Abstract
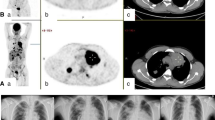
Patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) have a poor prognosis. Chimeric antigen receptor (CAR) modified T cells targeting CD19 hold great promise to improve the complete response rates of DLBCL patients compared with conventional therapies. Here, we conducted a clinical trial to evaluate the efficacy and safety of CAR-T cells. Five patients with relapsed or refractory DLBCL were treated with autologous T cells expressing the 19-41BBz chimeric antigen receptor (CAR) specifically targeted the CD19 antigen (IM19 CAR-T). The development of cytokine release syndrome (CRS) was observed. And the efficacy of IM19 CAR-T cell treatment was measured with positron emission tomography (PET)–computed tomography (CT). Of the four patients evaluable for response, two obtained complete responses (CRs), one obtained partial response (PR), and one had stable disease (SD). Remarkably, among the five patients, only one developed grade 2 CRS while the others only elicited grade 1 CRS. Additionally, the efficacy and safety of IM19 CAR-T cells were correlated with the peak blood level and persistence of CAR-T cells, as well as the immunophenotype of T-cell subsets. Overall, this study indicates the feasibility and effectiveness of IM19 CAR-T cells in the treatment of refractory or relapsed diffuse large B-cell lymphoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The dataset(s) supporting the findings of this study are included within the article.
References
Chaganti S, Illidge T, Barrington S, Mckay P, Linton K, Cwynarski K et al. Guidelines for the management of diffuse large B-cell lymphoma. Br J Haematol. 2016; 174:43–56.
Sehn LH, Gascoyne RD. Diffuse large B-cell lymphoma: optimizing outcome in the context of clinical and biologic heterogeneity. Blood. 2015;125:22.
Ng AK. Diffuse large B-cell lymphoma. Semin Radiat Oncol. 2007;17:169–75.
Seshadri T, Stakiw J, Pintilie M, Keating A, Crump M, Kuruvilla J. Utility of subsequent conventional dose chemotherapy in relapsed/refractory transplant-eligible patients with diffuse large B-cell lymphoma failing platinum-based salvage chemotherapy. Hematology. 2008;13:261–6.
Crump M, Neelapu SS, Farooq U, Van DNE, Kuruvilla J, Westin J, et al. Outcomes in refractory diffuse large B-cell lymphoma: results from the international SCHOLAR-1 study. Blood. 2017;16:S101–S101.
Locke FL, Neelapu SS, Bartlett NL, Siddiqi T, Chavez JC, Hosing CM, et al. Phase 1 Results of ZUMA-1: A Multicenter Study of KTE-C19 Anti-CD19 CAR T Cell Therapy in Refractory Aggressive Lymphoma. Mol Ther. 2017;25:285–95.
Kochenderfer JN, Dudley ME, Kassim SH, Somerville RPT, Carpenter RO, Stetler-Stevenson M, et al. Chemotherapy-refractory diffuse large B-cell lymphoma and indolent B-cell malignancies can be effectively treated with autologous T cells expressing an anti-CD19 chimeric antigen receptor. J Clin Oncol. 2015;33:540–9.
Gardner RA, Finney O, Annesley C, Brakke H, Summers C, Leger K et al. Intent to treat leukemia remission by CD19CAR T cells of defined formulation and dose in children and young adults. Blood. 2017;129:3322–31.
Davila ML, Riviere I, Wang X, Bartido S, Park J, Curran K. et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med. 2014;6:224ra25–224ra25.
Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR, et al. Chimeric antigen receptor–modified T cells for acute lymphoid leukemia. New Engl J Med. 2013;368:1509.
Kochenderfer JN, Dudley ME, Feldman SA, Wilson WH, Spaner DE, Maric I, et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor–transduced T cells. Blood. 2012;119:2709–20.
Barrington SF, Mikhaeel NG, Kostakoglu L, Meignan M, Hutchings M, Müeller SP, et al. Role of imaging in the staging and response assessment of lymphoma: consensus of the international conference on malignant lymphomas imaging working group. J Clin Oncol Off J Am Soc Clin Oncol. 2014;32:3048–58.
Lee DW, Gardner R, Porter DL, Louis CU, Ahmed N, Jensen M, et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124:188–95.
Yip A, Webster RM. The market for chimeric antigen receptor T cell therapies. Nat Rev Drug Discov. 2018;17:161–62.
Turtle CJ, Hanafi L-A, Berger C, Gooley TA, Cherian S, Hudecek M, et al. CD19 CAR–T cells of defined CD4 + :CD8 + composition in adult B cell ALL patients. J Clin Investig. 2016;126:2123–38.
