWW Document]. Epidemiol. Cancer Stat. Gr. Dep. Heal. Sci. Area 3 Seebohm Rowntree Build. YORK YO10 5DD. 2018. https://www.hmrn.org/statistics. Accessed 20 July 2018).
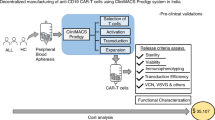
Kaiser AD, Assenmacher M, Schröder B, Meyer M, Orentas R, Bethke, et al. Towards a commercial process for the manufacture of genetically modified T cells for therapy. Cancer Gene Ther. 2015;22:72–78. https://doi.org/10.1038/cgt.2014.78.
Levine BL, Miskin J, Wonnacott K, Keir C. Global manufacturing of CAR T cell therapy. Mol Ther - Methods Clin Dev. 2017;4:92–101. https://doi.org/10.1016/j.omtm.2016.12.006.
Shah, N. Pharmaceutical supply chains: key issues and strategies for optimisation. In: Computers and chemical engineering. Pergamon; 2004. p. 929–941 2004. https://doi.org/10.1016/j.compchemeng.2003.09.022.
Levine BL. Performance-enhancing drugs: design and production of redirected chimeric antigen receptor (CAR) T cells. Cancer Gene Ther. 2015;22:79–84. https://doi.org/10.1038/cgt.2015.5.
Kite Pharma. First CAR T Therapy for certain types of relapsed or refractory B-cell lymphoma [WWW Document]. 2018. https://www.yescartahcp.com/. Accessed 7 Mar 2019.
Novartis. Kymriah® (tisagenlecleucel), first-in-class CAR-T therapy from Novartis, receives second FDA approval to treat appropriate r/r patients with large B-cell lymphoma [WWW Document]. 2018. https://www.novartis.com/news/media-releases/kymriahr-tisagenlecleucel-first-class-car-t-therapy-from-novartis-receives-second-fda-approval-treat-appropriate-rr-patients-large-b-cell-lymphoma. Accessed 24 Jul 2018.
Novartis. KYMRIAH Treatment Process, Dosing & Administration | HCP [WWW Document]. 2018. https://www.hcp.novartis.com/products/kymriah/acute-lymphoblastic-leukemia-children/dosing-and-administration/. Accessed 7 Mar 2019).
U.S. Food and Drug Administration. Summary Basis for Regulatory Action- ATryn 1–16. 2017.
Pantelides CC. Unified frameworks for optimal process planning and scheduling. In: Second conference on foundations of computer aided operations. New York: Cache Publications; 1994. p. 253–274.
Herbert S. Understanding the critical impact of logistics on scale-up & commercialization. Cell Gene Ther Insights. 2017;3:655–662. https://doi.org/10.18609/cgti.2017.067.
Lamb M, Margolin RE, Vitale J. Personalized supply chains for cell therapies. Cell Gene Ther Insights. 2017;3:815–833. https://doi.org/10.18609/cgti.2017.081.
Papathanasiou M. Advances in enabling smart technologies across the cell therapy supply chain. Cell Gene Ther Insights. 2018;4:495–500. https://doi.org/10.18609/cgti.2018.050.
Branke J, Farid SS, Shah N. Industry 4.0: a vision for personalized medicine supply chains? Cell Gene Ther. Insights. 2016;2:263–270. https://doi.org/10.18609/cgti.2016.027.
Hampson G. Regenerative medicines and cell therapy products: is the nice approach fit for purpose? In: CAR-TCR Europe Summit 2018. Office of Health Economics; 2018.
Blankenship K. Kite’s CAR-T manufacturing gets another boost with new Maryland facility [WWW Document]. 2019. https://www.fiercepharma.com/pharma/new-maryland-facility-to-boost-kite-s-car-t-manufacturing-ability. Accessed 2 May 2019.
Susarla N, Karimi IA. Integrated supply chain planning for multinational pharmaceutical enterprises. Comput Chem Eng 2012;42:168–177. https://doi.org/10.1016/j.compchemeng.2012.03.002.
Blau G, Mehta B, Bose S, Pekny J, Sinclair G, Keunker K, Bunch P. Risk management in the development of new products in highly regulated industries. In: Computers and chemical engineering. Pergamon; 2000. p. 659–664. https://doi.org/10.1016/S0098-1354(00)00388-4.