Gattinoni L, Klebanoff CA, Restifo NP. Paths to stemness: building the ultimate antitumour T cell. Nat Rev Cancer. 2012;12:671–84.
Klebanoff CA, Scott CD, Leonardi AJ, Yamamoto TN, Cruz AC, Ouyang C, et al. Memory T cell-driven differentiation of naive cells impairs adoptive immunotherapy. J Clin Investig. 2016;126:318.
Mahnke YD, Brodie TM, Sallusto F, Roederer M, Lugli E. The who’s who of T-cell differentiation: human memory T-cell subsets. Eur J Immunol. 2013;43:2797.
Gattinoni L, Klebanoff CA, Palmer DC, Wrzesinski C, Kerstann K, Yu Z, et al. Acquisition of full effector function in vitro paradoxically impairs the in vivo antitumor efficacy of adoptively transferred CD8 + T cells. J Clin Investig. 2005;115:1616.
Sommermeyer D, Hudecek M, Kosasih PL, Gogishvili T, Maloney DG, Turtle CJ, et al. Chimeric antigen receptor-modified T cells derived from defined CD8|[plus]| and CD4|[plus]| subsets confer superior antitumor reactivity in vivo. Leukemia. 2016;30:492.
Gattinoni L, Lugli E, Ji Y, Pos Z. A human memory T cell subset with stem cell-like properties. Nat Med. 2011;17:1290.
Klebanoff CA, Gattinoni L, Torabiparizi P, Kerstann K, Cardones AR, Finkelstein SE, et al. Central memory self/tumor-reactive CD8 + T cells confer superior antitumor immunity compared with effector memory T cells. Proc Natl Acad Sci USA. 2005;102:9571–6.
Sabatino M, Hu J, Sommariva M, Gautam S, Fellowes V, Hocker JD, et al. Generation of clinical-grade CD19-specific CAR-modified CD8 + memory stem cells for the treatment of human B-cell malignancies. Blood. 2016;128:519.
Lee DW, Kochenderfer JN, Stetlerstevenson M, Cui YK, Delbrook C, Feldman SA, et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet. 2015;385:517.
Maude SL, Frey N, Shaw PA, Aplenc R, Barrett DM, Bunin NJ, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. New Engl J Med. 2014;371:1507.
Acknowledgements
This work was supported by Beijing Natural Science Foundation (7132183), China Health Promotion Foundation (CHPF-zlkysx-001), Key project of Peking University Third Hospital (BYSY2015004), Zhongguancun Frontier Reserve Project, Beijing Science and Technology SME Promotion Project, Beijing Science and Technology Rising Star Project and Beijing Eagle Talent Project. CAR-T cells in this study were provided by Beijing Immunochina Medical Science & Technology Co., Ltd. The clinical trial in this study was also sponsored by Beijing Immunochina Medical Science & Technology Co., Ltd.
Author information
Authors and Affiliations
Contributions
JHM, QFF, LXA, and HT conceived and designed the study; BF, WW, HK, YP, DF, WJ, and JHM performed the clinical examination; JHM, BF, WW, QFF, LXA, HT, and LGH analyzed and interpreted the data; QFF, LXA, and HT designed the CAR and prepared the CAR-T cell product; HT, LGH, BF, and WW wrote the paper. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This study was performed in accordance with the Declaration of Helsinki and approved by the Peking University Third Hospital Institutional Review Board. All patients enrolled in this trial gave written informed consent after a discussion of the possible risks and adverse effects of the therapy.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Trial registration: NCT03344705, Registered November 14, 2017, Prospective clinical trial. https://www.clinicaltrials.gov/ct2/show/NCT03344705?term=immunochina&rank=1
Rights and permissions
About this article
Cite this article
Bao, F., Wan, W., He, T. et al. Autologous CD19-directed chimeric antigen receptor-T cell is an effective and safe treatment to refractory or relapsed diffuse large B-cell lymphoma. Cancer Gene Ther 26, 248–255 (2019). https://doi.org/10.1038/s41417-018-0073-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41417-018-0073-7
This article is cited by
-
Current Challenges and Potential Strategies for Designing a New Generation of Chimeric Antigen Receptor-T cells with High Anti-tumor Activity in Solid Tumors
Current Tissue Microenvironment Reports (2023)
-
Autologous stem cell transplantation in tandem with Anti-CD30 CAR T-cell infusion in relapsed/refractory CD30+ lymphoma
Experimental Hematology & Oncology (2022)
-
Update in Diagnosis and Management of Primary Cutaneous B-Cell Lymphomas
American Journal of Clinical Dermatology (2022)