George ED, Farid SS. Strategic biopharmaceutical portfolio development: an analysis of constraint-induced implications. Biotechnol Prog 2008;24:698–713. https://doi.org/10.1021/bp070410s.
Settanni E, Harrington TS, Srai JS. Pharmaceutical supply chain models: a synthesis from a systems view of operations research. Oper Res Perspect. 2017;4:74–95. https://doi.org/10.1016/J.ORP.2017.05.002.
Lakhdar K, Farid SS, Titchener-Hooker NJ, Papageorgiou LG. Medium term planning of biopharmaceutical manufacture with uncertain fermentation titers. Biotechnol Prog 2006;22:1630–1636. https://doi.org/10.1021/bp0601950.
Lakhdar K, Savery J, Papageorgiou LG, Farid SS. Multiobjective long-term planning of biopharmaceutical manufacturing facilities. Biotechnol Prog 2007;23:1383–1393. https://doi.org/10.1021/bp0701362.
Siganporia CC, Ghosh S, Daszkowski T, Papageorgiou LG, Farid SS. Capacity planning for batch and perfusion bioprocesses across multiple biopharmaceutical facilities. Biotechnol Prog 2014;30:594–606. https://doi.org/10.1002/btpr.1860.
Gatica G, Papageorgiou LG, Shah N. Capacity planning under uncertainty for the pharmaceutical industry. Chem Eng Res Des 2003;81:665–678. https://doi.org/10.1205/026387603322150516.
UN. United Nations Population Division [WWW Document]. 2018.
Griffiths, R. and Lakelin, M. Successfully managing the unique demands of cell therapy supply chains 2017, p. 9. Available at: www.pciservices.com (Accessed 15 October 2018).
Acknowledgements
The authors would like to acknowledge expert opinion received through multiple conversations with the User Steering Committee of the Future Targeted Healthcare Manufacturing Hub. Funding from the UK Engineering & Physical Sciences Research Council (EPSRC) for the Future Targeted Healthcare Manufacturing Hub hosted at University College London with UK university partners is gratefully acknowledged (Grant Reference: EP/P006485/1). Financial and in-kind support from the consortium of industrial users and sector organisations is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A
Appendix A
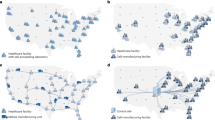
For the patient population forecast studies, we assume a constant average growth of approximately 0.4% [34] and constant incidence rates per 100,000 people ((HMRN (Haematological Malignancy Research Network), 2018)), as presented in Table A.1 in the supplementary information. Based on these assumptions, we present indicative projections (Table A.2, supplementary information) for the liquid cancer patient population in the UK for the next 15 years. In order to estimate the patient population, we only consider liquid cancer types as currently CAR T therapies are considered to be more advanced in that space. Furthermore, the types of liquid cancer here have been chosen based on data availability. Patient population numbers for types not present in the current set are either scarcely provided or not available.
The three final rows of Table A.2 (please see supplementary information) represent: (1) the total number of patients with liquid cancer in the UK for the next 15 years, (2) a scenario where estimates are 20% lower and (3) a scenario where estimates are 20% higher. Figure A1 of the supplementary information represents the ±20% case. Nevertheless, CAR T cell therapies will most probably not be the first line of therapy, therefore limiting the number of eligible patients. Figure A.1 (please see supplementary information) illustrates a potential scenario, where only 10% of the total patient population will be eligible for CAR T cell therapy treatment. Despite this constraint, patient numbers are estimated to increase almost fivefold by 2031, thus challenging CART manufacturing and supply chain scale up.
Rights and permissions
About this article
Cite this article
Papathanasiou, M.M., Stamatis, C., Lakelin, M. et al. Autologous CAR T-cell therapies supply chain: challenges and opportunities?. Cancer Gene Ther 27, 799–809 (2020). https://doi.org/10.1038/s41417-019-0157-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-019-0157-z
This article is cited by
-
Chimeric antigen receptor T cells therapy in solid tumors
Clinical and Translational Oncology (2023)
-
A new immunotherapy strategy targeted CD30 in peripheral T-cell lymphomas: CAR-modified T-cell therapy based on CD30 mAb
Cancer Gene Therapy (2022)
-
A digital platform for the design of patient-centric supply chains
Scientific Reports (2022)
-
Cancer gene therapy 2020: highlights from a challenging year
Cancer Gene Therapy (2022)
-
Industrializing engineered autologous T cells as medicines for solid tumours
Nature Reviews Drug Discovery (2021)